Abstract
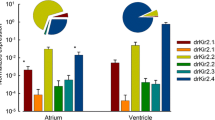
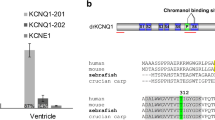
Zebrafish provide a translational model of human cardiac function. Their similar cardiac electrophysiology enables screening of human cardiac repolarization disorders, drug arrhythmogenicity, and novel antiarrhythmic therapeutics. However, while zebrafish cardiac repolarization is driven by delayed rectifier potassium channel current (IKr), the relative role of alternate channel transcripts is uncertain. While human ether-a-go-go-related-gene-1a (hERG1a) is the dominant transcript in humans, expression of the functionally distinct alternate transcript, hERG1b, modifies the electrophysiological and pharmacologic IKr phenotype. Studies of zebrafish IKr are frequently translated without consideration for the presence and impact of hERG1b in humans. Here, we performed phylogenetic analyses of all available KCNH genes from Actinopterygii (ray-finned fishes). Our findings confirmed zebrafish cardiac zkcnh6a as the paralog of human hERG1a (hKCNH2a), but also revealed evidence of a hERG1b (hKCNH2b)-like N-terminally truncated gene, zkcnh6b, in zebrafish. zkcnh6b is a teleost-specific variant that resulted from the 3R genome duplication. qRT-PCR showed dominant expression of zkcnh6a in zebrafish atrial and ventricular tissue, with low levels of zkcnh6b. Functional evaluation of zkcnh6b in a heterologous system showed no discernable function under the conditions tested, and no influence on zkcnh6a function during the zebrafish ventricular action potential. Our findings provide the first descriptions of the zkcnh6b gene, and show that, unlike in humans, zebrafish cardiac repolarization does not rely upon co-assembly of zERG1a/zERG1b. Given that hERG1b modifies IKr function and drug binding in humans, our findings highlight the need for consideration when translating hERG variant effects and toxicological screens in zebrafish, which lack a functional hERG1b-equivalent gene.






Similar content being viewed by others
Data availability
The data underlying this article are available in the article and in its online supplementary material.
References
Abi-Gerges N et al (2011) hERG subunit composition determines differential drug sensitivity. Br J Pharmacol 164(2b):419–432
Ablain J et al (2015) A CRISPR/Cas9 vector system for tissue-specific gene disruption in zebrafish. Dev Cell 32(6):756–764
Abramochkin DV, Hassinen M, Vornanen M (2018) Transcripts of Kv7.1 and MinK channels and slow delayed rectifier K(+) current (I(Ks)) are expressed in zebrafish (Danio rerio) heart. Pflugers Arch 470(12):1753–1764
Alexander J, Stainier DY, Yelon D (1998) Screening mosaic F1 females for mutations affecting zebrafish heart induction and patterning. Dev Genet 22(3):288–299
Amores A et al (1998) Zebrafish hox clusters and vertebrate genome evolution. Science 282(5394):1711–4
Arnaout R et al (2007) Zebrafish model for human long QT syndrome. Proc Natl Acad Sci U S A 104(27):11316–11321
Baker K et al (1997) Defective “pacemaker” current (Ih) in a zebrafish mutant with a slow heart rate. Proc Natl Acad Sci U S A 94(9):4554–4559
Bakkers J (2011) Zebrafish as a model to study cardiac development and human cardiac disease. Cardiovasc Res 91(2):279–288
Bhatt M et al (2022) The “www” of Xenopus laevis Oocytes: The Why, When, What of Xenopus laevis Oocytes in Membrane Transporters Research. Membranes (Basel) 12(10):927
Bournele D, Beis D (2016) Zebrafish models of cardiovascular disease. Heart Fail Rev 21(6):803–813
Bowley G et al (2022) Zebrafish as a tractable model of human cardiovascular disease. Br J Pharmacol 179(5):900–917
Brette F et al (2008) Characterization of isolated ventricular myocytes from adult zebrafish (Danio rerio). Biochem Biophys Res Commun 374(1):143–146
Briggs JP (2002) The zebrafish: a new model organism for integrative physiology. Am J Physiol Regul Integr Comp Physiol 282(1):R3-9
Casadei R et al (2011) Identification of housekeeping genes suitable for gene expression analysis in the zebrafish. Gene Expr Patterns 11(3–4):271–6
Chen JN et al (1996) Mutations affecting the cardiovascular system and other internal organs in zebrafish. Development 123:293–302
Chiu CH et al (2004) Bichir HoxA cluster sequence reveals surprising trends in ray-finned fish genomic evolution. Genome Res 14(1):11–7
Cubeddu LX (2016) Drug-induced Inhibition and Trafficking Disruption of ion Channels: Pathogenesis of QT Abnormalities and Drug-induced Fatal Arrhythmias. Curr Cardiol Rev 12(2):141–154
Curran ME et al (1995) A molecular basis for cardiac arrhythmia: HERG mutations cause long QT syndrome. Cell 80(5):795–803
Dascal N (1987) The use of Xenopus oocytes for the study of ion channels. CRC Crit Rev Biochem 22(4):317–87
Echeazarra L et al (2020) Adult and Developing Zebrafish as Suitable Models for Cardiac Electrophysiology and Pathology in Research and Industry. Front Physiol 11:607860
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32(5):1792–7
Feng L et al (2021) Long QT Syndrome KCNH2 Variant Induces hERG1a/1b Subunit Imbalance in Patient-Specific Induced Pluripotent Stem Cell-Derived Cardiomyocytes. Circ Arrhythm Electrophysiol 14(4):e009343
Gauvrit S et al (2022) Modeling Human Cardiac Arrhythmias: Insights from Zebrafish. J Cardiovasc Dev Dis 9(1):13
Genge CE et al (2016) Functional Divergence in Teleost Cardiac Troponin Paralogs Guides Variation in the Interaction of TnI Switch Region with TnC. Genome Biol Evol 8(4):994–1011
Genge CE et al (2016) The zebrafish heart as a model of mammalian cardiac function. Rev Physiol Biochem Pharmacol 171:99–136
Glickman NS, Yelon D (2002) Cardiac development in zebrafish: coordination of form and function. Semin Cell Dev Biol 13(6):507–513
Goversen B et al (2019) The influence of hERG1a and hERG1b isoforms on drug safety screening in iPSC-CMs. Prog Biophys Mol Biol 149:86–98
Gurdon JB et al (1971) Use of frog eggs and oocytes for the study of messenger RNA and its translation in living cells. Nature 233(5316):177–82
Harchi AE et al (2018) Investigation of hERG1b Influence on hERG Channel Pharmacology at Physiological Temperature. J Pharmacol Pharmacother 9:92–103
Hassel D et al (2008) Deficient zebrafish ether-a-go-go-related gene channel gating causes short-QT syndrome in zebrafish reggae mutants. Circulation 117(7):866–875
Hassinen M et al (2015) Inward rectifier potassium current (I K1) and Kir2 composition of the zebrafish (Danio rerio) heart. Pflugers Arch 467(12):2437–2446
Haverinen J, Hassinen M, Vornanen M (2022) Effect of Channel Assembly (KCNQ1 or KCNQ1 + KCNE1) on the Response of Zebrafish IKs Current to IKs Inhibitors and Activators. J Cardiovasc Pharmacol 79(5):670–677
Hoshijima K, Jurynec MJ, Grunwald DJ (2016) Precise Editing of the Zebrafish Genome Made Simple and Efficient. Dev Cell 36(6):654–667
Huang Q et al (2014) Evolutionary analysis of voltage-gated potassium channels by Bayes method. J Mol Neurosci 53(1):41–9
Hull CM et al (2019) Investigating the utility of adult zebrafish ex vivo whole hearts to pharmacologically screen hERG channel activator compounds. Am J Physiol Regul Integr Comp Physiol 317(6):R921–R931
Hurvich CM, Tsai C-L (1989) Regression and Time Series Model Selection in Small Samples. Biometrika 76(2):11
Hwang WY et al (2013) Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat Biotechnol 31(3):227–229
Johnstone KA et al (2011) Expression of olfactory receptors in different life stages and life histories of wild Atlantic salmon (Salmo salar). Mol Ecol 20(19):4059–69
Jones DT, Taylor WR, Thornton JM (1992) The rapid generation of mutation data matrices from protein sequences. Comput Appl Biosci 8(3):275–82
Jones EM et al (2004) Cardiac IKr channels minimally comprise hERG 1a and 1b subunits. J Biol Chem 279(43):44690–44694
Jones DK et al (2014) hERG 1b is critical for human cardiac repolarization. Proc Natl Acad Sci U S A 111(50):18073–18077
Jou CJ et al (2013) An in vivo cardiac assay to determine the functional consequences of putative long QT syndrome mutations. Circ Res 112(5):826–830
Kasahara M et al (2007) The medaka draft genome and insights into vertebrate genome evolution. Nature 447(7145):714–719
Ke Y et al (2014) Role of the cytoplasmic N-terminal Cap and Per-Arnt-Sim (PAS) domain in trafficking and stabilization of Kv11.1 channels. J Biol Chem 289(20):13782–91
Kibbe WA (2007) OligoCalc: an online oligonucleotide properties calculator. Nucleic Acids Res 35(Web Server issue):W43-6
Langheinrich U, Vacun G, Wagner T (2003) Zebrafish embryos express an orthologue of HERG and are sensitive toward a range of QT-prolonging drugs inducing severe arrhythmia. Toxicol Appl Pharmacol 193(3):370–382
Larsen AP et al (2008) Characterization of hERG1a and hERG1b potassium channels-a possible role for hERG1b in the I (Kr) current. Pflugers Arch 456(6):1137–48
Lee HC, Lin CY, Tsai HJ (2021) Zebrafish, an In Vivo Platform to Screen Drugs and Proteins for Biomedical Use. Pharmaceuticals (Basel) 14(6):500
Lees-Miller JP et al (1997) Electrophysiological characterization of an alternatively processed ERG K+ channel in mouse and human hearts. Circ Res 81(5):719–726
Leong IU et al (2010) Identification and expression analysis of kcnh2 genes in the zebrafish. Biochem Biophys Res Commun 396(4):817–824
Leong IU et al (2010) Zebrafish as a model for long QT syndrome: the evidence and the means of manipulating zebrafish gene expression. Acta Physiol (Oxf) 199(3):257–276
Martínez-Torres A, Pereida-Jaramillo E (2022). The Use of Xenopus Oocytes to Study the Biophysics and Pharmacological Properties of Receptors and Channels. In: Fainsod A, Moody SA (eds) Xenopus: From basic biology to disease models in the genomic era. 1st edn. pp143-151
McNally BA, Pendon ZD, Trudeau MC (2017) hERG1a and hERG1b potassium channel subunits directly interact and preferentially form heteromeric channels. J Biol Chem 292(52):21548–21557
Meyer A, Schartl M (1999) Gene and genome duplications in vertebrates: the one-to-four (-to-eight in fish) rule and the evolution of novel gene functions. Curr Opin Cell Biol 11(6):699–704
Milan DJ et al (2003) Drugs that induce repolarization abnormalities cause bradycardia in zebrafish. Circulation 107(10):1355–1358
Milan DJ et al (2006) In vivo recording of adult zebrafish electrocardiogram and assessment of drug-induced QT prolongation. Am J Physiol Heart Circ Physiol 291(1):H269–H273
Mitcheson JS (2008) hERG potassium channels and the structural basis of drug-induced arrhythmias. Chem Res Toxicol 21(5):1005–1010
Moss AJ (1986) Prolonged QT-interval syndromes. JAMA 256(21):2985–2987
Nasevicius A, Ekker SC (2000) Effective targeted gene “knockdown” in zebrafish. Nat Genet 26(2):216–220
Nemtsas P et al (2010) Adult zebrafish heart as a model for human heart? An electrophysiological study. J Mol Cell Cardiol 48(1):161–171
Ohno S (1999) Gene duplication and the uniqueness of vertebrate genomes circa 1970–1999. Semin Cell Dev Biol 10(5):517–22
Peal DS et al (2011) Novel chemical suppressors of long QT syndrome identified by an in vivo functional screen. Circulation 123(1):23–30
Pelster B, Burggren WW (1996) Disruption of hemoglobin oxygen transport does not impact oxygen-dependent physiological processes in developing embryos of zebra fish (Danio rerio). Circ Res 79(2):358–362
Phartiyal P, Jones EMC, Robertson GA (2007) Heteromeric assembly of human ether-a-go-go-related gene (hERG) 1a/1b channels occurs cotranslationally via N-terminal interactions. J Biol Chem 282(13):9874–9882
Poss KD, Wilson LG, Keating MT (2002) Heart regeneration in zebrafish. Science 298(5601):2188–2190
Rayani K et al (2018) Zebrafish as a model of mammalian cardiac function: Optically mapping the interplay of temperature and rate on voltage and calcium dynamics. Prog Biophys Mol Biol 138:69–90
Robertson GA, January CT (2006) HERG trafficking and pharmacological rescue of LQTS-2 mutant channels. Handb Exp Pharmacol 171:349–55
Rozen S, Skaletsky H (2000) Primer3 on the WWW for general users and for biologist programmers. Methods Mol Biol 132:365–86
Sale H et al (2008) Physiological properties of hERG 1a/1b heteromeric currents and a hERG 1b-specific mutation associated with Long-QT syndrome. Circ Res 103(7):e81-95
Sanguinetti MC et al (1995) A mechanistic link between an inherited and an acquired cardiac arrhythmia: HERG encodes the IKr potassium channel. Cell 81(2):299–307
Scholz EP et al (2009) Biophysical properties of zebrafish ether-a-go-go related gene potassium channels. Biochem Biophys Res Commun 381(2):159–164
Schwerte T, Fritsche R (2003) Understanding cardiovascular physiology in zebrafish and Xenopus larvae: the use of microtechniques. Comp Biochem Physiol A Mol Integr Physiol 135(1):131–145
Shi YP et al (2019) Extracellular protons accelerate hERG channel deactivation by destabilizing voltage sensor relaxation. J Gen Physiol 151(2):231–246
Shi YP et al (2020) The hERG channel activator, RPR260243, enhances protective I(Kr) current early in the refractory period reducing arrhythmogenicity in zebrafish hearts. Am J Physiol Heart Circ Physiol 319(2):H251–H261
Simpson KE et al (2022) CRISPR-Cas9-mediated Precise Knock-in Edits in Zebrafish Hearts. J Vis Exp (187). https://doi.org/10.3791/64209
Simpson KE et al (2020) Utility of Zebrafish Models of Acquired and Inherited Long QT Syndrome. Front Physiol 11:624129
Stainier DY et al (1996) Mutations affecting the formation and function of the cardiovascular system in the zebrafish embryo. Development 123:285–292
Tamura K et al (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–9
TeBay C, Hill AP, Windley MJ (2022) Metabolic and electrolyte abnormalities as risk factors in drug-induced long QT syndrome. Biophys Rev 14(1):353–367
Tisdale JE et al (2020) Drug-Induced Arrhythmias: A Scientific Statement From the American Heart Association. Circulation 142(15):e214–e233
Tristani-Firouzi M et al (2001) Molecular biology of K(+) channels and their role in cardiac arrhythmias. Am J Med 110(1):50–59
Trudeau MC et al (1995) HERG, a human inward rectifier in the voltage-gated potassium channel family. Science 269(5220):92–95
Trudeau MC et al (2011) hERG1a N-terminal eag domain-containing polypeptides regulate homomeric hERG1b and heteromeric hERG1a/hERG1b channels: a possible mechanism for long QT syndrome. J Gen Physiol 138(6):581–592
van Opbergen CJM et al (2018) Optogenetic sensors in the zebrafish heart: a novel in vivo electrophysiological tool to study cardiac arrhythmogenesis. Theranostics 8(17):4750–4764
Vandesompele J et al (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3(7):RESEARCH0034
Viskin S (1999) Long QT syndromes and torsade de pointes. Lancet 354(9190):1625–1633
Vornanen M, Hassinen M (2016) Zebrafish heart as a model for human cardiac electrophysiology. Channels (Austin) 10(2):101–110
Wu CC et al (2016) Spatially Resolved Genome-wide Transcriptional Profiling Identifies BMP Signaling as Essential Regulator of Zebrafish Cardiomyocyte Regeneration. Dev Cell 36(1):36–49
Zhou Z, Gong Q, January CT (1999) Correction of defective protein trafficking of a mutant HERG potassium channel in human long QT syndrome. Pharmacological and temperature effects. J Biol Chem 274(44):31123–6
Acknowledgements
This research was supported by a Canadian Institutes of Health Research Project grant (TWC) and a Natural Sciences and Engineering Research Council of Canada Discovery grant (TWC). C.M.H. was supported by a Natural Sciences and Engineering Research Council of Canada Alexander Graham Bell Canada Graduate Scholarship.
Funding
This research was conducted using grant support from the Canadian Institutes of Health Research (grant no. 156168 held by TWC) and the Natural Sciences and Engineering Research Council of Canada (grant no. RG-PIN-2020–04429 held by TWC). C.M.H. was supported by a Natural Sciences and Engineering Research Council of Canada Alexander Graham Bell Canada Graduate Scholarship.
Author information
Authors and Affiliations
Contributions
The first three authors contributed equally to this manuscript and may re-order the sequence of the first authors to reflect this in their curriculum vitae. C.E.G, P.M, and J.K performed research, analyzed data, contributed to methods, and wrote the paper. C.M.H conceived the study, M.Y, K.S, and D.H contributed to methods, and T.W.C conceived and designed the study, and wrote the paper.
Corresponding author
Ethics declarations
Ethical Approval
Work with zebrafish (Danio rerio) was conducted in accordance with Protocol#1264 K-18 approved by the Simon Fraser University Animal Care Committee and in accordance with the Canadian Council on Animal Care protocols and procedures.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Genge, C.E., Muralidharan, P., Kemp, J. et al. Zebrafish cardiac repolarization does not functionally depend on the expression of the hERG1b-like transcript. Pflugers Arch - Eur J Physiol 476, 87–99 (2024). https://doi.org/10.1007/s00424-023-02875-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-023-02875-z