Abstract
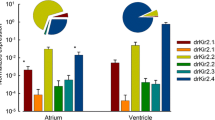
Zebrafish are increasingly used as a model for human cardiac electrophysiology, arrhythmias, and drug screening. However, K+ ion channels of the zebrafish heart, which determine the rate of repolarization and duration of cardiac action potential (AP) are still incompletely known and characterized. Here, we provide the first evidence for the presence of the slow component of the delayed rectifier K+channels in the zebrafish heart and characterize electrophysiological properties of the slow component of the delayed rectifier K+current, IKs. Zebrafish atrium and ventricle showed strong transcript expression of the kcnq1 gene, which encodes the Kv7.1 α-subunit of the slow delayed rectifier K+ channel. In contrast, the kcne1 gene, encoding the MinK β-subunit of the delayed rectifier, was expressed at 21 and 17 times lower level in ventricle and atrium, respectively, in comparison to the kcnq1. IKs was observed in 62% of ventricular myocytes with mean (± SEM) density of 1.23 ± 0.37 pA/pF at + 30 mV. Activation rate of IKs was 38% faster (τ50 = 1248 ± 215 ms) than kcnq1:kcne1 channels (1725 ± 792 ms) expressed in 3:1 ratio in Chinese hamster ovary cells. Microelectrode experiments demonstrated the functional relevance of IKs in the zebrafish heart, since 100 μM chromanol 293B produced a significant prolongation of AP in zebrafish ventricle. We conclude that AP repolarization in zebrafish ventricle is contributed by IKs, which is mainly generated by homotetrameric Kv7.1 channels not coupled to MinK ancillary β-subunits. This is a clear difference to the human heart, where MinK is an essential component of the slow delayed rectifier K+channel.






Similar content being viewed by others
References
Abramochkin DV, Vornanen M (2016) Seasonal changes of cholinergic response in the atrium of arctic navaga cod (Eleginus navaga). J Comp Physiol B 187:329–338
Alday A, Alonso H, Gallego M, Urrutia J, Letamendia A, Callol C, Casis O (2014) Ionic channels underlying the ventricular action potential in zebrafish embryo. Pharmacol Res 84:26–31. https://doi.org/10.1016/j.phrs.2014.03.011
Banyasz T, Jian Z, Horvath B, Khabbaz S, Izu LT, Chen-Izu Y (2014) Beta-adrenergic stimulation reverses the IKr-IKs dominant pattern during cardiac action potential. Pflüg Arch 466:2067–2076. https://doi.org/10.1007/s00424-014-1465-7
Barhanin J, Lesage F, Guillemare E, Fink M, Lazdunski M, Romey G (1996) KvLQT1 and IsK (minK) proteins associate to form the IKs cardiac potassium current. Nature 384:78–80
Bett GCL, Morales MJ, Beahm DL, Duffey ME, Rasmusson RL (2006) Ancillary subunits and stimulation frequency determine the potency of chromanol 293B block of the KCNQ1 potassium channel. J Physiol 576:755–767. https://doi.org/10.1113/jphysiol.2006.116012
Bosch RF, Gaspo R, Busch AE, Lang HJ, Li GR, Nattel S (1998) Effects of the chromanol 293B, a selective blocker of the slow, component of the delayed rectifier K+ current, on repolarization in human and guinea pig ventricular myocytes. Cardiovasc Res 38:441–450. https://doi.org/10.1016/s0008-6363(98)00021-2
Brette F, Luxan G, Cros C, Dixey H, Wilson C, Shiels HA (2008) Characterization of isolated ventricular myocytes from adult zebrafish (Danio rerio). Biochem Biophys Res Commun 374:143–146. https://doi.org/10.1016/j.bbrc.2008.06.109
Busch AE, Busch GL, Ford E, Suessbrich H, Lang HJ, Greger R, Kunzelmann K, Attali B, Stühmer W (1997) The role of the IsK protein in the specific pharmacological properties of the IKs channel complex. Br J Pharmacol 122:187–189
Chakraborty C, Hsu CH, Wen ZH, Lin CS, Agoramoorthy G (2009) Zebrafish: a complete animal model for in vivo drug discovery and development. Curr Drug Metab 10:116–124
Chen H, Kim LA, Rajan S, Xu S, Goldstein SAN (2003) Charybdotoxin binding in the IKs pore demonstrates two MinK subunits in each channel complex. Neuron 40:15–23
Colatsky TJ, Fermini B, Gintant GA, Pierson JB, Sager P, Sekino Y, Strauss DG, Stockbridge N (2016) The comprehensive in vitro proarrhythmia assay (CiPA) initiative—update on progress. J Pharmacol Toxicol Methods 81:15–20
Crumb WJ, Vicente J, Johannesen L, Strauss DG (2016) An evaluation of 30 clinical drugs against the comprehensive in vitro proarrhythmia assay (CiPA) proposed ion channel panel. J Pharmacol Toxicol Methods 81:251–262
Demolombe S, Baro I, Pereon Y, Bliek J, Mohammad-Panah R, Pollard H, Morid S, Mannens M, Wilde A, Barhanin J et al (1998) A dominant negative isoform of the long QT syndrome 1 gene product. J Biol Chem 273:6837–6843
Fermini B, Hancox JC, Abi-Gerges N, Bridgland-Taylor M, Chaudhary KW, Colatsky T, Correll K, Crumb W, Damiano B, Erdemli G (2016) A new perspective in the field of cardiac safety testing through the comprehensive in vitro proarrhythmia assay paradigm. J Biomol Screen 21:1–11
Gintant GA, Sager PT, Stockbridge N (2016) Evolution of strategies to improve preclinical cardiac safety testing. Nat RevDrug Discov 15:457–471
Guo X, Gao X, Wang Y, Peng L, Zhu Y, Wang S (2012) IKs protects from ventricular arrhythmia during cardiac ischemia and reperfusion in rabbits by preserving the repolarization reserve. PLoS One 7:e31545. https://doi.org/10.1371/journal.pone.0031545
Hassinen M, Laulaja S, Paajanen V, Haverinen J, Vornanen M (2011) Thermal adaptation of the crucian carp (Carassius carassius) cardiac delayed rectifier current, IKs, by homomeric assembly of Kv7.1 subunits without MinK. Am J Physiol 301:R255–R265. https://doi.org/10.1152/ajpregu.00067.2011
Hassinen M, Haverinen J, Hardy ME, Shiels HA, Vornanen M (2015) Inward rectifier potassium current (IK1) and Kir2 composition of the zebrafish (Danio rerio) heart. Pflüg Arch Eur J Physiol 467:2437–2446
Hassinen M, Haverinen J, Vornanen M (2015) Molecular basis and drug sensitivity of the delayed rectifier (IKr) in the fish heart. Comp Biochem Physiol C 176-177:44–51. https://doi.org/10.1016/j.cbpc.2015.07.005
Hassinen M, Haverinen J, Vornanen M (2017) Small functional If current in sinoatrial pacemaker cells of the brown trout (Salmo trutta fario) heart despite strong expression of HCN channel transcripts. Am J Physiol 313:R711–R722. https://doi.org/10.1152/ajpregu.00227.2017
Haverinen J, Vornanen M (2009) Responses of action potential and K+ currents to temperature acclimation in fish hearts: phylogeny or thermal preferences? Physiol Biochem Zool 82:468–482. https://doi.org/10.1086/590223
Haverinen J, Abramochkin DV, Kamkin A, Vornanen M (2017) The maximum heart rate in brown trout (Salmo trutta fario) is not limited by firing rate of pacemaker cells. Am J Physiol 312:R165–R171. https://doi.org/10.1152/ajpregu.00403.2016
Haverinen J, Hassinen M, Korajoki H, Vornanen M (2018) Cardiac voltage-gated sodium channel expression and electrophysiological characterization of the sodium current in the zebrafish (Danio rerio) ventricle. Prog Biophys Mol Biol 000:1–11 https://doi.org/10.1016/j.pbiomolbio.2018.04.002
Jost N, Virag L, Bitay M, Takacs J, Lengyel C, Biliczki P, Nagy Z, Bogats G, Lathrop DA, Papp JG et al (2005) Restricting excessive cardiac action potential and QT prolongation: a vital role for IKs in human ventricular muscle. Circulation 112:1392–1399
Jurkiewicz NK, Sanguinetti MC (1993) Rate-dependent prolongation of cardiac action potentials by a methanesulfonanilide class III antiarrhythmic agent. Specific block of rapidly activating delayed rectifier K+ current by dofetilide. Circ Res 72:75–83
Kari G, Rodeck U, Dicker AP (2007) Zebrafish: an emerging model system for human disease and drug discovery. Clin Pharmcol Ther 82:70–80
Kramer J, Obejero-Paz CA, Myatt G, Kuryshev YA, Bruening-Wright A, Verducci JS, Brown AM (2013) MICE models: superior to the HERG model in predicting Torsade de Pointes. Sci Rep 3:2100
Kurokawa J, Chen L, Kass RS (2003) Requirement of subunit expression for cAMP-mediated regulation of a heart potassium channel. Proc Natl Acad Sci U S A 100:2122–2127
Lerche C, Seebohm G, Wagner CI, Scherer CR, Dehmelt L, Abitbol I, Gerlach U, Brendel J, Attali B, Busch AE (2000) Molecular impact of MinK on the enantiospecific block of IKs by chromanols. Br J Pharmacol 131:1503–1506
Lerche C, Bruhova I, Lerche H, Steinmeyer K, Wei AD, Strutz-Seebohm N, Lang F, Busch AE, Zhorov BS, Seebohm G (2007) Chromanol 293B binding in KCNQ1 (Kv7.1) channels involves electrostatic interactions with a potassium ion in the selectivity filter. Mol Pharmacol 71:1503–1511
Liu DW, Antzelevitch C (1995) Characteristics of the delayed rectifier current (IKr and IKs) in canine ventricular epicardial, midmyocardial, and endocardial myocytes. A weaker IKs contributes to the longer action potential of the M cell. Circ Res 76:351–365
Lundquist AL, Manderfield LJ, Vanoye CG, Rogers CS, Donahue BS, Chang PA, Drinkwater DC, Murray KT, George AL (2005) Expression of multiple KCNE genes in human heart may enable variable modulation of IKs. J Mol Cell Cardiol 38:277–287
Marx SO, Kurokawa J, Reiken S, Motoike H, D'Armiento J, Marks AR, Kass RS (2002) Requirement of a macromolecular signaling complex for beta adrenergic receptor modulation of the KCNQ1-KCNE1 potassium channel. Science 295:496–499. https://doi.org/10.1126/science.1066843
McGrath P, Li CQ (2008) Zebrafish: a predictive model for assessing drug-induced toxicity. Drug Discov Today 13:394–401
Morin TJ, Kobertz WR (2008) Counting membrane-embedded KCNE beta-subunits in functioning K+ channel complexes. Proc Natl Acad Sci U S A 105:1478–1482. https://doi.org/10.1073/pnas.0710366105
Nakajo K, Ulbrich MH, Kubo Y, Isacoff EY (2010) Stoichiometry of the KCNQ1 - KCNE1 ion channel complex. Proc Natl Acad Sci U S A 107:18862–18867. https://doi.org/10.1073/pnas.1010354107
Nemtsas P, Wettwer E, Christ T, Weidinger G, Ravens U (2010) Adult zebrafish heart as a model for human heart? An electrophysiological study. J Mol Cell Cardiol 48:161–171. https://doi.org/10.1016/j.yjmcc.2009.08.034
Plant LD, Xiong D, Dai H, Goldstein SA (2014) Individual IKs channels at the surface of mammalian cells contain two KCNE1 accessory subunits. Proc Natl Acad Sci U S A 111:E1438–E1446. https://doi.org/10.1073/pnas.1323548111
Pongs O, Schwarz JR (2010) Ancillary subunits associated with voltage-dependent K+ channels. Physiol Rev 90:755–796
Qi M, Chen YH (2015) Zebrafish as a model for cardiac development and diseases. Hum Gen Ebryol 5:e112. https://doi.org/10.4172/2161-0436.1000e112
Romey G, Attali B, Chouabe C, Abitbol I, Guillemare E, Barhanin J, Lazdunski M (1997) Molecular mechanism and functional significance of the MinK control of the KvLQT1 channel activity. J Biol Chem 272:16713–16716
Rosati B, Dong M, Cheng L, Liou SR, Yan Q, Park JY, Shiang E, Sanguinetti MC, Wang HS, McKinnon D (2008) Evolution of ventricular myocyte electrophysiology. Physiol Genomics 35:262–272. https://doi.org/10.1152/physiolgenomics.00159.2007
Sanguinetti MC, Jurkiewicz NK, Scott A, Siegl PK (1991) Isoproterenol antagonizes prolongation of refractory period by the class III antiarrhythmic agent E-4031 in guinea pig myocytes. Mechanism of action. Circ Res 68:77–84
Sanguinetti MC, Curran ME, Zou A, Shen J, Specter PS, Atkinson DL, Keating MT (1996) Coassembly of KVLQT1 and minK (IsK) proteins to form cardiac IKs potassium channel. Nature 384:80–83
Takumi T, Ohkubo H, Nakanishi S (1988) Cloning of a membrane protein that induces a slow voltage-gated potassium current. Science 242:1042–1045
Tristani-Firouzi M, Chen J, Mitcheson JS, Sanguinetti MC (2001) Molecular biology of K+ channels and their role in cardiac arrhythmias. Am J Med 110:50–59
Trudeau MC, Warmke JW, Ganetzky B, Robertson GA (1995) HERG, a human inward rectifier in the voltage-gated potassium channel family. Science 269:92–95
Tsai CT, Wu CK, Chiang FT, Tseng CD, Lee JK, Yu CC, Wang YC, Lai LP, Lin JL, Hwang JJ (2011) In-vitro recording of adult zebrafish heart electrocardiogram—a platform for pharmacological testing. Clin Chim Acta 412:1963–1967. https://doi.org/10.1016/j.cca.2011.07.002
Vernlund L (2011) Characterization of expression of the KCNE gene family in zebrafish, Danio rerio. Honors Theses. Paper 15. http://docs.rwu.edu/honors_theses/15
Viswanathan PC, Shaw RM, Rudy Y (1999) Effects of IKr and IKs heterogeneity on action potential duration and its rate dependence. Circulation 99:2466–2474
Vornanen M (1997) Sarcolemmal Ca influx through L-type Ca channels in ventricular myocytes of a teleost fish. Am J Phys 272:R1432–R1440
Vornanen M, Hassinen M (2016) Zebrafish heart as a model for human cardiac electrophysiology. Channels 10:101–110
Vornanen M, Ryökkynen A, Nurmi A (2002) Temperature-dependent expression of sarcolemmal K+ currents in rainbow trout atrial and ventricular myocytes. Am J Phys 282:R1191–R1199
Wang W, Xia J, Kass RS (1998) MinK-KvLQT1 fusion proteins, evidence for multiple stoichiometries of the assembled IKs channel. J Biol Chem 273:34069–34074
Wang K, Terrenoire CE, Sampson KJ, Iyer V, Osteen JD, Lu J, Keller G, Kotton DN, Kass RS (2011) Biophysical properties of slow potassium channels in human embryonic stem cell derived cardiomyocytes implicate subunit stoichiometry. J Physiol 589:6093–6104
Westerfield M (2007) The zebrafish book: a guide for the laboratory use of zebrafish Danio (Brachydanio rerio), 5th edn. Univ. of Oregon Press, Eugene
Wiener R, Haitin Y, Shamgar L, Fernandez-Alonso MC, Martos A, Chomsky-Hecht O, Rivas G, Attali B, Hirsch JA (2008) The KCNQ1 (Kv7.1) COOH terminus, a multitiered scaffold for subunit assembly and protein interaction. J Biol Chem 283:5815–5830. https://doi.org/10.1074/jbc.M707541200
Wu C, Sharma K, Laster K, Hersi M, Torres C, Lukas TJ, Moore EJ (2014) Kcnq1-5 (Kv7.1-5) potassium channel expression in the adult zebrafish. BMC Physiol 14:1
Xu H, Guo W, Nerbonne JM (1999) Four kinetically distinct depolarization-activated K+ currents in adult mouse ventricular myocytes. J Gen Physiol 113:661–677
Yamada Y, Chen X, Kobayashi T, Kamada Y, Nagashima M, Tsutsuura M, Seki S, Yamakage M, Namiki A, Tohse N (2002) A truncated splice variant of KCNQ1 cloned from rat heart. Biochem Biophys Res Commun 294:199–204
Yue L, Feng J, Li GR, Nattel S (1996) Transient outward and delayed rectifier currents in canine atrium: properties and role of isolation methods. Am J Physiol 270:H2157–H2168. https://doi.org/10.1152/ajpheart.1996.270.6.H2157
Acknowledgements
Anita Kervinen is acknowledged for technical assistance and taking care of the fish.
Funding
This study was funded by the Russian Science Foundation (14-15-00268 to DVA) and the Jane and Aatos Erkko Foundation (64579 to MV).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abramochkin, D.V., Hassinen, M. & Vornanen, M. Transcripts of Kv7.1 and MinK channels and slow delayed rectifier K+ current (IKs) are expressed in zebrafish (Danio rerio) heart. Pflugers Arch - Eur J Physiol 470, 1753–1764 (2018). https://doi.org/10.1007/s00424-018-2193-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-018-2193-1