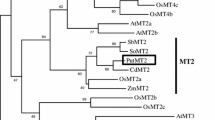
Abstract
Main conclusion
Physcomitrella patens contains four metallothionein-like genes. Three were shown to confer metal tolerance in yeast. Transcript profiling suggests their roles in senescence and reproductive development or cadmium and oxidative stress.
Abstract
Metallothioneins (MTs) have been suggested to play various roles including metal detoxification, nutrient remobilization, ROS scavenging, stress tolerance, and plant development. However, little is known about the forms and functions of MTs in bryophytes. The moss Physcomitrella patens genome was found to contain four MT-like genes. Amino acid sequence composition showed that the P. patens MTs (PpMTs) were clustered with Type 1 plant MTs, and could be further classified into two sub-types, herein referred to as sub-type 1: PpMT1.1a and PpMT1.1b and sub-type 2: PpMT1.2a and PpMT1.2b. Transcript abundance of PpMT1.1b and PpMT1.2b was upregulated in the gametophore compared to protonema, and all, except PpMT1.2a, were highly induced in senescing gametophytes. PpMT1.1a and PpMT1.1b transcripts were upregulated in protonema treated with cadmium and hydrogen peroxide. Unlike many higher plant MTs, the PpMT transcript abundance was not strongly induced in response to copper and zinc. These results suggest that PpMTs may play a role in protecting P. patens from cadmium and oxidative stress and may be involved in tissues senescence and reproductive development. The PpMTs, except PpMT1.2b, were also able to confer metal tolerance and accumulation when heterologously expressed in the ∆cup1 yeast. A P. patens mutant lacking PpMT1.2a through targeted gene disruption was generated. However, it did not show any alteration in growth phenotypes under senescence-induced conditions or hypersensitivity to cadmium, copper, zinc, H2O2, and NaCl stresses. Further characterization of additional P. patens mutants lacking single or multiple PpMTs may provide insight into the physiological roles of bryophytic MTs.








Similar content being viewed by others
Abbreviations
- EST:
-
Expressed sequence tag
- GPD:
-
Glyceraldehyde-3-phosphate dehydrogenase
- MT:
-
Metallothionein
- ROS:
-
Reactive oxygen species
References
Aldous AR (2002) Nitrogen translocation in Sphagnum mosses: effects of atmospheric nitrogen deposition. New Phytol 156(2):241–253
Andrews GK, Huet-Hudson YM, Paria BC, McMaster MT, De SK, Dey SK (1991) Metallothionein gene expression and metal regulation during preimplantation mouse embryo development (MT mRNA during early development). Dev Biol 145:13–27
Benatti MR, Yookongkaew N, Meetam M, Guo WJ, Punyasuk N, Abu Qamar S, Goldsbrough P (2014) Metallothionein deficiency impacts copper accumulation and redistribution in leaves and seeds of Arabidopsis. New Phytol 202(3):940–951
Breeze E, Wagstaff C, Harrison E, Bramke I, Rogers H, Stead A, Thomas B, Buchanan-Wollaston V (2004) Gene expression patterns to define stages of post-harvest senescence in Alstroemeria petals. Plant Biotechnol J 2(2):155–168
Chaturvedi AK, Patel MK, Mishra A, Tiwari V, Jha B (2014) The SbMT-2 gene from a halophyte confers abiotic stress tolerance and modulates ROS scavenging in transgenic tobacco. PLoS One 9(10):e111379
Cho SH, Hoang QT, Kim YY, Shin HY, Ok SH, Bae JM, Shin JS (2006) Proteome analysis of gametophores identified a metallothionein involved in various abiotic stress responses in Physcomitrella patens. Plant Cell Rep 25(5):475–488
Clendennen SK, May GD (1997) Differential gene expression in ripening banana fruit. Plant Physiol 115(2):463–469
Cobbett CS (2000) Phytochelatin biosynthesis and function in heavy-metal detoxification. Curr Opin Plant Biol 3(3):211–216
Cobbett C, Goldsbrough P (2002) Phytochelatins and metallothioneins: roles in heavy metal detoxification and homeostasis. Annu Rev Plant Biol 53(1):159–182
Cove D (2005) The moss Physcomitrella patens. Annu Rev Genet 39:339–358
Dalton T, Palmiter RD, Andrews GK (1994) Transcriptional induction of the mouse metallothionein-I gene in hydrogen peroxide-treated Hepa cells involves a composite major late transcription factor/antioxidant response element and metal response promoter elements. Nucleic Acids Res 22(23):5016–5023
Davidson AJ, Harborne JB, Longton RE (1990) The acceptability of mosses as food for generalist herbivores, slugs in the Arionidae. Bot J Linn Soc 104(1–3):99–113
Demidchik V, Straltsova D, Medvedev SS, Pozhvanov GA, Sokolik A, Yurin V (2014) Stress-induced electrolyte leakage: the role of K+-permeable channels and involvement in programmed cell death and metabolic adjustment. J Exp Bot 65(5):1259–1270
Ecker DJ, Butt TR, Sternberg EJ, Neeper MP, Debouck C, Gorman JA, Crooke ST (1986) Yeast metallothionein function in metal ion detoxification. J Biol Chem 261(36):16895–16900
Furst P, Hu S, Hackett R, Hamer D (1988) Copper activates metallothionein gene transcription by altering the conformation of a specific DNA binding protein. Cell 55(4):705–717
Guo WJ, Bundithya W, Goldsbrough PB (2003) Characterization of the Arabidopsis metallothionein gene family: tissue-specific expression and induction during senescence and in response to copper. New Phytol 159(2):369–381
Guo WJ, Meetam M, Goldsbrough PB (2008) Examining the specific contributions of individual Arabidopsis metallothioneins to copper distribution and metal tolerance. Plant Physiol 146(4):1697–1706
Hanley-Bodoin L, Lane BG (1983) A novel protein programmed by the mRNA conserved in dry wheat embryos. FEBS J 135(1):9–15
Heuchel R, Radtke F, Georgiev O, Stark G, Aguet M, Schaffner W (1994) The transcription factor MTF-1 is essential for basal and heavy metal-induced metallothionein gene expression. EMBO J 13(12):2870–2875
Hidalgo J, Belloso E, Hernandez J, Gasull T, Molinero A (1997) Role of glucocorticoids on rat brain metallothionein-I and-III response to stress. Stress 1(4):231–240
Kagi JHR, Schaeffer A (1988) Biochemistry of metallothionein. Biochemistry 27(23):8509–8515
Kawashima I, Kennedy TD, Chino M, Lane BG (1992) Wheat Ec metallothionein genes: like mammalian Zn2+ metallothionein genes, wheat Zn2+ metallothionein genes are conspicuously expressed during embryogenesis. Eur J Biochem 209(3):971–976
Knight CD, Cove DJ, Cuming AC, Quatrano RS (2002) Moss gene technology. Plant Mol Biol 2:285–301
Kopriva S, Wiedemann G, Reski R (2007) Sulfate assimilation in basal land plants -what does genomic sequencing tell us? Plant Biol 9(5):556–564
Kumar G, Kushwaha HR, Panjabi-Sabharwal V, Kumari S, Joshi R, Karan R, Mittal S, Pareek S, Pareek A (2012) Clustered metallothionein genes are co-regulated in rice and ectopic expression of OsMT1e-P confers multiple abiotic stress tolerance in tobacco via ROS scavenging. BMC Plant Biol 12(1):107
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33(7):1870–1874
Lane B, Kajioka R, Kennedy T (1987) The wheat-germ Ec protein is a zinc-containing metallothionein. Biochem Cell Biol 65(11):1001–1005
Leszczyszyn OI, Imam HT, Blindauer CA (2013) Diversity and distribution of plant metallothioneins: a review of structure, properties and functions. Metallomics 5(9):1146–1169
Li Z, Zhang Y, Zhang X, Peng Y, Merewitz E, Ma X, Huang L, Yan Y (2016) The alterations of endogenous polyamines and phytohormones induced by exogenous application of spermidine regulate antioxidant metabolism, metallothionein and relevant genes conferring drought tolerance in white clover. Environ Exp Bot 124:22–38
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382
Lunde C, Baumann U, Shirley NJ, Drew DP, Fincher GB (2006) Gene structure and expression pattern analysis of three monodehydroascorbate reductase (Mdhar) genes in Physcomitrella patens: implications for the evolution of the MDHAR family in plants. Plant Mol Biol 60:259–275
Mir G, Domènech J, Huguet G, Guo WJ, Goldsbrough P, Atrian S, Molinas M (2004) A plant type 2 metallothionein (MT) from cork tissue responds to oxidative stress. J Exp Bot 55(408):2483–2493
Moulick A, Mukhopadhyay D, Talapatra S, Ghoshal N, Raychaudhuri SS (2013) Molecular cloning, modeling, and characterization of type 2 metallothionein from Plantago ovata Forsk. Sequencing 2013:10
Mukae K, Inoue Y, Moriyasu Y (2015) ATG5-knockout mutants of Physcomitrella provide a platform for analyzing the involvement of autophagy in senescence processes in plant cells. Plant Signal Behav 10(11):e1086859
Mumberg D, Müller R, Funk M (1995) Yeast vectors for the controlled expression of heterologous proteins in different genetic backgrounds. Gene 156(1):119–122
Murphy A, Taiz L (1995) Comparison of metallothionein gene expression and nonprotein thiols in ten Arabidopsis ecotypes (correlation with copper tolerance). Plant Physiol 109(3):945–954
Murphy A, Zhou J, Goldsbrough PB, Taiz L (1997) Purification and immunological identification of metallothioneins 1 and 2 from Arabidopsis thaliana. Plant Physiol 113(4):1293–1301
Nakajima H, Itoh K, Otake H, Fujimoto K (2011) Spectral properties of the Cu-hyperaccumulating moss Scopelophila cataractae. J Photochem Photobiol B 104(3):467–472
Nevrtalova E, Baloun J, Hudzieczek V, Cegan R, Vyskot B, Dolezel J, Safar J, Milde D, Hobza R (2014) Expression response of duplicated metallothionein 3 gene to copper stress in Silene vulgaris ecotypes. Protoplasma 251(6):1427–1439
Nishimura H, Nishimura N, Tohyama C (1989) Immunohistochemical localization of metallothionein in developing rat tissues. J Histochem Cytochem 37(5):715–722
Palmiter RD, Findley SD, Whitmore TE, Durnam DM (1992) MT-III, a brain-specific member of the metallothionein gene family. Proc Natl Acad Sci USA 89(14):6333–6337
Pan Y, Pan Y, Zhai J, Xiong Y, Li J, Du X, Su C, Zhang X (2016) Cucumber metallothionein-like 2 (CsMTL2) exhibits metal-binding properties. Genes 7(12):106
Petraglia A, De Benedictis M, Degola F, Pastore G, Calcagno M, Ruotolo R, Mengoni A, Sanità di Toppi L (2014) The capability to synthesize phytochelatins and the presence of constitutive and functional phytochelatin synthases are ancestral (plesiomorphic) characters for basal land plants. J Exp Bot 65(4):1153–1163
Rensing SA, Lang D, Zimmer AD, Terry A, Salamov A, Shapiro H, Nishiyama T, Perroud P, Lindquist E, Kamisugi Y (2008) The Physcomitrella genome reveals evolutionary insights into the conquest of land by plants. Science 319(5859):64–69
Rother M, Krauss GJ, Grass G, Wesenberg D (2006) Sulphate assimilation under Cd2+ stress in Physcomitrella patens–combined transcript, enzyme and metabolite profiling. Plant Cell Environ 29(9):1801–1811
Rydin H, Clymo R (1989) Transport of carbon and phosphorus compounds about Sphagnum. Proc R Soc Lond B Biol Sci 237(1286):63–84
Sanchez-Vera V, Kenchappa CS, Landberg K, Bressendorff S, Schwarzbach S, Martin T, Mundy J, Petersen M, Thelander M, Sundberg E (2017) Autophagy is required for gamete differentiation in the moss Physcomitrella patens. Autophagy 13(11):1939–1951
Sassmann S, Adlassnig W, Puschenreiter M, Cadenas EJP, Leyvas M, Lichtscheidl IK, Lang I (2015) Free metal ion availability is a major factor for tolerance and growth in Physcomitrella patens. Environ Exp Bot 110:1–10
Schaefer DG, Zryd JP (2001) The moss Physcomitrella patens, now and then. Plant Physiol 127(4):1430–1438
Schiller M, Hegelund JN, Pedas P, Kichey T, Laursen KH, Husted S, Schjoerring JK (2014) Barley metallothioneins differ in ontogenetic pattern and response to metals. Plant, Cell Environ 37(2):353–367
Schultze P, Worgotter E, Braun W, Wagner G, Vasak M, Kagi J, Wuthrich K (1995) Conformation of [Cd7]–metallothionein-2 from rat liver in aqueous solution determined by nuclear magnetic resonance spectroscopy. J Mol Biol 203(1):251–268
Singh RK, Anandhan S, Singh S, Patade VY, Ahmed Z, Pande V (2011) Metallothionein-like gene from Cicer microphyllum is regulated by multiple abiotic stresses. Protoplasma 248(4):839–847
Spagnuolo V, Zampella M, Giordano S, Adamo P (2011) Cytological stress and element uptake in moss and lichen exposed in bags in urban area. Ecotoxicol Environ Saf 74(5):1434–1443
Steffens B, Sauter M (2009) Epidermal cell death in rice is confined to cells with a distinct molecular identity and is mediated by ethylene and H2O2 through an auto amplified signal pathway. Plant Cell 21(1):184–196
Steffens B, Geske T, Sauter M (2011) Aerenchyma formation in the rice stem and its promotion by H2O2. New Phytol 190(2):369–378
Sun SQ, Wang GX, He M, Cao T (2011) Effects of Pb and Ni stress on oxidative stress parameters in three moss species. Ecotoxicol Environ Saf 74(6):1630–1635
Thiele D, Hamer D (1986) Tandemly duplicated upstream control sequences mediate copper-induced transcription of the Saccharomyces cerevisiae copper-metallothionein gene. Mol Cell Biol 6(4):1158–1163
Uyar G, Avcil E, Ören M, Karaca F, Oncel MS (2009) Determination of heavy metal pollution in Zonguldak (Turkey) by moss analysis (Hypnum cupressiforme). Environ Eng Sci 26(1):183–194
White CN, Rivin CJ (1995) Characterization and expression of a cDNA encoding a seed-specific metallothionein in maize. Plant Physiol 108(2):831–832
Wu W, Zhang Q, Ervin EH, Yang Z, Zhang X (2017) Physiological mechanism of enhancing salt stress tolerance of perennial ryegrass by 24-epibrassinolide. Front Plant Sci 8:1017
Xue T, Li X, Zhu W, Wu C, Yang G, Zheng C (2008) Cotton metallothionein GhMT3a, a reactive oxygen species scavenger, increased tolerance against abiotic stress in transgenic tobacco and yeast. J Exp Bot 60(1):339–349
Yang Z, Wu Y, Li Y, Ling HQ, Chu C (2009) OsMT1a, a type 1 metallothionein, plays the pivotal role in zinc homeostasis and drought tolerance in rice. Plant Mol Biol 70(1–2):219–229
Yang M, Zhang F, Wang F, Dong Z, Cao Q, Chen M (2015) Characterization of a type 1 metallothionein gene from the stresses-tolerant plant Ziziphus jujuba. Int J Mol Sci 16(8):16750–16762
Yi J, Moon S, Lee YS, Zhu L, Liang W, Zhang D, Jung K, An G (2016) Defective tapetum cell death 1 (DTC1) regulates ROS levels by binding to metallothionein during tapetum degeneration. Plant Physiol 170(3):1611–1623
Zhang M, Takano T, Liu S, Zhang X (2014) Abiotic stress response in yeast and metal-binding ability of a type 2 metallothionein-like protein (PutMT2) from Puccinellia tenuiflora. Mol Biol Rep 41(9):5839–5849
Zhou J, Goldsbrough PB (1995) Structure, organization and expression of the metallothionein gene family in Arabidopsis. Mol Gen Genet 248(3):318–328
Ziller A, Fraissinet-Tachet L (2018) Metallothionein diversity and distribution in the tree of life: a multifunctional protein. Metallomics 10(11):1549–1559
Acknowledgements
This research was supported by a DPST research grant [grant no. 022/2557] to MM from the Institute for the Promotion of Teaching Science and Technology (IPST) and by a research assistantship grant to OP from Faculty of Science, Mahidol University. We acknowledge Dr. Thomas N. Stewart for proofreading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that they have no competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Suppl. Fig. S1
Relative PpACT2 transcript abundance in different tissues or ages of moss or in response to different stress treatments. a Total RNA was extracted from 4-week-old (4 W) or 8-week-old (8 W) protonema (Pro) or gametophore (Gam) tissues cultured on BCDAT medium. b Total RNA was extracted from 4-week-old protonema cultured in normal liquid medium (control) or supplemented with 50 µM CdCl2 or 2 mM H2O2 for 24 h. Relative ACT2 expression was normalized to the geometric mean of three reference genes including histone PpHis3.2, elongation factor 1 alpha PpeF1a, and beta-tubulin 1 PpTub1. Error bars represent SE (n = 3). ns, not significantly different (one-tailed t test, P < 0.05)
Suppl. Fig. S2
Dose response of colony growth inhibition of WT P. patens to ZnSO4, CuSO4 and CdCl2 supplemented to BCDAT agar medium. Equal amounts of protonema were inoculated on medium-containing various concentrations of ZnSO4, CuSO4 and CdCl2. After 4 weeks, moss colonies were photographed and colony areas were quantified by ImageJ software (NIH, USA). Relative colony area was calculated in comparison to the control condition. The fitted curve and the effective concentration (EC) were determined using the built-in “log(inhibitor) - normalized response” equation with a variable slope in GraphPad Prism (GraphPad Software Inc., USA)
Suppl. Fig. S3
Semi-quantitative RT-PCR analysis of PpMT transcript abundance in 4-week-old gametophore tissues of P. patens cultured in normal BCDAT agar medium for 4 weeks using the PpMT gene-specific primers. ACT2 was included as a loading control
Suppl. Fig. S4
Electrolyte leakage of WT P. patens gametophore tissues following treatment with ZnSO4, CuSO4, CdCl2 or H2O2 for 24 h in BCDAT broth medium. Asterisk indicates values that are significantly different from the control (one-tailed t test; *, P < 0.1 or **, P < 0.05). Error bars represent SE (n = 3)
Suppl. Fig. S5
Semi-quantitative RT-PCR analysis of transcript abundance corresponding to the endogenous CUP1 or heterologously expressed MT genes in yeast strains transformed with p424-GDP empty vector (EV) or the vector harboring different transgenes. The PCR amplicons were amplified using CUP1-specific or PpMT gene-specific primers. Yeast ACT1 was used as a loading control
Suppl. Fig. S6
Cadmium sensitivity of S. cerevisiaea ∆ycf1 or b ∆cup1 strains expressing PpMT genes in comparison to controls: empty vector (EV), S. cerevisiae CUP1 (CUP1), or A. thaliana MT2a (AtMT2a). Cultures were grown on normal SC (-trp) agar medium or supplemented with CdCl2 at the indicated concentration
Rights and permissions
About this article
Cite this article
Pakdee, O., Songnuan, W., Panvisavas, N. et al. Functional characterization of metallothionein-like genes from Physcomitrella patens: expression profiling, yeast heterologous expression, and disruption of PpMT1.2a gene. Planta 250, 427–443 (2019). https://doi.org/10.1007/s00425-019-03173-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-019-03173-8