Abstract
Motility of detrusor smooth muscle includes adrenergic relaxation and cholinergic contraction. Since the latter may be deregulated in overactive bladder (OAB) pathophysiology, anticholinergics are the standard therapy but occasionally less tolerated due to side effects such as dry mouth and constipation. β3 adrenoceptor agonists also alleviate OAB symptoms by relaxing the detrusor muscle. Their age dependence, however, is far from understood. To address this issue, we induced contractions with KCl (60 mM) and carbachol (from 10 nM to 100 μM) in the presence of the β3 adrenoceptor agonist CL316,243 (from 0.1 to 10 μM) in both human and rat muscle strips. Our results confirmed that both contractions were attenuated by β3 adrenoceptor activation in both species, but with differing age dependence. In humans, specimens from mid-life subjects showed a significantly more pronounced effect of CL316,243 in attenuating carbachol-induced contractions than those from aged subjects (Cohen’s d of maximal attenuation: 1.82 in mid-life versus 0.13 in aged) without altering EC50. Conversely, attenuation of KCl responses by CL316,243 increased during ageing (Spearman correlation coefficient = -0.584, P<0.01). In rats, both KCl- and carbachol-induced contractions were significantly more attenuated by CL316,243 in samples from adolescent as compared to aged samples. Immunohistochemistry in human detrusor sections proved β3 adrenoreceptor abundance to remain unaltered during ageing. In conclusion, our findings suggest differential age-dependent changes in human β3 adrenoceptor-dependent attenuation of detrusor contraction in terms of electromechanical versus pharmacomechanical coupling; they may help understand the differential responsiveness of OAB patients to β3 agents.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Overactive bladder (OAB) is a clinical syndrome composed of urinary urgency, increased micturition frequency, and nocturia, with or without urge incontinence [1] affecting 10–17% of the adult population in Japan, Europe, and North America [24, 33]. Standard antimuscarinic therapy allows for the intended bladder relaxation but entails some unfavourable side effects such as dry mouth and constipation owing to muscarinic receptors expressed throughout the parasympathetic nerve system [16]. Novel compounds with M3 receptor subtype specificity made no exception to this, since side effects were mostly linked to M3 receptors, too. Hence, up to 90% of OAB patients discontinue antimuscarinic medication within 12 months [8, 42, 45]. Consequently, research has set out to discover alternative routes of alleviating OAB symptoms. One major achievement consisted of detecting the β3 adrenoceptor as the preponderant sympathetic receptor subtype in the human bladder [20, 22, 27, 39, 50, 51, 56], but see [52]. Subsequently, observing that β3-adrenergic compounds relieved the overactive bladder [48] fostered the development of mirabegron as the first β3 adrenoceptor agonist approved for OAB [9].
Pharmacological treatment of OAB needs to respect the issue of receptor expression changes during normal bladder ageing, which potentially interferes with the effectiveness of drugs. In fact, some studies demonstrated age-related reduced functional responsiveness of β adrenoceptors in both rats [14, 18, 38] and humans [27]. In particular, the β2/3 adrenoceptor agonist BRL37,344 possessed less efficacy in detrusor samples from both aged rats [18] and elderly humans [27]. These data probably imply that reduced responsiveness to β3 adrenoceptor activation could be a feature of normal ageing. In line with this, Li et al. suggested there were a diminished number of binding sites in aged bladder tissue [27], but Niclauß and colleagues found the radioligand [3H]-dihydroalprenolol used in that study to be unsuitable for β3 adrenoceptors [37]. Moreover, immunostaining using β3-specific antibodies revealed sustained expression of all β adrenoceptors throughout the bladder with increasing age [28]. Beyond that, ageing mice even harboured enhanced β3 adrenoceptor levels, at least on the transcriptional level [13]. Thus, while OAB treatment with β3-adrenergic compounds has become clinical reality, its mechanism is far from understood, and the issue of age-dependent efficacy and tolerability was only recently addressed [36].
Based on the current knowledge, we still lack a comprehensive picture of β3 adrenoceptor expression and function across species and, more importantly, during ageing. Here, we aimed to study the functional responsiveness of human and rat detrusor smooth muscle strips to the highly selective β3 adrenoceptor agonist CL316,243 [4, 6, 59] towards high KCl and carbachol challenge. Whereas CL316,243 responsiveness consistently decreased in rats during ageing, human tissue revealed this compound to affect high KCl and carbachol responses differentially and age-dependently. Since β3 adrenoceptor immunoreactivity in humans was not altered during ageing, we conclude that intracellular cascades may be responsible for these effects.
Materials and methods
Preparation of human detrusor samples
Human detrusor samples were obtained from 21 patients (69 ± 9 years old, mean ± SD, range 55 to 85 years, median 70 years, 12 males, nine females). All in vitro experiments in this study using human material were approved by the local ethics committee (University Medicine of Rostock), and the informed consent to participate in this study was obtained from each patient. Since it was essential to get fresh tissue directly cooled within the operation theatre, we pursued the following approach: Once informed of an upcoming sample, we provided a beaker with 4 °C cold HEPES-buffered and calcium-reduced storage solution containing (in mM) 145 NaCl, 4.5 KCl, 1.2 NaH2PO4, 0.1 CaCl2, 1.0 MgSO4, 0.025 Na2-EDTA, and 5 HEPES (pH=7.4). This solution had been proven to show pH stability for at least 1 day. During surgery (radical cystectomy due to urothelium carcinoma), a tissue sample was excised from the macroscopically unaffected wall of the detrusor muscle (roughly 2 cm in width). This sample was immediately submerged into the storage solution of the above composition and transferred to the laboratory. Then, the detrusor samples were freed from mucosal and adipose tissue and cut into 4–8 muscle strips of some 1 cm length and 3 mm width. Subsequently, nylon threads were sutured to these specimens to fix them later in an organ bath chamber at 37 °C (Panlab ML0146/C, ADInstruments, Spechbach, Germany) filled with bicarbonate-buffered and normal-calcium solution that contained (in mM) 120 NaCl, 4.7 KCl, 2.5 CaCl2, 1.2 MgCl2, 30 NaHCO3, 1.2 KH2PO4, 0.5 Na2-EDTA, 5.5 glucose, 2 sodium pyruvate (pH=7.4), and was gassed with carbogen (95% O2 and 5% CO2). Since each organ bath contained four chambers (with 25 ml each), we used one or two organ baths for human bladder experiments. The whole procedure from bladder removal to insertion in the organ bath chamber lasted less than 3 h.
Preparation of rat bladder samples
Rat bladder specimens were obtained from 112 Wistar rats (RRID: RGD_737929, 43 males and 69 females, minimum 40 days, first quartile 58 days, median 70 days, third quartile 137 days, maximum 560 days, Charles-River, Sulzfeld, Germany). Rats were decapitated in deep anaesthesia with diethylether, and the bladder was quickly removed and submerged in the 4 °C cold HEPES-buffered and calcium-reduced storage solution of the above composition. Each bladder was cut into four strips (some 5 mm length in and 1 mm in width) to fix them thereafter in an organ bath chamber as described before for human detrusor strips (i.e. all four specimens into one organ bath). To ensure a preferably atraumatic preparation, we did not remove the mucosa from rat bladder tissue. The whole procedure from bladder removal to insertion in the organ bath chamber lasted less than 30 min.
Isometric contractions and relaxations in vitro
Human and rat specimens were carefully inserted into the organ bath without any tension. Once submerged in an organ bath chamber, the specimen was exposed to buoyant force and body temperature (37 °C). After roughly 10 min, we slightly stretched the specimens (~2 mN) to ensure that the force transducers (Panlab MLT0201), coupled with bridge amplifiers (Panlab ML224) and connected to an analogue-to-digital converter (PowerLab 4/30, LabChart 7, ADInstruments), were sufficiently sensitive to record both isometric contractions and relaxations. From our previous studies [25, 32] we learned that human tissue often needed prolonged equilibration periods of several hours to produce robust and reproducible KCl responses. Therefore, equilibration time was >30 min in rat and >6 h in human tissue. Since intra-surgical excision procedures and sample transport conditions showed a higher variability in human than in rat tissue, experiments with human tissue were carried out only in specimens developing a stable baseline tone (113 out of 123, baseline tone 3.9 ± 1.8 mN, mean ± SD, n=113).
We always used the following protocol: At the beginning, the specimens were challenged with 60 mM KCl for 10 min (termed Kpre) to obtain a robust isometric contraction by adding either 500 μl of 3 M or 1000 μl of 1.5 M stock solution (i.e. dilution 1:50 or 1:25, respectively) to the organ bath (25 ml). We did not compensate for the concomitant hypertonicity, for smooth muscle cells do not contract under these conditions [26]. After washout, specimens were allowed to recover for 30–40 min before assessment of the effect of the β3 adrenoceptor agonist CL316,243 (referred to as CL hereafter, obtained from Tocris, UK, cat. no. 1499), because it was proven to be more selective towards β3 adrenoceptors than the clinically used compound mirabegron (selectivity >10,000-fold, [15]), which was also true for human cell lines [4, 59]. To this end, we applied 25 μl of CL stock solution (0.1, 1, or 10 mM) into the organ bath chamber to yield the final CL concentrations of 0.1, 1, or 10 μM (dilution 1:1000). Following 10 min of incubation with CL, 60 mM KCl (10 min) was added to the organ bath as before (termed KCL). After 30–40 min recovery, specimens were challenged with 60 mM KCl (10 min) to test for CL washout (i.e. Kpost). Since these experiments, however, revealed that CL showed incomplete washout, we did not alternate CL and vehicle tests in the same specimen. Rather, we carried out parallel time control experiments, and a given chamber was either dedicated to being a CL or a time control experiment. Since specimen size as well as orientation of smooth muscle cells affect contractile force, we analysed KCl responses in a paired condition (i.e. without/before versus with/after CL).
Having assessed the effect of CL on KCl-induced contraction, we tested up to five different concentrations of carbachol (referred to as CCh hereafter, ranging from 10−8 to 10−4 M in a randomised fashion), each lasting for 10 min, by adding 25 μl of the respective stock solution (dilution 1:1000) into the organ bath chamber to obtain a concentration-response curve (CRC). However, concentrations greater than 10−6 were suspected to render further CCh testing impossible owing to desensitisation. In these cases, only three different concentrations were analysed (i.e. stopping the experiment after the highest concentration of >10−6 M). Each CCh-induced response was normalised to the KCl response of the very specimen to produce a CCh CRC either in the presence or absence of CL. In addition, we performed Boltzmann fits by minimising the sum of squared residuals (i.e. the sum of the squared difference between observed and fitted values using the EXCEL solver.xlam add-in) to yield the median effective CCh concentration (EC50) as well as the maximum effect (Emax).
Immunohistochemistry of human β3 adrenoceptors
Human detrusor samples (n=10 patients) were fixed in 0.1 M phosphate-buffered saline (PBS) containing 4% paraformaldehyde for at least 1 day, cryoprotected in 30% sucrose for 3 to 5 days, frozen in 2-methylbutane, and then stored at −80 °C. Horizontal sections (14 μm) were cut on a cryostat and stored (−80 °C). For immunohistochemistry (IHC), heat-induced antigen retrieval (10 min cooking time, 0.05% Tween-20 in 10 mM citrate buffer, pH 6.0) was carried out to enhance the immunofluorescent signal. After cooling down for 20 min and 3×10 min washing in PBS, sections were first incubated for 20 min with 0.1% triton-X (in PBS), washed with PBS (2×10 min), and then incubated for 60 min with 5% normal goat serum (NGS). Sections were then allowed to incubate with the primary antibody (rabbit anti-β3 1:200 in 1% NGS/PBS, Alomone AAR-017, RRID: AB_2039720 [46]) at 4 °C overnight. Following 3×10 min washes with PBS, the second antibody (Cy3 goat anti-rabbit, RRID: AB_2534029, 1:200 in 1% NGS/PBS) was allowed to bind, before sections were embedded with ProLongTM Gold Antifade Mountant with DNA stain DAPI (P36931, Thermo Fisher Scientific). To avoid investigator bias in IHC studies, one experimenter (T.K.) chose the specimens, while another experimenter (S.R.) who was blinded regarding age performed both staining and analysis of IHC experiments including selection of viewing fields.
Statistical analysis
All contractions and relaxations were assessed as absolute force data (in mN) as well as percentages of the first KCl response (referred to as %Kpre). All data in the manuscript are given as mean values ± standard deviation (SD). Prior to statistical evaluation, all technical replicates from the same animal were converted into one median per sample. Then, data were analysed using the Mann-Whitney U test, Wilcoxon signed rank test, or two-way analysis of variance with post-hoc tests (for concentration-response curves) using SigmaStat 3.5 software. Owing to the exploratory nature of the study, we did not perform power analysis beforehand to calculate sample sizes, and P values are deemed descriptive. Statistically significant differences were indicated by asterisks (*P<0.05, **P<0.01) in all figures.
Results
The β3 agonist CL316,243 dampens human detrusor contraction in an age-dependent manner
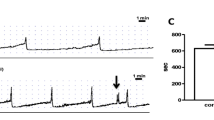
Aiming to explore the role of the β3 adrenoceptor in human detrusor muscle, we first evoked isometric contractions by 60 mM KCl in this tissue with or without the specific β3 adrenoceptor agonist CL316,243 (CL hereafter). Figure 1a shows a representative trace of two subsequent KCl-induced contractions (black traces, termed Kpre and Kpost) recorded from a specimen from a 66-year-old patient, which demonstrate their reproducibility. In an adjacent chamber of the organ bath, we tested a second specimen from the same patient for its responsiveness of KCl-induced contractions towards pre-applied CL (10 μM). In this patient, however, CL failed to depress the KCl response (blue traces in Fig. 1a; termed KCL). On the other hand, detrusor specimens from a 71-year-old patient also showed reproducible KCl responses (Kpre and Kpost in Fig. 1b), but here CL reduced its amplitude (blue traces in Fig. 1b; termed KCL), and, in addition, the KCl response after CL washout (Kpost) remained smaller when compared with that at the beginning of the experiment (Kpre). In a total of 37 specimens taken from nine patients, CL failed to significantly affect the subsequent KCl response (KCL), but reduced Kpost after CL washout (P<0.01, Wilcoxon signed rank test; blue box-whisker plots in Fig. 1c). In contrast, there was no such drop of the KCl response in parallel time control experiments (n=18 specimens from nine patients, black box-whisker plots in Fig. 1c). Since we wondered whether this statistically significant, but rather small effect could be relevant, we correlated the patients’ age to the relative size of KCL (Fig. 1d), which left us with a statistically significant negative correlation (Spearman correlation coefficient = −0.584, P<0.01; Fig. 1d). This result prompted us to analyse the CL effect in greater detail. In fact, this compound regularly reduced baseline tone (see arrowhead in Fig. 1b). Moreover, out of 37 specimens, four (from three patients) presented spontaneous activity before the application of CL: in all these cases, CL abolished spontaneous activity (see example in Fig. 1e). On average, activation of β3 adrenoceptors significantly depressed the baseline tone by 0.13 ± 0.13 mN or 1.8 ± 2.4% of Kpre (n=37, P<0.01, Wilcoxon signed rank test), which, however, showed no statistically significant correlation to ageing (Spearman correlation coefficient = 0.211, P=0.121; Fig. 1f).
CL316,243 inhibits KCl-induced contraction in human detrusor. a, b KCl-induced contractions in a 66-year-old patient (a) and a 71-year-old patient (b). Upper traces (black) demonstrate reproducible KCl-induced contractions (termed Kpre and Kpost). Lower traces (blue) show three subsequent KCl-induced contractions; the second one was recorded in the presence of CL316,243 (termed KCL). Note that KCL is not reduced in the 71-year-old patient, but in the 66-year-old patient. c The KCl response under CL (KCL) was not different from Kpre, but Kpost was significantly different from Kpre (Wilcoxon signed rank test). In time control experiments, no significant differences were found. n-numbers in brackets refer to muscle strips, N-numbers refer to patients. d The KCL amplitude was negatively correlated to age. e Representative trace showing the acute CL effect on baseline tone and spontaneous activity. f The CL effect on baseline tone was significant, but without statistically significant age dependence. The black circle indicates the subject of e
Since equilibration time in human tissue was up to 15 h, the question arose whether this could have affected contractility. To address this issue, we plotted the first mean KCl response (i.e. one value per subject) to the equilibration time and found no statistically significant correlation (Pearson correlation coefficient = 0.0598, n=17 patients, P=0.343). Furthermore, we quantified the baseline tone at the beginning and at the end of the prolonged equilibration phase showing a small but statistically significant reduction (−0.74 ± 1.5 mN, mean ± SD, n=113). To exclude that this reduction could have contaminated the KCl response under CL, we separately calculated the baseline tone reduction for specimens that were to be challenged later with CL (−0.57 ± 0.97 mN, n=52) versus those which were to serve later as controls (−0.88 ± 1.89 mN, n=61). These analyses demonstrated that the specimens of the CL group were even slightly more stable than those serving as controls. We therefore think it unlikely that the prolonged equilibration in human tissue could have affected the observed CL effects on contraction.
Next, we asked whether the β3 adrenoceptor agonist CL also affects CCh-induced contractions in human detrusor tissue. To control for systematic effects of strip size on the CCh response, we normalised this CCh response to Kpre of the same specimen and obtained CRCs with and without CL. A representative example of this experiment (Fig. 2a) shows the traces of a specimen that exhibited a KCl response of around 12 mN and a CCh response of about 30 mN (i.e. 250% Kpre; black traces). When applying CL prior to CCh in another specimen from this 59-year-old patient, CCh elicited a reduced response (140% of Kpre; blue traces in Fig. 2a) indicating a clear β3 adrenoceptor-dependent attenuation of the CCh response. In a specimen from a 71-year-old patient, however, the CCh response was unresponsive to the CL pretreatment (roughly 350% with or without CL; black and blue traces in Fig. 2b, respectively). To address the ageing effect statistically, we divided our patient cohort into two sub-cohorts of about the same sample size, i.e. either below 65 years (referred to as mid-life) or above (referred to as aged). In the first sub-cohort, the Boltzmann fit revealed the native CRC (i.e. without CL, black symbols in Fig. 2c) with an EC50 of 0.8 μM and a maximum effect (Emax) of 200% (95% C.I. 127–273%). In contrast, CL significantly compressed this curve cutting the maximum almost into halves (Emax = 110%, 95% C.I. 71–149%; Fig. 2c and Table 1; P<0.01, two-way-ANOVA with post-hoc Tukey test), but without altering EC50 (0.8 μM). Hence, although CL did not affect KCl responses in this patient cohort, CCh-induced contractions proved to be responsive to this compound (Cohen’s d of 1.82 in mid-life versus 0.13 in aged subjects). In aged humans, however, both CCh CRCs (i.e. with and without CL) were rather comparable (Fig. 2d, Table 1). Taken together, β3 adrenoceptor activation by CL reduced the potency of CCh to evoke contractions only in the mid-life sub-cohort, but not the aged. Moreover, CL left the EC50 unaltered implying a non-competitive mechanism of action.
CL316,243 inhibits CCh-induced contraction in human detrusor. a, b CCh-induced contractions were normalised to KCl-induced contractions (indicated by dotted lines). In a 59-year-old patient (a), administration of CL (blue traces) markedly reduced the CCh-response in comparison to the KCl response. In a 71-year-old patient (b), CCh responses were hardly affected by prior CCh administration. c Concentration-response curve of CCh in patients <65 years with (blue) and without (black) prior CL application. Data are presented as mean ± SD (n=1–11), N-number refers to patients. d Concentration-response curve of CCh in patients ≥65 years with (blue) and without (black) prior CL application. Data are presented as mean ± SD (n=1–23), N-number refers to patients
The β3 agonist CL316,243 dampens rat bladder contraction in an age-dependent manner
Having established a differential responsiveness of human detrusor tissue towards β3 adrenoceptor activation, we wished to ascertain whether rat tissue confirmed these results. Figure 3 illustrates the influence of CL on rat bladder KCl-induced contractions. Whereas successive applications of 60 mM KCl in time control experiments elicited reproducible results in a 54-day-old animal (black trace in Fig. 3a), CL markedly reduced the KCl response in an adjacent bladder specimen, and moreover, entailed poor washout (blue trace in Fig. 3a). Moreover, CL effectuated a statistically significant drop of baseline tone (−8 ± 8% at 10 μM, mean ± SD; see arrowhead in the blue trace in Fig. 3a). Like the human tissue study, we divided our animal cohort (from 40 days to 1.5 years of age) into two sub-cohorts of about the same sample size, here either below 65 days (referred to as adolescent) or above (referred to as mid-life). In 106 specimens from 27 adolescent rats, we found a statistically significant and dose-dependent attenuating effect of CL on KCl-induced contraction (P<0.01, Wilcoxon signed rank test, blue box-whisker plots in Fig. 3b), while time controls showed a statistically significant increase (P<0.01, Wilcoxon signed rank test, black box-whisker plots in Fig. 3b). In contrast, CL affected the KCl response in the mid-life group only at the highest concentration (Fig. 3c). Since the absolute KCl response depends on parameters such as strip size or orientation of smooth muscle cells within the specimens, we wished to control for this and separately calculated Kpre and Kpost for each CL subgroup. These analyses showed that all averaged amplitudes were quite comparable (Table 2). Two-way ANOVA (factors age, and CL subgroup) on Kpre revealed a significant age effect (P<0.05), but no significant CL effect (P=0.392). Kpre was 13.3 ± 3.8 mN (n=27) in adolescent rats and 16.7 ± 7.6 mN (n=22) in mid-life rats. We infer from these data that specimen preparation was quite homogenous within the same age group, but there was a statistically significant difference between adolescent and mid-life rats.
CL316,243 inhibits KCl-induced contraction in rat detrusor. a Representative traces of KCl-induced contractions in a 54-day-old rat from a time-control experiment (black) and a CL testing experiment (blue). Note the drop of the baseline tone after CL application (arrowhead). b CL dose-dependently reduced the subsequent CCh-induced contractions in animals <65 days (Wilcoxon signed rank test). All n-numbers refer to animals. c CL had a statistically significant effect on KCl-induced contractions in animals ≥65 days only at 10 μM
We then set out to assess the impact of CL on CCh responses in the rat bladder. Here, in a bladder specimen from a 39-day-old animal, 60 mM KCl and 0.8 μM CCh produced contractions of comparable size (black traces in Fig. 4a). When, however, CL was present prior to CCh administration, the 0.8 μM CCh response was half as large as the 60 mM KCl response (blue traces in Fig. 4a). To study the CL effect on the CCh response systematically, we randomly elicited the CCh response in 96 animals to obtain CRCs with or without CL pre-treatment (0.1, 1, and 10 μM). After dividing our animal cohort into two sub-cohorts (as above), the two-way-ANOVA followed by Tukey post-hoc test revealed a significant treatment effect of all three CL concentrations tested in the adolescent group (all F values >8 and P<0.01 for 0.1, 1, and 10 μM), but not the mid-life group (Table 1 and Fig. 4b–e). Importantly, the EC50 remained almost constant pointing again to a non-competitive mechanism of CL in inhibiting the CCh response. In summary, adolescent but not mid-life rat bladder specimens consistently responded to CL with reduced KCl and CCh responses.
CL316,243 inhibits CCh-induced contraction in rat detrusor. a CCh-induced contractions were normalised to KCl-induced contractions (indicated by dotted lines). In a 39-day-old rat, the CCh response was almost as large as the KCl response (black), but administration of CL (blue traces) markedly reduced the CCh response in comparison to the KCl response. b–e Concentration-response curves with (blue) and without (black) prior CL application. A significant CL effect was obtained with 1 μM (b) and 10 μM (d), but not in animals ≥65 days (c and e). Data are presented as mean ± SD, all n-numbers refer to animals
Immunohistochemical detection of β3 adrenoceptors in human detrusor
We next wondered whether β3 adrenoceptor expression levels maintain with age. To this end, we carried out immunohistochemistry in human sections from ten patients (nine males, one female) with previously used anti-β3 antibodies [46]. Detecting β3 immunoreactivity throughout the detrusor smooth muscle in all ten patients tested (Fig. 5a–d), we observed a somewhat lesser staining in sections from aged patients (Fig. 5c). However, nuclear staining with DAPI was also diminished in this cohort indicative of a loss of nuclei (Fig. 5d). To discern a difference between mid-life and aged tissue samples, we analysed one group with a mean age of around 55 years and another one with a mean age of around 80 years with comparable regions of interest (Fig. 5e, f). Normalised to DAPI nuclear stain as a measure of cell density, we failed to find a statistically significant difference between both groups (Fig. 5g). From these data, we cannot infer a decline in β3 adrenoceptor abundance during ageing in males.
Immunohistochemistry of β3 adrenoceptors in human detrusor. a–d β3 immunoreactivity in human detrusor from a 54-year-old patient (a) and a 78-year-old patient (c). Note the difference in staining which resembles the fainter nuclear staining with DAPI in the older patient (d) compared with the younger one (b). Scale bar = 50 μm. e–f Analysis of two different age groups (P<0.01, Mann-Whitney U test) (e) with similar regions of interest (f) showed similar β3 immunoreactivity when normalised to DAPI (f). n-numbers refer to patients
Discussion
The present study aimed at elucidating the age-dependent role of β3 adrenoceptor-mediated attenuation of urinary bladder contraction in human and rat tissue. We found that high KCl and carbachol (CCh) evoked tonic contractions in human detrusor muscle strips, which were differentially affected by prior β3 adrenoceptor activation with the selective agonist CL316,243 (CL). Whereas the KCl responsiveness was greater in tissue from aged patients, the CCh responsiveness was greater in tissue from mid-life patients. In contrast, tissue from adolescent rats was generally more sensitive to CL than tissue from mid-life rats. Immunohistochemistry confirmed the presence of β3 adrenoceptors on human detrusor smooth muscle cells but failed to detect a difference in receptor abundance between mid-life and aged tissue.
Role of β3 adrenoceptors in resting detrusor
One major finding of our study was that the β3-specific agonist CL reduced both basal tone and spontaneous contractions. This corroborates previous reports on non-selective β agonists such as isoprenaline [22, 23], as well as β3-selective agents showing higher potencies than β1 or β2 agents such as dobutamine, clenbuterol, or procaterol [5, 23, 57]. Although not specifically addressed, age-dependent changes seem rather unlikely in all these studies, as they included tissue samples from 5- to 82-year-old patients. Since we did not use β3 adrenoceptor antagonists, we cannot exclude β3-independent effects on resting detrusor. Based on our data on age-dependent changes in CL sensitivity, we infer that β3-mediated relaxation of resting human detrusor was irrespective of the donor’s age.
With respect to intracellular signalling, β adrenoceptors couple positively to adenylate cyclase which by itself promotes detrusor relaxation [10]. In theory, this would directly counteract negative coupling to this enzyme through M2 receptors. From experimental animals, we know that both β3 adrenoceptors and M2 receptors are expressed on smooth muscle cells to provide a functional antagonism [20, 21, 29, 30, 35, 43, 44, 49, 58]. Whilst there is evidence that in human detrusor, M3 receptors, although less available, outweigh the M2-mediated contraction [11], some M2-dependent tone may be presumed which would imply acetylcholine release, e.g. arising from nerve sources during urothelial stretching [47]. Albeit being of minor relevance in humans, we infer from our data that an M2-dependent basal tone also exists in humans and that β3 activation may increase both compliance and capacity as demonstrated in rats [54].
We found no evidence for an age-dependent decline in β3 adrenoceptor abundance in human detrusor. It is well established that β3 adrenoceptors are expressed on human detrusor smooth muscle; its mRNA substantially outreaches—by far more than 90%—the β1 and β2 isoforms [20, 22, 27, 39, 50, 51, 56]. Several immunohistochemical studies confirmed β3 protein being located on these cells [28, 40, 46]. One major drawback of immunohistochemical studies on β adrenoceptors is target specificity [41]. Here, we chose the antibody AAR-017 [46] because it was raised against the human and rat receptor, and it showed the same results as the validated antibody MC-A4198 [28, 40, 46]. Although the patients’ ages ranged from 18 to 77 years in these studies, age dependence was rarely analysed, except for one study where β3 adrenoceptor expression remained stable, at least in a small female cohort [28]. Consistent with this study, we found comparable β3 staining in mid-life and aged tissue—while having a clear male preponderance in our sample. Hence, the present study not only confirms available data, but also adds to the literature by demonstrating maintained β3 expression in the elderly male.
Role of β3 adrenoceptors in KCl-induced detrusor contraction
The effects of β3 adrenoceptors on resting detrusor do not necessarily predict to what extent externally evoked contractions are concerned. KCl-induced contractions were slightly more attenuated by CL in aged tissue than in mid-life tissue, whereas CCh-induced contractions showed higher CL sensitivity in mid-life than in aged tissue. This implies that different contraction mechanisms differ in their β3 responsiveness, as previously suggested [7]. In particular, KCl depolarises the cell membrane and leads to contraction by Ca2+ influx through voltage-gated Ca2+ channels (reviewed by [34]). CCh, in turn, activates the muscarinic receptor and leads to contraction by Ca2+ release from internal stores or by Ca2+ sensitisation through Gq/11 and G12/13 coupling, respectively (reviewed by [55]). Although cross-talk may exist between these pathways to a certain extent, differential β3-mediated effects occurring during ageing point to interference among intracellular signalling cascades rather than to age-dependent changes in receptor abundance. In the following paragraphs, we will discuss KCl-induced contractions; CCh-induced contractions will be discussed thereafter.
Almost all studies so far analysed β3 effects on KCl-precontracted detrusor and largely disregarded the impact of age, which impedes reconciling their findings with our own data. At least, the maximally achieved effects were comparable with ours, even though with relatively high concentrations: CL or BRL37244 (both at 100 μM) relieved the KCl-induced contraction by around 35% in a mid-life cohort [2, 3]. Others normalised the KCl response relieving effect to broad-spectrum relaxants such as isoprenaline or papaverine and obtained conspicuously higher maximum effects ranging from 56% to almost 100% [27, 51]. One of these even claimed a significantly reduced effect in aged tissue as opposed to young adult “control” tissue [27]. However, this was certainly due to (1) reduced isoprenaline effects in the aged tissue, and (2) missing time control experiments [27], and thus awaits further confirmation. In view of our data, we draw the conclusion that β3-mediated inhibition of KCl-induced contractions is at least preserved during ageing.
In contrast to humans, rats showed reduced CL attenuating effects of KCl-induced contractions during ageing. Nishimoto and co-workers had similarly observed that aged rat bladder was less sensitive to isoproterenol, forskolin, or cholera toxin [38], which was confirmed using β3-selective agonists [18]. To date, it is not clear whether species differences in intracellular signalling cascades are responsible for the differential CL effects. At least, three aspects need to be considered: (1) G protein abundance in rat bladder is affected by age [14], (2) the Rho kinase inhibitor blocks CCh-induced contractions by ~45% in humans [7, 25], compared with ~60% in rats [7], and (3) the combined inhibition of Rho kinase and myosin light-chain kinase fails to abolish contraction in humans [25]. When comparing human and rat experiments, one should also take into consideration that the investigated ages were not overlapping and that experimental designs were not identical in both species. While the mucosa has been removed from human bladder strips, it was intact in bladder strips. We thus cannot fully exclude mucosa-mediated effects in rat specimens. Therefore, we can only speculate that the human detrusor exhibits multiple parallel contraction pathways, which can compensate for each other in case of a shortage of one of these, e.g. during ageing.
Role of β3 adrenoceptors in CCh-induced detrusor contraction
In contrast to KCl, our study demonstrated an age-dependent decline of β3-mediated inhibition of CCh-induced contractions in human and rat detrusor samples. Except for mirabegron, previous studies in humans revealed submicromolar to low micromolar sensitivity of β3 agonists, which was compatible with our EC50 of ~1 μM [5, 23, 39, 50]. Age dependence, however, was not addressed in these studies: analysing 50 patients from 5 to 82 years, Igawa and co-workers observed stronger CL-mediated relaxation in normal than in hyperreflexic detrusor, but without acknowledging the patient’s age as a relevant covariate [23]. The remainder could not address this issue owing to their small sample sizes (12 patients aged from 60 to 68 years in [50]; seven patients aged from 53 to 76 years in [39]; 12 patients aged from 28 to 75 years in [5]). To what extent different experimental paradigms in human studies affect age dependence needs to be clarified. Taken together, we confirmed this age-dependent decline in rat tissue, consistent with (1) functional β3 adrenoceptor expression on rat detrusor [20, 30, 44] and (2) reduced adenylate cyclase activity in aged rats [53], probably due to a differential change in G protein abundance [14]. Future studies will need to address whether these issues also hold for human detrusor.
What is the clinical implication of our study? A recent review clarified that anticholinergics and β3 agonists showed similar efficacy, but tolerability issues clearly favoured the latter [31]. Since OAB is more prevalent in the elderly, reduced β3 responsiveness of bladder smooth muscle cells could be of clinical interest, even though—at present—there is little evidence for this [36]. As a matter of fact, direct detrusor relaxation by β3 agonists is only one mechanism of action, while others such as β3 adrenoceptor-mediated effects on afferent nerve fibres [12] may preserve their function during aging. Our data demonstrated a relevant difference in β3 sensitivity of electromechanical versus pharmacomechanical coupling. While the first is maintained in the elderly, the latter is reduced. This will become particularly relevant as far as both ionotropic and metabotropic receptors of the same transmitter are concerned, e.g. in the purinergic system [17, 19]. Conceivably, this will also help understand the responsiveness of OAB patients to β3 agents.
Data availability
Data will be made available on request.
References
Abrams P, Cardozo L, Fall M, Griffiths D, Rosier P, Ulmsten U, van Kerrebroeck P, Victor A, Wein A (2002) The standardisation of terminology of lower urinary tract function: report from the Standardisation Subcommittee of the International Continence Society. Neurourol Urodyn 21:167–178. https://doi.org/10.1002/nau.10052
Badawi JK, Seja T, Uecelehan H, Honeck P, Kwon ST, Bross S, Langbein S (2007) Relaxation of human detrusor muscle by selective beta-2 and beta-3 agonists and endogenous catecholamines. Urology 69(4):785–790. https://doi.org/10.1016/j.urology.2007.01.059
Badawi JK, Uecelehan H, Hatzinger M, Michel MS, Haferkamp A, Bross S (2005) Relaxant effects of beta-adrenergic agonists on porcine and human detrusor muscle. Acta Physiol Scand 185(2):151–159. https://doi.org/10.1111/j.1365-201X.2005.01474.x
Baker JG (2005) The selectivity of beta-adrenoceptor antagonists at the human beta1, beta2 and beta3 adrenoceptors. Br J Pharmacol 144(3):317–322. https://doi.org/10.1038/sj.bjp.0706048
Biers SM, Reynard JM, Brading AF (2006) The effects of a new selective beta3-adrenoceptor agonist (GW427353) on spontaneous activity and detrusor relaxation in human bladder. BJU Int 98(6):1310–1314. https://doi.org/10.1111/j.1464-410X.2006.06564.x
Bloom JD, Dutia MD, Johnson BD, Wissner A, Burns MG, Largis EE, Dolan JA, Claus TH (1992) Disodium (R,R)-5-[2-[[2-(3-chlorophenyl)-2-hydroxyethyl]-amino] propyl]-1,3-benzodioxole-2,2-dicarboxylate (CL 316,243). A potent beta-adrenergic agonist virtually specific for beta 3 receptors. A promising antidiabetic and antiobesity agent. J Med Chem 35:3081–3084. https://doi.org/10.1021/jm00094a025
Cernecka H, Kersten K, Maarsingh H, Elzinga CR, de Jong IJ, Korstanje C, Michel MC, Schmidt M (2015) β3-Adrenoceptor-mediated relaxation of rat and human urinary bladder: roles of BKCa channels and Rho kinase. Naunyn Schmiedebergs Arch Pharmacol 388(7):749–759. https://doi.org/10.1007/s00210-015-1128-z
Chancellor MB, Migliaccio-Walle K, Bramley TJ, Chaudhari SL, Corbell C, Globe D (2013) Long-term patterns of use and treatment failure with anticholinergic agents for overactive bladder. Clin Ther 35(11):1744–1751. https://doi.org/10.1016/j.clinthera.2013.08.017
Chapple CR, Cardozo L, Nitti VW, Siddiqui E, Michel MC (2014) Mirabegron in overactive bladder: a review of efficacy, safety, and tolerability. Neurourol Urodyn 33(1):17–30. https://doi.org/10.1002/nau.22505
Chess-Williams R (2002) Muscarinic receptors of the urinary bladder: detrusor, urothelial and prejunctional. Auton Autacoid Pharmacol 22(3):133–145. https://doi.org/10.1046/j.1474-8673.2002.00258.x
Chess-Williams R, Chapple CR, Yamanishi T, Yasuda K, Sellers DJ (2001) The minor population of M3-receptors mediate contraction of human detrusor muscle in vitro. J Auton Pharmacol 21(5-6):243–248. https://doi.org/10.1046/j.1365-2680.2001.00231.x
D` Agostino G, Condino AM, Calvi P (2015) Involvement of β3-adrenoceptors in the inhibitory control of cholinergic activity in human bladder: direct evidence by [(3)H]-acetylcholine release experiments in the isolated detrusor. Eur J Pharmacol 758:115–122. https://doi.org/10.1016/j.ejphar.2015.03.074
de Oliveira MG, Alexandre EC, Bonilla-Becerra SM, Bertollotto GM, Oliveira Justo AF, Mónica FZ, Antunes E (2019) Autonomic dysregulation at multiple sites is implicated in age-associated underactive bladder in female mice. Neurourol Urodyn 38:1212–1221. https://doi.org/10.1002/nau.23990
Derweesh IH, Wheeler MA, Weiss RM (2000) Alterations in G-proteins and beta -adrenergic responsive adenylyl cyclase in rat urinary bladder during aging. J Pharmacol Exp Ther 294:969–974
Dolan JA, Muenkel HA, Burns MG, Pellegrino SM, Fraser CM, Pietri F, Strosberg AD, Largis EE, Dutia MD, Bloom JD, Bass AS, Tanikella TK, Cobuzzi A, Lai FM, Claus TH (1994) Beta-3 adrenoceptor selectivity of the dioxolane dicarboxylate phenethanolamines. J Pharmacol Exp Ther 269(3):1000–1006
Erdem N, Chu FM (2006) Management of overactive bladder and urge urinary incontinence in the elderly patient. Am J Med 119:29S–36S. https://doi.org/10.1016/j.amjmed.2005.12.014
Fong Z, Griffin CS, Hollywood MA, Thornbury KD, Sergeant GP (2019) β3-Adrenoceptor agonists inhibit purinergic receptor-mediated contractions of the murine detrusor. Am J Physiol Cell Physiol 317(1):C131–C142. https://doi.org/10.1152/ajpcell.00488.2018
Frazier EP, Schneider T, Michel MC (2006) Effects of gender, age and hypertension on beta-adrenergic receptor function in rat urinary bladder. Naunyn Schmiedebergs Arch Pharmacol 373:300–309. https://doi.org/10.1007/s00210-006-0077-y
Fry CH, Young JS, Jabr RI, McCarthy C, Ikeda Y, Kanai AJ (2012) Modulation of spontaneous activity in the overactive bladder: the role of P2Y agonists. Am J Physiol Renal Physiol 302(11):F1447–F1454. https://doi.org/10.1152/ajprenal.00436.2011
Fujimura T, Tamura K, Tsutsumi T, Yamamoto T, Nakamura K, Koibuchi Y, Kobayashi M, Yamaguchi O (1999) Expression and possible functional role of the beta3-adrenoceptor in human and rat detrusor muscle. J Urol 161(2):680–685
Hegde SS, Choppin A, Bonhaus D, Briaud S, Loeb M, Moy TM, Loury D, Eglen RM (1997) Functional role of M2 and M3 muscarinic receptors in the urinary bladder of rats in vitro and in vivo. Br J Pharmacol 120(8):1409–1418. https://doi.org/10.1038/sj.bjp.0701048
Igawa Y, Yamazaki Y, Takeda H, Hayakawa K, Akahane M, Ajisawa Y, Yoneyama T, Nishizawa O, Andersson KE (1999) Functional and molecular biological evidence for a possible beta3-adrenoceptor in the human detrusor muscle. Br J Pharmacol 126(3):819–825. https://doi.org/10.1038/sj.bjp.0702358
Igawa Y, Yamazaki Y, Takeda H, Kaidoh K, Akahane M, Ajisawa Y, Yoneyama T, Nishizawa O, Andersson KE (2001) Relaxant effects of isoproterenol and selective beta3-adrenoceptor agonists on normal, low compliant and hyperreflexic human bladders. J Urol 165(1):240–244. https://doi.org/10.1097/00005392-200101000-00071
Irwin DE, Milsom I, Hunskaar S, Reilly K, Kopp Z, Herschorn S, Coyne K, Kelleher C, Hampel C, Artibani W, Abrams P (2006) Population-based survey of urinary incontinence, overactive bladder, and other lower urinary tract symptoms in five countries: results of the EPIC study. Eur Urol 50:1306–1314. https://doi.org/10.1016/j.eururo.2006.09.019
Kirschstein T, Protzel C, Porath K, Sellmann T, Köhling R, Hakenberg OW (2014) Age-dependent contribution of Rho kinase in carbachol-induced contraction of human detrusor smooth muscle in vitro. Acta Pharmacol Sin 35(1):74–81. https://doi.org/10.1038/aps.2013.126
Kirschstein T, Rehberg M, Bajorat R, Tokay T, Porath K, Köhling R (2009) High K+-induced contraction requires depolarization-induced Ca2+ release from internal stores in rat gut smooth muscle. Acta Pharmacol Sin 30(8):1123–1131. https://doi.org/10.1038/aps.2009.98
Li G, Li K, Li Z, Wang P (2003) Age-dependent changes in beta-adrenoceptor function in human detrusors and possible mechanisms. Chin Med J (Engl) 116:1511–1514
Limberg BJ, Andersson KE, Kullmann FA, Burmer G, de Groat WC, Rosenbaum JS (2010) β-Adrenergic receptor subtype expression in myocyte and non-myocyte cells in human female bladder. Cell Tissue Res 342:295–306. https://doi.org/10.1007/s00441-010-1053-x
Longhurst PA, Leggett RE, Briscoe JA (1995) Characterization of the functional muscarinic receptors in the rat urinary bladder. Br J Pharmacol 116(4):2279–2285. https://doi.org/10.1111/j.1476-5381.1995.tb15065.x
Longhurst PA, Levendusky M (1999) Pharmacological characterization of beta-adrenoceptors mediating relaxation of the rat urinary bladder in vitro. Br J Pharmacol 127(7):1744–1750. https://doi.org/10.1038/sj.bjp.0702709
Lozano-Ortega G, Ng DB, Szabo SM, Deighton AM, Riveros B, Guttschow A, Gooch KL, Gomes CM (2020) Management of patients with overactive bladder in Brazil: a retrospective observational study using data from the Brazilian Public Health System. Adv Ther 37(5):2344–2355. https://doi.org/10.1007/s12325-020-01318-w
Mader F, Müller S, Krause L, Springer A, Kernig K, Protzel C, Porath K, Rackow S, Wittstock T, Frank M, Hakenberg OW, Köhling R, Kirschstein T (2018) Hyperpolarization-activated cyclic nucleotide-gated non-selective (HCN) ion channels regulate human and murine urinary bladder contractility. Front Physiol 9:753. https://doi.org/10.3389/fphys.2018.00753
Madhuvrata P, Cody JD, Ellis G, Herbison GP, Hay-Smith EJC (2012) Which anticholinergic drug for overactive bladder symptoms in adults. Cochrane Database Syst Rev 1:CD005429. https://doi.org/10.1002/14651858.CD005429.pub2
Malysz J, Petkov GV (2020) Urinary bladder smooth muscle ion channels: expression, function, and regulation in health and disease. Am J Physiol Renal Physiol 319(2):F257–F283. https://doi.org/10.1152/ajprenal.00048.2020
Matsui M, Griffin MT, Shehnaz D, Taketo MM, Ehlert FJ (2003) Increased relaxant action of forskolin and isoproterenol against muscarinic agonist-induced contractions in smooth muscle from M2 receptor knockout mice. J Pharmacol Exp Ther 305(1):106–113. https://doi.org/10.1124/jpet.102.044701
Nakagomi H, Mitsui T, Shimura H, Ihara T, Kira S, Sawada N, Takeda M (2022) Mirabegron for overactive bladder in frail patients 80 years or over (HOKUTO study). BMC Urol 22(1):40. https://doi.org/10.1186/s12894-022-00989-7
Niclauß N, Michel-Reher MB, Alewijnse AE, Michel MC (2006) Comparison of three radioligands for the labelling of human beta-adrenoceptor subtypes. Naunyn Schmiedebergs Arch Pharmacol 374:99–105. https://doi.org/10.1007/s00210-006-0104-z
Nishimoto T, Latifpour J, Wheeler MA, Yoshida M, Weiss RM (1995) Age-dependent alterations in beta-adrenergic responsiveness of rat detrusor smooth muscle. J Urol 153:1701–1705. https://doi.org/10.1016/S0022-5347(01)67508-6
Nomiya M, Yamaguchi O (2003) A quantitative analysis of mRNA expression of alpha 1 and beta-adrenoceptor subtypes and their functional roles in human normal and obstructed bladders. J Urol 170:649–653. https://doi.org/10.1097/01.ju.0000067621.62736.7c
Otsuka A, Kawasaki H, Matsumoto R, Shinbo H, Kurita Y, Iwashita T, Ozono S (2013) Expression of β-adrenoceptor subtypes in urothelium, interstitial cells and detrusor of the human urinary bladder. Low Urin Tract Symptoms 5(3):173–180. https://doi.org/10.1111/luts.12007
Pradidarcheep W, Stallen J, Labruyère WT, Dabhoiwala NF, Michel MC, Lamers WH (2009) Lack of specificity of commercially available antisera against muscarinergic and adrenergic receptors. Naunyn Schmiedebergs Arch Pharmacol 379(4):397–402. https://doi.org/10.1007/s00210-009-0393-0
Robinson D, O'Kane M, Cardozo L (2023) Adherence to overactive bladder syndrome treatments recent developments and future perspectives. Int J Womens Health 15:799–811. https://doi.org/10.2147/IJWH.S369588
Rouget C, Rekik M, Camparo P, Botto H, Rischmann P, Lluel P, Palea S, Westfall TD (2014) Modulation of nerve-evoked contractions by β3-adrenoceptor agonism in human and rat isolated urinary bladder. Pharmacol Res 80:14–20. https://doi.org/10.1016/j.phrs.2013.12.006
Seguchi H, Nishimura J, Zhou Y, Niiro N, Kumazawa J, Kanaide H (1998) Expression of beta3-adrenoceptors in rat detrusor smooth muscle. J Urol 159(6):2197–2201. https://doi.org/10.1016/S0022-5347(01)63305-6
Sexton CC, Notte SM, Maroulis C, Dmochowski RR, Cardozo L, Subramanian D, Coyne KS (2011) Persistence and adherence in the treatment of overactive bladder syndrome with anticholinergic therapy: a systematic review of the literature. Int J Clin Pract 65(5):567–585. https://doi.org/10.1111/j.1742-1241.2010.02626.x
Silva I, Costa AF, Moreira S, Ferreirinha F, Magalhães-Cardoso MT, Calejo I, Silva-Ramos M, Correia-de-Sá P (2017) Inhibition of cholinergic neurotransmission by β3-adrenoceptors depends on adenosine release and A1-receptor activation in human and rat urinary bladders. Am J Physiol Renal Physiol 313(2):F388–F403. https://doi.org/10.1152/ajprenal.00392.2016
Stenqvist J, Aronsson P, Carlsson T, Winder M, Tobin G (2020) In vivo paracrine effects of ATP-induced urothelial acetylcholine in the rat urinary bladder. Auton Neurosci. 227:102689. https://doi.org/10.1016/j.autneu.2020.102689
Svalø J, Nordling J, Bouchelouche K, Andersson KE, Korstanje C, Bouchelouche P (2013) The novel β3-adrenoceptor agonist mirabegron reduces carbachol-induced contractile activity in detrusor tissue from patients with bladder outflow obstruction with or without detrusor overactivity. Eur J Pharmacol 699(1-3):101–105. https://doi.org/10.1016/j.ejphar.2012.11.060
Takasu T, Ukai M, Sato S, Matsui T, Nagase I, Maruyama T, Sasamata M, Miyata K, Uchida H, Yamaguchi O (2007) Effect of (R)-2-(2-aminothiazol-4-yl)-4'-{2-[(2-hydroxy-2-phenylethyl)amino]ethyl} acetanilide (YM178), a novel selective beta3-adrenoceptor agonist, on bladder function. J Pharmacol Exp Ther 321(2):642–647. https://doi.org/10.1124/jpet.106.115840
Takeda M, Obara K, Mizusawa T, Tomita Y, Arai K, Tsutsui T, Hatano A, Takahashi K, Nomura S (1999) Evidence for beta3-adrenoceptor subtypes in relaxation of the human urinary bladder detrusor: analysis by molecular biological and pharmacological methods. J Pharmacol Exp Ther 288(3):1367–1373
Tyagi P, Thomas CA, Yoshimura N, Chancellor MB (2009) Investigations into the presence of functional Beta1, Beta2 and Beta3-adrenoceptors in urothelium and detrusor of human bladder. Int Braz J Urol 35(1):76–83. https://doi.org/10.1590/s1677-55382009000100012
Uhlén M, Fagerberg L, Hallström BM, Lindskog C, Oksvold P, Mardinoglu A et al (2015) Proteomics. Tissue-based map of the human proteome. Science 347(6220):1260419. https://doi.org/10.1126/science.1260419
Wheeler MA, Pontari M, Nishimoto T, Weiss RM (1990) Changes in lipid composition and isoproterenol- and ethanol-stimulated adenylate cyclase activity in aging Fischer rat bladders. J Pharmacol Exp Ther 254(1):277–283
Woods M, Carson N, Norton NW, Sheldon JH, Argentieri TM (2001) Efficacy of the beta3-adrenergic receptor agonist CL-316243 on experimental bladder hyperreflexia and detrusor instability in the rat. J Urol 166(3):1142–1147
Wray S, Burdyga T, Noble K (2005) Calcium signalling in smooth muscle. Cell Calcium 38(3-4):397–407. https://doi.org/10.1016/j.ceca.2005.06.018
Wuest M, Eichhorn B, Grimm MO, Wirth MP, Ravens U, Kaumann AJ (2009) Catecholamines relax detrusor through beta 2-adrenoceptors in mouse and beta 3-adrenoceptors in man. J Pharmacol Exp Ther 328(1):213–222. https://doi.org/10.1124/jpet.108.142562
Yamanishi T, Yasuda K, Kitahara S, Nakai H, Yoshida K, Iizuka H (2006) Effects of 138-355, a beta3-adrenoceptor selective agonist, on relaxation of the human detrusor muscle in vitro. Neurourol Urodyn 25(7):815–819. https://doi.org/10.1002/nau.20231
Yamazaki Y, Takeda H, Akahane M, Igawa Y, Nishizawa O, Ajisawa Y (1998) Species differences in the distribution of beta-adrenoceptor subtypes in bladder smooth muscle. Br J Pharmacol 124(3):593–599. https://doi.org/10.1038/sj.bjp.0701870
Yanagisawa T, Sato T, Yamada H, Sukegawa J, Nunoki K (2000) Selectivity and potency of agonists for the three subtypes of cloned human beta-adrenoceptors expressed in Chinese hamster ovary cells. Tohoku J Exp Med 192(3):181–193. https://doi.org/10.1620/tjem.192.181
Acknowledgements
We thank Hanka Schmidt, Silke Birkmann, and Andreas Prestel for their excellent technical assistance.
Funding
Open Access funding enabled and organized by Projekt DEAL. This work was supported by intramural grants to TK (Project no. 889129 and 889584).
Author information
Authors and Affiliations
Contributions
OWH, RK, and TK conceived the study. CP, KP, SR, KK, and TK performed research. CP, KP, and TK analysed data. CP, RK, and TK wrote the paper.
Corresponding author
Ethics declarations
Ethics approval
All in vitro experiments with human material were approved by the local ethics committee (University Medicine of Rostock), and informed consent to participate in this study was obtained from each patient. All animal experiments were performed according to the international and national guidelines for the use of experimental animals. All experimenters involved in anaesthesia and killing have been accredited (certificate from the Federation of European Laboratory Animal Science Associations) and approved by the animal committee (Landesamt für Landwirtschaft, Lebensmittelsicherheit und Fischerei Mecklenburg-Vorpommern, approval no. 7221.3-1.2-007/10).
Consent for publication
All authors have seen and approved the manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Petereit, C., Porath, K., Rackow, S. et al. Age-dependent effects of the β3 adrenoceptor agonist CL316,243 on human and rat detrusor muscle strips. Pflugers Arch - Eur J Physiol 476, 243–256 (2024). https://doi.org/10.1007/s00424-023-02877-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-023-02877-x