Abstract
The cytoskeletal protein doublecortin (DCX) is a marker for neuronal cells retaining high potential for structural plasticity, originating from both embryonic and adult neurogenic processes. Some of these cells have been described in the subcortical white matter of neonatal and postnatal mammals. In mice and humans it has been shown they are young neurons migrating through the white matter after birth, reaching the cortex in a sort of protracted neurogenesis. Here we show that DCX+ cells in the white matter of neonatal and young Cetartiodactyla (dolphin and sheep) form large clusters which are not newly generated (in sheep, and likely neither in dolphins) and do not reach the cortical layers, rather appearing “trapped” in the white matter tissue. No direct contact or continuity can be observed between the subventricular zone region and the DCX+ clusters, thus indicating their independence from any neurogenic source (in dolphins further confirmed by the recent demonstration that periventricular neurogenesis is inactive since birth). Cetartiodactyla include two orders of large-brained, relatively long-living mammals (cetaceans and artiodactyls) which were recognized as two separate monophyletic clades until recently, yet, despite the evident morphological distinctions, they are monophyletic in origin. The brain of Cetartiodactyla is characterized by an advanced stage of development at birth, a feature that might explain the occurrence of “static” cell clusters confined within their white matter. These results further confirm the existence of high heterogeneity in the occurrence, distribution and types of structural plasticity among mammals, supporting the emerging view that multiple populations of DCX+, non-newly generated cells can be abundant in large-brained, long-living species.

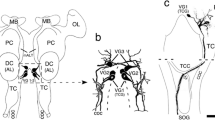
(adapted from Price et al. 2005; in blue, families considered in this study). b Animal species analyzed. c Comparable anatomical levels (L1–L4, anterior to posterior) in dolphin and sheep brain. Dark gray, gray matter; light gray, white matter









Similar content being viewed by others
References
Bagley C (1922) Cortical motor mechanism of the sheep brain. Arch Neurol Psych 7:417–453
Bonfanti L (2006) PSA-NCAM in mammalian structural plasticity and neurogenesis. Prog Neurobiol 80:129–164. https://doi.org/10.1016/j.pneurobio.2006.08.003
Bonfanti L (2016) Adult neurogenesis 50 years later: limits and opportunities in mammals. Front Neurosci 10:44. https://doi.org/10.3389/fnins.2016.00044
Bonfanti L, Nacher J (2012) New scenarios for neuronal structural plasticity in non-neurogenic brain parenchyma: the case of cortical layer II immature neurons. Prog Neurobiol 98:1–15. https://doi.org/10.1016/j.pneurobio.201205.002
Bonfanti L, Olive S, Poulain DA, Theodosis DT (1992) Mapping of the distribution of polysialylated neural cell adhesion molecule throughout the central nervous system of the adult rat: an immunohistochemical study. Neuroscience 49:419–436. https://doi.org/10.1016/0306-4522(92)90107-D
Bonner JT (1980) The evolution of culture in animals. Princeton University Press, Princeton
Borgatti G (1956) Centri nervosi e sistema nervoso centrale. In: Borgatti G, Martini E, Rowinski P, Usuelli F Fisiologia degli animali domestici. Tinarelli, Bologna, p 1382
Brown JP, Couillard-Despres S, Cooper-Kuhn CM, Winkler J, Aigner L, Kuhn HG (2003) Transient expression of doublecortin during adult neurogenesis. J Comp Neurol 467:1–10. https://doi.org/10.1002/cne.10874
Brus M, Keller M, Lévy F (2013a) Temporal features of adult neurogenesis: differences and similarities across mammalian species. Front Neurosci 7:135–144. https://doi.org/10.3389/fnins.2013.00135
Brus M, Meurisse M, Gheusi G, Keller M, Lledo P, Levy F (2013b) Dynamics of olfactory and hippocampal neurogenesis in adult sheep. J Comp Neurol 521:169–188. https://doi.org/10.1002/cne.23169
Chaudhry FA, Reimer RJ, Bellocchio EE, Danbolt NC, Edwards RH, Storm-Mathisen J (1998) The vesicular GABA transporter, VGAT, localizes to synaptic vesicles in sets of glycinergic as well as GABAergic neurons. J Neurosci 18:9733–9750
Cozzi B, Huggenberger S, Oelschläger HHA (2017) The anatomy of dolphins. Insights into body structure and function. In: Chapter 6: brain, spinal cord, and cranial nerves. Academic Press, London, pp 191–285
Dekaban AS, Sadowsky D (1978) Changes in brain weights during the span of human life: relation of brain weights to body heights and body weights. Ann Neurol 4:345–356. https://doi.org/10.1002/ana.410040410
Faykoo-Martinez M, Toor I, Holmes MM (2017) Solving the neurogenesis puzzle: looking for pieces outside the traditional box. Front Neurosci 11:505. https://doi.org/10.3389/fnins.2017.00505
Feliciano DM, Bordey A, Bonfanti L (2015) Noncanonical sites of adult neurogenesis in the mammalian brain. Cold Spring Harb Perspect Biol 7:a018846. https://doi.org/10.1101/cshperspect.a018846
Finlay BL, Uchiyama R (2017) The timing of brain maturation, early experience, and the human social niche. In: Kaas J (ed) Evolution of nervous systems 2e, vol 3. Elsevier, Oxford, pp 123–148
Fung SJ, Joshi D, Allen KM, Sivagnanasundaram S, Rothmond DA, Saunders R, Noble PL, Webster MJ, Shannon Weickert CS (2011) Developmental patterns of doublecortin expression and white matter neuron density in the postnatal primate prefrontal cortex and schizophrenia. PLoS One 6(9):e25194. https://doi.org/10.1371/journal.pone.0025194
Gallyas F (1979) Silver staining of myelin by means of physical development. Neurol Res 1:203–209
Garwicz M, Christensson M, Psounib E (2009) A unifying model for timing of walking onset in humans and other mammals. Proc Natl Acad Sci USA 106:21889–21893. https://doi.org/10.1073/pnas.0905777106
Gatesy J, Milinkovitch M, Waddell V, Stanhope M (1999) Stability of cladistic relationships between Cetacea and higher-level Artiodactyl taxa. Syst Biol 48:6–20
Geraci JR, Lounsbury VJ (2005) Marine mammals ashore: a field guide for strandings, 2nd edn. National Aquarium in Baltimore, Baltimore
Gleeson JG, Lin PT, Flanagan LA, Walsh CA (1999) Doublecortin is a microtubule-associated protein and is expressed widely by migrating neurons. Neuron 23:257–271. https://doi.org/10.1016/S0896-6273(00)80778-3
Gómez-Climent MA, Castillo-Gómez E, Varea E, Guirado R, Blasco-Ibáñez JM, Crespo C, Martínez-Guijarro FJ, Nácher J (2008) A population of prenatally generated cells in the rat paleocortex maintains an immature neuronal phenotype into adulthood. Cereb Cortex 18:2229–2240. https://doi.org/10.1093/cercor/bhm255
Graur D, Higgins DG (1994) Molecular evidence for the inclusion of cetaceans within the order Artiodactyla. Mol Biol Evol 11:357–364
Hahn ME, Jensen C, Dudek DC (1979) Development and evolution of brain size: behavioral implications. Academic Press, New York
Inta D, Alfonso J, von Engelhardt J, Kreuzberg MM, Meyer AH, van Hooft JA, Monyer H (2008) Neurogenesis and widespread forebrain migration of distinct GABAergic neurons from the postnatal subventricular zone. Proc Natl Acad Sci USA 105:20994–20999. https://doi.org/10.1073/pnas.0807059105
Kazu RS, Maldonado J, Mota B, Manger PR, Herculano-Houzel S (2014) Cellular scaling rules for the brain of Artiodactyla include a highly folded cortex with few neurons. Front Neuroanat 8:128. https://doi.org/10.3389/fnana.2014.00128
Kee N, Sivalingam S, Boonstra R, Wojtowicz JM (2002) The utility of Ki-67 and BrdU as proliferative markers of adult neurogenesis. J Neurosci Methods 115:97–105
Kern A, Siebert U, Cozzi B, Hof PR, Oelschlager HHA (2011) Stereology of the neocortex in Odontocetes: qualitative, quantitative, and functional implications. Brain Behav Evol 77:79–90. https://doi.org/10.1159/000323674
König R, Benedetti B, Rotheneichner P, O′Sullivan A, Kreutzer C, Belles M, Nacher J, Weiger TM, Aigner L, Couillard-Després S (2016) Distribution and fate of DCX/PSA-NCAM expressing cells in the adult mammalian cortex: a local reservoir for adult cortical neuroplasticity? Front Biol 11:193–213. https://doi.org/10.1007/s11515-016-1403-5
Kuhn GH, Blomgren K (2011) Developmental dysregulation of adult neurogenesis. Eur J Neurosci 33:1115–1122. https://doi.org/10.1111/j.1460-9568.2011.07610.x
Le Magueresse C, Alfonso A, Khodosevich K, Arroyo Martin AA, Bark C, Monyer H (2011) ‘Small axonless neurons’: postnatally generated neocortical interneurons with delayed functional maturation. J Neurosci 31:16731–16747. https://doi.org/10.1523/JNEUROSCI.4273-11.2011
Le Magueresse C, Alfonso J, Bark C, Eliava M, Khrulev S, Monyer H (2012) Subventricular zone-derived neuroblasts use vasculature as a scaffold to migrate radially to the cortex in neonatal mice. Cereb Cortex 22:2285–2296. https://doi.org/10.1093/cercor/bhr302
Lipp HP, Bonfanti L (2016) Adult neurogenesis in mammals: variations and confusions. Brain Behav Evol 87:205–221. https://doi.org/10.1159/000446905
Lois C, Garcìa-Verdugo JM, Alvarez-Buylla A (1996) Chain migration of neuronal precursors. Science 271:978–981. https://doi.org/10.1126/science.271.5251.978
Luzzati F, Peretto P, Aimar P, Ponti G, Fasolo A, Bonfanti L (2003) Glia-independent chains of neuroblasts through the subcortical parenchyma of the adult rabbit brain. Proc Natl Acad Sci USA 100:13036–13041. https://doi.org/10.1073/pnas.1735482100
Luzzati F, Bonfanti L, Fasolo A, Peretto P (2009) DCX and PSA-NCAM expression identifies a population of neurons preferentially distributed in associative areas of different pallial derivatives and vertebrate species. Cereb Cortex 19:1028–1041. https://doi.org/10.1093/cercor/bhn145
McIntosh GH, Baghurst I, Potter BJ, Hetzel BS (1979) Foetal brain development in the sheep. Neuropath Appl Neurobiol 5:103–114. https://doi.org/10.1111/j.1365-2990.1979.tb00664.x
Mullen RJ, Buck CR, Smith AM (1992) NeuN, a neuronal specific nuclear protein in vertebrates. Development 116:201–211
Murphy WJ, Pevzner PA, O’Brien SJ (2004) Mammalian phylogenomics comes of age. Trends Genet 20:631–639. https://doi.org/10.1016/j.tig.2004.09.005
Nacher J, Crespo C, McEwen BS (2001) Doublecortin expression in the adult rat telencephalon. European J Neurosci 14:629–644. https://doi.org/10.1046/j.0953-816x.2001.01683.x
Paredes MF, James D, Gil-Perotin S, Kim H, Cotter JA, Ng C, Sandoval K, Rowitch DH, Xu D, McQuillen PS, Garcia-Verdugo J-M, Huang EJ, Alvarez-Buylla A (2016) Extensive migration of young neurons into the infant human frontal lobe. Science. https://doi.org/10.1126/science.aaf7073
Parolisi R, Peruffo A, Messina S, Panin M, Montelli S, Giurisato M, Cozzi B, Bonfanti L (2015) Forebrain neuroanatomy of the neonatal and juvenile dolphin (T. truncatus and S. coeruloalba). Front Neuroanat 9:140. https://doi.org/10.3389/fnana.2015.00140
Parolisi R, Cozzi B, Bonfanti L (2017) Non-neurogenic SVZ-like niche in dolphins, mammals devoid of olfaction. Brain Struct Funct 222:2625–2639. https://doi.org/10.1007/s00429-016-1361-3
Peretto P, Merighi A, Fasolo A, Bonfanti L (1999) The subependymal layer in rodents: a site of structural plasticity and cell migration in the adult mammalian brain. Brain Res Bull 49:221–243. https://doi.org/10.1016/S0361-9230(99)00037-4
Peretto P, Giachino C, Aimar P, Fasolo A, Bonfanti L (2005) Chain formation and glial tube assembly in the shift from neonatal to adult subventricular zone of the rodent forebrain. J Comp Neurol 487:407–427. https://doi.org/10.1002/cne.20576
Piumatti M, Palazzo O, La Rosa C, Crociara P, Parolisi R, Luzzati F, Lévy F, Bonfanti L (2018) Non-newly generated, “immature” neurons in the sheep brain are not restricted to cerebral cortex. J Neurosci E-pub. https://doi.org/10.1523/JNEUROSCI.1781-17.2017
Ponti G, Aimar P, Bonfanti L (2006) Cellular composition and cytoarchitecture of the rabbit subventricular zone (SVZ) and its extensions in the forebrain. J Comp Neurol 498:491–507. https://doi.org/10.1002/cne.21043
Ponti G, Peretto P, Bonfanti L (2008) Genesis of neuronal and glial progenitors in the cerebellar cortex of peripubertal and adult rabbits. PLoS One 3:e2366. https://doi.org/10.1371/journal.pone.0002366
Poth C, Fung C, Gunturkun O, Ridgway SH, Oelschlager HHA (2005) Neuron numbers in sensory cortices of five delphinids compared to a physeterid, the pygmy sperm whale. Brain Res Bull 66:357–360. https://doi.org/10.1016/j.brainresbull.2005.02.001
Price SA, Bininda-Emonds OR, Gittleman JL (2005) A complete phylogeny of the whales, dolphins and even-toed hoofed mammals (Cetartiodactyla). Biol Rev Camb Philos Soc 80:445–473
Puelles L, Kuwana E, Puelles E, Bulfone A, Shimamura K, Keleher J, Smiga S, Rubenstein JL (2000) Pallial and subpallial derivatives in the embryonic chick and mouse telencephalon, traced by the expression of the genes Dlx-2, Emx-1, Nkx-2.1, Pax-6, and Tbr-1. J Comp Neurol 424:409–438
Rauschmann MA, Huggenberger S, Kossatz LS, Oelschlager HHA (2006) Head morphology in perinatal dolphins: a window into phylogeny and ontogeny. J Morphol 267:1295–1315. https://doi.org/10.1002/jmor.10477
Reidenberg JS, Laitman JT (2002) Prenatal development in cetaceans. In: Perrin WF, Wursig B, Thewissen JGM (eds) Encyclopedia of marine mammals. Academic Press, San Diego, pp 998–1007
Riccio O, Murthy S, Szabo G, Vutskits L, Kiss JZ, Vitalis T, Lebrand C, Dayer AG (2012) New pool of cortical interneuron precursors in the early postnatal dorsal white matter. Cereb Cortex 22:86–98. https://doi.org/10.1093/cercor/bhr086
Ridgway SH (1990) The central nervous system of the bottlenose dolphin. In: The Bottlenose Dolphin, eds S. Leatherwood and R.R. Reeves, Waltham MA, Academic Press, 69–97
Sanai N, Nguyen T, Ihrie RA, Mirzadeh Z, Tsai HH, Wong M, Gupta N, Berger MS, Huang E, Garcia-Verdugo JM, Rowitch DH, Alvarez-Buylla A (2011) Corridors of migrating neurons in the human brain and their decline during infancy. Nature 478:382–386. https://doi.org/10.1038/nature10487
Schwab ME (2010) Functions of Nogo proteins and their receptors in the nervous system. Nat Rev Neurosci 11:799–811. https://doi.org/10.1038/nrn2936
Snaidero N, Simons M (2014) Myelination at a glance. J Cell Sci 127:2999–3004. https://doi.org/10.1242/jcs.151043
Taylor CR, Shi SR, Chaiwun B, Young L, Imam SA, Cote RJ (1994) Strategies for improving the immunohistochemical staining of various intranuclear prognostic markers in formalin-paraffin sections: androgen receptor, estrogen receptor, progesterone receptor, p53 protein, proliferating cell nuclear antigen, and Ki-67 antigen revealed by antigen retrieval techniques. Hum Pathol 25:263–270
Urbán N, Guillemot F (2014) Neurogenesis in the embryonic and adult brain: same regulators, different roles. Front Cell Neurosci 8:396. https://doi.org/10.3389/fncel.2014.00396
van Kann E, Cozzi B, Hof PR, Oelschläger HHA (2017) Qualitative and quantitative analysis of primary neocortical areas in selected mammals. Brain Behav Evol E-pub. https://doi.org/10.1159/000477431
Varea E, Belles M, Vidueira S, Blasco-Ibáñez JM, Crespo C, Pastor ÁM, Nacher J (2011) PSA-NCAM is expressed in immature, but not recently generated, neurons in the adult cat cerebral cortex layer II. Front Neurosci 5:17. https://doi.org/10.3389/fnins.2011.00017
Vincze A, Mazlo M, Seress L, Komoly S, Abraham H (2008) A correlative light and electron microscopic study of postnatal myelination in the murine corpus callosum. Int J Dev Neurosci 26:575–584. https://doi.org/10.1016/j.ijdevneu.2008.05.003
Wiedenmann B, Franke WW (1985) Identification and localization of synaptophysin, an integral membrane glycoprotein of Mr 38,000 characteristic of presynaptic vesicles. Cell 41:1017–1028
Workman AD, Charvet CJ, Clancy B, Darlington RB, Finlay BL (2013) Modeling transformations of neurodevelopmental sequences across mammalian species. J Neurosci 33:7368–7383. https://doi.org/10.1523/JNEUROSCI.5746-12.2013
Xu CJ, Wang JL, Jin WL (2015) The neural stem cell microenvironment: Focusing on axon guidance molecules and myelin-associated factors. J Mol Neurosci 56:887–897. https://doi.org/10.1007/s12031-015-0538-1
Zamenhof S, van Marthens E (1976) Neonatal and adult brain parameters in mice selected for adult brain weight. Dev Psychobiol 9:587–593. https://doi.org/10.1002/dev.420090612
Acknowledgements
The authors thank Bruno Cozzi (expertise in dolphin and sheep brain neuroanatomy) and Barbara Finlay and Christine Charvet (expertise in neurodevelopmental “translating time” modeling) for helpful discussion; Alessandra Pecora for technical help and Enrica Boda for kindly providing cryostat mouse sections.
Funding
The present work has been supported by MIUR-PRIN2015 (Grant 2015Y5W9YP).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Ethical approval
All experiments have been carried out on fixed tissue material obtained from institutions which hold the appropriate authorizations. Dolphin tissues were stored in the Mediterranean Marine Mammal Tissue Bank (MMMTB) of the University of Padova, Italy, a CITES-recognized (IT020) research center and tissue bank, sponsored by the Italian Ministry of the Environment and the University of Padova, with the aim of harvesting tissues from wild and captive cetaceans and distributing them to qualified research centers worldwide. Sheep tissues came from the INRA research center (Nouzilly; Indre et Loire, France—ethical permissions reported in Brus et al. 2013b).
Research involving human participants and/or animals
Not applicable.
Rights and permissions
About this article
Cite this article
La Rosa, C., Parolisi, R., Palazzo, O. et al. Clusters of DCX+ cells “trapped” in the subcortical white matter of early postnatal Cetartiodactyla (Tursiops truncatus, Stenella coeruloalba and Ovis aries). Brain Struct Funct 223, 3613–3632 (2018). https://doi.org/10.1007/s00429-018-1708-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-018-1708-z