Abstract
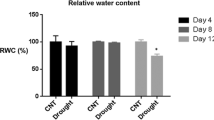
Wild watermelon (Citrullus lanatus) is a xerophyte native to the Kalahari Desert, Africa. To better understand the molecular mechanisms of drought resistance in this plant, we examined changes in the proteome in response to water deficit. Wild watermelon leaves showed decreased transpiration and a concomitant increase in leaf temperature under water deficit conditions. Comparison of the proteome of stressed plants with that of unstressed plants by two-dimensional gel electrophoresis revealed that the intensity of 40 spots increased in response to the stress, and the intensity of 11 spots decreased. We positively identified 23 stress-induced and 6 stress-repressed proteins by mass spectrometry and database analyses. Interestingly, 15 out of the 23 up-regulated proteins (65% of annotated up-regulated proteins) were heat shock proteins (HSPs). Especially, 10 out of the 15 up-regulated HSPs belonged to the small heat shock protein (sHSP) family. Other stress-induced proteins included those related to antioxidative defense and carbohydrate metabolism. Fifteen distinct cDNA sequences encoding the sHSP were characterized from wild watermelon. Quantitative real-time PCR analysis of the representative sHSP genes revealed strong transcriptional up-regulation in the leaves under water deficit. Moreover, immunoblot analysis confirmed that protein abundance of sHSPs was massively increased under water deficit. Overall, these observations suggest that the defense response of wild watermelon may involve orchestrated regulation of a diverse array of functional proteins related to cellular defense and metabolism, of which HSPs may play a pivotal role on the protection of the plant under water deficit in the presence of strong light.





Similar content being viewed by others
Abbreviations
- 2DE:
-
Two-dimensional gel electrophoresis
- CLsHSP:
-
Citrullus lanatus small heat shock protein
- HSP:
-
Heat shock protein
- LC/MS/MS:
-
Liquid chromatography–tandem mass spectrometry
- MG:
-
Methylglyoxal
- MS:
-
Mass spectrometry
- PGM:
-
Phosphoglucomutase
- PHGPX:
-
Phospholipid hydroperoxide glutathione peroxidase
- RFO:
-
Raffinose family oligosaccharide
- sHSP:
-
Small heat shock protein
References
Agarwal M, Katiyar-Agarwal S, Sahi C, Gallie DR, Grover A (2001) Arabidopsis thaliana Hsp100 proteins: kith and kin. Cell Stress Chaperones 6:219–224
Akashi K, Miyake C, Yokota A (2001) Citrulline, a novel compatible solute in drought-tolerant wild watermelon leaves, is an efficient hydroxyl radical scavenger. FEBS Lett 508:438–442
Akashi K, Nishimura N, Ishida Y, Yokota A (2004) Potent hydroxyl radical-scavenging activity of drought-induced type-2 metallothionein in wild watermelon. Biochem Biophys Res Commun 323:72–78
Akashi K, Yoshimura K, Nanasato Y, Takahara K, Munekage Y, Yokota A (2008) Wild plant resources for studying molecular mechanisms of drought/strong light stress tolerance. Plant Biotechnol 25:257–263
Apuya NR, Yadegari R, Fischer RL, Harada JJ, Zimmerman JL, Goldberg RB (2001) The Arabidopsis embryo mutant schlepperless has a defect in the chaperonin-60α gene. Plant Physiol 126:717–730
Bogeat-Triboulot MB, Brosche M, Renaut J, Jouve L, Le Thiec D, Fayyaz P, Vinocur B, Witters E, Laukens K, Teichmann T, Altman A, Hausman JF, Polle A, Kangasjarvi J, Dreyer E (2007) Gradual soil water depletion results in reversible changes of gene expression, protein profiles, ecophysiology, and growth performance in Populus euphratica, a poplar growing in arid regions. Plant Physiol 143:876–892
Bonhomme L, Monclus R, Vincent D, Carpin S, Claverol S, Lomenech AM, Labas V, Plomion C, Brignolas F, Morabito D (2009) Genetic variation and drought response in two Populus x euramericana genotypes through 2-DE proteomic analysis of leaves from field and glasshouse cultivated plants. Phytochemistry 70:988–1002
Boston RS, Viitanen PV, Vierling E (1996) Molecular chaperones and protein folding in plants. Plant Mol Biol 32:191–222
Bray EA (2004) Genes commonly regulated by water-deficit stress in Arabidopsis thaliana. J Exp Bot 55:2331–2341
Broin M, Rey P (2003) Potato plants lacking the CDSP32 plastidic thioredoxin exhibit overoxidation of the BAS1 2-cysteine peroxiredoxin and increased lipid peroxidation in thylakoids under photooxidative stress. Plant Physiol 132:1335–1343
Buchanan BB, Balmer Y (2005) Redox regulation: a broadening horizon. Annu Rev Plant Biol 56:187–220
Chaves MM, Pereira JS, Maroco J, Rodrigues ML, Ricardo CP, Osorio ML, Carvalho I, Faria T, Pinheiro C (2002) How plants cope with water stress in the field. Photosynthesis and growth. Ann Bot (Lond) 89:907–916
Dangoor I, Peled-Zehavi H, Levitan A, Pasand O, Danon A (2009) A small family of chloroplast atypical thioredoxins. Plant Physiol 149:1240–1250
Dixon DP, Cummins L, Cole DJ, Edwards R (1998) Glutathione-mediated detoxification systems in plants. Curr Opin Plant Biol 1:258–266
Espartero J, Sanchez-Aguayo I, Pardo JM (1995) Molecular characterization of glyoxalase-I from a higher plant; upregulation by stress. Plant Mol Biol 29:1223–1233
Flexas J, Medrano H (2002) Energy dissipation in C3 plants under drought. Funct Plant Biol 29:1209–1215
Frydman J (2001) Folding of newly translated proteins in vivo: the role of molecular chaperones. Annu Rev Biochem 70:603–647
Gazanchian A, Hajheidari M, Sima NK, Salekdeh GH (2007) Proteome response of Elymus elongatum to severe water stress and recovery. J Exp Bot 58:291–300
Goloubinoff P, Mogk A, Zvi AP, Tomoyasu T, Bukau B (1999) Sequential mechanism of solubilization and refolding of stable protein aggregates by a bichaperone network. Proc Natl Acad Sci USA 96:13732–13737
Gueta-Dahan Y, Yaniv Z, Zilinskas BA, Ben-Hayyim G (1997) Salt and oxidative stress: similar and specific responses and their relation to salt tolerance in citrus. Planta 203:460–469
Hajheidari M, Abdollahian-Noghabi M, Askari H, Heidari M, Sadeghian SY, Ober ES, Salekdeh GH (2005) Proteome analysis of sugar beet leaves under drought stress. Proteomics 5:950–960
Haritatos E, Keller F, Turgeon R (1996) Raffinose oligosaccharide concentrations measured in individual cell and tissue type in Cucumis melo L leaves: implications for phloem loading. Planta 198:614–622
Hasegawa PM, Bressan RA, Zhu JK, Bohnert HJ (2000) Plant cellular and molecular responses to high salinity. Annu Rev Plant Physiol Plant Mol Biol 51:463–499
Ingle RA, Schmidt UG, Farrant JM, Thomson JA, Mundree SG (2007) Proteomic analysis of leaf proteins during dehydration of the resurrection plant Xerophyta viscosa. Plant Cell Environ 30:435–446
Jeffery CJ (1999) Moonlighting proteins. Trends Biochem Sci 24:8–11
Kajikawa M, Yokota A, Akashi K (2010) Establishment of a transgenic hairy root system in wild and domesticated watermelon (Citrullus lanatus) for studying root vigor under drought. Plant Cell Rep 29:771
Kawasaki S, Miyake C, Kohchi T, Fujii S, Uchida M, Yokota A (2000) Responses of wild watermelon to drought stress: accumulation of an ArgE homologue and citrulline in leaves during water deficits. Plant Cell Physiol 41:864–873
Kennedy MC, Mende-Mueller L, Blondin GA, Beinert H (1992) Purification and characterization of cytosolic aconitase from beef liver and its relationship to the iron-responsive element binding protein. Proc Natl Acad Sci USA 89:11730–11734
Kohzuma K, Cruz JA, Akashi K, Hoshiyasu S, Munekage Y, Yokota A, Kramer DM (2009) The long-term responses of the photosynthetic proton circuit to drought. Plant Cell Environ 32:209–219
Larcher W (1995) Plants under stress. Springer, Berlin, pp 321–448
Lawlor DW, Cornic G (2002) Photosynthetic carbon assimilation and associated metabolism in relation to water deficits in higher plants. Plant Cell Environ 25:275–294
Lennartz K, Plucken H, Seidler A, Westhoff P, Bechtold N, Meierhoff K (2001) HCF164 encodes a thioredoxin-like protein involved in the biogenesis of the cytochrome b(6)f complex in Arabidopsis. Plant Cell 13:2539–2551
Lytovchenko A, Sweetlove L, Pauly M, Fernie AR (2002) The influence of cytosolic phosphoglucomutase on photosynthetic carbohydrate metabolism. Planta 215:1013–1021
Mewes HW, Dietmann S, Frishman D, Gregory R, Mannhaupt G, Mayer KF, Munsterkotter M, Ruepp A, Spannagl M, Stumpflen V, Rattei T (2008) MIPS: analysis and annotation of genome information in 2007. Nucleic Acids Res 36:D196–D201
Meyer Y, Siala W, Bashandy T, Riondet C, Vignols F, Reichheld JP (2008) Glutaredoxins and thioredoxins in plants. Biochim Biophys Acta 1783:589–600
Milioni D, Hatzopoulos P (1997) Genomic organization of hsp90 gene family in Arabidopsis. Plant Mol Biol 35:955–961
Mitchell DE, Madore MA (1992) Patterns of assimilate production and translocation in muskmelon (Cucumis melo L.): II. Low temperature effects. Plant Physiol 99:966–971
Mogk A, Schlieker C, Friedrich KL, Schonfeld HJ, Vierling E, Bukau B (2003) Refolding of substrates bound to small Hsps relies on a disaggregation reaction mediated most efficiently by ClpB/DnaK. J Biol Chem 278:31033–31042
Nishizawa A, Yabuta Y, Shigeoka S (2008) Galactinol and raffinose constitute a novel function to protect plants from oxidative damage. Plant Physiol 147:1251–1263
Ort DR (2001) When there is too much light. Plant Physiol 125:29–32
Peterbauer T, Richter A (2001) Biochemistry and physiology of raffinose family oligosaccharides and galactosyl cyclitols in seeds. Seed Sci Res 1:185–197
Pinheiro C, Chaves MM, Ricardo CP (2001) Alterations in carbon and nitrogen metabolism induced by water deficit in the stems and leaves of Lupinus albus L. J Exp Bot 52:1063–1070
Pratt WB, Toft DO (2003) Regulation of signaling protein function and trafficking by the hsp90/hsp70-based chaperone machinery. Exp Biol Med (Maywood) 228:111–133
Riccardi F, Gazeau P, de Vienne D, Zivy M (1998) Protein changes in response to progressive water deficit in maize. Quantitative variation and polypeptide identification. Plant Physiol 117:1253–1263
Roitsch T (1999) Source-sink regulation by sugar and stress. Curr Opin Plant Biol 2:198–206
Salekdeh GH, Siopongco J, Wade LJ, Ghareyazie B, Bennett J (2002) Proteomic analysis of rice leaves during drought stress and recovery. Proteomics 2:1131–1145
Siddique M, Gernhard S, von Koskull-Doring P, Vierling E, Scharf KD (2008) The plant sHSP superfamily: five new members in Arabidopsis thaliana with unexpected properties. Cell Stress Chaperones 13:183–197
Takahara K, Akashi K, Yokota A (2005) Purification and characterization of glutamate N-acetyltransferase involved in citrulline accumulation in wild watermelon. FEBS J 272:5353–5364
Thao NP, Chen L, Nakashima A, Hara S, Umemura K, Takahashi A, Shirasu K, Kawasaki T, Shimamoto K (2007) RAR1 and HSP90 form a complex with Rac/Rop GTPase and function in innate-immune responses in rice. Plant Cell 19:4035–4045
Valliyodan B, Nguyen HT (2006) Understanding regulatory networks and engineering for enhanced drought tolerance in plants. Curr Opin Plant Biol 9:189–195
van Wyk B-E, Gericke N (2000) People’s plants. A guide to useful plants of Southern Africa. Briza Publications, Pretoria
Wang W, Vinocur B, Shoseyov O, Altman A (2004) Role of plant heat-shock proteins and molecular chaperones in the abiotic stress response. Trends Plant Sci 9:244–252
Waters ER, Aevermann BD, Sanders-Reed Z (2008) Comparative analysis of the small heat shock proteins in three angiosperm genomes identifies new subfamilies and reveals diverse evolutionary patterns. Cell Stress Chaperones 13:127–142
Wickens GE (1998) Ecophysiology of economic plants in arid and semi-arid lands. Springer, Berlin
Yadav SK, Singla-Pareek SL, Ray M, Reddy MK, Sopory SK (2005) Methylglyoxal levels in plants under salinity stress are dependent on glyoxalase I and glutathione. Biochem Biophys Res Commun 337:61–67
Yamaguchi-Shinozaki K, Shinozaki K (2006) Transcriptional regulatory networks in cellular responses and tolerance to dehydration and cold stresses. Annu Rev Plant Biol 57:781–803
Yokota A, Kawasaki S, Iwano M, Nakamura C, Miyake C, Akashi K (2002) Citrulline and DRIP-1 protein (ArgE homologue) in drought tolerance of wild watermelon. Ann Bot (Lond) 89:825–832
Yoshimura K, Masuda A, Kuwano M, Yokota A, Akashi K (2008) Programmed proteome response for drought avoidance/tolerance in the root of a C3 xerophyte (wild watermelon) under water deficits. Plant Cell Physiol 49:226–241
Young JC, Moarefi I, Hartl FU (2001) Hsp90: a specialized but essential protein-folding tool. J Cell Biol 154:267–273
Acknowledgments
We thank Drs Taise Shimaoka and Kaori Kohzuma for helpful technical assistance on the proteome analysis. This research was supported partly by a grant from the Japan Society for the Promotion of Science, and by funds from the Nara Institute of Science and Technology (NAIST), both awarded to K. Akashi. This research was also supported partly by the Asia-Africa Science and Technology Strategic Cooperation Promotion Program from the Japan Science and Technology Agency, the NAIST Global COE program, and the Nissan Science Foundation.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
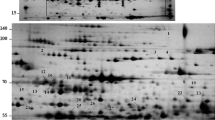
Supplementary Fig. 1 Three replicates of 2DE images of proteome in the leaf soluble fraction of wild watermelon. Leaf soluble proteins from three unstressed plants (a-c), and those from three plants under water deficit for three days (d-f), were extracted individually, and separated by 2DE with the isoelectric focusing in the first horizontal dimension, and SDS/PAGE in the second vertical dimension. These three sets of 2DE images, together with the data presented in Fig. 2, were used for the quantitative analysis of the spot intensity, and for the LC/MS/MS analyses for protein identification.
Supplementary Fig. 2 Amino acid alignment of CLsHSPs. Alignment was generated by ClustalW program, and visually inspected and edited. In each block of the alignments, amino acid positions counted from their N-terminus were presented at the rightmost columns
425_2010_1341_MOESM1_ESM.pdf
Supplementary Fig. 1 Three replicates of 2DE images of proteome in the leaf soluble fraction of wild watermelon. Leaf soluble proteins from three unstressed plants (a–c), and those from three plants under water deficit for three days (d–f), were extracted individually, and separated by 2DE with the isoelectric focusing in the first horizontal dimension, and SDS/PAGE in the second vertical dimension. These three sets of 2DE images, together with the data presented in Fig. 2, were used for the quantitative analysis of the spot intensity, and for the LC/MS/MS analyses for protein identification (PDF 682 kb)
425_2010_1341_MOESM2_ESM.pdf
Supplementary Fig. 2 Amino acid alignment of CLsHSPs. Alignment was generated by ClustalW program, and visually inspected and edited. In each block of the alignments, amino acid positions counted from their N-terminus were presented at the rightmost columns. (PDF 191 kb)
Rights and permissions
About this article
Cite this article
Akashi, K., Yoshida, K., Kuwano, M. et al. Dynamic changes in the leaf proteome of a C3 xerophyte, Citrullus lanatus (wild watermelon), in response to water deficit. Planta 233, 947–960 (2011). https://doi.org/10.1007/s00425-010-1341-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-010-1341-4