Abstract
Main conclusion
G6PDH negatively regulates viral accumulation in Nicotiana benthamiana through RBOHB-associated ROS signaling. Anti-oxidative metabolism and phytohormone-mediated immunity responses play important roles in virus infection.
Abstract
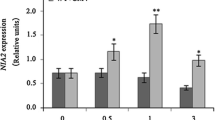
Glucose-6-phosphate dehydrogenase (G6PDH) is an enzyme in the pentose phosphate pathway, which plays an important role in maintaining intracellular redox homeostasis and has functions in plant growth, development and stress tolerance. However, the role of G6PDH in plants response to virus infection is poorly understood. In this study, NbG6PDH was found to be down-regulated after Chilli veinal mottle virus (ChiVMV-GFP) infection in Nicotiana benthamiana. Subcellular localization of NbG6PDH showed that it was punctate distributed in the protoplasm. Silencing of NbG6PDH reduced the sensitivity of N. benthamiana plants to ChiVMV-GFP. By contrast, transient overexpression of NbG6PDH promoted the accumulation of the virus. The results of physiological indexes showed that glutathione (GSH), catalase (CAT) and proline played an important role in maintaining plants physiological homeostasis. The results of gene expression detection showed that jasmonic acid/ethylene (JA/ET) signaling pathway was significantly correlated with the response of N. benthamiana to ChiVMV-GFP infection, and the changes of N. benthamiana respiratory burst oxidase homologues B (NbRBOHB) indicated that the NbG6PDH-dependent ROS may be regulated by NbRBOHB. Pretreatment of the inducer of reactive oxygen species (ROS) promoted virus infection, whereas inhibitor of ROS alleviated virus infection. Thus, our results indicate that the promoting effect of NbG6PDH on ChiVMV-GFP infection may be related to the NbRBOHB-regulated ROS production.








Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- CAT:
-
Catalase
- ChiVMV:
-
Chilli veinal mottle virus
- dpi:
-
Day post infection
- ET:
-
Ethylene
- GSH:
-
Glutathione
- MDA:
-
Malondialdehyde
- POD:
-
Peroxidase
- JA:
-
Jasmonic acid
- SOD:
-
Superoxide dismutase
- RBOH:
-
Respiratory burst oxidase homologues
- ROS:
-
Reactive oxygen species
References
Adhikari A, Adhikari S, Ghosh S et al (2020) Imbalance of redox homeostasis and antioxidant defense status in maize under chromium (VI) stress. Environ Exp Bot 169:103873. https://doi.org/10.1016/j.envexpbot.2019.103873
Alazem M, Lin N-S (2015) Roles of plant hormones in the regulation of host-virus interactions. Mol Plant Pathol 16:529–540. https://doi.org/10.1111/mpp.12204
Asai S, Yoshioka M, Nomura H et al (2011) A plastidic glucose-6-phosphate dehydrogenase is responsible for hypersensitive response cell death and reactive oxygen species production. J Gen Plant Pathol 77:152–162. https://doi.org/10.1007/s10327-011-0304-3
Basu S, Kumar G (2021) Exploring the significant contribution of silicon in regulation of cellular redox homeostasis for conferring stress tolerance in plants. Plant Physiol Biochem 166:393–404. https://doi.org/10.1016/j.plaphy.2021.06.005
Chen Y-J, Deng X-G, Peng X-J et al (2014) Turnip crinkle virus with nonviral gene cancels the effect of silencing suppressors of P19 and 2b in Arabidopsis thaliana. Physiol Mol Plant Pathol 88:94–100. https://doi.org/10.1016/j.pmpp.2014.10.002
Chen L-J, Zou W-S, Wu G et al (2018) Tobacco alpha-expansin EXPA4 plays a role in Nicotiana benthamiana defence against Tobacco mosaic virus. Planta 247:355–368. https://doi.org/10.1007/s00425-017-2785-6
Cimini S, Gualtieri C, Anca M et al (2019) Redox balance-DDR-miRNA triangle: relevance in genome stability and stress responses in plants. Front Plant Sci 10:989. https://doi.org/10.3389/fpls.2019.00989
Dal Santo S, Stampfl H, Krasensky J et al (2012) Stress-induced GSK3 regulates the redox stress response by phosphorylating glucose-6-phosphate dehydrogenase in Arabidopsis. Plant Cell 24:3380–3392. https://doi.org/10.1105/tpc.112.101279
Ding Y, Sun T, Ao K et al (2018) Opposite roles of salicylic acid receptors NPR1 and NPR3/NPR4 in transcriptional regulation of plant immunity. Cell 173:1454-1467.e15. https://doi.org/10.1016/j.cell.2018.03.044
Gajewska E, Skłodowska M, Słaba M, Mazur J (2006) Effect of nickel on antioxidative enzyme activities, proline and chlorophyll contents in wheat shoots. Biol Plant 50:653–659. https://doi.org/10.1007/s10535-006-0102-5
Gao F, Jin J, Zou W et al (2016) Geographically driven adaptation of chilli veinal mottle virus revealed by genetic diversity analysis of the coat protein gene. Arch Virol 161:1329–1333. https://doi.org/10.1007/s00705-016-2761-7
Genty B, Briantais J-M, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta Gen Subj 990:87–92. https://doi.org/10.1016/S0304-4165(89)80016-9
Halim VA, Altmann S, Ellinger D et al (2009) PAMP-induced defense responses in potato require both salicylic acid and jasmonic acid. Plant J 57:230–242. https://doi.org/10.1111/j.1365-313X.2008.03688.x
He Q, Li P, Zhang W, Bi Y (2020) Cytoplasmic glucose-6-phosphate dehydrogenase plays an important role in the silicon-enhanced alkaline tolerance in highland barley. Funct Plant Biol 48:119–130. https://doi.org/10.1071/FP20084
Hu C, Wei C, Ma Q et al (2021) Ethylene response factors 15 and 16 trigger jasmonate biosynthesis in tomato during herbivore resistance. Plant Physiol 185:1182–1197. https://doi.org/10.1093/plphys/kiaa089
Huang J, Han R, Ji F et al (2022) Glucose-6-phosphate dehydrogenase and abscisic acid mediate programmed cell death induced by aluminum toxicity in soybean root tips. J Hazard Mater 425:127964. https://doi.org/10.1016/j.jhazmat.2021.127964
Hyodo K, Hashimoto K, Kuchitsu K et al (2017) Harnessing host ROS-generating machinery for the robust genome replication of a plant RNA virus. Proc Natl Acad Sci USA 114:E1282–E1290. https://doi.org/10.1073/pnas.1610212114
Jin J, Wang X, Wang J et al (2021) Glucose-6-phosphate dehydrogenase is involved in the tolerance of soybean seedlings to low nitrogen stress. Agronomy 11(14):637. https://doi.org/10.3390/agronomy11040637
Landi S, Capasso G, Esposito S (2021) Different G6PDH isoforms show specific roles in acclimation to cold stress at various growth stages of barley (Hordeum vulgare) and Arabidopsis thaliana. Plant Physiol Biochem 169:190–202. https://doi.org/10.1016/j.plaphy.2021.11.017
Lee H-R, An HJ, You YG et al (2013) Development of a novel codominant molecular marker for chili veinal mottle virus resistance in Capsicum annuum L. Euphytica 193:197–205. https://doi.org/10.1007/s10681-013-0897-z
Lee J-H, An J-T, Siddique MI et al (2017) Identification and molecular genetic mapping of Chili veinal mottle virus (ChiVMV) resistance genes in pepper (Capsicum annuum). Mol Breed 37:121. https://doi.org/10.1007/s11032-017-0717-6
Li Y, Zhang J, Zhao F et al (2016) The interaction between Turnip crinkle virus p38 and Cucumber mosaic virus 2b and its critical domains. Virus Res 222:94–105. https://doi.org/10.1016/j.virusres.2016.06.005
Li N, Wang X, Ma B et al (2021) A leucoanthocyanidin dioxygenase gene (RtLDOX2) from the feral forage plant Reaumuria trigyna promotes the accumulation of flavonoids and improves tolerance to abiotic stresses. J Plant Res 134:1121–1138. https://doi.org/10.1007/s10265-021-01315-2
Lichtenthaler H, Wellburn A (1982) Determination of total carotenoids and chlorophylls A and B of leaf in different solvents. Biochem Soc Trans 11:591–592. https://doi.org/10.1042/bst0110591
Liu Y, Schiff M, Dinesh-Kumar SP (2002) Virus-induced gene silencing in tomato. Plant J 31:777–786. https://doi.org/10.1046/j.1365-313X.2002.01394.x
Liu Y, Wu R, Wan Q et al (2007) Glucose-6-phosphate dehydrogenase plays a pivotal role in nitric oxide-involved defense against oxidative stress under salt stress in red kidney bean roots. Plant Cell Physiol 48:511–522. https://doi.org/10.1093/pcp/pcm020
Liu MM, Yu HY, Ouyang B et al (2020) NADPH oxidases and the evolution of plant salinity tolerance. Plant Cell Environ 43:2957–2968. https://doi.org/10.1111/pce.13907
Lopez-Delacalle M, Silva CJ, Mestre TC et al (2021) Synchronization of proline, ascorbate and oxidative stress pathways under the combination of salinity and heat in tomato plants. Environ Exp Bot 183:104351. https://doi.org/10.1016/j.envexpbot.2020.104351
Luo X, Dai Y, Zheng C et al (2021) The ABI4-RbohD/VTC2 regulatory module promotes reactive oxygen species (ROS) accumulation to decrease seed germination under salinity stress. New Phytol 229:950–962. https://doi.org/10.1111/nph.16921
Ma X, Wang W, Bittner F et al (2016) Dual and opposing roles of xanthine dehydrogenase in defense-associated reactive oxygen species metabolism in Arabidopsis. Plant Cell 28:1108–1126. https://doi.org/10.1105/tpc.15.00880
Mandadi KK, Scholthof K-BG (2013) Plant immune responses against viruses: how does a virus cause disease? Plant Cell 25:1489–1505. https://doi.org/10.1105/tpc.113.111658
Marcec MJ, Gilroy S, Poovaiah BW, Tanaka K (2019) Mutual interplay of Ca2+ and ROS signaling in plant immune response. Plant Sci 283:343–354. https://doi.org/10.1016/j.plantsci.2019.03.004
Muthamilarasan M, Prasad M (2013) Plant innate immunity: an updated insight into defense mechanism. J Biosci 38:433–449. https://doi.org/10.1007/s12038-013-9302-2
Pieterse CMJ, Van der Does D, Zamioudis C et al (2012) Hormonal modulation of plant immunity. Annu Rev Cell Dev Biol 28:489–521. https://doi.org/10.1146/annurev-cellbio-092910-154055
Sanabam R, Chanu NT, Sharma SK et al (2018) Genetic diversity of Chilli veinal mottle virus infecting different chilli landraces in North East India indicates the possibility of transboundary movement of virus. 3 Biotech 8(8):357. https://doi.org/10.1007/s13205-018-1382-0
Shan C, Liang Z, Sun Y et al (2011) The protein kinase MEK1/2 participates in the regulation of ascorbate and glutathione content by jasmonic acid in Agropyron cristatum leaves. J Plant Physiol 168:514–518. https://doi.org/10.1016/j.jplph.2010.08.014
Tian Y, Peng K, Bao Y et al (2021) Glucose-6-phosphate dehydrogenase and 6-phosphogluconate dehydrogenase genes of winter wheat enhance the cold tolerance of transgenic Arabidopsis. Plant Physiol Biochem 161:86–97. https://doi.org/10.1016/j.plaphy.2021.02.005
Verma N, Prasad SM (2021) Regulation of redox homeostasis in cadmium stressed rice field cyanobacteria by exogenous hydrogen peroxide and nitric oxide. Sci Rep 11:2893. https://doi.org/10.1038/s41598-021-82397-9
Völz R, Park J-Y, Kim S et al (2020) The rice/maize pathogen Cochliobolus spp. infect and reproduce on Arabidopsis revealing differences in defensive phytohormone function between monocots and dicots. Plant J 103:412–429. https://doi.org/10.1111/tpj.14743
Wang J, Nan Z, Christensen MJ, Li C (2018) Glucose-6-phosphate dehydrogenase plays a vital role in Achnatherum inebrians plants host to Epichloë gansuensis by improving growth under nitrogen deficiency. Plant Soil 430:37–48. https://doi.org/10.1007/s11104-018-3710-x
Wang R, He F, Ning Y, Wang G-L (2020) Fine-tuning of RBOH-mediated ROS signaling in plant immunity. Trends Plant Sci 25:1060–1062. https://doi.org/10.1016/j.tplants.2020.08.001
Waszczak C, Carmody M, Kangasjärvi J (2018) Reactive oxygen species in plant signaling. Annu Rev Plant Biol 69:209–236. https://doi.org/10.1146/annurev-arplant-042817-040322
Xi DH, Yang H, Jiang Y et al (2010) Interference between Tobacco necrosis virus and Turnip crinkle virus in Nicotiana benthamiana. J Phytopathol 158:263–269. https://doi.org/10.1111/j.1439-0434.2009.01607.x
Xiong X-P, Sun S-C, Zhang X-Y et al (2020) GhWRKY70D13 regulates resistance to Verticillium dahliae in cotton through the ethylene and jasmonic acid signaling pathways. Front Plant Sci 11:69. https://doi.org/10.3389/fpls.2020.00069
Xu H, Hu L, Liu T et al (2020) Caffeine targets G6PDH to disrupt redox homeostasis and inhibit renal cell carcinoma proliferation. Front Cell Dev Biol 8:556162. https://doi.org/10.3389/fcell.2020.556162
Yang T, Qiu L, Huang W et al (2020) Chilli veinal mottle virus HCPro interacts with catalase to facilitate virus infection in Nicotiana tabacum. J Exp Bot 71(18):5656–5668. https://doi.org/10.1093/jxb/eraa304
Yang C, Dolatabadian A, Fernando D (2021) The wonderful world of intrinsic and intricate immunity responses in plants against pathogens. Can J Plant Pathol 44(1):1–20. https://doi.org/10.1080/07060661.2021.1960610
Zang Z, Wang Z, Zhao F et al (2021) Maize ethylene response factor ZmERF061 is required for resistance to Exserohilum turcicum. Front Plant Sci 12:630413. https://doi.org/10.3389/fpls.2021.630413
Zhang Y, Li X (2019) Salicylic acid: biosynthesis, perception, and contributions to plant immunity. Curr Opin Plant Biol 50:29–36. https://doi.org/10.1016/j.pbi.2019.02.004
Zhao Y, Cui Y, Huang S et al (2020) Genome-wide analysis of the glucose-6-phosphate dehydrogenase family in soybean and functional identification of GmG6PDH2 involvement in salt stress. Front Plant Sci 11:214. https://doi.org/10.3389/fpls.2020.00214
Zhu Y, Guo M-J, Song J-B et al (2021) Roles of endogenous melatonin in resistance to Botrytis cinerea infection in an Arabidopsis model. Front Plant Sci 12:683228. https://doi.org/10.3389/fpls.2021.683228
Acknowledgements
This work was supported by the National Natural Science Foundation of China (32070167, 31772131), International Cooperation Project of Chengdu Science and Technology Bureau (2020-GH02-00026-HZ), Special Project on Traditional Chinese Medicine of Sichuan Provincial Administration of Traditional Chinese Medicine (2021MS522) and the Fundamental Research Funds for the Central Universities (2022SCUH0006).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, D., Peng, Q., Cheng, Y. et al. Glucose-6-phosphate dehydrogenase promotes the infection of Chilli veinal mottle virus through affecting ROS signaling in Nicotiana benthamiana. Planta 256, 96 (2022). https://doi.org/10.1007/s00425-022-04010-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-022-04010-1