Abstract
It is increasingly appreciated that the acidic microenvironment of a tumour contributes to its evolution and clinical outcomes. However, our understanding of the mechanisms by which tumour cells detect acidosis and the signalling cascades that it induces is still limited. Acid-sensing ion channels (ASICs) are sensitive receptors for protons; therefore, they are also candidates for proton sensors in tumour cells. Although in non-transformed tissue, their expression is mainly restricted to neurons, an increasing number of studies have reported ectopic expression of ASICs not only in brain cancer but also in different carcinomas, such as breast and pancreatic cancer. However, because ASICs are best known as desensitizing ionotropic receptors that mediate rapid but transient signalling, how they trigger intracellular signalling cascades is not well understood. In this review, we introduce the acidic microenvironment of tumours and the functional properties of ASICs, point out some conceptual problems, summarize reported roles of ASICs in different cancers, and highlight open questions on the mechanisms of their action in cancer cells. Finally, we propose guidelines to keep ASIC research in cancer on solid ground.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Acidosis as a characteristic of the tumour microenvironment
Cancer is a complex and polymorphic disease characterized by significant differences between cancer and non-cancer cells. While carcinogenesis has already been extensively studied on a cellular level over the years, recent attention has increasingly focused on the unique tumour microenvironment (TME) that differs substantially from its physiological counterpart. The TME plays a crucial role in cancer development, progression, and behaviour [65]. The most-extensively studied characteristic of the TME is hypoxia, defined as a decreased oxygen supply at tissue, resulting from inadequate vascularization of rapidly proliferating tumour cells [35]. Another common, yet less explored, characteristic is its acidity: Cancer cells are typically surrounded by a milieu containing a substantially higher concentration of protons. Tumour tissue has been reported to exhibit pH values as low as 6.6 or even lower [30, 76].
In general, pH homeostasis is crucial for cellular function, as alterations in pH can impact the function and activity of enzymes, ion channels, transporters, and other cellular proteins. Acidity is a characteristic feature of various pathophysiological states, not only cancer. Particularly in cancer, multiple mechanisms contribute to an increased intracellular proton concentration (decrease of the intracellular pH, pHi) as well as to the extrusion of protons. Given that pH regulation is a highly dynamic process, protons are constantly released to and removed from the extracellular space. At a certain point in carcinogenesis, proton release into the extracellular space surpasses its removal, primarily due to inadequate vascularization. This imbalance finally leads to an accumulation of protons in the extracellular compartment, causing a drop of the extracellular pH (pHe) [32] (Fig. 1a). Consequently, an acidic TME is a key characteristic in cancer, exhibiting a heterogeneous spatial pH distribution within the tumour mass [37], so that some cancer cells face an acidic pHe while others do not.
Disturbed acid–base regulation in cancers. a The increased metabolic activity of cancer cells leads to increased CO2 production and generation of protons. Due to increased O2 consumption and decreased delivery of O2, the TME often becomes hypoxic and glycolysis is increased; the Warburg effect further increases glycolysis and production of lactate and protons. Due to the increased diffusion distance, protons accumulate in the TME and pHe decreases. MCT, monocarboxylate transporter. b Left, in non-tumour cells, the proton concentration within the cells is slightly higher than outside of the cells. Right, in cancer cells, the activity of acid-extruding transporters is increased and the proton gradient can be inversed. However, the pHi is often still more acidic than in non-tumour cells. The actual pH values vary depending on the local position of a cell within a tumour. NHE, Na+-H+-exchanger; NBC, Na+-bicarbonate cotransporter
There are several reasons for the increased intracellular release of protons, including enhanced glycolysis and mitochondrial respiration. But enhanced glycolysis appears to be characteristic for cancer cells: Their increased growth, especially within the core of the tumour mass, results in limited access to oxygen due to an inadequate vascularization, leading to hypoxia [75]. Hypoxia shifts cellular metabolism in both cancer and non-cancer cells towards glycolysis. This adaptation allows cells to account for their ATP requirements without relying on oxygen molecules. In contrast to 38 molecules of ATP per glucose produced through oxidative phosphorylation, the 2 molecules of ATP per glucose resulting from the Pasteur effect seem to be rather inefficient and contribute to the increased acid production to meet cellular ATP demands [45] (Fig. 1a). Remarkably, while most non-tumour cells predominantly shift their metabolism towards lactic acid fermentation during oxygen deprivation, cancer cells as well as highly proliferating non-cancer cells tend to exhibit this behaviour even without limited access to oxygen, referred to as Warburg effect [29].
Hence, it is interesting that cancer cells respond differently to an acidic TME than non-tumour cells and generally exhibit better resistance to this extracellular stressor. To understand this phenomenon, it is essential to appreciate that pHi and pHe are tightly coupled, with an electrochemical driving force favouring intracellular acidification relative to the extracellular compartment. For a pHi of 7.2, which corresponds to 63 nM protons, and a pHe of 7.4, which corresponds to 40 nM protons, the equilibrium potential EH for protons is ~ -12 mV, indicating an inwardly directed driving force for protons at a membrane potential more negative than EH. To avoid a drastic drop of the pHi, which is a potent inductor of apoptosis, active acid extrusion mechanisms are indispensable. These mechanisms involve either extruding protons, for example facilitated by Na+/H+-exchangers (NHEs) or monocarboxylate transporters (MCTs), or importing HCO3−-ions, for example through Na+/HCO3−-cotransporters (NBCs), which then buffer the excess of intracellular protons. Consequently, compared to typical physiological conditions with a pHe ~ 7.4 and a pHi ~ 7.2, this gradient is often reversed in cancer cells [30] (Fig. 1b). In cancer cells, acid-extruding mechanisms are typically upregulated compared to their non-tumour counterparts [70], contributing to the reversed pH gradient. However, it is crucial to point out that within the TME this is a rather relative phenomenon, resulting in cancer cells still exhibiting a lower or at least equal pHi compared to non-cancer cells in their typical microenvironment (Fig. 1b).
Although still not as extensively studied as the hypoxic TME, there is a growing number of studies focusing on the impact of an acidic TME on cancer cell behaviour [72]. Recent publications demonstrate that alterations of the pHe affect different aspects of cancer cell behaviour, including migration [19], invasion [26], metastatic potential [40], proliferation, stemness and cell death [18], and can also promote somatic evolution of cancer cells [64]. However, the precise mechanisms by which cancer cells sense pH alterations remain poorly understood. There are various possibilities on how cells can sense the pHe. One possibility involves the coupling of the pHi to the pHe: Secondary changes in pHi could potentially impact entire metabolic pathways due to pH-optima of key enzymes, as shown for glycolysis [60]. Alternatively, intracellularly located direct proton or HCO3−-sensors, including the FAK-related kinase PYK2 [53] and the soluble adenylate cyclase [16] might act as sensors.
Given that the pHe to pHi gradient is often reversed in cancer cells [48], direct sensing of the pHe may be more significant in cancer cells. Extracellular proton sensing has already been demonstrated for G-protein-coupled receptors, including GPR68, GPR4 and GPR5 [41], whereas the receptor protein tyrosine phosphatase-γ (RPTPγ) has recently been identified as a sensor for the extracellular HCO3−-concentration [99]. Another group of sensors, although less extensively studied, includes acid-sensing ion channels (ASICs). Their activation by extracellular protons and modulation by intracellular protons [52], make them interesting pH-sensing targets, particularly in cancer cells.
Functional properties of acid-sensing ion channels
An ASIC subunit has two transmembrane domains and a large extracellular domain (ECD), to which protons and modulators bind. The relatively short N and C termini are in the cytoplasm (Fig. 2). Three ASIC subunits assemble into functional homo- or heterotrimers. ASICs are weakly selective Na+ channels (PNa/PK = 5–14) [33] such that the opening of an ASIC depolarizes a cell.
Ionotropic and metabotropic signalling of ASIC1a. A sudden drop in pH activates ASIC1a, leading to a transient influx of Na+ and depolarization of the cell. This depolarization could contribute to the excitatory postsynaptic potential at nerve cell-tumour cell synapses. In addition, it can trigger the activation of Cavs and transient influx of Ca2+. Within seconds, ASIC1a desensitizes, and the pore is shut. This will also lead to the closure of Cavs. It has been proposed that during sustained acidosis, the N and C termini of ASIC1a move apart, enabling the interaction of the C terminus with RIPK1 to trigger cell death
There are six principal ASIC isoforms in humans, namely ASIC1a, ASIC1b, ASIC2a, ASIC2b, ASIC3, and ASIC4. Most studies to date have reported a role for ASIC1a in cancer cells, and a few for ASIC2a and ASIC3 as well. Homomeric ASIC1a and ASIC3 have high proton sensitivity: They start to be activated at pH 6.8—7.1 and are half-maximally activated at pH50 ~ 6.6 [33]. Homomeric ASIC2a has a much lower pH sensitivity (pH50 < 5.0) [4, 13, 42], but forms heteromers with ASIC1a and ASIC3. Heteromeric ASIC1a/2a and ASIC3/2a have a relatively low pH50 (pH50 < 6.0), but start to activate at pH ~ 6.7 [42, 93]. Thus, homomeric ASIC1a and ASIC3 and perhaps heteromeric ASIC1a/2a and ASIC3/2a are sensitive enough to sense the pH values that are typical for the acidic TME (pH 7.0 – 6.6; Table 1).
In non-transformed tissues, ASIC expression is mainly confined to neurons. Homomeric ASIC1a and heteromeric ASIC1a/2a are the most abundant ASICs in the central nervous system [2, 5, 90], but ASIC1a/2a heteromers are probably less abundant in the human than in the rodent brain [22, 51], for which we have most data. ASIC3 is primarily expressed in the peripheral nervous system [39]. In the brain, ASIC1a localizes to the postsynaptic membrane [85, 95], and upon binding of protons, which are released during synaptic transmission, it opens and allows the transient influx of Na+, contributing to excitatory postsynaptic currents in different brain regions [23, 31, 46]. Thus, the main function of ASICs appears to be the mediation of phasic responses to synaptic fluctuations in the proton concentration. Intriguingly, in recent years it has become apparent that many tumours are innervated and that neuronal input drives tumour progression [56, 63, 77,78,79, 89]. Thus, it is an exciting possibility that ectopic expression of ASIC1a in tumour tissues contributes to the response of tumour cells to synaptically released protons. However, this hypothesis has not yet been explored.
ASIC1a may also localize to extra-synaptic sites, for example on the soma of neurons, where it could mediate tonic responses to the ambient proton concentration. The problem with this idea is that within a few seconds of binding of protons, ASIC1a enters a non-conducting, desensitized state, from which it can no longer be activated (Fig. 2). Even at a slightly reduced pH, ASIC1a enters the desensitized state without apparent opening, such that all channels are desensitized at pH < 7.0 [4]. This process, known as steady-state desensitization (SSD), severely limits the ability of homomeric ASIC1a to signal during sustained acidosis [33]. Recovery from desensitization (return to a closed conformation) requires that the pH returns to values > 7.0 [4]. Thus, ASIC1a is ideally suited for detecting transient acidification but not the acidic pH of the TME. The same applies for ASIC1a/2a. Nevertheless, so far, most studies on ASICs in cancer cells have explored the response of ASICs to sustained acidification. Only for homomeric ASIC3 and heteromeric ASIC3/2a, activation and SSD curves slightly overlap, generating a small sustained “window” current near pH 7.0 for ASIC3, and at slightly more acidic pH for ASIC3/2a [93]. This window current leads to a small but sustained influx of Na+, slightly depolarizing the cell. This is important, for example, for triggering cardiac pain associated with myocardial ischemia [71, 93].
Pharmacological inhibition is one way to test the involvement of ASICs in specific cellular events. The canonical ASIC inhibitor is amiloride. It provides a convenient, low-cost drug to test the involvement of ASICs. However, because amiloride is not specific for ASICs and has relatively low potency for ASICs (IC50 ~ 10 μM), it can only provide first and inconclusive evidence for the involvement of ASICs. In particular, it needs to be considered that amiloride also inhibits NHEs with a similar IC50 as ASICs [28]. Therefore, other, more specific ASIC inhibitors should also be used. The standard toxin that inhibits homomeric ASIC1a with high potency (IC50 ~ 5 nM) and relatively high specificity is psalmotoxin 1 (PcTx1) [14, 25]. In addition, the snail toxin MitTx is an ASIC1a agonist that can be used to activate ASIC1a homomers at neutral pH [8].
ASICs belong to the DEG/ENaC gene family [44]. Other members of this family in humans are the bile-acid-sensitive ion channel BASIC (also named INaC or ASIC5) [68, 88] and the α-, β-, γ-, and δ-subunits of the epithelial Na+ channel (ENaC). BASIC is relatively closely related to ASICs [86], but is insensitive to protons [87]. Although ASIC-BASIC heteromers have never been described, they cannot be formally excluded. In contrast, recent phylogenetic analyses have shown that ENaC belongs to a clade of the DEG/ENaC gene family that is separated by > 500 million years of evolution from the clade containing ASICs [1, 24]. Because both subgroups have evolved independently for such a long time, it is unlikely that they still form functional heteromers. Although such ASIC-ENaC heteromers have been postulated in some cancer cells (see below), their existence is questionable in the absence of robust experimental support.
How could the activation of ASICs trigger downstream signalling in cancer cells?
The main mechanistic link between ASIC activation and proliferation, migration, and invasion of cancer cells, which has been postulated so far, is Ca2+ signalling (see below). Homomeric ASIC1a and human homomeric ASIC1b are the only ASICs that are also permeable to Ca2+ [6, 39, 81]. The Ca2+ permeability of ASIC1a homomers is low (PNa/PCa ~ 20) [6], however, and the effect of ASIC1a activation on the intracellular Ca2+ concentration ([Ca2+]i) is often overestimated. Even though the Ca2+ permeability of human ASIC1b homomers seems to be higher (PNa/PCa ~ 2.5) [39], the significance of this finding remains unclear, as ASIC1b has not yet been described in cancer cells. Most studies report that the activation of ASIC1a homomers is insufficient to substantially increase [Ca2+]i by itself [67, 73, 96], but may do so via the activation of voltage-gated Ca2+ channels (Cavs) [58, 96] (Fig. 2). Therefore, while opening homomeric ASIC1a might increase the local Ca2+ concentration in a nanodomain close to the channel, the bulk of Ca2+ influx occurs via Cavs and relies on the presence of these channels in cancer cells. Since carcinomas are derived from epithelial cells, which are typically non-excitable, it is important to test for the presence of Cavs in carcinoma cells when an increase in [Ca2+]i is claimed. In any case, due to the desensitization of ASIC1a and inactivation of Cavs, the influx of Ca2+ triggered by ASIC1a will be transient. Moreover, it is possible that Cavs, even if present, are inactive at the depolarized membrane potential that is typical for cancer cells [94].
It is also important to keep in mind that Ca2+ signals in cells are typically not only transient but also local. Ca2+ diffusion within a cell is strongly limited by Ca2+-binding proteins. In addition, there are efficient ways to remove Ca2+ from the cytoplasm, such as the plasma membrane Ca2+-ATPase and the Na+-Ca2+-exchanger (NCX). Although it has been reported that ASIC1a activation induces Ca2+ waves that propagate to mitochondria [3], our understanding of the temporal and spatial Ca2+ signals triggered by ASIC opening in cells is limited and it is unclear how the transient influx of Ca2+ triggered by ASIC activation can lead to dysregulation of Ca2+ signalling, which needs to be assumed to explain at least some of the presumed effects of ASICs on cancer cells.
Recently, a completely new metabotropic function, which is independent of ion conduction, was proposed for ASIC1a [84]. Several independent studies have reported that sustained activation of ASIC1a, for example during the acidosis associated with ischemic stroke, leads to neurodegeneration [11, 92]. But how can a completely desensitizing ion channel signal sustained acidosis and induce cell death? Interestingly, it has recently been reported that upon prolonged activation, ASIC1a directly interacts and activates receptor interacting protein kinase 1 (RIPK1) [84]. RIPK1 is known to be part of the necroptosis pathway [17, 50], and it has been proposed that activation of RIPK1 by ASIC1a leads to neurodegeneration. Moreover, it has been proposed that the ASIC1a-RIPK1 interaction is triggered by slow conformational changes in the cytoplasmic termini of ASICs during sustained acidosis that break electrostatic interactions between the cytoplasmic N and C termini, liberating the C terminus for interaction with RIPK1 [83] (Fig. 2). Interestingly, pretreatment with inhibitors of RIPK1 was necessary to prevent acid-induced cell death, suggesting activation of RIPK1 at the onset of acidotoxicity in neurons [84].
However, experimental support for this model is ambiguous. One study using fluorescence resonance energy transfer (FRET) to interrogate motions of the cytoplasmic N and C termini of ASIC1 did not find evidence for the predicted lateral movement (Fig. 2), but found that they slightly moved toward the plasma membrane upon extracellular acidification [20]. Another study found that the N and C termini are not close enough to each other to directly interact [21]. Although these methods have limitations, they highlight that we still need to better understand the dynamics of the ASIC1a-RIPK1 interaction. Moreover, it is unclear whether activation of RIPK1 by ASIC1a triggers necroptosis or another cell death pathway (see also below) [18]. Thus, many aspects of the sustained, metabotropic signalling by ASIC1a remain unclear.
In summary, while ASIC1a may have metabotropic functions in addition to its ionotropic functions, to date, only a direct interaction with RIPK1 to induce cell death has been reported. Moreover, how ASIC1a could selectively initiate specific cellular signalling pathways by triggering Ca2+ influx is largely unknown. With these ideas in mind, we will now review the reported roles of ASICs in different types of cancer. We will largely exclude reports from our review in which the role of ASICs has been investigated only at neutral pH 7.4, because the downstream signalling involved is obscure. Figure 3 provides an overview of the mRNA abundance of ASIC1a in different cancer types (red) and normal tissue (blue).
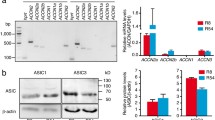
ASIC1 (alias: ACCN2) expression level in different cancer types. Differential expression of ACCN2 (ASIC1) between tumor and adjacent normal tissues was analyzed using data from “The Cancer Genome Atlas” (TCGA), accessed through TIMER2.0 [54] on November 15, 2023. ACCN2 is expressed across multiple cancer types, with the highest expression in Lower Grade Glioma (LGG) and Glioblastoma (GBM). Additionally, ACCN2 shows a significantly elevated expression in breast invasive carcinoma (BRCA), cholangiocarcinoma (CHOL), colon adenocarcinoma (COAD), head and neck squamous cell carcinoma (HNSC), kidney chromophobe carcinoma (KICH), liver hepatocellular carcinoma (LIHC), lung adenocarcinoma (LUAD), lung squamous cell carcinoma (LUSC), pheochromocytoma and paraganglioma (PCPG), prostate adenocarcinoma (PRAD), rectum adenocarcinoma (READ) and thyroid carcinoma (THCA). Conversely a statistically significant decrease in ACCN2 expression was observed in bladder urothelial carcinoma (BLCA), kidney renal clear cell carcinoma (KIRC) and kidney renal papillary cell carcinoma (KIRP) compared to non-tumor tissue. * p < 0.05, ** p < 0.005, *** p < 0.001)
ASICs in brain cancer – increased migration vs. increased cell death
Already > 20 years ago, the presence of a small, constitutive, amiloride-sensitive cation current was reported in different glioblastoma multiforme (GBM) cell lines as well as in primary GBM cultures and in freshly isolated medulloblastoma (MB) cells. Moreover, while ASIC1a was highly expressed in these cells, ASIC2 was often downregulated [7, 10]. However, the evidence that this constitutive current was related to ASICs was inconclusive, and rested mainly on its sensitivity to amiloride (IC50 10–30 μM) [7, 10] and to PcTx1 [9]. On the other hand, the findings that the K+ fraction of this current was approximately fourfold higher than the Na+ fraction [9] and that this current was present at pH 7.4 speak against a contribution of ASICs. Nevertheless, in subsequent studies, it was reported that the inhibition or knockdown of ASIC1a or epithelial sodium channel (ENaC) subunits impaired the proliferation and migration of GBM cells in vitro [43, 66]. The authors proposed that the cells express an ASIC1a-ENaC heteromer [7, 43, 66] and that its aberrant and constitutive activity is caused by a lack of ASIC2 surface expression [80]. Surprisingly, however, this channel had not been reconstituted in heterologous expressions systems and these studies never attempted to assess the presence of a typical ASIC current by application of low pH. Therefore, the evidence for the existence of ASIC-ENaC heteromers remains inconclusive and, in the absence of robust experimental support, highly speculative. In addition, the effects of this small cation conductance on migration and proliferation were all measured at pH 7.4, implying that ASIC-ENaC heteromers would not serve as proton sensors in GBM and other brain tumour cells.
Glioma cell lines cultured in serum-containing medium, which are often used in in vitro studies such as these early studies on ASICs in GBM, are poorly representative of primary tumours [49]. In contrast, glioblastoma stem cell lines (GSCs), cultured in serum-free conditions as tumourspheres, better represent the primary GBM [47]. Functional analysis of ASICs in such GSCs revealed the presence of typical ASIC currents, mediated by ASIC1 and ASIC3; like previously reported for GBM cells, ASIC2 was downregulated in GSCs [73]. In contrast, a constitutive amiloride-sensitive Na+ current could not be detected [73]. While slight acidosis (pH 6.6) strongly increased migration of GSCs in a tumoursphere outgrowth assay in vitro, neither pharmacological inhibition or genetic knockout of ASIC1a nor overexpression of ASIC2a affected migration of GSCs in vitro [19]. The increased migration in acidic medium was rather mediated by phosphoinositide 3-kinase (PI3K) [19], a known mediator of cancer cell migration. Another study using GBM cells cultured in serum-containing medium, also reported functional expression of a typical ASIC current, but found that inhibition or knockdown of ASIC1a reduced the increase in migration at slightly acidic pH (7.0) [69]. Thus, there is controversial evidence on the role of ASICs in migration of GBM cells in vitro. However, recent studies did not confirm a role of ASIC1a in proliferation of GBM cells or GSCs [18, 69].
Which other role could ASICs play in GBM? Because sustained activation of ASIC1a induces neurodegeneration, it has been investigated if it does so in GSCs as well. Surprisingly, it was indeed found that ASIC1a activation at slight acidosis (pH 6.6) significantly reduced tumoursphere formation and induced necrotic morphology of the tumour cells. In contrast, sphere size or proliferation was not affected [18], suggesting that acidosis induces cell death at the onset of acidification, in which GSCs appear to be vulnerable to cell death induction. Whether this mechanism is relevant for the evolution of a tumour in situ is unknown, and future studies need to address this question in xenograft animal models. Furthermore, it is unclear whether this mode of cell death will be beneficial or detrimental for patients, given the ambivalent nature of necroptosis and its possible pro- and anti-tumoural effects [62].
Interestingly, a recent study, which has not yet been peer-reviewed, reported reduced tumour growth and prolonged survival of mice after transplanting the murine glioma cell line GL261 into the brain of mice with ASIC1a gene deletion [59]. Importantly, ASIC had been knocked-out in the host tissue and not in the tumour cells. ASIC expression in GL261 cells was not assessed. The authors proposed that the acidic TME increased ASIC signalling also in neighbouring neurons leading to an increased connectivity between neurons and glioma [59], which is known to drive glioma progression [77,78,79]. However, the mechanisms and signalling pathways underlying this regulatory role of ASIC1a on neuron-glioma connectivity remained unclear.
The expression of functional ASIC1a homomers has recently also been reported for DAOY cells, which are derived from the paediatric tumour MB; ASIC2 and ASIC3 were not expressed [61]. Furthermore, it was found that DAOY cells were resistant to acid-induced cell death and that they expressed low levels of RIPK3, a kinase downstream of RIPK1 in the necroptosis pathway. Strikingly, overexpressing RIPK3 in DAOY cells rendered them vulnerable to acid-induced cell death in an ASIC1a-dependent fashion [61]. Thus, it appears that sustained activation of ASIC1a can, in principle, induce regulated cell death also in MB cells.
In summary, although the expression of ASICs in malignant gliomas has been increasingly studied over the recent years, their exact functions remain unclear. While early studies suggested a role for unconventional ASIC-ENaC heteromers in pro-tumoral proliferation and migration, more recent studies could not confirm these observations. Possible anti-tumoral effects through an ASIC1a-mediated necroptosis-related cell death pathway have instead been proposed. While ASICs are also expressed in MB tissue, their expression in other types of brain tumours, such as meningiomas or ependymomas has not yet been investigated.
ASICs in breast and prostate cancer – increase of reactive oxygen species
One of the first studies to examine the role of ASICs in breast cancer found that ASIC1a is overexpressed in this epithelia-derived carcinoma [34]. It was shown that acidosis (pH 6.6) increases the levels of reactive oxygen species (ROS) in MCF-7 and LM-4142 breast cancer cell lines in an ASIC1a-dependent manner. Moreover, it was shown that LM-4142 cells express functional ASIC (amplitude: > 1 nA at pH 5), that acidosis increases [Ca2+]i in an ASIC1-dependent manner, and that Ca2+ chelation prevents the formation of ROS in these cells. It has been proposed that acidosis activates ASIC1a, leading to an influx of Ca2+, probably via Cavs. The increased [Ca2+]i would trigger ROS formation, which would then activate the protein kinases AKT and ERK1/2 and finally nuclear factor κB (NF- κB) [34] (Fig. 4). These effects increased the invasion of LM-4142 cells in an in vitro Matrigel assay and increased tumour growth and metastasis in a xenograft mouse model [34]. Although not all causal relationships had been delineated, this study proposed a clear mechanism for the role of ASIC1a in breast cancer cell lines. The critical link between ASIC activation and downstream signalling is Ca2+ signalling and ROS production. While the relationship between dysregulated Ca2+ signalling and increased ROS production is well established [38, 55], the main source of ROS are the mitochondria [27, 82], and it is surprising that the transient and local increase in [Ca2+]i in the cytosol, which is expected from ASIC1a activation (the time course of the Ca2+ signal was not reported in this study), was sufficient for robust ROS formation in breast cancer cells. While a recent study reported that ASIC1a activity increases [Ca2+] in mitochondria of mouse neurons, it also reported that ASIC1a activation decreased rather than increased ROS production [3]. Therefore, ROS formation as a consequence of ASIC1a activation and its mechanistic details need to be further explored.
Proposed signalling pathways of ASIC1a in different cancers. In glioblastoma stem cells, it appears that under certain conditions ASIC1a can induce a programmed cell death involving RIPK1. For breast cancer cells, it has been reported that ASIC1a triggers the formation of ROS and ensuing activation of AKT and NF-κB. A similar pathway may be active in prostate cancer cells. For pancreatic cancer, it has been proposed that ASIC1a triggers the activation of RhoA and the ensuing EMT. For details, see text
In a follow-up study, it was shown that 22Rv1 prostate cancer cells express ASIC1a, and that while AKT is constitutively active in these cells, acidosis (pH 6.6) generates ROS and activates ERK1 in an ASIC1a-dependent manner [12]. ERK would then phosphorylate IκB kinase (IKK), leading to the degradation of IκB and activation of NF-κB. These effects increased invasion in an in vitro Transwell assay [12]. Again, the critical link between ASIC1a activation and ROS production requires further confirmation.
ASICs in pancreatic cancer – activation of RhoA
It was reported that PANC-1 and BxPC-3 pancreatic cancer cells express functional ASIC1a and ASIC3 [101]. The ASIC current had a small amplitude (< 100 pA at pH 6.4), however, was only 50% blocked by 100 μM amiloride, and did not have the typical transient kinetics of ASIC1a and ASIC3 currents [101]. Overall, there is only weak evidence for the expression of functional ASICs in these cell lines. Nevertheless, downregulation of ASIC1 or ASIC3 expression by siRNA in these cells completely rescued the increased invasion at acidic pH (pH 6.4) in an in vitro Transwell assay and the increased migration in an in vitro scratch assay. Similarly, amiloride completely rescued the increased migration of the cells [101]. It is surprising that while knockdown of either ASIC1a or ASIC3 should only partially reduce ASICs expression and while amiloride only partially reduced the small ASIC currents, each manipulation separately led to a complete rescue of increased invasion and migration. Moreover, it was reported that the expression of mesenchymal markers increased at pH 6.4 and that this increased expression was completely abolished by knockdown of either ASIC1 or ASIC3 by siRNA [101]. Acidic pH induced a Ca2+ signal that lasted for 60 – 350 s, depending on the pH and the cell line. However, this Ca2+ signal was only modestly reduced by knockdown of either ASIC1 or ASIC3 or by amiloride [101]. Moreover, it was found that acidosis increases the activity of the small G protein RhoA, which is involved in cytoskeletal regulation. Although this increased RhoA activity was abolished by knockdown of either ASIC1 or ASIC3, by amiloride, or by chelating Ca2+ [101], the mechanistic details linking ASIC activation, Ca2+ signalling, and RhoA activation remain unclear (Fig. 4). Knockdown of RhoA reduced the increased invasion and migration, and reversed the increased expression of mesenchymal markers [101]. In summary, although this study provided a first indication that ASIC1a and ASIC3 might be involved in epithelial-mesenchymal transition (EMT) in pancreatic cancer cells, many questions regarding the specific role of ASICs remain open.
ASICs in colorectal cancer – activation of NFAT
It has been reported that ASIC2a is highly expressed in some colorectal cancer cell lines, but weakly expressed in others [100]. Overexpression of ASIC2a in weakly expressing cells increased invasion in a Transwell assay at pH 6.5 (but not at pH 7.4), whereas downregulating it in highly expressing cells decreased invasion at pH 6.5. Moreover, overexpression of ASIC2a increased and downregulation decreased proliferation and colony formation, respectively, surprisingly also at neutral pH 7.4 [100]. Moreover, it was shown that nuclear translocation of nuclear factor of activated T cells (NFAT1) increased after overexpression of ASIC2a and decreased after its downregulation, and it was proposed that an increase in [Ca2+]i by activation of ASIC2a triggered nuclear translocation of NFAT1 [100]. ASIC currents were not recorded in this study, and it is unclear why the expression of the ASIC2a subunit, which has low H+-sensitivity, should increase [Ca2+]i. In addition, Ca2+ signals were not measured. Therefore, the mechanistic link between ASIC2a expression and NFAT activation remains to be elucidated.
ASICs in lung cancer – increased proliferation and migration
It was reported that ASIC1, ASIC2, and ASIC3 are expressed in A549 lung cancer cells [91]. Typical transient ASIC currents were observed in these cells (~ 1 nA at pH 6.0) and were partially sensitive to PcTx1. Surprisingly, acidosis (pH < 7.0) not only increased migration of these cells in a scratch assay but also metabolic activity as measured via an MTT assay. Moreover, overexpression of ASIC1a increased whereas PcTx1 reduced proliferation and migration at pH 6.0 [91]. Finally, pH 6.0 induced a Ca2+ signal in these cells, which was reduced by PcTx1 [91] and it was proposed that ASIC activation induced the Ca2+ signal that then led to increased proliferation and migration.
ASICs in hepatocellular carcinoma – activation of PI3K and AKT
Two independent studies reported that ASIC1a is expressed in hepatocellular carcinoma (HCC) cell lines and that it is upregulated in drug resistant cells; inhibition of ASICs by amiloride or knockdown of ASIC1a by shRNA enhanced chemosensitivity whereas overexpression of ASIC1a enhanced chemoresistance of HCC cells [97, 98]. Moreover, it was reported that pH 6.5 induced a Ca2+ signal in HCC cells, which was inhibited by amiloride but not the Cav-inhibitor verapamil [98]. It was shown that ASIC1a knockdown reduced p-AKT in HCC cells and it was proposed that direct Ca2+ influx via ASIC1a activated PI3K [98]. It remained unclear, however, how knockdown of ASIC1a in cells kept at neutral pH can reduce phosphorylation of AKT [98]. Moreover, it was reported that ASIC1a promoted EMT in HCC cells, and it was proposed that EMT was mediated via the AKT/GSK3β/Snail pathway [97]. Increasing the complexity of the proposed effects, a different study reported an inverse cascade where in hepatic stellate cells, the PI3K/AKT pathway increases ASIC1a surface expression enhancing endoplasmic reticulum stress [102]. In another study, it was found that increased migration of liver cancer cells in an in vitro scratch assay and increased invasion in a transwell assay were reduced by PcTx1 or by knockdown of ASIC1a via shRNA. Overexpression of ASIC1a had the inverse effect [97]. It was proposed that ASIC1a activated the PI3K/AKT/MTOR pathway to induce the secretion of matrix metalloproteinases (MMPs) [97]. In this study, no ASIC currents or Ca2+ signals elicited by low pH were recorded and the mechanistic details of how ASIC activation might trigger the downstream effects were not elucidated.
ASICs in gastric carcinoma
Using the gastric carcinoma cell line AGS, it was found that ASIC1a knock-down by shRNA reduced migration in a scratch assay at pH 7.4 and pH 6.5 and reduced tumour weight in a xenograft mouse model [15]. No mechanism for the observed effects was reported.
Summary
In several cell models of diverse types of cancer, a role for ASIC1a in proliferation, migration and invasion has been proposed. Overall, a plethora of signalling cascades have been reported to be activated downstream of ASIC activation in different carcinomas – increase in ROS, activation of RhoA, NFAT, PI3K, or AKT—all due to an increase in [Ca2+]i triggered by ASICs. The diversity of these effects all induced by supposedly transient Ca2+ signals shed some doubt on their specificity. Moreover, in vitro results have been obtained using established cell lines that grow as monolayers in serum-containing medium. These cells do not well represent the tumour heterogeneity and biology. Therefore, we need to test the role of ASICs also in more advanced models, such as tumourspheres and, if feasible, organoids. As an exception, recent studies using GSC tumourspheres did not find any role for ASICs in proliferation and migration [18, 19], but rather an induction of a regulated cell death pathway [18].
Guidelines to address the challenges and conceptual problems of ASIC research in cancer
To strengthen the mechanistic link between ASIC activation and downstream signalling in cancer, we propose the following guidelines for studies addressing the mechanistic role of ASICs in cancer:
-
1)
When the expression of an ASIC gene is assessed by qPCR, it should be normalized to an appropriate housekeeping gene and these values should be reported to allow the assessment of the absolute expression level. It is insufficient to only report ASIC expression at different pH normalized to expression at pH 7.4.
-
2)
Western blots and immunocytochemistry are, in principle, appropriate methods to test for the presence of an ASIC, but it should be kept in mind that antibodies are often unspecific and, therefore, whenever possible, good controls (for example knock-out cells) should be employed.
-
3)
Functional ASICs should be electrophysiologically characterized in the target tissue or cell.
-
4)
The ASIC subtypes involved in functional ASICs should be identified by their electrophysiological characteristics and by pharmacological tools.
-
5)
To proof the involvement of ASICs in a specific cellular event (for example, proliferation or migration), more than one inhibitor should be used. In addition, ideally, the outcome of disruption of the respective ASIC gene, for example using CRISPR-Cas9, should be assessed. Successful knockout or knockdown (via shRNA) is ideally verified by electrophysiology.
-
6)
The functional role of ASICs for a cancer/cell should be addressed at acidic pH, in the range between 7.3 and 6.5 and the pH of the cell culture should be controlled following evidence-based guidelines [57].
-
7)
If increases of [Ca2+]i are proposed to be involved in the function of an ASIC, they should be directly demonstrated using Ca2+ imaging and the triggering function of ASIC1a (or other ASICs) for Ca2+ signals should be demonstrated with appropriate inhibitors of ASIC1a.
-
8)
The presence of active Cavs in cancer cells should be tested by application of a high K+-solution to depolarize the cell and the contribution of Cavs to Ca2+ signals should be tested with appropriate inhibitors.
-
9)
Cell models should be used which are as representative of the parental tumour as possible. In addition, the expression of a respective ASIC subunit in a certain tumour type should be ascertained by analysis of online databases containing expression data from tumour tissue.
-
10)
If Kaplan–Meier analysis is performed, it should be clearly indicated what “High expression” and “Low expression” means. Ideally, the two groups are separated at the median expression level.
Conclusions
In summary, while an increasing number of studies report a role for ASICs in proliferation, migration, death, and even drug resistance of cancer cells, the signalling pathways that have been proposed to be involved in these effects are surprisingly diverse and heterogenous and the evidence linking ASIC activation to downstream signalling events is often surprising and not sufficiently robust. Therefore, our understanding of ASIC for downstream signalling cascades needs to be increased and the mechanistic role of ASICs in tumour cell function needs to be further established before we can evaluate the significance of ASICs as a target in cancer cells.
Data availability
No datasets were generated or analysed during the current study.
References
Aguilar-Camacho JM, Foreman K, Jaimes-Becerra A, Aharoni R, Gründer S, Moran Y (2023) Functional analysis in a model sea anemone reveals phylogenetic complexity and a role in cnidocyte discharge of DEG/ENaC ion channels. Commun Biol 6:17. https://doi.org/10.1038/s42003-022-04399-1
Askwith CC, Wemmie JA, Price MP, Rokhlina T, Welsh MJ (2004) Acid-sensing ion channel 2 (ASIC2) modulates ASIC1 H+-activated currents in hippocampal neurons. J Biol Chem 279:18296–18305
Azoulay IS, Qi X, Rozenfeld M, Liu F, Hu Q, Ben Kasus Nissim T, Stavsky A, Zhu MX, Xu TL, Sekler I (2022) ASIC1a senses lactate uptake to regulate metabolism in neurons. Redox Biol 51:102253. https://doi.org/10.1016/j.redox.2022.102253
Babini E, Paukert M, Geisler HS, Gründer S (2002) Alternative splicing and interaction with di- and polyvalent cations control the dynamic range of acid-sensing ion channel 1 (ASIC1). J Biol Chem 277:41597–41603
Baron A, Voilley N, Lazdunski M, Lingueglia E (2008) Acid sensing ion channels in dorsal spinal cord neurons. J Neurosci 28:1498–1508
Bässler EL, Ngo-Anh TJ, Geisler HS, Ruppersberg JP, Gründer S (2001) Molecular and functional characterization of acid-sensing ion channel (ASIC) 1b. J Biol Chem 276:33782–33787
Berdiev BK, Xia J, McLean LA, Markert JM, Gillespie GY, Mapstone TB, Naren AP, Jovov B, Bubien JK, Ji HL, Fuller CM, Kirk KL, Benos DJ (2003) Acid-sensing ion channels in malignant gliomas. J Biol Chem 278:15023–15034. https://doi.org/10.1074/jbc.M300991200
Bohlen CJ, Chesler AT, Sharif-Naeini R, Medzihradszky KF, Zhou S, King D, Sanchez EE, Burlingame AL, Basbaum AI, Julius D (2011) A heteromeric Texas coral snake toxin targets acid-sensing ion channels to produce pain. Nature 479:410–414. https://doi.org/10.1038/nature10607
Bubien JK, Ji HL, Gillespie GY, Fuller CM, Markert JM, Mapstone TB, Benos DJ (2004) Cation selectivity and inhibition of malignant glioma Na+ channels by psalmotoxin 1. Am J Physiol Cell Physiol 287:C1282-1291. https://doi.org/10.1152/ajpcell.00077.2004
Bubien JK, Keeton DA, Fuller CM, Gillespie GY, Reddy AT, Mapstone TB, Benos DJ (1999) Malignant human gliomas express an amiloride-sensitive Na+ conductance. Am J Physiol 276:C1405-1410. https://doi.org/10.1152/ajpcell.1999.276.6.C1405
Chassagnon IR, McCarthy CA, Chin YK, Pineda SS, Keramidas A, Mobli M, Pham V, De Silva TM, Lynch JW, Widdop RE, Rash LD, King GF (2017) Potent neuroprotection after stroke afforded by a double-knot spider-venom peptide that inhibits acid-sensing ion channel 1a. Proc Natl Acad Sci U S A 114:3750–3755. https://doi.org/10.1073/pnas.1614728114
Chen B, Liu J, Ho TT, Ding X, Mo YY (2016) ERK-mediated NF-kappaB activation through ASIC1 in response to acidosis. Oncogenesis 5:e279. https://doi.org/10.1038/oncsis.2016.81
Chen CC, England S, Akopian AN, Wood JN (1998) A sensory neuron-specific, proton-gated ion channel. Proc Natl Acad Sci U S A 95:10240–10245
Chen X, Kalbacher H, Gründer S (2005) The tarantula toxin psalmotoxin 1 inhibits acid-sensing ion channel (ASIC) 1a by increasing its apparent H+ affinity. J Gen Physiol 126:71–79
Chen X, Sun X, Wang Z, Zhou X, Xu L, Li F, Zhang X, Pan J, Qi L, Qian H, Mao Z (2018) Involvement of acid-sensing ion channel 1a in gastric carcinoma cell migration and invasion. Acta Biochim Biophys Sin (Shanghai) 50:440–446. https://doi.org/10.1093/abbs/gmy026
Chen Y, Cann MJ, Litvin TN, Iourgenko V, Sinclair ML, Levin LR, Buck J (2000) Soluble adenylyl cyclase as an evolutionarily conserved bicarbonate sensor. Science 289:625–628. https://doi.org/10.1126/science.289.5479.625
Cho YS, Challa S, Moquin D, Genga R, Ray TD, Guildford M, Chan FK (2009) Phosphorylation-driven assembly of the RIP1-RIP3 complex regulates programmed necrosis and virus-induced inflammation. Cell 137:1112–1123. https://doi.org/10.1016/j.cell.2009.05.037
Clusmann J, Franco KC, Suarez DAC, Katona I, Minguez MG, Boersch N, Pissas KP, Vanek J, Tian Y, Gründer S (2022) Acidosis induces RIPK1-dependent death of glioblastoma stem cells via acid-sensing ion channel 1a. Cell Death Dis 13:702. https://doi.org/10.1038/s41419-022-05139-3
Cortes Franco KD, Brakmann IC, Feoktistova M, Panayotova-Dimitrova D, Gründer S, Tian Y (2023) Aggressive migration in acidic pH of a glioblastoma cancer stem cell line in vitro is independent of ASIC and K(Ca)3.1 ion channels, but involves phosphoinositide 3-kinase. Pflugers Arch 475:405–416. https://doi.org/10.1007/s00424-022-02781-w
Couch T, Berger KD, Kneisley DL, McCullock TW, Kammermeier P, Maclean DM (2021) Topography and motion of acid-sensing ion channel intracellular domains. Elife 10. https://doi.org/10.7554/eLife.68955
Cullinan MM, Klipp RC, Camenisch A, Bankston JR (2023) Dynamic landscape of the intracellular termini of acid-sensing ion channel 1a. eLife 12:RP90755. https://doi.org/10.7554/eLife.90755
Delaunay A, Gasull X, Salinas M, Noel J, Friend V, Lingueglia E, Deval E (2012) Human ASIC3 channel dynamically adapts its activity to sense the extracellular pH in both acidic and alkaline directions. Proc Natl Acad Sci U S A 109:13124–13129. https://doi.org/10.1073/pnas.1120350109
Du J, Reznikov LR, Price MP, Zha XM, Lu Y, Moninger TO, Wemmie JA, Welsh MJ (2014) Protons are a neurotransmitter that regulates synaptic plasticity in the lateral amygdala. Proc Natl Acad Sci U S A 111:8961–8966. https://doi.org/10.1073/pnas.1407018111
Elkhatib W, Yanez-Guerra LA, Mayorova TD, Currie MA, Singh A, Perera M, Gauberg J, Senatore A (2023) Function and phylogeny support the independent evolution of an ASIC-like Deg/ENaC channel in the placozoa. Commun Biol 6:951. https://doi.org/10.1038/s42003-023-05312-0
Escoubas P, De Weille JR, Lecoq A, Diochot S, Waldmann R, Champigny G, Moinier D, Menez A, Lazdunski M (2000) Isolation of a tarantula toxin specific for a class of proton-gated Na+ channels. J Biol Chem 275:25116–25121
Estrella V, Chen T, Lloyd M, Wojtkowiak J, Cornnell HH, Ibrahim-Hashim A, Bailey K, Balagurunathan Y, Rothberg JM, Sloane BF, Johnson J, Gatenby RA, Gillies RJ (2013) Acidity generated by the tumor microenvironment drives local invasion. Cancer Res 73:1524–1535. https://doi.org/10.1158/0008-5472.CAN-12-2796
Fontana F, Limonta P (2021) The multifaceted roles of mitochondria at the crossroads of cell life and death in cancer. Free Radic Biol Med 176:203–221. https://doi.org/10.1016/j.freeradbiomed.2021.09.024
Frelin C, Barbry P, Vigne P, Chassande O, Cragoe EJ Jr, Lazdunski M (1988) Amiloride and its analogs as tools to inhibit Na+ transport via the Na+ channel, the Na+/H+ antiport and the Na+/Ca2+ exchanger. Biochimie 70:1285–1290. https://doi.org/10.1016/0300-9084(88)90196-4
Gatenby RA, Gillies RJ (2004) Why do cancers have high aerobic glycolysis? Nat Rev Cancer 4:891–899. https://doi.org/10.1038/nrc1478
Gerweck LE, Seetharaman K (1996) Cellular pH gradient in tumor versus normal tissue: potential exploitation for the treatment of cancer. Cancer Res 56:1194–1198
Gonzalez-Inchauspe C, Urbano FJ, Di Guilmi MN, Uchitel OD (2017) Acid-sensing ion channels activated by evoked released protons modulate synaptic transmission at the mouse calyx of held synapse. J Neurosci 37:2589–2599. https://doi.org/10.1523/JNEUROSCI.2566-16.2017
Griffiths JR (1991) Are cancer cells acidic? Br J Cancer 64:425–427. https://doi.org/10.1038/bjc.1991.326
Gründer S, Pusch M (2015) Biophysical properties of acid-sensing ion channels (ASICs). Neuropharmacology 94:9–18. https://doi.org/10.1016/j.neuropharm.2014.12.016
Gupta SC, Singh R, Asters M, Liu J, Zhang X, Pabbidi MR, Watabe K, Mo YY (2016) Regulation of breast tumorigenesis through acid sensors. Oncogene 35:4102–4111. https://doi.org/10.1038/onc.2015.477
Harris B, Saleem S, Cook N, Searle E (2022) Targeting hypoxia in solid and haematological malignancies. J Exp Clin Cancer Res 41:318. https://doi.org/10.1186/s13046-022-02522-y
Hattori T, Chen J, Harding AM, Price MP, Lu Y, Abboud FM, Benson CJ (2009) ASIC2a and ASIC3 heteromultimerize to form pH-sensitive channels in mouse cardiac dorsal root ganglia neurons. Circ Res 105:279–286 (CIRCRESAHA.109.202036). https://doi.org/10.1161/CIRCRESAHA.109.202036
Helmlinger G, Yuan F, Dellian M, Jain RK (1997) Interstitial pH and pO2 gradients in solid tumors in vivo: high-resolution measurements reveal a lack of correlation. Nat Med 3:177–182. https://doi.org/10.1038/nm0297-177
Hempel N, Trebak M (2017) Crosstalk between calcium and reactive oxygen species signaling in cancer. Cell Calcium 63:70–96. https://doi.org/10.1016/j.ceca.2017.01.007
Hoagland EN, Sherwood TW, Lee KG, Walker CJ, Askwith CC (2010) Identification of a calcium permeable human acid-sensing ion channel 1 transcript variant. J Biol Chem 285:41852–41862. https://doi.org/10.1074/jbc.M110.171330
Ibrahim Hashim A, Cornnell HH, Coelho Ribeiro Mde L, Abrahams D, Cunningham J, Lloyd M, Martinez GV, Gatenby RA, Gillies RJ (2011) Reduction of metastasis using a non-volatile buffer. Clin Exp Metastasis 28:841–849. https://doi.org/10.1007/s10585-011-9415-7
Imenez Silva PH, Camara NO, Wagner CA (2022) Role of proton-activated G protein-coupled receptors in pathophysiology. Am J Physiol Cell Physiol 323:C400–C414. https://doi.org/10.1152/ajpcell.00114.2022
Joeres N, Augustinowski K, Neuhof A, Assmann M, Gründer S (2016) Functional and pharmacological characterization of two different ASIC1a/2a heteromers reveals their sensitivity to the spider toxin PcTx1. Sci Rep 6:27647. https://doi.org/10.1038/srep27647
Kapoor N, Bartoszewski R, Qadri YJ, Bebok Z, Bubien JK, Fuller CM, Benos DJ (2009) Knockdown of ASIC1 and epithelial sodium channel subunits inhibits glioblastoma whole cell current and cell migration. J Biol Chem 284:24526–24541. https://doi.org/10.1074/jbc.M109.037390
Kellenberger S, Schild L (2002) Epithelial sodium channel/degenerin family of ion channels: a variety of functions for a shared structure. Physiol Rev 82:735–767
Kierans SJ, Taylor CT (2021) Regulation of glycolysis by the hypoxia-inducible factor (HIF): implications for cellular physiology. J Physiol 599:23–37. https://doi.org/10.1113/JP280572
Kreple CJ, Lu Y, Taugher RJ, Schwager-Gutman AL, Du J, Stump M, Wang Y, Ghobbeh A, Fan R, Cosme CV, Sowers LP, Welsh MJ, Radley JJ, LaLumiere RT, Wemmie JA (2014) Acid-sensing ion channels contribute to synaptic transmission and inhibit cocaine-evoked plasticity. Nat Neurosci 17:1083–1091. https://doi.org/10.1038/nn.3750
Lee J, Kotliarova S, Kotliarov Y, Li A, Su Q, Donin NM, Pastorino S, Purow BW, Christopher N, Zhang W, Park JK, Fine HA (2006) Tumor stem cells derived from glioblastomas cultured in bFGF and EGF more closely mirror the phenotype and genotype of primary tumors than do serum-cultured cell lines. Cancer Cell 9:391–403. https://doi.org/10.1016/j.ccr.2006.03.030
Lee S, Mele M, Vahl P, Christiansen PM, Jensen VE, Boedtkjer E (2015) Na+, HCO3- -cotransport is functionally upregulated during human breast carcinogenesis and required for the inverted pH gradient across the plasma membrane. Pflugers Arch 467:367–377. https://doi.org/10.1007/s00424-014-1524-0
Li A, Walling J, Kotliarov Y, Center A, Steed ME, Ahn SJ, Rosenblum M, Mikkelsen T, Zenklusen JC, Fine HA (2008) Genomic changes and gene expression profiles reveal that established glioma cell lines are poorly representative of primary human gliomas. Mol Cancer Res 6:21–30. https://doi.org/10.1158/1541-7786.MCR-07-0280
Li J, McQuade T, Siemer AB, Napetschnig J, Moriwaki K, Hsiao YS, Damko E, Moquin D, Walz T, McDermott A, Chan FK, Wu H (2012) The RIP1/RIP3 necrosome forms a functional amyloid signaling complex required for programmed necrosis. Cell 150:339–350. https://doi.org/10.1016/j.cell.2012.06.019
Li M, Inoue K, Branigan D, Kratzer E, Hansen JC, Chen JW, Simon RP, Xiong ZG (2010) Acid-sensing ion channels in acidosis-induced injury of human brain neurons. J Cereb Blood Flow Metab 30:1247–1260 (jcbfm201030). https://doi.org/10.1038/jcbfm.2010.30
Li MH, Leng TD, Feng XC, Yang T, Simon RP, Xiong ZG (2016) Modulation of acid-sensing Ion Channel 1a by intracellular pH and its role in ischemic stroke. J Biol Chem 291:18370–18383. https://doi.org/10.1074/jbc.M115.713636
Li S, Sato S, Yang X, Preisig PA, Alpern RJ (2004) Pyk2 activation is integral to acid stimulation of sodium/hydrogen exchanger 3. J Clin Invest 114:1782–1789. https://doi.org/10.1172/JCI18046
Li T, Fan J, Wang B, Traugh N, Chen Q, Liu JS, Li B, Liu XS (2017) TIMER: a web server for comprehensive analysis of tumor-infiltrating immune cells. Cancer Res 77:e108–e110. https://doi.org/10.1158/0008-5472.CAN-17-0307
Madreiter-Sokolowski CT, Thomas C, Ristow M (2020) Interrelation between ROS and Ca(2+) in aging and age-related diseases. Redox Biol 36:101678. https://doi.org/10.1016/j.redox.2020.101678
Mauffrey P, Tchitchek N, Barroca V, Bemelmans AP, Firlej V, Allory Y, Romeo PH, Magnon C (2019) Progenitors from the central nervous system drive neurogenesis in cancer. Nature 569:672–678. https://doi.org/10.1038/s41586-019-1219-y
Michl J, Park KC, Swietach P (2019) Evidence-based guidelines for controlling pH in mammalian live-cell culture systems. Commun Biol 2:144. https://doi.org/10.1038/s42003-019-0393-7
Neuhof A, Tian Y, Reska A, Falkenburger BH, Gründer S (2021) Large acid-evoked currents, mediated by ASIC1a, accompany differentiation in human dopaminergic neurons. Front Cell Neurosci 15:668008. https://doi.org/10.3389/fncel.2021.668008
Park G, Jin Z, Ge Q, Pan Y, Du J (2023) Neuronal acid-sensing ion channel 1a regulates neuron-to-glioma synapses. bioRxiv. https://doi.org/10.1101/2023.08.31.555794
Persi E, Duran-Frigola M, Damaghi M, Roush WR, Aloy P, Cleveland JL, Gillies RJ, Ruppin E (2018) Systems analysis of intracellular pH vulnerabilities for cancer therapy. Nat Commun 9:2997. https://doi.org/10.1038/s41467-018-05261-x
Pissas KP, Schilling M, Tian Y, Gründer S (2023) Functional characterization of acid-sensing ion channels in the cerebellum-originating medulloblastoma cell line DAOY and in cerebellar granule neurons. Pflugers Arch. https://doi.org/10.1007/s00424-023-02839-3
Qin X, Ma D, Tan YX, Wang HY, Cai Z (2019) The role of necroptosis in cancer: a double-edged sword? Biochim Biophys Acta Rev Cancer 1871:259–266. https://doi.org/10.1016/j.bbcan.2019.01.006
Restaino AC, Walz A, Vermeer SJ, Barr J, Kovacs A, Fettig RR, Vermeer DW, Reavis H, Williamson CS, Lucido CT, Eichwald T, Omran DK, Jung E, Schwartz LE, Bell M, Muirhead DM, Hooper JE, Spanos WC, Drapkin R, Talbot S, Vermeer PD (2023) Functional neuronal circuits promote disease progression in cancer. Sci Adv 9:eade4443. https://doi.org/10.1126/sciadv.ade4443
Rolver MG, Holland LKK, Ponniah M, Prasad NS, Yao J, Schnipper J, Kramer S, Elingaard-Larsen L, Pedraz-Cuesta E, Liu B, Pardo LA, Maeda K, Sandelin A, Pedersen SF (2023) Chronic acidosis rewires cancer cell metabolism through PPARalpha signaling. Int J Cancer 152:1668–1684. https://doi.org/10.1002/ijc.34404
Roma-Rodrigues C, Mendes R, Baptista PV, Fernandes AR (2019) Targeting tumor microenvironment for cancer therapy. Int J Mol Sci 20. https://doi.org/10.3390/ijms20040840
Rooj AK, McNicholas CM, Bartoszewski R, Bebok Z, Benos DJ, Fuller CM (2012) Glioma-specific cation conductance regulates migration and cell cycle progression. J Biol Chem 287:4053–4065. https://doi.org/10.1074/jbc.M111.311688
Samways DS, Harkins AB, Egan TM (2009) Native and recombinant ASIC1a receptors conduct negligible Ca2+ entry. Cell Calcium 45:319–325
Schaefer L, Sakai H, Mattei M, Lazdunski M, Lingueglia E (2000) Molecular cloning, functional expression and chromosomal localization of an amiloride-sensitive Na(+) channel from human small intestine. FEBS Lett 471:205–210
Sheng Y, Wu B, Leng T, Zhu L, Xiong Z (2021) Acid-sensing ion channel 1 (ASIC1) mediates weak acid-induced migration of human malignant glioma cells. Am J Cancer Res 11:997–1008
Stock C, Ludwig FT, Schwab A (2012) Is the multifunctional Na(+)/H(+) exchanger isoform 1 a potential therapeutic target in cancer? Curr Med Chem 19:647–660. https://doi.org/10.2174/092986712798992101
Sutherland SP, Benson CJ, Adelman JP, McCleskey EW (2001) Acid-sensing ion channel 3 matches the acid-gated current in cardiac ischemia-sensing neurons. Proc Natl Acad Sci U S A 98:711–716
Swietach P, Boedtkjer E, Pedersen SF (2023) How protons pave the way to aggressive cancers. Nat Rev Cancer. https://doi.org/10.1038/s41568-023-00628-9
Tian Y, Bresenitz P, Reska A, El Moussaoui L, Beier CP, Gründer S (2017) Glioblastoma cancer stem cell lines express functional acid sensing ion channels ASIC1a and ASIC3. Sci Rep 7:13674. https://doi.org/10.1038/s41598-017-13666-9
Vaithia A, Vullo S, Peng Z, Alijevic O, Kellenberger S (2019) Accelerated current decay kinetics of a rare human acid-sensing ion channel 1a variant that is used in many studies as wild type. Front Mol Neurosci 12. https://doi.org/10.3389/fnmol.2019.00133
Vaupel P, Hockel M, Mayer A (2007) Detection and characterization of tumor hypoxia using pO2 histography. Antioxid Redox Signal 9:1221–1235. https://doi.org/10.1089/ars.2007.1628
Vaupel P, Kallinowski F, Okunieff P (1989) Blood flow, oxygen and nutrient supply, and metabolic microenvironment of human tumors: a review. Cancer Res 49:6449–6465
Venkataramani V, Tanev DI, Strahle C, Studier-Fischer A, Fankhauser L, Kessler T, Korber C, Kardorff M, Ratliff M, Xie R, Horstmann H, Messer M, Paik SP, Knabbe J, Sahm F, Kurz FT, Acikgoz AA, Herrmannsdorfer F, Agarwal A, Bergles DE, Chalmers A, Miletic H, Turcan S, Mawrin C, Hanggi D, Liu HK, Wick W, Winkler F, Kuner T (2019) Glutamatergic synaptic input to glioma cells drives brain tumour progression. Nature 573:532–538. https://doi.org/10.1038/s41586-019-1564-x
Venkataramani V, Yang Y, Schubert MC, Reyhan E, Tetzlaff SK, Wissmann N, Botz M, Soyka SJ, Beretta CA, Pramatarov RL, Fankhauser L, Garofano L, Freudenberg A, Wagner J, Tanev DI, Ratliff M, Xie R, Kessler T, Hoffmann DC, Hai L, Dorflinger Y, Hoppe S, Yabo YA, Golebiewska A, Niclou SP, Sahm F, Lasorella A, Slowik M, Doring L, Iavarone A, Wick W, Kuner T, Winkler F (2022) Glioblastoma hijacks neuronal mechanisms for brain invasion. Cell 185(2899–2917):e2831. https://doi.org/10.1016/j.cell.2022.06.054
Venkatesh HS, Morishita W, Geraghty AC, Silverbush D, Gillespie SM, Arzt M, Tam LT, Espenel C, Ponnuswami A, Ni L, Woo PJ, Taylor KR, Agarwal A, Regev A, Brang D, Vogel H, Hervey-Jumper S, Bergles DE, Suva ML, Malenka RC, Monje M (2019) Electrical and synaptic integration of glioma into neural circuits. Nature 573:539–545. https://doi.org/10.1038/s41586-019-1563-y
Vila-Carriles WH, Kovacs GG, Jovov B, Zhou ZH, Pahwa AK, Colby G, Esimai O, Gillespie GY, Mapstone TB, Markert JM, Fuller CM, Bubien JK, Benos DJ (2006) Surface expression of ASIC2 inhibits the amiloride-sensitive current and migration of glioma cells. J Biol Chem 281:19220–19232. https://doi.org/10.1074/jbc.M603100200
Waldmann R, Champigny G, Bassilana F, Heurteaux C, Lazdunski M (1997) A proton-gated cation channel involved in acid-sensing. Nature 386:173–177
Wallace DC (2012) Mitochondria and cancer. Nat Rev Cancer 12:685–698. https://doi.org/10.1038/nrc3365
Wang JJ, Liu F, Yang F, Wang YZ, Qi X, Li Y, Hu Q, Zhu MX, Xu TL (2020) Disruption of auto-inhibition underlies conformational signaling of ASIC1a to induce neuronal necroptosis. Nat Commun 11:475. https://doi.org/10.1038/s41467-019-13873-0
Wang YZ, Wang JJ, Huang Y, Liu F, Zeng WZ, Li Y, Xiong ZG, Zhu MX, Xu TL (2015) Tissue acidosis induces neuronal necroptosis via ASIC1a channel independent of its ionic conduction. Elife 4. https://doi.org/10.7554/eLife.05682
Wemmie JA, Chen J, Askwith CC, Hruska-Hageman AM, Price MP, Nolan BC, Yoder PG, Lamani E, Hoshi T, Freeman JH Jr, Welsh MJ (2002) The acid-activated ion channel ASIC contributes to synaptic plasticity, learning, and memory. Neuron 34:463–477
Wiemuth D, Assmann M, Gründer S (2014) The bile acid-sensitive ion channel (BASIC), the ignored cousin of ASICs and ENaC. Channels 8(1):29–34. https://doi.org/10.4161/chan.27493
Wiemuth D, Gründer S (2010) A single amino acid tunes Ca2+ inhibition of brain liver intestine Na+ channel (BLINaC). J Biol Chem 285:30404–30410 (M110.153064). https://doi.org/10.1074/jbc.M110.153064
Wiemuth D, Sahin H, Falkenburger BH, Lefevre CM, Wasmuth HE, Gründer S (2012) BASIC–a bile acid-sensitive ion channel highly expressed in bile ducts. Faseb J 26:4122–4130 (fj.12-207043). https://doi.org/10.1096/fj.12-207043
Winkler F, Venkatesh HS, Amit M, Batchelor T, Demir IE, Deneen B, Gutmann DH, Hervey-Jumper S, Kuner T, Mabbott D, Platten M, Rolls A, Sloan EK, Wang TC, Wick W, Venkataramani V, Monje M (2023) Cancer neuroscience: state of the field, emerging directions. Cell 186:1689–1707. https://doi.org/10.1016/j.cell.2023.02.002
Wu LJ, Duan B, Mei YD, Gao J, Chen JG, Zhuo M, Xu L, Wu M, Xu TL (2004) Characterization of acid-sensing ion channels in dorsal horn neurons of rat spinal cord. J Biol Chem 279:43716–43724
Wu Y, Gao B, Xiong QJ, Wang YC, Huang DK, Wu WN (2017) Acid-sensing ion channels contribute to the effect of extracellular acidosis on proliferation and migration of A549 cells. Tumour Biol 39:1010428317705750. https://doi.org/10.1177/1010428317705750
Xiong ZG, Zhu XM, Chu XP, Minami M, Hey J, Wei WL, MacDonald JF, Wemmie JA, Price MP, Welsh MJ, Simon RP (2004) Neuroprotection in ischemia: blocking calcium-permeable acid-sensing ion channels. Cell 118:687–698
Yagi J, Wenk HN, Naves LA, McCleskey EW (2006) Sustained currents through ASIC3 ion channels at the modest pH changes that occur during myocardial ischemia. Circ Res 99:501–509
Yang M, Brackenbury WJ (2013) Membrane potential and cancer progression. Front Physiol 4:185. https://doi.org/10.3389/fphys.2013.00185
Zha XM, Costa V, Harding AM, Reznikov L, Benson CJ, Welsh MJ (2009) ASIC2 subunits target acid-sensing ion channels to the synapse via an association with PSD-95. J Neurosci 29:8438–8446
Zha XM, Wemmie JA, Green SH, Welsh MJ (2006) Acid-sensing ion channel 1a is a postsynaptic proton receptor that affects the density of dendritic spines. Proc Natl Acad Sci U S A 103:16556–16561
Zhang Y, Cao N, Gao J, Liang J, Liang Y, Xie Y, Zhou S, Tang X (2022) ASIC1a stimulates the resistance of human hepatocellular carcinoma by promoting EMT via the AKT/GSK3beta/Snail pathway driven by TGFbeta/Smad signals. J Cell Mol Med 26:2777–2792. https://doi.org/10.1111/jcmm.17288
Zhang Y, Zhang T, Wu C, Xia Q, Xu D (2017) ASIC1a mediates the drug resistance of human hepatocellular carcinoma via the Ca(2+)/PI3-kinase/AKT signaling pathway. Lab Invest 97:53–69. https://doi.org/10.1038/labinvest.2016.127
Zhou Y, Skelton LA, Xu L, Chandler MP, Berthiaume JM, Boron WF (2016) Role of receptor protein tyrosine phosphatase gamma in sensing extracellular CO2 and HCO3. J Am Soc Nephrol 27:2616–2621. https://doi.org/10.1681/ASN.2015040439
Zhou ZH, Song JW, Li W, Liu X, Cao L, Wan LM, Tan YX, Ji SP, Liang YM, Gong F (2017) The acid-sensing ion channel, ASIC2, promotes invasion and metastasis of colorectal cancer under acidosis by activating the calcineurin/NFAT1 axis. J Exp Clin Cancer Res 36:130. https://doi.org/10.1186/s13046-017-0599-9
Zhu S, Zhou HY, Deng SC, Deng SJ, He C, Li X, Chen JY, Jin Y, Hu ZL, Wang F, Wang CY, Zhao G (2017) ASIC1 and ASIC3 contribute to acidity-induced EMT of pancreatic cancer through activating Ca(2+)/RhoA pathway. Cell Death Dis 8:e2806. https://doi.org/10.1038/cddis.2017.189
Zuo L, Zhu Y, Hu L, Liu Y, Wang Y, Hu Y, Wang H, Pan X, Li K, Du N, Huang Y (2019) PI3-kinase/akt pathway-regulated membrane transportation of acid-sensing ion channel 1a/Calcium ion influx/endoplasmic reticulum stress activation on PDGF-induced HSC activation. J Cell Mol Med 23:3940–3950. https://doi.org/10.1111/jcmm.14275
Acknowledgements
We thank all present and former members of the Gründer lab for many helpful discussions.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
S.G., J.V., and K.-P.P. jointly wrote the original draft and S.G. reviewed and edited the original draft with important input from the other authors.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the special issue on Physiology of systemic and cellular pH regulation in Pflügers Archiv—European Journal of Physiology.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gründer, S., Vanek, J. & Pissas, KP. Acid-sensing ion channels and downstream signalling in cancer cells: is there a mechanistic link?. Pflugers Arch - Eur J Physiol 476, 659–672 (2024). https://doi.org/10.1007/s00424-023-02902-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-023-02902-z