Abstract
The proposed role of CDH1 (E-cadherin gene) methylation as a mechanism of gene inactivation in invasive lobular carcinoma (ILC) remains inconclusive. For many years, CDH1 promoter hypermethylation has been regarded as a mechanism for gene inactivation in ILC. However, this assumption has primarily relied on non-quantitative assays, which have reported CDH1 methylation frequencies ranging from 26 to 93% at CpG sites within the island region. Few studies employing quantitative methods and covering CpG island shores, regions of relatively low CpG density situated proximal to conventional promoter CpGs, have been conducted, revealing lower percentages of methylation ranging from 0 to 51%. Therefore, using the quantitative pyrosequencing method, we examined CDH1 methylation in the island region and shores in E-cadherin deficient ILC cases (15 with CDH1 mutation and 22 non-mutated), 19 cases of invasive breast carcinomas non-special type (IBC-NSTs), and five cases of usual ductal hyperplasia (UDH). Our analysis revealed CDH1 methylation frequencies ranging from 3 to 64%, with no significant increase in methylation levels in any group of ILCs (median = 12%) compared to IBC-NST (median = 15%). In addition, considering the poorly studied association between the number of tumor-infiltrating lymphocytes (TILs) and CDH1 methylation in breast cancer, we undertook a thorough analysis within our dataset. Our findings revealed a positive correlation between CDH1 methylation and the presence of TILs (r = 0.5; p-value < 0.05), shedding light on an aspect of breast cancer biology warranting further investigation. These findings challenge CDH1 methylation as a CDH1 inactivation mechanism in ILC and highlight TILs as a potential confounding factor in gene methylation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Invasive lobular carcinoma (ILC) is the second most common type of invasive breast cancer, accounting for around 10–15% of all cases. ILC is characterized by its unique growth pattern. The key molecular hallmark is the loss of the epithelial cell-cell adhesion molecule E-cadherin, encoded by CDH1, which occurs in 85% of ILC [1]. The molecular mechanisms involved in the decrease or even loss of this protein vary. The CDH1 gene can be inactivated by mutations (50–60% cases) and loss of heterozygosity [1].
For many years, CDH1 promoter hypermethylation has been accepted as a mechanism for gene inactivation in ILC. This assumption largely stems from non-quantitative assays, predominantly methylation-specific PCR (MSP), which reported CDH1 methylation frequencies ranging from 26 to 93% [2,3,4,5,6,7]. However, it has been demonstrated that MSP can yield a significant number of false-positive results [8].
The comprehensive TCGA study by Ciriello et al. [9] challenged the hypothesis of frequent CDH1 methylation in ILC. Analyzing 111 ILCs via Illumina Infinium DNA methylation HumanMethylation 27 (HM27) and HumanMethylation 450 (HM450) platforms, the authors unveiled similar CDH1 methylation patterns in ILCs and invasive breast carcinomas non-special type (IBC-NSTs), the latter characterized by preserved E-cadherin expression. While the study encompassed both CDH1 wild-type and mutated ILCs, it lacked a dedicated analysis of methylation data stratified by mutation status or CDH1 mRNA expression, positive in 13% of samples. More recently, Alexander et al. [10] also failed to identify significant CDH1 promoter methylation in nine ILC cases exhibiting varying levels of E-cadherin expression through methylation EPIC BeadChip 850K array analysis.
It is noteworthy that a variable that has been underexplored in CDH1 methylation studies in breast cancer is the role of tumor-infiltrating lymphocytes (TILs), despite Lombaerts et al. [5] first describing lymphocyte infiltration as a factor to consider in 2004 due to its potential influence on the detection of CDH1 promoter methylation in breast tumors.
In light of these discoveries, we hypothesized that if CDH1 methylation contributes to CDH1 gene inactivation in ILCs, it would be more prevalent in tumors lacking E-cadherin expression and devoid of CDH1 mutation. In this selected group of cases, CDH1 methylation could present itself as a viable alternative mechanism for inducing inactivation, complementing the role typically fulfilled by gene mutations. To test this hypothesis and to evaluate the possible impact of the abundance of TILs on methylation results, we analyze a group of ductal and lobular carcinomas with different CDH1 mutational status and TIL abundance by pyrosequencing.
Methods
The study received approval from the Local Ethics Committee (Ramón y Cajal Research Ethics Committee reference 223/18). A total of 61 cases were selected from the Pathology Department of Ramón y Cajal University Hospital (Madrid, Spain). The selection process for the studied cases was based on data availability from prior sequencing studies conducted in our laboratory, along with more recent cases diagnosed within the past year through the Pathology Department. These latter cases underwent thorough DNA sequencing as part of their evaluation process. Clinical data were obtained from clinical databases. Histological evaluation, immunohistochemistry, and sequencing were carried out as previously reported [11].
TIL evaluation was conducted in regions where DNA was extracted for methylation analysis, following the recommendations of the TILs Working Group [12].
Genomic DNA (2 μg) from all tumors was used for sodium bisulfite treatment using the EpiTect Bisulfite kit (Qiagen). This approach ensures the complete conversion of unmethylated cytosine to uracil, enabling the detection of methylated CpGs. Four sets of primers were designed, covering 18 CpG dinucleotides in the regulatory regions of the gene CDH1–N-shore, CpG Island, and S-shore, using the PyroMark Assay Design 2.0 software (Qiagen) (Supplementary Table 1). Quantitative pyrosequencing was employed to assess the DNA methylation of these regulatory regions. PCR amplification was conducted with PyroMark PCR Kit (Qiagen) as per the manufacturer’s instructions. Pyrosequencing was performed using the PyroMark Gold Q24 Reagents (Qiagen) on a PyroMark Q24 platform. Data analysis utilized the PyroMark Q24 2.0.6. software (Qiagen). Median methylation values for each CpG were compared among the three groups of tumors (Kruskal-Wallis or ANOVA test). To examine differences in methylation levels across studied regions, the median of the mean methylation values of the CpG sites per region were compared among groups (Kruskal-Wallis or ANOVA test). All statistical tests and plots were conducted using the R software.
Results and discussion
To ascertain the prevalence of CDH1 methylation in ILCs characterized by both the absence of CDH1 mutation in the exonic region and E-cadherin expression, we conducted quantitative pyrosequencing on a cohort of 22 ILC cases that had undergone comprehensive massive parallel sequencing, revealing a lack of CDH1 mutations and complete E-cadherin expression absence [11, 13, 14]. For comparative purposes, we analyzed 15 ILC cases with CDH1 mutations and full E-cadherin expression loss, along with 19 IBC-NSTs marked by preserved E-cadherin expression and no CDH1 mutation and five cases of usual ductal hyperplasia (UDH). The main clinicopathological and molecular data of the patients are presented in Supplementary Table 2. In summary, tumors were diagnosed in patients aged between 36 and 92 years old (median = 63 years old). Concerning the histological type, 30% of the ILC tumors exhibited pleomorphic lobular characteristics. In terms of molecular subtype, 86% of the tumors were luminal (32 ILC and 16 IBC-NST), 12% were triple-negative (4 ILC and 3 IBC-NST), and 2% were luminal HER2+ (1 ILC).
Pyrosequencing is a high-resolution method for the detection of DNA methylation and provides quantitative information for each CpG site under study, allowing for the control of bisulfite conversion efficiency. Pyrosequencing is the technique with the best reproducibility (even higher than methylation array) and can work well even on minute amounts of highly fragmented DNA [15].
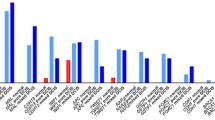
Within this cohort of 56 primary tumors, we comprehensively examined the methylation status of 18 CpG dinucleotides situated in the CpG island (103 bp) of the CDH1 gene and in the Northern and Southern shore (N-shore, S-shore) regions. Analyzing the differences in methylation frequency according to molecular phenotype (triple-negative vs. luminal), we did not observe statistically significant differences, neither in general nor in any of the methylation zones (N-shore, island, and S-shore) (p-value > 0.05). The median methylation frequency in ILCs was 12%, while in IBC-NSTs, it was 15% (p-value > 0.05). The CpG island region encompassed the majority of sites explored in prior MSP studies as well as four CpGs as scrutinized by Ciriello et al. [9] and five CpGs by Alexander et al. [10] through methylation arrays (Supplementary Tables 3, 4, 5). Notably, the observed methylation values in the CpG island were generally modest (ranging between 3 and 18%) (Fig. 1a). CpG methylation values statistically differ among the studied groups for CpG sites at positions 68737141, 68737278, 68737296, and 68737299 located in the CpG island region. Interestingly, these CpGs exhibited slightly heightened methylation levels in IBC-NSTs (p-value < 0.05) (Supplementary Fig. 1 B-E). Furthermore, there were significant differences in methylation levels in the whole island region between the group of mutated ILCs and IBC-NSTs, the latter being higher (Fig. 1b).
Tissue- and cancer-specific differentially methylated regions can occur not only within CpG islands themselves but also within CpG island shores, regions of relatively low CpG density, situated proximal to conventional promoter CpGs (up to 2 kb distant). This suggests the potential involvement of shore methylation in tissue differentiation, epigenetic reprogramming, and cancer [16]. Intriguingly, the analysis of CDH1 shore methylation has not been analyzed in MSP studies (Supplementary Table 3). Therefore, we extended our primer design to CpGs located in both N-shore and S-shore (Supplementary Table 3). Methylation levels in these regions were, in general, higher than in the CpG island (ranging from 4 to 35% and 14 to 64%, respectively) (Fig. 1a). There were significant differences between the studied groups for CpG site at position 68737077 in terms of CpG site-specific comparisons (Supplementary Fig. 1A), but there were no significant differences for whole region assessments (Fig. 1b).
We further examined these CpG sites in the five non-tumoral tissue samples, revealing lower methylation percentages compared to tumor samples across all regions (Fig. 1a).
In an effort to corroborate our findings, we compare our results with those reported by Ciriello et al. [9] and Alexander et al. [10]. Unfortunately, the available datasets from Ciriello et al. lack explicit specification of methylation beta values corresponding to the individual probes, offering a graphical overview instead. Since they did not make a differential analysis between the methylation status of CDH1-mutated and non-mutated cases, we compared the methylation frequencies at each CpG site for both ILC groups combined (with and without CDH1 mutation). Conversely, the dataset provided by Alexander et al. [10] allowed us to compare methylation levels in ILCs according to CDH1 mutation status, although the small number of cases lacking CDH1 mutation (n = 4) was a significant limitation of data reproducibility. In general, we observed similarity in methylation levels when compared to those outlined by Ciriello et al. [9], while we demonstrated lower methylation levels in contrast to those observed in the study by Alexander et al. [10] (Supplementary Tables 4 and 5). Furthermore, Fridrichova et al. [17] reported CDH1 methylation levels assessed by pyrosequencing across seven identical CpGs situated within the CpG island among 24 ILC cases, 178 invasive ductal carcinoma, and four other breast cancer patients. Although the mutational status of ILC cases was not assessed in this study, consistent with our current results, there were no disparities in DNA methylation across these groups, and the average value in tumors and paired lymph node metastasis remained below 10.5% [17].
While we did not observe substantial differences in CDH1 methylation across diverse tumor subtypes, noteworthy instances of elevated methylation were noted in selected tumors, such as cases 2, 8, 11, 50, or 56, among others. The relevant aspect to be considered is that CDH1 methylation can occur in TILs, thereby introducing a confounding element that can lead to false positive outcomes, particularly when using MSP [5]. To confirm this hypothesis, we conducted a correlation analysis between TILs and methylation levels across different CpGs, unveiling a modest yet statistically significant correlation between TILs and methylation levels across all examined regions (p-value < 0.05) (Fig. 2). In our observations, the percentage of TILs was found to be slightly higher in the IBC-NST group (median = 30%), exhibiting a statistically significant difference compared to the mutated-ILC group (median = 5%) (p-value < 0.05). This disparity could influence the higher methylation percentage observed in the IBC-NST group across all studied regions (Fig. 1; Supplementary Fig. 1). Additionally, UDH displayed low methylation percentages (median 11%, 6%, and 16% methylation in N-shore, Island, and S-shore regions, respectively) (Fig. 1a), alongside a considerably low TILs percentage ranging from 0 to 10% (median = 4%), which could influence the low methylation levels.
In conclusion, our findings, facilitated by high-resolution quantitative detection methodology, indicated that the frequency and extent of CDH1 gene methylation in ILCs are not higher than those observed in IBC-NSTs. This result held true irrespective of the presence or absence of CDH1 mutations, thereby challenging the notion of CDH1 methylation as a pervasive mechanism for CDH1 gene inactivation. Moreover, our analysis suggested the potential impact of TIL abundance on CDH1 methylation analysis. Importantly, the conspicuous loss of E-cadherin in the non-mutated ILC subgroup might be driven by mechanisms beyond DNA methylation. The intricate interplay of additional genetic and epigenetic mechanisms, along with non-genetic determinants such as cellular signaling pathways, environmental factors, and cellular context, holds promise in shedding light on alternative mechanisms to the loss of CDH1 for the lobular phenotype [18].
Data availability
Additional information on this article can be found in supplementary material.
Abbreviations
- ILC:
-
Invasive lobular carcinoma
- IBC-NSTs:
-
Invasive breast carcinoma non-special type
- TILs:
-
Tumor-infiltrating lymphocytes
- MSP:
-
Methylation-specific PCR
- HM27:
-
HumanMethylation 27
- HM450:
-
HumanMethylation 450
- N-shore:
-
Northern shore
- S-shore:
-
Southern shore
References
WHO Classification of Tumours Editorial Board, International Agency for Research on Cancer, World Health Organization (2019) WHO classification of tumours. Breast Tumours. International Agency for Research on Cancer, Lyon
Droufakou S, Deshmane V, Roylance R et al (2001) Multiple ways of silencing E-cadherin gene expression in lobular carcinoma of the breast. Int J Cancer 92:404–408. https://doi.org/10.1002/ijc.1208
Sarrió D, Moreno-Bueno G, Hardisson D et al (2003) Epigenetic and genetic alterations of APC and CDH1 genes in lobular breast cancer: relationships with abnormal E-cadherin and catenin expression and microsatellite instability: E-Cadherin, Catenins and APC. Int J Cancer 106:208–215. https://doi.org/10.1002/ijc.11197
Zou D, Yoon H-S, Perez D et al (2009) Epigenetic silencing in non-neoplastic epithelia identifies E-cadherin (CDH1) as a target for chemoprevention of lobular neoplasia. J Pathol 218:265–272. https://doi.org/10.1002/path.2541
Lombaerts M, Middeldorp JW, van der Weide E et al (2004) Infiltrating leukocytes confound the detection of E-cadherin promoter methylation in tumors. Biochem Biophys Res Commun 319:697–704. https://doi.org/10.1016/j.bbrc.2004.05.041
Shinozaki M, Hoon DSB, Giuliano AE et al (2005) Distinct hypermethylation profile of primary breast cancer is associated with sentinel lymph node metastasis. Clin Cancer Res 11:2156–2162. https://doi.org/10.1158/1078-0432.CCR-04-1810
Liu J, Sun X, Qin S et al (2016) CDH1 promoter methylation correlates with decreased gene expression and poor prognosis in patients with breast cancer. Oncol Lett 11:2635–2643. https://doi.org/10.3892/ol.2016.4274
Bücker L, Lehmann U (2022) CDH1 (E-cadherin) Gene methylation in human breast cancer: critical appraisal of a long and twisted story. Cancers 14:4377. https://doi.org/10.3390/cancers14184377
Ciriello G, Gatza ML, Beck AH et al (2015) Comprehensive molecular portraits of invasive lobular breast cancer. Cell 163:506–519. https://doi.org/10.1016/j.cell.2015.09.033
Alexander J, Mariani O, Meaudre C et al (2022) Assessment of the molecular heterogeneity of E-cadherin expression in invasive lobular breast cancer. Cancers 14:295. https://doi.org/10.3390/cancers14020295
González-Martínez S, Pizarro D, Pérez-Mies B et al (2022) Differences in the molecular profile between primary breast carcinomas and their cutaneous metastases. Cancers 14:1151. https://doi.org/10.3390/cancers14051151
Salgado R, Denkert C, Demaria S et al (2015) The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: recommendations by an International TILs Working Group 2014. Ann Oncol 26:259–271. https://doi.org/10.1093/annonc/mdu450
Rosa-Rosa J, Caniego-Casas T, Leskela S et al (2019) High frequency of ERBB2 activating mutations in invasive lobular breast carcinoma with pleomorphic features. Cancers 11:74. https://doi.org/10.3390/cancers11010074
Pérez-Mies B, Caniego-Casas T, Carretero-Barrio I et al (2022) The clonal relationship between the ductal and lobular components of mixed ductal-lobular carcinomas suggested a ductal origin in most tumors. Am J Surg Pathol 46:1545–1553. https://doi.org/10.1097/PAS.0000000000001936
The BLUEPRINT consortium, Bock C, Halbritter F et al (2016) Quantitative comparison of DNA methylation assays for biomarker development and clinical applications. Nat Biotechnol 34:726–737. https://doi.org/10.1038/nbt.3605
Irizarry RA, Ladd-Acosta C, Wen B et al (2009) The human colon cancer methylome shows similar hypo- and hypermethylation at conserved tissue-specific CpG island shores. Nat Genet 41:178–186. https://doi.org/10.1038/ng.298
Fridrichova I, Smolkova B, Kajabova V et al (2015) CXCL12 and ADAM23 hypermethylation are associated with advanced breast cancers. Transl Res 165:717–730. https://doi.org/10.1016/j.trsl.2014.12.006
Dopeso H, Gazzo AM, Derakhshan F et al (2024) Genomic and epigenomic basis of breast invasive lobular carcinomas lacking CDH1 genetic alterations. npj Precis Onc 8:33. https://doi.org/10.1038/s41698-024-00508-x
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. This study was funded by grants from the Instituto de Salud Carlos III (ISCIII) (PI19/01331 and PI22/01892) and was realized thanks to the Short-Term Scientific Mission Grant awarded by the Cost action CA19138 to SG-M.
Author information
Authors and Affiliations
Contributions
SG-M carried out most of the experimental process, the statistical analyses, the preparation of the original manuscript, and contributed to the conceptualization stage. VHK contributed to the experimental part, including primer design. JP and BS acted as corresponding authors were responsible for the conceptualization, contributed to the process of writing and revising the manuscript, and ensured its consistency. BP-M, ICB, TC-C, and JP performed the histological examination of the tumors. DS, GM-B, MG, JP-G, and JC reviewed and provided critical input to the original manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Disclaimer
The funders played no role in study design, data collection, analysis and interpretation of data, or the writing of this manuscript.
Ethics approval
The study received approval from the Local Ethics Committee (Ramón y Cajal Research Ethics Committee reference 223/18) at the Ramón y Cajal University Hospital, 28034 Madrid, Spain.
Competing interests
Consulting/advisor: Roche, AstraZeneca, Seattle Genetics, Daiichi Sankyo, Lilly, Merck Sharp&Dohme, Leuko, Bioasis, Clovis Oncology, Boehringer Ingelheim, Ellipses, Hibercell, BioInvent, Gemoab, Gilead, Menarini, Zymeworks, Reveal Genomics, Scorpion Therapeutics, Expres2ion Biotechnologies, Jazz Pharmatheuticals, Abbvie. Honoraria: Roche , Novartis , Eisai, Pfizer, Lilly, Merck Sharp&Dohme, Daiichi Sankyo, Astrazeneca, Gilead, Steamline Therapeutics. Research funding to the Institution: Roche, Ariad pharmaceuticals, AstraZeneca, Baxalta GMBH/Servier Affaires, Bayer healthcare, Eisai, F.Hoffman-La Roche, Guardanth health, Merck Sharp&Dohme, Pfizer, Piqur Therapeutics, Queen Mary University of London, IQVIA. Stock: MAJ3 Capital, Leuko (relative). Travel, accommodation, expenses: Roche, Novartis, Eisai, Pfizer, Daiichi Sankyo, Astrazeneca, Gilead, Merck Sharp&Dohme, Steamline. Patents: Pharmaceutical Combinations of A Pi3k Inhibitor And A Microtubule Destabilizing Agent. Javier Cortés Castán, Alejandro Piris Giménez, Violeta Serra Elizalde. WO 2014/199294 A. ISSUED. Her2 as a predictor of response to dual HER2 blockade in the absence of cytotoxic therapy. Aleix Prat, Antonio Llombart, Javier Cortés.US 2019/ 0338368 A1. LICENSED
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(XLSX 917 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
González-Martínez, S., Kajabova, V.H., Pérez-Mies, B. et al. CDH1 methylation analysis in invasive lobular breast carcinomas with and without gene mutation. Virchows Arch 485, 291–297 (2024). https://doi.org/10.1007/s00428-024-03814-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-024-03814-8