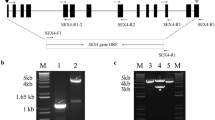
Abstract
The genes encoding the major putative rice plastidic translocators involved in the carbon flow related to starch metabolism were identified by exhaustive database searches. The genes identified were two for the triose phosphate/phosphate translocator (TPT), five for the glucose 6-phosphate/phosphate translocator (GPT) including putatively non-functional ones, four for the phosphoenolpyruvate/phosphate translocator (PPT), three for the putative ADP-glucose translocator (or Brittle-1 protein, BT1), two for the plastidic nucleotide transport protein (NTT), and one each for the plastidic glucose translocator (pGlcT) and the maltose translocator (MT). The expression patterns of the genes in various photosynthetic and non-photosynthetic organs were examined by quantitative real-time PCR. OsBT1-1 was specifically expressed in the seed and its transcript level tremendously increased at the onset of vigorous starch production in the endosperm, suggesting that the ADP-glucose synthesized in the cytosol is a major precursor for starch biosynthesis in the endosperm amyloplast. In contrast, all of the genes for OsTPT, OsPPT, and OsNTT were mainly expressed in source tissues, suggesting that their proteins play essential roles in the regulation of carbohydrate metabolism in chloroplasts. Substantial expression of the four OsGPT genes and the OspGlcT gene in both source and sink organs suggests that the transport of glucose phosphate and glucose is physiologically important in both photosynthetic and non-photosynthetic tissues. The present study shows that comprehensive analysis of expression patterns of the plastidic translocator genes is a valuable tool for the elucidation of the functions of the translocators in the regulation of starch metabolism in rice.



Similar content being viewed by others
Abbreviations
- BT1:
-
Brittle-1
- DAF:
-
Days after flowering
- GPT:
-
Glucose 6-phosphate/phosphate translocator
- MT:
-
Maltose translocator
- NTT:
-
Plastidic nucleotide transport protein
- pGlcT:
-
Plastidic glucose translocator
- PPT:
-
Phosphoenolpyruvate/phosphate translocator
- TPT:
-
Triose phosphate/phosphate translocator
References
Achard P, Lagrange T, El-Zanaty AF, Mache R (2003) Architecture and transcriptional activity of the initiator element of the TATA-less RPL21 gene. Plant J 35:743–752
Ball SG, Morell MK (2003) From bacterial glycogen and to starch: understanding the biogenesis of the plant starch granule. Annu Rev Plant Biol 54:207–233
Bhave MR, Lawrence S, Barton C, Hannah LC (1990) Identification and molecular characterization of shrunken-2 cDNA clones of maize. Plant Cell 2:581–588
Butowt R, Granot D, Rodriguez-Garcia MI (2003) A putative plastidic glucose translocator is expressed in heterotrophic tissues that do not contain starch, during olive (Olea europea L.) fruit ripening. Plant Cell Physiol 44:1152–1161
Cao H, Shannon JC (1997) BT1, a possible adenylate translocator, is developmentally expressed in maize endosperm but not detected in starchy tissues from several other species. Physiol Plant 100:400–406
Eicks M, Maurino V, Knappe S, Flügge UI, Fischer K (2002) The plastidic pentose phosphate translocator represents a link between the cytosolic and the plastidic pentose phosphate pathways in plants. Plant Physiol 128:512–522
Emes MJ, Bowsher CG, Hedley C, Burrell MM, Scrase-Field ES, Tetlow IJ (2003) Starch synthesis and carbon partitioning in developing endosperm. J Exp Bot 54:569–575
Ferro M, Salvi D, Riviere-Rolland H, Vermat T, Seigneurin-Berny D, Grunwald D, Garin J, Joyard J, Rolland N (2002) Integral membrane proteins of the chloroplast envelope: identification and subcellular localization of new transporters. Proc Natl Acad Sci USA 99:11487–11492
Fischer K, Weber A (2002) Transport of carbon in non-green plastids. Trends Plant Sci 7:345–351
Fischer K, Arbinger B, Kammerer B, Busch C, Brink S, Wallmeier H, Sauer N, Eckerskorn C, Flügge UI (1994) Cloning and in vivo expression of functional triose phosphate/phosphate translocators from C3- and C4-plants: evidence for the putative participation of specific amino acid residues in the recognition of phosphoenolpyruvate. Plant J 5:215–226
Fischer K, Kammerer B, Gutensohn M, Arbinger B, Weber A, Hausler RE, Flügge UI (1997) A new class of plastidic phosphate translocators: a putative link between primary and secondary metabolism by the phosphoenolpyruvate/phosphate antiporter. Plant Cell 9:453–462
Flügge U (1999) Phosphate translocators in plastids. Annu Rev Plant Physiol Plant Mol Biol 50:27–45
Geigenberger P, Stamme C, Tjaden J, Schulz A, Quick PW, Betsche T, Kersting HJ, Neuhaus HE (2001) Tuber physiology and properties of starch from tubers of transgenic potato plants with altered plastidic adenylate transporter activity. Plant Physiol 125:1667–1678
Goff SA, Ricke D, Lan TH, Presting G, Wang R, Dunn M, Glazebrook J, Sessions A, Oeller P, Varma H, Hadley D, Hutchison D, Martin C, Katagiri F, Lange BM, Moughamer T, Xia Y, Budworth P, Zhong J, Miguel T, Paszkowski U, Zhang S, Colbert M, Sun WL, Chen L, Cooper B, Park S, Wood TC, Mao L, Quail P, Wing R, Dean R, Yu Y, Zharkikh A, Shen R, Sahasrabudhe S, Thomas A, Cannings R, Gutin A, Pruss D, Reid J, Tavtigian S, Mitchell J, Eldredge G, Scholl T, Miller RM, Bhatnagar S, Adey N, Rubano T, Tusneem N, Robinson R, Feldhaus J, Macalma T, Oliphant A, Briggs S (2002) A draft sequence of the rice genome (Oryza sativa L. ssp. japonica). Science 296:92–100
Herold A, Leegood R, McNeil PH, Robinson SP (1981) Accumulation of maltose during photosynthesis in protoplasts isolated from spinach leaves treated with mannose. Plant Physiol 67:85–88
Higo K, Ugawa Y, Iwamoto M, Korenaga T (1999) Plant cis-acting regulatory elements (PLACE) database: 1999. Nucleic Acids Res 27:297–300
Huber SC, Hanson KR (1992) Carbon partitioning and growth of a starchless mutant of Nicotiana sylvestris. Plant Physiol 99:1449–1454
James MG, Denyer K, Myers AM (2003) Starch synthesis in the cereal endosperm. Curr Opin Plant Biol 6:215–222
Kammerer B, Fischer K, Hilpert B, Schubert S, Gutensohn M, Weber A, Flügge UI (1998) Molecular characterization of a carbon transporter in plastids from heterotrophic tissues: the glucose 6-phosphate/phosphate antiporter. Plant Cell 10:105–117
Kampfenkel K, Mohlmann T, Batz O, Van Montagu M, Inzé D, Neuhaus HE (1995) Molecular characterization of an Arabidopsis thaliana cDNA encoding a novel putative adenylate translocator of higher plants. FEBS Lett 374:351–355
Kawagoe Y, Kubo A, Satoh H, Takaiwa F, Nakamura Y (2005) Roles of isoamylase and ADP-glucose pyrophosphorylase in starch granule synthesis in rice endosperm. Plant J 42:164–174
Kikuchi S, Satoh K, Nagata T, Kawagashira N, Doi K, Kishimoto N, Yazaki J, Ishikawa M, Yamada H, Ooka H, Hotta I, Kojima K, Namiki T, Ohneda E, Yahagi W, Suzuki K, Li CJ, Ohtsuki K, Shishiki T, Otomo Y, Murakami K, Iida Y, Sugano S, Fujimura T, Suzuki Y, Tsunoda Y, Kurosaki T, Kodama T, Masuda H, Kobayashi M, Xie Q, Lu M, Narikawa R, Sugiyama A, Mizuno K, Yokomizo S, Niikura J, Ikeda R, Ishibiki J, Kawamata M, Yoshimura A, Miura J, Kusumegi T, Oka M, Ryu R, Ueda M, Matsubara K, Kawai J, Carninci P, Adachi J, Aizawa K, Arakawa T, Fukuda S, Hara A, Hashizume W, Hayatsu N, Imotani K, Ishii Y, Itoh M, Kagawa I, Kondo S, Konno H, Miyazaki A, Osato N, Ota Y, Saito R, Sasaki D, Sato K, Shibata K, Shinagawa A, Shiraki T, Yoshino M, Hayashizaki Y, Yasunishi A; Rice Full-Length cDNA Consortium; National Institute of Agrobiological Sciences Rice Full-Length cDNA Project Team; Foundation of Advancement of International Science Genome Sequencing & Analysis Group; RIKEN (2003) Collection, mapping, and annotation of over 28,000 cDNA clones from japonica rice. Science 301:376–379
Knappe S, Flügge UI, Fischer K (2003a) Analysis of the plastidic phosphate translocator gene family in Arabidopsis and identification of new phosphate translocator-homologous transporters, classified by their putative substrate-binding site. Plant Physiol 131:1178–1190
Knappe S, Lottgert T, Schneider A, Voll L, Flügge UI, Fischer K (2003b) Characterization of two functional phosphoenolpyruvate/phosphate translocator (PPT) genes in Arabidopsis–AtPPT1 may be involved in the provision of signals for correct mesophyll development. Plant J 36:411–420
Knight JS, Gray JC (1994) Expression of genes encoding the tobacco chloroplast phosphate translocator is not light-regulated and is repressed by sucrose. Mol Gen Genet 242:586–594
Kofler H, Hausler RE, Schulz B, Groner F, Flügge UI, Weber A (2000) Molecular characterization of a new mutant allele of the plastid phosphoglucomutase in Arabidopsis, and complementation of the mutant with the wild-type cDNA. Mol Gen Genet 263:978–986
Koo AJ, Ohlrogge JB (2002) The predicted candidates of Arabidopsis plastid inner envelope membrane proteins and their expression profiles. Plant Physiol 130:823–836
Leroch M, Kirchberger S, Haferkamp I, Wahl M, Neuhaus E, Tjaden J (2005) Identification and characterization of a novel plastidic adenine nucleotide uniporter from Solanum tuberosum. J Biol Chem 280:17992–18000
Mohlmann T, Tjaden J, Henrichs G, Quick WP, Hausler R, Neuhaus HE (1997) ADP-glucose drives starch synthesis in isolated maize endosperm amyloplasts: characterization of starch synthesis and transport properties across the amyloplast envelope. Biochem J 324:503–509
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Naeem M, Tetlow IJ, Emes MJ (1997) Starch synthesis in amyloplasts purified from developing potato tubers. Plant J 11:1095–1103
Nakamura Y (2002) Towards a better understanding of the metabolic system for amylopectin biosynthesis in plants: rice endosperm as a model tissue. Plant Cell Physiol 43:718–725
Nakamura M, Tsunoda T, Obokata J (2002) Photosynthesis nuclear genes generally lack TATA-boxes: a tobacco photosystem I gene responds to light through an initiator. Plant J 29:1–10
Neuhaus HE, Wagner R (2000) Solute pores, ion channels, and metabolite transporters in the outer and inner envelope membranes of higher plants plastids. Biochim Biophys Acta 1465:307–323
Neuhaus HE, Thom E, Mohlmann T, Steup M, Kampfenkel K (1997) Characterization of a novel eukaryotic ATP/ADP translocator located in the plastid envelope of Arabidopsis thaliana L. Plant J 11:73–82
Niittyla T, Messerli G, Trevisan M, Chen J, Smith AM, Zeeman SC (2004) A previously unknown maltose transporter essential for starch degradation in leaves. Science 303:87–89
Overlach S, Diekmann W, Raschke K (1993) Phosphate translocator of isolated guard-cell chloroplasts from Pisum sativum L. transports glucose-6-phosphate. Plant Physiol 101:1201–1207
Page RD (1996) Tree view: an application to display phylogenetic trees on personal computers. Comput Appl Biosci 12:357–358
Patron NJ, Greber B, Fahy BF, Laurie DA, Parker ML, Denyer K (2004) The lys5 mutations of barley reveal the nature and importance of plastidial ADP-Glc transporters for starch synthesis in cereal endosperm. Plant Physiol 135:2088–2097
Reiser J, Linka N, Lemke L, Jeblick W, Neuhaus HE (2004) Molecular physiological analysis of the two plastidic ATP/ADP transporters from Arabidopsis. Plant Physiol 136:3524–3536
Satoh H, Nishi A, Yamashita K, Takemoto Y, Tanaka Y, Hosaka Y, Sakurai A, Fujita N, Nakamura Y (2003) Starch-branching enzyme I-deficient mutation specifically affects the structure and properties of starch in rice endosperm. Plant Physiol 133:1111–1121
Schulz B, Frommer WB, Flugge UI, Hummel S, Fischer K, Willmitzer L (1993) Expression of the triose phosphate translocator gene from potato is light dependent and restricted to green tissues. Mol Gen Genet 238:357–361
Schunemann D, Schott K, Borchert S, Heldt HW (1996) Evidence for the expression of the triosephosphate translocator gene in green and non-green tissue of tomato and potato. Plant Mol Biol 31:101–111
Schwacke R, Schneider A, van der Graaff E, Fischer K, Catoni E, Desimone M, Frommer WB, Flügge UI, Kunze R (2003) ARAMEMNON, a novel database for Arabidopsis integral membrane proteins. Plant Physiol 131:16–26
Shannon JC, Pien FM, Cao H, Liu KC (1998) Brittle-1, an adenylate translocator, facilitates transfer of extraplastidial synthesized ADP-glucose into amyloplasts of maize endosperms. Plant Physiol 117:1235–1252
Sullivan TD, Kaneko Y (1995) The maize brittle1 gene encodes amyloplast membrane polypeptides. Planta 196:477–484
Sullivan TD, Strelow LI, Illingworth CA, Phillips RL, Nelson OE Jr (1991) Analysis of maize brittle-1 alleles and a defective suppressor-mutator-induced mutable allele. Plant Cell 3:1337–1348
Tetlow IJ, Blisset KJ, Emes MJ (1994) Starch synthesis and carbohydrate oxidation in amyloplasts from developing wheat endosperm. Planta 194:454–460
Tetlow IJ, Bowsher CG, Emes MJ (1996) Reconstitution of the hexose phosphate translocator from the envelope membrane of wheat endsperm amyloplasts. Biochem J 319:717–723
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Tjaden J, Mohlmann T, Kampfenkel K, Henrich G, Neuhaus HE (1998) Alterd plastidic ATP/ADP-transporter activity influences potato (Solanum tuberosum L.) tuber morphology, yield and composition of tuber starch. Plant J 16:531–540
Tyson RH, ap Rees T (1988) Starch synthesis by isolated amyloplasts from wheat endosperm. Planta 175:33–38
Ware D, Jaiswal P, Ni J, Pan X, Chang K, Clark K, Teytelman L, Schmidt S, Zhao W, Cartinhour S, McCouch S, Stein L (2002) Gramene: a resource for comparative grass genomics. Nucleic Acids Res 30:103–105
Weber A (2004) Solute transporters as connecting elements between cytosol and plastid stroma. Curr Opin Plant Biol 7:247–253
Weber A, Servaites JC, Geiger DR, Kofler H, Hille D, Groner F, Hebbeker U, Flügge UI (2000) Identification, purification, and molecular cloning of a putative plastidic glucose translocator. Plant Cell 12:787–802
Weber A, Schneidereit J, Voll L (2004) Using mutants to probe the in vivo function of plastid envelope membrane metabolite transporters. J Exp Bot 55:1231–1244
Weise SE, Weber A, Sharkey TD (2004) Maltose is the major form of carbon exported from the chloroplast at night. Planta 218:474–482
Winkler HH, Neuhaus HE (1999) Non-mitochondrial ATP transport. Trends Biochem Sci 24:64–68
Yano M, Isono Y, Satoh H, Omura T (1984) Gene analysis of sugary and shrunken mutants of rice, Oryza sativa L. Jpn J Breed 34:43–49
Zimmermann P, Hirsch-Hoffmann M, Hennig L, Gruissem W (2004) GENEVESTIGATOR. Arabidopsis microarray database and analysis toolbox. Plant Physiol 136:2621–2632
Acknowledgements
We are grateful to Dr. Perigio B. Francisco Jr. (Akita Prefectural University, Japan) for his critical reading of the manuscript. Analysis of DNA sequencing was conducted with the CREST-Akita Plant Molecular Science Satellite Laboratory in Life Science Research Support Center in Akita Prefectural University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Toyota, K., Tamura, M., Ohdan, T. et al. Expression profiling of starch metabolism-related plastidic translocator genes in rice. Planta 223, 248–257 (2006). https://doi.org/10.1007/s00425-005-0128-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-005-0128-5