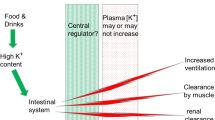
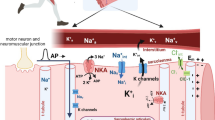
Abstract
Skeletal muscle cells can both gain and lose volume during periods of exercise and rest. Muscle cells do not behave as perfect osmometers because the cell volume changes are less than predicted from the change in extracellular osmolality. Therefore, there are mechanisms involved in regulating cell volume, and they are different for regulatory volume decreases and regulatory volume increases. Also, after an initial rapid change in cell volume, there is a gradual and partial recovery of cell volume that is effected by ion and water transport mechanisms. The mechanisms have been studied in non-contracting muscle cells, but remain to be fully elucidated in contracting muscle. Changes in muscle cell volume are known to affect the strength of contractile activity as well as anabolic/catabolic signaling, perhaps indicating that cell volume should be a regulated variable in skeletal muscle cells. Muscles contracting at moderate to high intensity gain intracellular volume because of increased intracellular osmolality. Concurrent increases in interstitial (extracellular) muscle volume occur from an increase in osmotically active molecules and increased vascular filtration pressure. At the same time, non-contracting muscles lose cell volume because of increased extracellular (blood) osmolality. This review provides the physiological foundations and highlights key concepts that underpin our current understanding of volume regulatory processes in skeletal muscle, beginning with consideration of osmosis more than 200 years ago and continuing through to the process of regulatory volume decrease and regulatory volume increase.








Similar content being viewed by others
Availability of data and material
This is a review with no new data.
Code availability
Not applicable.
Abbreviations
- ADP:
-
Adenosine diphosphate
- ATP:
-
Adenosine triphosphate
- ATPase:
-
Adenosine triphosphatase
- Ca2 + :
-
Calcium cation
- Cl− :
-
Chloride anion
- K+ :
-
Potassium cation
- MCT:
-
Monocarboxylate transport protein
- Mg2 + :
-
Magnesium cation
- Na+ :
-
Sodium cation
- NaCl:
-
Sodium chloride
- NKA:
-
Sodium, potassium ATPase
- NKCC:
-
Sodium, potassium, two chloride cotransporter
- Pi:
-
Inorganic phosphate
- PCr:
-
Phosphocreatine
- RVD:
-
Regulatory volume decrease
- RVI:
-
Regulatory volume increase
- T system:
-
Transverse tubule system
- TRPV2:
-
Transient receptor potential protein V2
References
Bergström J, Guarnieri G, Hultman E (1971) Carbohydrate metabolism and electrolyte changes in human muscle tissue during heavy work. J Appl Physiol 30:122–125. https://doi.org/10.1152/JAPPL.1971.30.1.122
Bernard C (1865) Introduction a l’etude de la medicine experimentale. Bailleire, Paris
Bernstein J (1902) Untersuchungen zur Thermodynamik der bioelektrischen Ströme—Erster Theil. Pflüger Arch Für Die Gesammte Physiol Des Menschen Und Der Thiere 92:521–562. https://doi.org/10.1007/BF01790181
Bonen A, Heynen M, Hatta H (2006) Distribution of monocarboxylate transporters MCT1–MCT8 in rat tissues and human skeletal muscle. Appl Physiol Nutr Metab 31:31–39. https://doi.org/10.1139/h05-002
Bressler BH, Matsuba K (1991) Tension and instantaneous stiffness of single muscle fibers immersed in Ringer solution of decreased tonicity. Biophys J 59:1002–1006. https://doi.org/10.1016/S0006-3495(91)82315-3
Brooks GA (2020) Lactate as a fulcrum of metabolism. Redox Biol. https://doi.org/10.1016/j.redox.2020.101454
Brown D (2017) The discovery of water channels (aquaporins). Ann Nutr Metab 70:37–42. https://doi.org/10.1159/000463061
Brücke E (1843) Beiträge zur Lehre von der Diffusion tropfbarflüssiger Körper durch poröse Scheidewände. Ann Phys 134:77–94. https://doi.org/10.1002/andp.18431340107
Cermak NM, LeBlanc PJ, Peters SJ et al (2009) Effect of extracellular osmolality on metabolism in contracting mammalian skeletal muscle in vitro. Appl Physiol Nutr Metab 34:1055–1064. https://doi.org/10.1139/H09-106
Chen Y, Simasko SM, Niggel J et al (1996) Ca2+ uptake in GH3 cells during hypotonic swelling: The sensory role of stretch-activated ion channels. Am J Physiol Cell Physiol. https://doi.org/10.1152/ajpcell.1996.270.6.c1790
Clarkson TW (1967) The transport of salt and water across isolated rat ileum. Evidence for at least two distinct pathways. J Gen Physiol 50:695–727. https://doi.org/10.1085/jgp.50.3.695
Clausen T (2003) Na+–K+ pump regulation and skeletal muscle contractility. Physiol Rev 83:1269–1324. https://doi.org/10.1152/physrev.00011.2003
Clausen T, Persson AEG (1998) Jens Christian Skou awarded the nobel prize in chemistry for the identification of the Na+, K+-pump. Acta Physiol Scand 163:1–2. https://doi.org/10.1046/j.1365-201x.1998.00367.x
Conway EJ, Boyle PJ (1939) A mechanism for the concentrating of potassium by cells, with experimental verification for muscle [7]. Nature 144:709–710
Denker BM, Smith BL, Kuhajda FP, Agre P (1988) Identification, purification, and partial characterization of a novel M(r) 28,000 integral membrane protein from erythrocytes and renal tubules. J Biol Chem 263:15634–15642. https://doi.org/10.1016/s0021-9258(19)37635-5
Dick DAT (1959) The rate of diffusion of water in the protoplasm of living cells. Exp Cell Res 17:5–12. https://doi.org/10.1016/0014-4827(59)90146-6
Dick DAT (1966) Cell water, molecular. Butterworths, Washington
Dill DB, Talbott JH, Edwards HT (1930) Studies in muscular activity: VI. Response of several individuals to a fixed task. J Physiol 69:267–305. https://doi.org/10.1113/jphysiol.1930.sp002649
Dölken G, Leisner E, Pette D (1975) Immunofluorescent localization of glycogenolytic and glycolytic enzyme proteins and of malate dehydrogenase isozymes in cross-striated skeletal muscle and heart of the rabbit. Histochemistry 43:113–121. https://doi.org/10.1007/BF00492440
Dutrochet H (1826) L’agent immédiat du mouvement vital dévoilé dans sa nature et dans son mode d’action chez les végétaux et les animaux. Dentu, Paris
Edwards GA, Ruska H, de Souza SP, Vallejo-Freire A (1956) Comparative cytophysiology of striated muscle with special reference to the role of the endoplasmic reticulum. J Biophys Biochem Cytol 2:143–156. https://doi.org/10.1083/jcb.2.4.143
Fenn WO (1936) Electrolytes in muscle. Cold Spring Harb Symp Quant Biol 4:252–259. https://doi.org/10.1101/sqb.1936.004.01.027
Fenn WO, Cobb DM (1934) The potassium equilibrium in muscle. J Gen Physiol 17:629–656. https://doi.org/10.1085/jgp.17.5.629
Fenn WO, Cobb DM (1936) Electrolyte changes in muscle during activity. Am J Physiol 115:345–356. https://doi.org/10.1152/ajplegacy.1936.115.2.345
Fenn W, Cobb D, Manery J, Bloor WR (1938) Electrolyte changes in cat muscle during stimulation. Am J Physiol 121:595–608. https://doi.org/10.1152/ajplegacy.1938.121.3.595
Forbes SC, Raymer GH, Kowalchuk JM, Marsh GD (2005) NaHCO3-induced alkalosis reduces the phosphocreatine slow component during heavy-intensity forearm exercise. J Appl Physiol 99:1668–1675. https://doi.org/10.1152/japplphysiol.01200.2004
González-Alonso J, Mortensen SP, Dawson EA et al (2006) Erythrocytes and the regulation of human skeletal muscle blood flow and oxygen delivery: role of erythrocyte count and oxygenation state of haemoglobin. J Physiol 572:295–305. https://doi.org/10.1113/jphysiol.2005.101121
Gosmanov AR, Thomason DB (2002) Insulin and isoproterenol differentially regulate mitogen-activated protein kinase-dependent Na+–K+–2Cl− cotransporter activity in skeletal muscle. Diabetes 51:615–623. https://doi.org/10.2337/diabetes.51.3.615
Gosmanov AR, Wong JA, Thomason DB (2002) Duality of G protein-coupled mechanisms for β-adrenergic activation of NKCC activity in skeletal muscle. Am J Physiol Cell Physiol. https://doi.org/10.1152/ajpcell.00096.2002
Gosmanov AAR, Lindinger MI, Thomason DBD (2003a) Riding the tides: K+ concentration and volume regulation by muscle Na+–K+–2Cl− cotransport activity. News Physiol Sci 18:196–200. https://doi.org/10.1152/nips.01446.2003
Gosmanov AR, Schneider EG, Thomason DB (2003b) NKCC activity restores muscle water during hyperosmotic challenge independent of insulin, ERK, and p38 MAPK. Am J Physiol Regul Integr Comp Physiol 284:655–665. https://doi.org/10.1152/ajpregu.00576.2002
Gosmanov AR, Fan Z, Mi X et al (2004) ATP-sensitive potassium channels mediate hyperosmotic stimulation of NKCC in slow-twitch muscle. Am J Physiol Cell Physiol 286:586–595. https://doi.org/10.1152/ajpcell.00247.2003
Grundfest H (1966) Heterogeneity of excitable membrane: electrophysiological and pharmacological evidence and some consequences. Ann NY Acad Sci 137:901–949. https://doi.org/10.1111/j.1749-6632.1966.tb50208.x
Gulati J, Babu A (1982) Tonicity effects on intact single muscle fibers: relation between force and cell volume. Science 215:1109–1112. https://doi.org/10.1126/science.6977845
Hallows KR, Mount PF, Pastor-Soler NM, Power DA (2010) Role of the energy sensor AMP-activated protein kinase in renal physiology and disease. Am J Physiol Ren Physiol. https://doi.org/10.1152/ajprenal.00005.2010
Hamilton MT, Ward DS, Watson PD (1993) Effect of plasma osmolality on steady-state fluid shifts in perfused cat skeletal muscle. Am J Physiol Regul Integr Comp Physiol. https://doi.org/10.1152/ajpregu.1993.265.6.r1318
Hargreaves M, Spriet LL (2020) Skeletal muscle energy metabolism during exercise. Nat Metab. 2:817–828. https://doi.org/10.1038/s42255-020-0251-4
Harris RC, Edwards RHT, Hultman E et al (1976) The time course of phosphorylcreatine resynthesis during recovery of the quadriceps muscle in man. Pflügers Arch Eur J Physiol 367:137–142. https://doi.org/10.1007/BF00585149
Harrison MH (1985) Effects of thermal stress and exercise on blood volume in humans. Physiol Rev 65:149–209. https://doi.org/10.1152/physrev.1985.65.1.149
Heidenhain R (1894) Neue Versuche über die Aufsaugung im Dünndarm. Pflugers Arch 56:579–631
Heigenhauser GJ, Lindinger MI (1988) The total ionic status of muscle during intense exercise. Adv Exp Med Biol 227:237–242. https://doi.org/10.1007/978-1-4684-5481-9_21
Hewson W (1773) On the figure and composition of the red particles of the blood, commonly called the red globules. Philos Trans R Soc Ser B Biol Sci 63:303–323
Hille B (1967) A Pharmacological Analysis of the Ionic Channels of Nerve. Digital Commons @ RU Student Theses and Dissertations. https://digitalcommons.rockefeller.edu/student_theses_and_dissertations/569/
Hille B (1968) Charges and potentials at the nerve surface. Divalent ions and pH. J Gen Physiol 51:221–236. https://doi.org/10.1085/jgp.51.2.221
Hille B (2018) The founding of Journal of General Physiology: Membrane permeation and ion selectivity. J Gen Physiol 150:389–400. https://doi.org/10.1085/jgp.201711937
Hodgkin AL, Huxley AF (1945) Resting and action potentials in single nerve fibres. J Physiol 104:176–195. https://doi.org/10.1113/jphysiol.1945.sp004114
Hodgkin AL, Huxley AF (1952) Movement of sodium and potassium ions during nervous activity. Cold Spring Harb Symp Quant Biol 17:43–52. https://doi.org/10.1101/SQB.1952.017.01.007
Hoffmann EK, Sjøholm C, Simonsen LO (1983) Na+, Cl− cotransport in Ehrlich ascites tumor cells activated during volume regulation (regulatory volume increase). J Membr Biol 76:269–280. https://doi.org/10.1007/BF01870369
Hoffmann EK, Lambert IH, Pedersen SF (2009) Physiology of cell volume regulation in vertebrates. Physiol Rev 89:193–277. https://doi.org/10.1152/physrev.00037.2007
Holmstrom PK, Karlsson Ö, Lindblom H et al (2021) Enhanced splenic volume and contraction in elite endurance athletes. J Appl Physiol 131:474–486. https://doi.org/10.1152/japplphysiol.01066.2020
Hoppe-Seyler F (1866) Medicinisch-Chemische Untersuchungen, Vol. 1: Aus dem Laboratorium für Angewandte Chemie zu Tübingen. Berlin, 1866-1871. http://onlinebooks.library.upenn.edu/webbin/book/lookupid?key=ha100795736
Ingber DE (1993) Cellular tensegrity: defining new rules of biological design that govern the cytoskeleton. J Cell Sci 104:613–627
Ingber DE, Heidemann SR, Lamoureux P, Buxbaum RE (2000) Opposing views on tensegrity as a structural framework for understanding cell mechanics. J Appl Physiol 89:1663–1678. https://doi.org/10.1152/jappl.2000.89.4.1663
Jones WM, Barer R (1948) Electron microscopy of the sarcolemma. Nature 161:1012. https://doi.org/10.1038/1611012a0
Kahle KT, Rinehart J, Lifton RP (2010) Phosphoregulation of the Na–K-2Cl and K-Cl cotransporters by the WNK kinases. Biochim Biophys Acta Mol Basis Dis 1802:1150–1158. https://doi.org/10.1016/j.bbadis.2010.07.009
Kleinzeller A (1999) Chapter 1 Charles Ernest Overton’s concept of a cell membrane. Curr Top Membr Transp 48:1–22. https://doi.org/10.1016/S0070-2161(08)61039-4
Kowalchuk JM, Heigenhauser GJF, Lindinger MI et al (1988a) Role of lungs and inactive muscle in acid-base control after maximal exercise. J Appl Physiol 65:2090–2096. https://doi.org/10.1152/jappl.1988.65.5.2090
Kowalchuk JM, Heigenhauser GJF, Lindinger MI et al (1988b) Factors influencing hydrogen ion concentration in muscle after intense exercise. J Appl Physiol 65:2080–2089. https://doi.org/10.1152/jappl.1988.65.5.2080
Kregenow FM (1971a) The response of duck erythrocytes to nonhemolytic hypotonic media: evidence for a volume-controlling mechanism. J Gen Physiol 58:372–395. https://doi.org/10.1085/jgp.58.4.372
Kregenow FM (1971b) The response of duck erythrocytes to hypertonic media: Further evidence for a volume-controlling mechanism. J Gen Physiol 58:396–412. https://doi.org/10.1085/jgp.58.4.396
Lang F (2007) Mechanisms and significance of cell volume regulation. J Am Coll Nutr 26:613S-623S. https://doi.org/10.1080/07315724.2007.10719667
Lang F, Görlach A, Vallon V (2009) Targeting SGK1 in diabetes. Expert Opin Ther Targets 13:1303–1311. https://doi.org/10.1517/14728220903260807
Lindinger MI, Cairns SP (2021) Regulation of muscle potassium: exercise performance, fatigue and health implications. Eur J Appl Physiol. https://doi.org/10.1007/s00421-020-04546-8
Lindinger M, Heigenhauser GJ (1988) Ion fluxes during tetanic stimulation in isolated perfused rat hindlimb. Am J Physiol Regul Integr Comp Physiol. https://doi.org/10.1152/ajpregu.1988.254.1.r117
Lindinger M, Ward S (2022) A century of exercise physiology—key concepts. Eur J Appl Physiol
Lindinger MI, Heigenhauser JF, Jones NL (1986) Acid-base and respiratory properties of a buffered bovine erythrocyte perfusion medium. Can J Physiol Pharmacol 64:550–555. https://doi.org/10.1139/y86-091
Lindinger MII, Heigenhauser GJFJF, Spriet LLL (1987) Effects of intense swimming and tetanic electrical stimulation on skeletal muscle ions and metabolites. J Appl Physiol 63:2331–2339. https://doi.org/10.1152/jappl.1987.63.6.2331
Lindinger MII, Heigenhauser GJFJF, McKelvie RSS, Jones NLL (1990) Role of nonworking muscle on blood metabolites and ions with intense intermittent exercise. Am J Physiol Regul Integr Comp Physiol. https://doi.org/10.1152/ajpregu.1990.258.6.r1486
Lindinger MI, Heigenhauser GJF, McKelvie RS, Jones NL (1992) Blood ion regulation during repeated maximal exercise and recovery in humans. Am J Physiol Regul Integr Comp Physiol. https://doi.org/10.1152/ajpregu.1992.262.1.r126
Lindinger M, Spriet L, Hultman E et al (1994) Plasma volume and ion regulation during exercise after low- and high- carbohydrate diets. Am J Physiol Regul Integr Comp Physiol 266:R1896–R1906. https://doi.org/10.1152/ajpregu.1994.266.6.r1896
Lindinger MI, McKelvie RS, Heigenhauser GJF (1995) K+ and Lac- distribution in humans during and after high-intensity exercise: role in muscle fatigue attenuation? J Appl Physiol 78:765–777. https://doi.org/10.1152/jappl.1995.78.3.765
Lindinger MI, Horn PL, Grudzien SP (1999) Exercise-induced stimulation of K+ transport in human erythrocytes. J Appl Physiol 87:2157–2167. https://doi.org/10.1152/jappl.1999.87.6.2157
Lindinger M, Hawke T, Lipskie S et al (2002) K+ transport and volume regulatory response by NKCC in resting rat hindlimb skeletal muscle. Cell Physiol Biochem 12:279–292. https://doi.org/10.1159/000067898
Lindinger MI, Leung M, Trajcevski KE, Hawke TJ (2011) Volume regulation in mammalian skeletal muscle: the role of sodium-potassium-chloride cotransporters during exposure to hypertonic solutions. J Physiol 589:2887–2899. https://doi.org/10.1113/jphysiol.2011.206730
Lindinger MI, Leung MJ, Hawke TJ (2013) Inward flux of lactate- through monocarboxylate transporters contributes to regulatory volume increase in mouse muscle fibres. PLoS ONE. https://doi.org/10.1371/journal.pone.0084451
Ling G (2004) What determines the normal water content of a living cell? Physiol Chem Phys Med NMR 36:1–9
Lombard J (2014) Once upon a time the cell membranes: 175 years of cell boundary research. Biol Direct. https://doi.org/10.1186/s13062-014-0032-7
Low SY, Rennie MJ, Taylor PM (1997) Involvement of integrins and the cytoskeleton in modulation of skeletal muscle glycogen synthesis by changes in cell volume. FEBS Lett 417:101–103. https://doi.org/10.1016/S0014-5793(97)01264-7
Lundvall J, Mellander S, Westling H, White T (1970) Dynamics of fluid transfer between the intra- and extravascular compartments during exercise. Acta Physiol Scand. https://doi.org/10.1111/j.1748-1716.1970.tb04854.x
McBride TA, Stockert BW, Gorin FA, Carlsen RC (2000) Stretch-activated ion channels contribute to membrane depolarization after eccentric contractions. J Appl Physiol 88:91–101. https://doi.org/10.1152/jappl.2000.88.1.91
McCartney N, Spriet LL, Heigenhauser GJF et al (1986) Muscle power and metabolism in maximal intermittent exercise. J Appl Physiol 60:1164–1169. https://doi.org/10.1152/jappl.1986.60.4.1164
Meyerhof O (1947) The main chemical phases of the recovery of muscle. Ann NY Acad Sci 47:815–834. https://doi.org/10.1111/j.1749-6632.1947.tb31738.x
Müller W (1976) Subsarcolemmal mitochondria and capillarization of soleus muscle fibers in young rats subjected to an endurance training—a morphometric study of semithin sections. Cell Tissue Res 174:367–389. https://doi.org/10.1007/BF00220682
Needham DM (1971) Machina Carnis. Cambridge University Press, Cambridge
Nernst W (1890) Ein osmotischer Versuch. Zeit Phys Chem 6:37–40
Olsen LA, Nicoll JX, Fry AC (2019) The skeletal muscle fiber: a mechanically sensitive cell. Eur J Appl Physiol 119:333–349. https://doi.org/10.1007/s00421-018-04061-x
Osterhout WJV (1935) How do electrolytes enter the cell? Proc Natl Acad Sci 21:125–132. https://doi.org/10.1073/pnas.21.2.125
Overton E (1895) Ueber die osmotischen Eigenschaften der lebenden Pflanzen- und Tierzelle. Vierteljahrsschrift der Naturforschenden Gesellschaft Zürich
Overton E (1896) [About the osmotic properties of the cell and its importance for toxicology and pharmacology] Ueber die osmotischen Eigenschaftern der Zelle in ihrer Bedeutung fur die Toxikologie und pharmakologie. Vierteljahrsschrift Der Naturforschenden Gesellschaft Zürich 41:383–406
Overton E (1899) Ueber die allgemeinen osmotischen Eigenschaften der Zelle, ihre vermutlichen Ursachen und ihre Bedeutung fur die Physiologie. Vierteljahrsschrift Der Naturforschenden Gesellschaft Zürich 44:88–135
Overton E (1902) Beiträge zur allgemeinen muskel- und nervenphysiologie—II. mittheilung. Ueber die unentbehrlichkeit von natrium- (oder lithium-)ionen für den contractionsact des muskels. Pflüger, Arch Für Die Gesammte Physiol Des Menschen Und Der Thiere 92:346–386. https://doi.org/10.1007/BF01659816
Pedersen TH, De Paoli F, Nielsen OB (2005) Increased excitability of acidified skeletal muscle: role of chloride conductance. J Gen Physiol 125:237–246. https://doi.org/10.1085/jgp.200409173
Pfeffer W (1877) Osmotische Untersuchungen. Engelmann, Leipzig
Pierce GN, Philipson KD (1985) Binding of glycolytic enzymes to cardiac sarcolemmal and sarcoplasmic reticular membranes. J Biol Chem 260:6862–6870. https://doi.org/10.1016/s0021-9258(18)88860-3
Pirkmajer S, Chibalin AV (2016) Na, K-ATPase regulation in skeletal muscle. Am J Physiol Endocrinol Metab 311:E1–E31. https://doi.org/10.1152/ajpendo.00539.2015
Pollack GH (2003) The role of aqueous interfaces in the cell. Adv Colloid Interface Sci 103:173–196. https://doi.org/10.1016/S0001-8686(02)00095-7
Poole D, Musch T, Colburn T (2022) A century of exercise physiology: key concepts in oxygen flux from capillary to mitochondria. Eur J Appl Physiol
Porret R (1816) Expériences Galvaniques Curieuses. Ann Chim Phys 2:137–140
Ranke J (1865) Tetanus. Eine physiologische Studie, vol 1. Wilhelm Engelmann
Rapp G, Ashley CC, Bagni MA et al (1998) Volume changes of the myosin lattice resulting from repetitive stimulation of single muscle fibers. Biophys J 75:2984–2995. https://doi.org/10.1016/S0006-3495(98)77739-2
Rennie MJ, Low SY, Taylor PM et al (1998) Amino acid transport during muscle contraction and its relevance to exercise. Adv Exp Med Biol 441:299–305. https://doi.org/10.1007/978-1-4899-1928-1_27
Robinson JR (1960) Metabolism of intracellular water. Physiol Rev 40:112–149. https://doi.org/10.1152/physrev.1960.40.1.112
Schmidt C (1851) Charakteristik der peidemischen Cholera gegenuber verwandten Transsudationasanomalieen. Ene physiologisch-chemische Untersuchung, G.A. Reyher, Leipzig
Schulze E, Likiernik A (1891) Ueber das Lecithin der Pflanzensamen. Bchm 15:405–414. https://doi.org/10.1515/bchm1.1891.15.5.405
Sejersted OM, Sjogaard G (2000) Dynamics and consequences of potassium shifts in skeletal muscle and heart during exercise. Physiol Rev 80:1411–1481. https://doi.org/10.1152/physrev.2000.80.4.1411
Sejersted OM, Vollestad NK, Medbo JI (1986) Muscle fluid and electrolyte balance during and following exercise. Acta Physiol Scand 128:119–127
Semb SO, Sejersted OM (1996) Fuzzy space and control of Na+, K+-pump rate in heart and skeletal muscle. Acta Physiol Scand. Blackwell Publishing Ltd, pp 213–225
Shephard RJ (2016) Responses of the human spleen to exercise. J Sports Sci 34:929–936. https://doi.org/10.1080/02640414.2015.1078488
Sitdikov RF, Urazaev KhA, Volkov EM et al (1989) The influence of hyperosmolarity and furosemide on the membrane potential and fiber volume in rats muscle. Byulleten Eksp Biol i Meditsiny 107:563–566
Sjogaard G (1983) Electrolytes in slow and fast muscle fibers of humans at rest and with dynamic exercise. Am J Physiol Regul Integr Comp Physiol 14:25–31. https://doi.org/10.1152/ajpregu.1983.245.1.r25
Sjogaard G, Saltin B (1982) Extra- and intracellular water spaces in muscles of man at rest and with dynamic exercise. Am J Physiol Regul Integr Comp Physiol. https://doi.org/10.1152/ajpregu.1982.243.3.r271
Sjogaard G, Adams RP, Saltin B (1985) Water and ion shifts in skeletal muscle of humans with intense dynamic knee extension. Am J Physiol Regul Integr Comp Physiol 17:190–196. https://doi.org/10.1152/ajpregu.1985.248.2.r190
Skou JC (1957) The influence of some cations on an adenosine triphosphatase from peripheral nerves. BBA Biochim Biophys Acta 23:394–401. https://doi.org/10.1016/0006-3002(57)90343-8
Spiegler KS (1958) Transport processes in ionic membranes. Trans Faraday Soc 54:1408–1428. https://doi.org/10.1039/tf9585401408
Spronck AC (1965) Évolution temporelle de l’hydrolyse de la phosphocréatine et de la synthèse d’hexosediphosphate pendant et après CINQ secousses simples, a o°c, chez le sartorius de rana temporaria, intoxiqué Par l’acide monoiodoacétique. Arch Physiol Biochem 73:241–259. https://doi.org/10.3109/13813456509084250
Suleymanian MA, Clemo HF, Cohen NM, Baumgarten CM (1995) Stretch-activated channel blockers modulate cell volume in cardiac ventricular myocytes. J Mol Cell Cardiol 27:721–728. https://doi.org/10.1016/S0022-2828(08)80062-4
Talwar S, Lynch JW (2015) Investigating ion channel conformational changes using voltage clamp fluorometry. Neuropharmacology 98:3–12. https://doi.org/10.1016/j.neuropharm.2015.03.018h
Taylor NAS, Notley SR, Lindinger MI (2021) Heat adaptation in humans: extrapolating from basic to applied science. Eur J Appl Physiol. https://doi.org/10.1007/s00421-021-04612-9
Teorell T (1949) Permeability. Annu Rev Physiol 11:545–564. https://doi.org/10.1146/annurev.ph.11.030149.002553
Tosteson DC (1955) The effects of sickling on ion transport. II. The effect of sickling on sodium and cesium transport. J Gen Physiol 39:55–67. https://doi.org/10.1085/jgp.39.1.55
Tosteson DC, Hoffman JF (1960) Regulation of cell volume by active cation transport in high and low potassium sheep red cells. J Gen Physiol 44:169–194. https://doi.org/10.1085/jgp.44.1.169
Traube M (1867) Experimente zur Theorie der Zellenbildung und Endosmose. Arch Anat Physiol Wiss Med 1:87–165
Trump ME, Heigenhauser GJF, Putman CT, Spriet LL (1996) Importance of muscle phosphocreatine during intermittent maximal cycling. J Appl Physiol 80:1574–1580. https://doi.org/10.1152/jappl.1996.80.5.1574
Urazaev AK, Surovtsev VA, Chikin AV et al (1987) Neǐrotroficheskiǐ kontrol’ transmembrannogo perenosa khlora v myshechnykh voloknakh mlekopitaiushchikh. Neirofiziologiya 19:766–771
Usher-Smith JA, Huang CLH, Fraser JA (2009) Control of cell volume in skeletal muscle. Biol Rev 84:143–159. https://doi.org/10.1111/j.1469-185X.2008.00066.x
Ward DS, Hamilton MT, Watson PD (1996) Measurement of tissue volume during non-steady state high-intensity muscle contraction. Am J Physiol Regul Integr Comp Physiol. https://doi.org/10.1152/ajpregu.1996.271.6.r1682
Watson PD (1993) Sieving of electrolytes at capillary wall of cat skeletal muscle by osmotic water flow. Am J Physiol Hear Circ Physiol. https://doi.org/10.1152/ajpheart.1993.265.6.h1869
Watson PD, Garner RP, Ward DS (1993) Water uptake in stimulated cat skeletal muscle. Am J Physiol Regul Integr Comp Physiol. https://doi.org/10.1152/ajpregu.1993.264.4.r790
Weech AA, Michaelis L (1928) Studies on permeability of membranes: V. the diffusion of non-electrolytes through the dried collodion membrane. J Gen Physiol 12:55–81. https://doi.org/10.1085/jgp.12.1.55
Weiss JN, Lamp ST (1987) Glycolysis preferentially inhibits ATP-sensitive K+ channels in isolated guinea pig cardiac myocytes. Science (80-) 238:67–69. https://doi.org/10.1126/science.2443972
Zanou N, Mondin L, Fuster C et al (2015) Osmosensation in TRPV2 dominant negative expressing skeletal muscle fibres. J Physiol 593:3849–3863. https://doi.org/10.1113/JP270522
Zuntz N, Schumburg, (1901) Zuntz und Schumburg, Studien zu einer Physiologie des Marsches. Dtsch Medizinische Wochenschrift 27:900. https://doi.org/10.1055/s-0029-1187243
Acknowledgements
MIL is grateful to Dr. Irena Rebalka for carefully reviewing and editing an earlier version of this paper, and to the reviewers who provided many valuable suggestions for improvements. MIL also expresses many thanks to all of the former graduate studies that have contributed to the inspiration and work for some of the research described in the paper. Figures 1, 2, 3, 4 and 7 were created using BioRender.com.
Funding
No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
MIL conceived, wrote and edited the submitted document in its entirety.
Corresponding author
Ethics declarations
Conflict of interest
The authors serves on the Editorial Board for the European Journal of Applied Physiology.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by Philip D. Chilibeck.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lindinger, M.I. A century of exercise physiology: key concepts in muscle cell volume regulation. Eur J Appl Physiol 122, 541–559 (2022). https://doi.org/10.1007/s00421-021-04863-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-021-04863-6