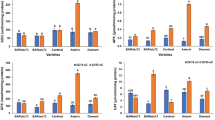
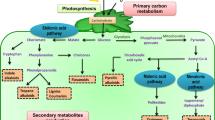
Abstract
Main conclusion
Ηeat and calcium treatments reprogram sweet cherry fruit metabolism during postharvest senescence as evidenced by changes in respiration, amino acid metabolism, sugars, and secondary metabolites shift.
Abstract
Heat and calcium treatments are used to improve postharvest fruit longevity; however, the exact mechanism remains poorly understood. To characterize the impact of these treatments on sweet cherries metabolism, ‘Lapins’ fruits were treated with heat or CaCl2 solutions and their combination and subsequently were exposed at room temperature, for up to 4 days, defined as senescence period. Single and combined heat and calcium treatments partially delayed fruit senescence, as evidenced by changes in fruit colour darkening, skin penetration force, and respiration activity. Calcium content was noticeably increased by heat in Ca-treated fruit. Several primary metabolites, including amino acids, organic acids, and alcohols, were decreased in response to both treatments, while many soluble sugars and secondary metabolites were increased within 1 day post-treatment. Changes of several metabolites in heat-treated fruits, especially esculetin, peonidin 3-O-glucoside and peonidin 3-O-galactoside, ribose, pyroglutamate, and isorhamnetin-3-O-rutinoside, were detected. The metabolome of fruit exposed to calcium also displayed substantial modulations, particularly in the levels of galactose, glycerate, aspartate, tryptophan, phospharate rutin, and peonidin 3-O-glucoside. The expression of several genes involved in TCA cycle (MDH1, IDH1, OGDH, SUCLA2, and SDH1-1), pectin degradation (ADPG1) as well as secondary (SK1, 4CL1, HCT, and BAN), amino acids (ALDH18A1, ALDH4A1, GS, GAD, GOT2, OPLAH, HSDH, and SDS), and sugar (PDHA1 and DLAT) metabolism were affected by both treatments. Pathway-specific analysis further revealed the regulation of fruit metabolic programming by heat and calcium. This work provides a comprehensive understanding of metabolic regulation in response to heat and calcium during fruit senescence.





Similar content being viewed by others
References
Ainalidou A, Tanou G, Belghazi M, Samiotaki M, Diamantidis G, Molassiotis A, Karamanoli K (2016) Integrated analysis of metabolites and proteins reveal aspects of the tissue-specific function of synthetic cytokinin in kiwifruit development and ripening. J Proteomics 143:318–333. https://doi.org/10.1016/j.jprot.2016.02.013
Alique R, Zamorano JP, Martínez MA, Alonso J (2005) Effect of heat and cold treatments on respiratory metabolism and shelf-life of sweet cherry, type picota cv “Ambrunés”. Postharvest Biol Technol 35:153–165. https://doi.org/10.1016/J.POSTHARVBIO.2004.07.003
Arapitsas P, Perenzoni D, Nicolini G, Mattivi F (2012) Study of sangiovese wines pigment profile by UHPLC-MS/MS. J Agric Food Chem 60:10461–10471. https://doi.org/10.1021/jf302617e
Batistič O, Kudla J (2012) Analysis of calcium signaling pathways in plants. Biochim Biophys Acta Gen Subj 1820:1283–1293. https://doi.org/10.1016/j.bbagen.2011.10.012
Braam J (1992) Regulated expression of the calmodulin-related TCH genes in cultured Arabidopsis cells: induction by calcium and heat shock. Proc Natl Acad Sci USA 89:3213–3216. https://doi.org/10.1073/pnas.89.8.3213
Brugière N, Dubois F, Masclaux C et al (2000) Immunolocalization of glutamine synthetase in senescing tobacco (Nicotiana tabacum L.) leaves suggests that ammonia assimilation is progressively shifted to the mesophyll cytosol. Planta 211:519–527. https://doi.org/10.1007/s004250000309
Catola S, Castagna A, Santin M et al (2017) The dominant allele Aft induces a shift from flavonol to anthocyanin production in response to UV-B radiation in tomato fruit. Planta 246:263–275. https://doi.org/10.1007/s00425-017-2710-z
Ding Y, Chang J, Ma Q et al (2015) Network analysis of postharvest senescence process in citrus fruits revealed by transcriptomic and metabolomic profiling. Plant Physiol 168:357–376. https://doi.org/10.1104/pp.114.255711
Dodd AN, Kudla J, Sanders D (2010) The language of calcium signaling. Annu Rev Plant Biol 61:593–620. https://doi.org/10.1146/annurev-arplant-070109-104628
Dumville JC, Fry SC (2003) Solubilisation of tomato fruit pectins by ascorbate: a possible non-enzymic mechanism of fruit softening. Planta 217:951–961. https://doi.org/10.1007/s00425-003-1061-0
Fait A, Fromm H, Walter D et al (2008) Highway or byway: the metabolic role of the GABA shunt in plants. Trends Plant Sci 13:14–19. https://doi.org/10.1016/j.tplants.2007.10.005
Fernie AR, Carrari F, Sweetlove LJ (2004) Respiratory metabolism: glycolysis, the TCA cycle and mitochondrial electron transport. Curr Opin Plant Biol 7:254–261. https://doi.org/10.1016/j.pbi.2004.03.007
Fotopoulos V, Ziogas V, Tanou G, Molassiotis A (2010) Involvement of AsA/DHA and GSH/GSSG ratios in gene and protein expression and in the activation of defence mechanisms under abiotic stress conditions. In: Ascorbate-glutathione pathway and stress tolerance in plants. In: Anjum NA, Ulmar S, Chan MT (eds) Ascorbate-glutathione pathway and stress tolerance in plants. Springer, Netherlands, Dordrecht, pp 265–302
González-Gómez D, Lozano M, Fernández-León MF et al (2009) Detection and quantification of melatonin and serotonin in eight sweet cherry cultivars (Prunus avium L.). Eur Food Res Technol 229:223–229. https://doi.org/10.1007/s00217-009-1042-z
Goulas V, Minas IS, Kourdoulas PM et al (2015) 1H NMR metabolic fingerprinting to probe temporal postharvest changes on qualitative attributes and phytochemical profile of sweet cherry fruit. Front Plant Sci 6:959. https://doi.org/10.3389/fpls.2015.00959
Hernández-Muñoz P, Almenar E, Del Valle V et al (2008) Effect of chitosan coating combined with postharvest calcium treatment on strawberry (Fragaria × ananassa) quality during refrigerated storage. Food Chem 110:428–435. https://doi.org/10.1016/J.FOODCHEM.2008.02.020
Hocking B, Tyerman SD, Burton RA, Gilliham M (2016) Fruit calcium: transport and physiology. Front Plant Sci 7:569. https://doi.org/10.3389/fpls.2016.00569
Jones JJB, Case VW (1990) Sampling, handling, and analyzing plant tissue samples. In: Westerman R (ed) Soil testing and plant analysis, SSSA book series, 2nd edn, No 3. Soil Science Society of America, Madison, WI, pp 389–427
Karagiannis E, Tanou G, Samiotaki M et al (2016) Comparative physiological and proteomic analysis reveal distinct regulation of peach skin quality traits by altitude. Front Plant Sci 7:1689. https://doi.org/10.3389/fpls.2016.01689
Karagiannis E, Michailidis M, Karamanoli K et al (2018a) Postharvest responses of sweet cherry fruit and stem tissues revealed by metabolomic profiling. Plant Physiol Biochem 127:478–484. https://doi.org/10.1016/j.plaphy.2018.04.029
Karagiannis E, Michailidis M, Tanou G et al (2018b) Ethylene—dependent and—independent superficial scald resistance mechanisms in ‘Granny Smith’ apple fruit. Sci Rep 8:11436. https://doi.org/10.1038/s41598-018-29706-x
Karchi H, Miron D, Ben-Yaakov S, Galili G (1995) The lysine-dependent stimulation of lysine catabolism in tobacco seed requires calcium and protein phosphorylation. Plant Cell 7:1963–1970. https://doi.org/10.1105/tpc.7.11.1963
Katz E, Fon M, Lee YJ et al (2007) The citrus fruit proteome: insights into citrus fruit metabolism. Planta 226:989–1005. https://doi.org/10.1007/s00425-007-0545-8
Klein JD, Ferguson IB (1987) Effect of high temperature on calcium uptake by suspension-cultured pear fruit cells. Plant Physiol 84:153–156. https://doi.org/10.1104/PP.84.1.153
Kotak S, Larkindale J, Lee U et al (2007) Complexity of the heat stress response in plants. Curr Opin Plant Biol 10:310–316. https://doi.org/10.1016/j.pbi.2007.04.011
Langer SE, Oviedo NC, Marina M et al (2018) Effects of heat treatment on enzyme activity and expression of key genes controlling cell wall remodeling in strawberry fruit. Plant Physiol Biochem 130:334–344. https://doi.org/10.1016/j.plaphy.2018.07.015
Lurie S, Pedreschi R (2014) Fundamental aspects of postharvest heat treatments. Hortic Res 1:14030. https://doi.org/10.1038/hortres.2014.30
Manganaris GA, Ilias IF, Vasilakakis M, Mignani I (2007a) The effect of hydrocooling on ripening related quality attributes and cell wall physicochemical properties of sweet cherry fruit (Prunus avium L). Int J Refrig 30:1386–1392. https://doi.org/10.1016/j.ijrefrig.2007.04.001
Manganaris GA, Vasilakakis M, Diamantidis G, Mignani I (2007b) The effect of postharvest calcium application on tissue calcium concentration, quality attributes, incidence of flesh browning and cell wall physicochemical aspects of peach fruits. Food Chem 100:1385–1392. https://doi.org/10.1016/j.foodchem.2005.11.036
Massot C, Bancel D, Lopez Lauri F et al (2013) High temperature inhibits ascorbate recycling and light stimulation of the ascorbate pool in tomato despite increased expression of biosynthesis genes. PLoS One 8:e84474. https://doi.org/10.1371/journal.pone.0084474
Michailidis M, Karagiannis E, Tanou G et al (2017) Metabolomic and physico-chemical approach unravel dynamic regulation of calcium in sweet cherry fruit physiology. Plant Physiol Biochem 116:68–79. https://doi.org/10.1016/j.plaphy.2017.05.005
Michailidis M, Karagiannis E, Tanou G et al (2018) Metabolic mechanisms underpinning vegetative bud dormancy release and shoot development in sweet cherry. Environ Exp Bot 155:1–11. https://doi.org/10.1016/j.envexpbot.2018.06.024
Minas IS, Vicente AR, Dhanapal AP, Manganaris GA, Goulas V, Vasilakakis M, Crisosto CH, Molassiotis A (2014) Ozone-induced kiwifruit ripening delay is mediated by ethylene biosynthesis inhibition and cell wall dismantling regulation. Plant Sci 229:76–85. https://doi.org/10.1016/j.plantsci.2014.08.016
Molassiotis A, Tanou G, Filippou P, Fotopoulos V (2013) Proteomics in the fruit tree science arena: new insights into fruit defense, development, and ripening. Proteomics 13:1871–1884. https://doi.org/10.1002/pmic.201200428
Nelles A (1985) Das Membraaspotential von Maiskoleoptilen unter dem Einfluß von Hitzestreß: Effect of heat stress on the membrane potential of corn coleoptile cells. Biochem und Physiol der Pflanz 180(6):458–463. https://doi.org/10.1016/S0015-3796(85)80056-1
Peng H, Yang T, Whitaker BD et al (2016) Calcium/calmodulin alleviates substrate inhibition in a strawberry UDP-glucosyltransferase involved in fruit anthocyanin biosynthesis. BMC Plant Biol 16:197. https://doi.org/10.1186/s12870-016-0888-z
Poovaiah BW, Reddy ASN (1993) Calcium and signal transduction in plants. CRC Crit Rev Plant Sci 12:185–211. https://doi.org/10.1080/07352689309701901
Ranjbar S, Rahemi M, Ramezanian A (2018) Comparison of nano-calcium and calcium chloride spray on postharvest quality and cell wall enzymes activity in apple cv. Red Delicious. Sci Hortic (Amst) 240:57–64. https://doi.org/10.1016/j.scienta.2018.05.035
Saure MC (2014) Why calcium deficiency is not the cause of blossom-end rot in tomato and pepper fruit—a reappraisal. Sci Hortic (Amst) 174:151–154. https://doi.org/10.1016/j.scienta.2014.05.020
Shafiee M, Taghavi TS, Babalar M (2010) Addition of salicylic acid to nutrient solution combined with postharvest treatments (hot water, salicylic acid, and calcium dipping) improved postharvest fruit quality of strawberry. Sci Hortic (Amst) 124:40–45. https://doi.org/10.1016/j.scienta.2009.12.004
Sinha R, Kumar D, Datta R et al (2015) Integrated transcriptomic and proteomic analysis of Arabidopsis thaliana exposed to glutathione unravels its role in plant defense. Plant Cell Tissue Organ Cult 120:975–988. https://doi.org/10.1007/s11240-014-0651-9
Stuttmann J, Hubberten H-M, Rietz S et al (2011) Perturbation of Arabidopsis amino acid metabolism causes incompatibility with the adapted biotrophic pathogen Hyaloperonospora arabidopsidis. Plant Cell 23:2788–2803. https://doi.org/10.1105/tpc.111.087684
Sultana N, Florance HV, Johns A, Smirnoff N (2015) Ascorbate deficiency influences the leaf cell wall glycoproteome in Arabidopsis thaliana. Plant Cell Environ 38:375–384. https://doi.org/10.1111/pce.12267
Sweetlove LJ, Beard KFM, Nunes-Nesi A et al (2010) Not just a circle: flux modes in the plant TCA cycle. Trends Plant Sci 15:462–470. https://doi.org/10.1016/j.tplants.2010.05.006
Tanou G, Minas IS, Karagiannis E, Tsikou D, St Audebert, Papadopoulou KK, Molassiotis A (2015) The impact of sodium nitroprusside and ozone in kiwifruit ripening physiology: a combined gene and protein expression profiling approach. Ann Bot 116:649–662. https://doi.org/10.1093/aob/mcv107
Tanou G, Minas IS, Scossa F, Belghazi M, Xanthopoulou A, Ganopoulos I, Madesis P, Fernie A, Molassiotis A (2017) Exploring priming responses involved in peach fruit acclimation to cold stress. Sci Rep 7:1–14
Tsantili E, Rouskas D, Christopoulos MV et al (2007) Effects of two pre-harvest calcium treatments on physiological and quality parameters in ‘Vogue’ cherries during storage. J Hortic Sci Biotechnol 82:657–663. https://doi.org/10.1080/14620316.2007.11512287
Vangdal E, Nordbø R, Flatland S (2005) Postharvest calcium and heat treatments of sweet cherries (Prunus avium L.). Acta Hortic 682:1133–1136. https://doi.org/10.17660/ActaHortic.2005.682.149
Vicente AR, Costa ML, Martínez GA et al (2005) Effect of heat treatments on cell wall degradation and softening in strawberry fruit. Postharvest Biol Technol 38:213–222. https://doi.org/10.1016/j.postharvbio.2005.06.005
Vitrac X, Larronde F, Krisa S et al (2000) Sugar sensing and Ca2 + -calmodulin requirement in Vitis vinifera cells producing anthocyanins. Phytochemistry 53:659–665. https://doi.org/10.1016/S0031-9422(99)00620-2
Vrhovsek U, Masuero D, Gasperotti M et al (2012) A versatile targeted metabolomics method for the rapid quantification of multiple classes of phenolics in fruits and beverages. J Agric Food Chem 60:8831–8840. https://doi.org/10.1021/jf2051569
Wang K, Shao X, Gong Y et al (2013) The metabolism of soluble carbohydrates related to chilling injury in peach fruit exposed to cold stress. Postharvest Biol Technol 86:53–61. https://doi.org/10.1016/j.postharvbio.2013.06.020
Wang Y, Xie X, Long LE (2014) The effect of postharvest calcium application in hydro-cooling water on tissue calcium content, biochemical changes, and quality attributes of sweet cherry fruit. Food Chem 160:22–30. https://doi.org/10.1016/J.FOODCHEM.2014.03.073
Wang Q-H, Zhao C, Zhang M et al (2017) Transcriptome analysis around the onset of strawberry fruit ripening uncovers an important role of oxidative phosphorylation in ripening. Sci Rep 7:41477. https://doi.org/10.1038/srep41477
Wang Y, Li W, Chang H et al (2019) Sweet cherry fruit miRNAs and effect of high CO2 on the profile associated with ripening. Planta 249:1799–1810. https://doi.org/10.1007/s00425-019-03110-9
Xu Z-S, Ma J, Wang F et al (2016) Identification and characterization of DcUCGalT1, a galactosyltransferase responsible for anthocyanin galactosylation in purple carrot (Daucus carota L.) taproots. Sci Rep 6:27356. https://doi.org/10.1038/srep27356
Yun Z, Gao H, Liu P et al (2013) Comparative proteomic and metabolomic profiling of citrus fruit with enhancement of disease resistance by postharvest heat treatment. BMC Plant Biol 13:1–44. https://doi.org/10.1186/1471-2229-13-44
Acknowledgements
MM is grateful to the Hellenic Foundation for Research and Innovation (ELIDEK) for a PhD scholarship during this work. Partially funding was obtained from ADP 2011–2018 project funded by the Autonomous Province of Trento.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Suppl. Fig. S1
Weight loss of sweet cherries ‘Lapins’ at 1 days at 20 ℃ after immersion in CaCl2 (0, 1, 2, and 4% CaCl2·2H2O), at three different solution temperatures (20, 40, and 50 °C) and for 2 treatment durations (5 and 10 min). Each value represents the mean of five replications per ten fruits and vertical bars represent the least significance difference (LSD), P ≤ 0.05. Red arrows indicate the treatments that were selected for conducting the final experiment (PPTX 45 kb)
Suppl. Table S1
Primary polar metabolites quantification (XLSX 45 kb)
Suppl. Table S2
Secondary metabolites quantification (XLSX 28 kb)
Suppl. Table S3
Molecular properties of primary and secondary metabolites (XLSX 37 kb)
Suppl. Table S4
Transcripts properties and primer sequence (XLSX 23 kb)
Suppl. Table S5
Relative abundance of transcripts (XLSX 21 kb)
Rights and permissions
About this article
Cite this article
Michailidis, M., Karagiannis, E., Tanou, G. et al. An integrated metabolomic and gene expression analysis identifies heat and calcium metabolic networks underlying postharvest sweet cherry fruit senescence. Planta 250, 2009–2022 (2019). https://doi.org/10.1007/s00425-019-03272-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-019-03272-6