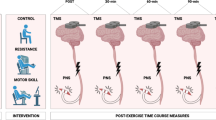
Abstract
The initial increases in force production with resistance training are thought to be primarily underpinned by neural adaptations. This notion is firmly supported by evidence displaying motor unit adaptations following resistance training; however, the precise locus of neural adaptation remains elusive. The purpose of this review is to clarify and critically discuss the literature concerning the site(s) of putative neural adaptations to short-term resistance training. The proliferation of studies employing non-invasive stimulation techniques to investigate evoked responses have yielded variable results, but generally support the notion that resistance training alters intracortical inhibition. Nevertheless, methodological inconsistencies and the limitations of techniques, e.g. limited relation to behavioural outcomes and the inability to measure volitional muscle activity, preclude firm conclusions. Much of the literature has focused on the corticospinal tract; however, preliminary research in non-human primates suggests reticulospinal tract is a potential substrate for neural adaptations to resistance training, though human data is lacking due to methodological constraints. Recent advances in technology have provided substantial evidence of adaptations within a large motor unit population following resistance training. However, their activity represents the transformation of afferent and efferent inputs, making it challenging to establish the source of adaptation. Whilst much has been learned about the nature of neural adaptations to resistance training, the puzzle remains to be solved. Additional analyses of motoneuron firing during different training regimes or coupling with other methodologies (e.g., electroencephalography) may facilitate the estimation of the site(s) of neural adaptations to resistance training in the future.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Resistance exercise is one of the most common exercise modalities, providing numerous functional and physiological benefits to various populations, from athletes to patients. Following a period of resistance training, the maximal volitional force generating capacity of skeletal muscles is typically increased. Though long-term resistance training is accompanied by modifications of muscle morphology, the initial (< 2–4 weeks) increases in force production are thought to be primarily underpinned by neural adaptations (Moritani and DeVries 1979; for reviews see Enoka 1988; Sale 1988; Carroll et al. 2001; Folland and Williams 2007). The existence of neural adaptations is purported due to several behavioural observations such as task-specificity of strength in the absence of significant morphological adaptations (e.g., Ansdell et al. 2020), the disproportionate increase in muscle force relative to muscle size (Moritani and DeVries 1979; Häkkinen et al. 1998) and the increase in voluntary activation (Lee et al. 2009) in the initial weeks of training, the phenomenon of cross-education, whereby unilateral resistance training of one limb increases force production of contralateral homologous muscle group (Manca et al. 2018), and increased muscle force generating capacity following weeks of imagined contractions (e.g., Zijdewind et al. 2003).
Whilst there is a paucity of methods allowing direct assessment of neural activity in awake humans, the nervous system can be accessed through recordings of motor unit action potentials. The motor unit (alpha motoneuron and all muscle fibres innervated by its axons), also known as the final common pathway of neural activation signals (Liddell and Sherrington 1925), is the transducer of synaptic sensory and descending inputs transmitted to the motoneuron pools into mechanical muscle actions (Heckman and Enoka 2012). The characteristics of motor unit action potentials, i.e., summation and time-course, determine the electromyogram (EMG; Farina et al. 2014; Enoka and Duchateau 2015), which can be recorded through the surface of the skin over the muscle (surface recordings) or from within the muscle (needle/fine-wire recordings; Adrian and Bronk 1928). With the existence of the high safety factor of transmission at the neuromuscular junction (Wood and Slater 2001), recordings of motor unit action potentials through EMG infer the discharges of individual motoneurons (Duchateau and Enoka 2011), making them the only nerve cells that can be recorded non-invasively in humans (Heckman and Enoka 2012). Initial studies investigating the compound (interference) EMG signal showed an increased amplitude in the early stages of resistance training concomitantly with increased muscle force generating capacity, suggesting neural contribution (Moritani and DeVries 1979; Häkkinen et al. 1998). However, the interference EMG amplitude is only a crude indicator of the neural drive to skeletal muscle (Farina et al. 2014; Del Vecchio et al. 2017), which precluded robust conclusion about the nature of adaptations. Subsequently, experiments employing advanced EMG recordings [e.g., intra-muscular or high-density surface EMG (HDsEMG)] and decomposition, that allow precise identification of motor unit discharge times, demonstrated that increased force production following resistance training is accompanied by decreased motor unit recruitment threshold and increased discharge rate (Van Cutsem et al. 1998; Kamen and Knight 2004; Vila-Chã et al. 2010; Del Vecchio et al. 2019), providing direct evidence of neural adaptations to resistance training at the level of individual motoneurons.
Despite a clear demonstration of adaptation in the final pathway of the nervous system, the precise site of early neural adaptation causing changes in motor unit activity that accompany increased muscle force generating capacity following short-term resistance training remains elusive, with several mechanisms proposed (Fig. 1). Understanding the aetiology of neural adaptations is a critical consideration for the use of resistance training as a rehabilitation strategy for clinical populations, e.g., following stroke (Kim et al. 2019b), and for optimising resistance training programmes in athletes. Therefore, the purpose of this review is to clarify and critically discuss the literature concerning the site(s) of putative neural adaptations to short-term resistance training when morphological adaptations are expected to be largely absent. Particular attention will be given to the constraints of the current methodology to elucidate the site of neural adaptations, with suggestions for future investigations. Given the focus of the review is on the mechanisms underpinning the phenomenon of increased force production following resistance training, the review will be largely concentrated on studies employing single-joint/single-muscle isometric contractions during assessments, which allow the level of experimental control needed to isolate specific site(s) of neural adaptations. Regarding training interventions used throughout the literature, the studies included in the present review used similar training intensities (i.e., 75–80% 1RM) and volumes (i.e., 6–12 reps, 4 sets). Single-joint, isometric resistance training is the most commonly employed training modality (e.g., Nuzzo et al. 2017; Casolo et al. 2019; Del Vecchio et al. 2019), though multi-joint dynamic and ballistic interventions have also been assessed (e.g., Schubert et al. 2008; Weier et al. 2012; Ansdell et al. 2020). Whilst it is recognised that these factors may influence both the degree and mechanisms of neural adaptation, there is limited evidence systematically comparing the effects of these factors on neural adaptations to strength training, and such a discussion would be beyond the scope of the review. The fact that the majority of mechanistic studies reviewed in this article rely on simplified models of resistance exercise highlights the difficulty in obtaining neurophysiological data in response to more ecologically valid modes of resistance training (e.g., dynamic, compound resistance training), where methodological constraints challenge the ability to capture such data in a task-specific manner (Brownstein et al. 2018; Ansdell et al. 2020). The aforementioned factors should thus be considered when extrapolating findings from mechanistic studies into applied practice.

Adapted from Glover and Baker (2020)
Possible sites of neural adaptation to resistance training. Many potential sites of neural adaptations to resistance training have been suggested. Changes in intracortical inhibitory interneurons (IN; A) have been demonstrated following resistance training in both human (Weier et al. 2012) and non-human primates (Glover and Baker 2020). Adaptations within the corticospinal tract (CST), the main conduit of movement signals in humans, have been equivocal, but may occur at the level of the corticomotoneuronal synapse (B) or via corticospinal projections to interneurons (C) (Nuzzo et al. 2017; Colomer-Poveda et al. 2019; Siddique et al. 2020). Though human data is lacking, experiments in primates suggest contribution of the reticulospinal tract (RST), a bilateral descending tract implicated in gross motor tasks, to increased force production following resistance training (Glover and Baker 2020), which may occur via corticoreticular connections (D), reciprocal reticular connections (E), reticulospinal projections to interneurons (F), or monosynaptic reticular projections to motoneurons (α-MNs; G). The potential neural substrate for resistance training adaptations is also the increased monoaminergic drive via brainstem projections, increasing the strength of persistent inward currents within motoneurons and thus up-regulating depolarisation and shortening the afterhyperpolarisation phase of motoneurons (H and I). Electrophysiological adaptations are also possible within the motor units themselves (J) and might be particularly potent in high-threshold motor units (Casolo et al. 2019). Finally, adjustments in the sensory feedback from muscle via Ia afferent neurons may occur with resistance training (Aagaard et al. 2002; Durbaba et al. 2013) either through monosynaptic connections to motoneurons (K) or via spinal interneurons (L).
Cortical or spinal adaptations: stimulation studies reveal inconsistent results
The early studies attempting to discern the site of neural adaptations to strength training employed stimulation of peripheral nerves and the study of reflex responses. The most commonly studied responses in the context of resistance training are the Hoffmann (H) reflex (Fig. 2a) and the V-wave (Fig. 2b), both of which involve stimulation of a mixed nerve and principally examine the monosynaptic spinal circuitry between the Ia afferent and alpha motoneuron (Fig. 1K). The H-reflex and V-wave differ in that the former is elicited with submaximal stimulation, whereas the latter is evoked with a supramaximal stimulation during a voluntary contraction (McNeil et al. 2013). Following resistance training, studies have generally shown no changes in the H-reflex amplitude when elicited at rest (Aagaard et al. 2002; Scaglioni et al. 2002), but an increase during strong contractions (Aagaard et al. 2002; Duclay et al. 2008; Schubert et al. 2008), consistent with the notion of task-specificity of neural adaptations. Similarly, V-waves, that can only be elicited during a contraction, have typically been shown to increase following resistance training (Sale et al. 1983; Aagaard et al. 2002; Fimland et al. 2009).
The responses commonly used to assess the site of neural adaptation to resistance training. Early studies have shown increased responses to percutaneous mixed nerve stimulation during a contraction following resistance training (e.g., Sale et al. 1983; Aagaard et al. 2002)—these responses are known as the H-reflex (a), which is a long-latency response to submaximal nerve stimulation often evoked with a small M-wave (note the short-latency response), and the V-wave (b), which is a long-latency response to supramaximal nerve stimulation (hence the presence of a short-latency maximal M-wave; for further details on methodology see Burke and Gandevia 1999). In recent decades, transcranial magnetic stimulation (for details on methodology see Rossini et al. 2015) has been used to infer the site of neural adaptation to resistance training; however, the response to such stimuli, known as the motor evoked potentials followed by the silent period (c), have yielded variable results when assessed after resistance training (for meta-analysis see Siddique et al. 2020). Since responses to transcranial magnetic stimulation alone cannot differentiate between the cortical and spinal site of adaptation, additional methods have had to be employed, such as responses to direct activation of corticospinal axons, e.g., lumbar evoked potentials (d), but they have been shown not to change following resistance training (Nuzzo et al. 2017; Ansdell et al. 2020). It is important to note that changes in responses to stimulation techniques following resistance training are likely to be specific to the training task (Kalmar 2018)—as a result there have been recent attempts to replicate the training task when assessing responses to stimulation (E; from Brownstein et al. 2018, with permission). Data displaying responses is from the personal archive of the authors—the average of 5 responses is displayed in colour with individual response overlaid in black
The authors of the aforementioned studies have often inferred that neural adaptations to resistance training are mediated at the ‘spinal’ level. However, the term ‘spinal’ is rather broad, and both H-reflex and V-wave are subject to technical limitations that preclude identification of the precise site of neural adaptation (for reviews see Burke and Gandevia 1999; Zehr 2002; Knikou 2008). Briefly, due to activation of Ia afferents, the H-reflex is subject to modulation of presynaptic inhibition (for review see Zehr 2002). Thus, increased H-reflex amplitude following resistance training (Aagaard et al. 2002) may be primarily associated to downregulated presynaptic inhibition of sensory inputs rather than adaptations within the motor pathway. Furthermore, H-reflex is sensitive to changes in axonal excitability (Bostock and Grafe 1985), meaning that increased amplitude of the reflex may not necessarily represent changes within the spinal cord. Though less likely to influence the V-wave (Burke and Gandevia 1999), changes in axonal excitability represent a potential confound regardless of whether one attempts to ensure a similar proportion of motoneuron pool activation when eliciting the H-reflex (e.g., standardised M-wave preceding H-reflex; Zehr 2002). Finally, both the H-reflex and V-wave have non-monosynaptic contributions (Fig. 1L; Burke et al. 1984; Marchand-Pauvert et al. 2002); thus, the changes in reflex response could be the result of alterations in synaptic efficacy in one or both of these circuits (Burke and Gandevia 1999). In essence, changes in H-reflex and V-wave do not necessarily measure motoneuron excitability as the latter typically assumes a predominantly monosynaptic contribution (McNeil et al. 2013).
The advent of transcranial magnetic stimulation (TMS; Barker et al. 1985), which allows non-invasive, painless activation of neurons within the motor cortex (for review see Rossini et al. 2015), has resulted in a proliferation of studies investigating motor control and neural alterations to various motor tasks, including resistance training (Carroll et al. 2011; Kidgell and Pearce 2011). The size of the response to TMS, the motor evoked potential (Fig. 2c), can be recorded at the muscle via EMG and represents an index of excitability of the corticospinal tract (see CST in Fig. 1, Bestmann and Krakauer 2015). Perhaps the most consistent experimental observation using TMS is that the motor cortical inhibition is decreased with resistance training (Fig. 1A; Goodwill et al. 2012; Weier et al. 2012; Leung et al. 2017), though contradicting evidence does exist (Beck et al. 2007; Ansdell et al. 2020). The majority of these studies utilised similar training intensities and volumes (75–80% 1RM, 6–12 repetitions, 4 sets; Goodwill et al. 2012; Weier et al. 2012; Leung et al. 2017; Ansdell et al. 2020), though other characteristics of the training differed (e.g., unilateral, Goodwill et al. 2012; Leung et al. 2017; bilateral, Weier et al. 2012; Ansdell et al. 2020; lower limb, Goodwill et al. 2012; Weier et al. 2012; Ansdell et al. 2020; upper limb, Leung et al. 2017), possibly explaining the discrepant results.
The activity of inhibitory interneurons in the motor cortex is principally assessed with paired-pulse TMS, whereby two pulses are delivered with a short interstimulus interval, and the responses are thought to be underpinned by the activity of receptors of gamma aminobutyric acid (Ziemann et al. 1996; Di Lazzaro et al. 2007), the main inhibitory neurotransmitter in the human central nervous system. This neurotransmitter has been heavily implicated in motor learning (Bachtiar and Stagg 2014), which supports the notion proposed a few decades ago that resistance training is a form of motor leaning (Sale 1988). Thus, one potential argument is that the neural adaptations and increased strength observed in the initial stages of resistance training may reflect the processes implicated in motor learning. In addition to paired-pulse responses to TMS, the duration of the silent period following the evoked response to TMS has also been shown to be reduced following resistance training (Christie and Kamen 2014; Siddique et al. 2020) and used to infer a reduction in the activity of intracortical interneurons; however, this interpretation of the phenomenon has been questioned, with suggestions that adjustments within the spinal network could be complicit (Yacyshyn et al. 2016; Škarabot et al. 2019b). Nevertheless, meta-analyses generally support the premise that resistance training alters excitability of the intracortical inhibitory interneurons, particularly when these are assessed during a voluntary contraction (Kidgell et al. 2017; Siddique et al. 2020). Similar reductions in intra-cortical inhibition have been demonstrated following acute aerobic exercise (Singh and Staines 2015; El-Sayes et al. 2019), perhaps suggestive of a mechanism linked to exercise in general, rather than specific to resistance training.
Evidence of changes in corticospinal excitability has been inconsistent, with an increase (Griffin and Cafarelli 2007; Weier et al. 2012), decrease (Carroll et al. 2002; Beck et al. 2007; Giboin et al. 2018) or no change (Carroll et al. 2009; Christie and Kamen 2014; Coombs et al. 2016) shown following short-term resistance training. The differences in experimental designs, particularly as they relate to the training protocol, and the lack of agreement between the training and testing task (Avela and Gruber 2011; Kalmar 2018) likely contribute to these discrepancies. For example, whilst the majority of studies employed training intensities between 70 and 100% maximum intensity, these were a mixture of isometric (Griffin and Cafarelli 2007; Christie and Kamen 2014; Giboin et al. 2018) and dynamic contractions (Carroll et al. 2002; Beck et al. 2007; Weier et al. 2012; Coombs et al. 2016). When these factors are coupled with inconsistent methodological approaches to measure indices of corticospinal excitability and/or inhibition, the equivocal nature of the literature is perhaps unsurprising. The mechanistic extrapolation is further complicated, because whilst TMS activates pyramidal neurons in the motor cortex through indirect activation, the response measured in the EMG activity (motor evoked potential) represents inputs from both cortical as well as spinal centres (Rossini et al. 2015). Indeed, changes in motor evoked potentials following resistance training could represent alterations within the motor cortex itself, within the spinal cord, or in the efficacy of the synapses leading to the motoneuron (e.g., Fig. 1B, C). Additional methods, such as assessing responses to direct activation of corticospinal axons at subcortical levels (Fig. 2d; Taylor and Gandevia 2004; Martin et al. 2008; Škarabot et al. 2019a) is required in conjunction with TMS to make a distinction as to whether the site of neural adaptations lies within the motor cortex or subcortically. Few studies have examined responses to direct activation of corticospinal axons following short-term resistance training, but their findings agree that neural adaptations are not mediated by intrinsic changes to motoneurons, efficacy of corticomotoneuronal synapses or transmission efficacy along descending pathways (Nuzzo et al. 2017; Ansdell et al. 2020).
Taken together, the general inconsistencies in the literature on the site of neural adaptations to resistance training inferred from stimulation techniques are widespread. However, the equivocal nature of findings from studies employing TMS does not necessarily exclude the implication of the motor cortex and/or the corticospinal tract in neural adaptations to resistance training. A consideration of the limitations of techniques used to study corticospinal changes following resistance training, as well as the context of their use, might provide explanations for equivocal results or offer alternative considerations. Firstly, the relationship between TMS-induced responses and behavioural outcomes is complex and not always directly interrelated. Indeed, TMS may activate elements of the motor output that are not necessarily directly related to volitional neural activity. For example, TMS responses provide information about the population of neurons activated by stimulation, which represent presynaptic interneural inputs and postsynaptic corticospinal excitability, which may not be directly relevant to motor behaviour (Bestmann and Krakauer 2015). Other technologies that permit inferences of central nervous system behaviour during volitional actions could overcome this limitation, as discussed in subsequent sections. Additionally, changes might occur in cortical areas outside the primary motor cortex, which may or may not cause changes in the population of neurons activated by stimulation (Bestmann and Krakauer 2015). Secondly, responses to stimulation techniques are known to be variable; this has been suggested to be due to inter-individual variability in synaptic efficacy of different neuronal populations and subtle changes in electrophysiological properties of neuronal populations within an individual (Orth et al. 2003). Because of this variability, methodological nuances can influence the sensitivity to detect changes, especially if these are subtle. Thirdly, the training and the assessment tasks typically differ in the generation of the motor command, which can mask potential changes in neural responses. For example, even when attempts have been made to replicate biomechanical characteristics of the training task when measuring TMS-induced responses (Fig. 2e; Brownstein et al. 2018), recordings were made during a low-intensity isometric contraction, which differed considerably to the training task involving dynamic squats with 80% of 1-repetition maximum (Ansdell et al. 2020). However, when the intent to produce force, and thus likely the motor command, was replicated in the assessment task motor evoked potentials showed a clear task-specific change (Giboin et al. 2018). Future studies could consider investigation of TMS-induced responses during the movement preparation phase, which represents an experimental lens into the motor command (Tanji and Evarts 1976; Cisek and Kalaska 2010). Finally, although the corticospinal tract represents the primary pathway controlling skeletal muscle, it is possible that the main site of neural adaptation lies outside the direct corticomotoneuronal connection. Other descending tracts could be considered sites of neural adaptation to resistance training, such as the reticulospinal tract. Several characteristics of the reticulospinal tract provide a rationale for its implication in the neural causes of strength increase: its bilateral nature could facilitate certain exercises (Jankowska et al. 2003); its collateralisation could enable the activation of muscle synergies during gross motor tasks (Peterson et al. 1975); as well as its direct and indirect (via an interneuron) projections with motoneurons (Riddle et al. 2009). Furthermore, when lesions were made in the pyramidal (Lawrence and Kuypers 1968) and corticospinal tract (Zaaimi et al. 2012, 2018) of non-human primates, a ‘compensatory’ increase in the efficacy of reticulospinal connections with the motoneuron was observed that accompanied the recovery of strength. These findings provide the neuroanatomical, neurophysiological and behavioural basis that make the reticulospinal tract a potent substrate for neural adaptations to resistance training. Indeed, direct stimulation of the reticulospinal tract in non-human primates reveals increased responses following resistance training suggesting adaptation in this tract, likely through monosynaptic (Fig. 1H) and disynaptic (via an interneuron; Fig. 1I) connections to motoneurons, decreased corticoreticular connection (Fig. 1D) and/or increased reciprocal reticular connection (Fig. 1E; Glover and Baker 2020). Though reticulospinal tract function is not possible to assess directly in humans, startle reaction time tasks (Baker and Perez 2017) and auditory startle cues combined with TMS (Tazoe and Perez 2017) and transcranial electrical stimulation (Furubayashi et al. 2000) have been used previously to infer reticulospinal function in humans and might be worth considering in future studies investigating neural adaptations to resistance training.
The range of potential adaptation aetiologies means that relying on TMS or other non-invasive neurostimulation paradigms alone might limit the inferences that can be made by a single experiment. As will be discussed in the next sections, technologies that allow inferences to be made regarding central nervous system behaviour during volitional actions might provide routes for further exploration of neural adaptation to resistance training.
High-density surface electromyography: potential for source identification
Stimulation techniques, including TMS, involve the study of evoked responses. Therefore, stimulation methods will always be, to some extent, limited in their ability to make inferences about behavioural outcomes as they do not allow capturing changes in volitional neural activity. On the other hand, recent technological advances allow a non-invasive study of the activity of large populations of motor units during voluntary contractions in the full recruitment range of a muscle through careful decomposition of HDsEMG (Fig. 3a; Holobar and Zazula 2007; Farina et al. 2016; Del Vecchio et al. 2020). Furthermore, due to the spatial ‘signature’ of each motor unit discharge, it is possible to longitudinally track motor units across recording sessions (Martinez-Valdes et al. 2017; Del Vecchio and Farina 2020), thus allowing direct comparison of potential changes in motor unit properties as a result of an intervention (e.g., training/rehabilitation protocol). Using this methodology, it has recently been shown that increased force production following short-term resistance training was accompanied by decreased recruitment threshold (Fig. 3b) and increased firing rate (Fig. 3c) of a large population of longitudinally-tracked motor units (Del Vecchio et al. 2019). These findings clearly support that early adaptations to resistance training are of neural origin. However, since motor unit activity represents transformation of synaptic sensory and descending inputs, establishing the origin of decreased recruitment and augmented firing rate is challenging.

Adapted from Del Vecchio et al. (2019), with permission. c Concomitantly with decreased recruitment thresholds, motor unit firing rate have also been shown to be augmented with short-term resistance training when the same motor units are tracked across time (Del Vecchio et al. 2019), whereas no such phenomenon is observed in the control group; consistent with the data previously obtained from fine-wire electromyography (Van Cutsem et al. 1998). The scatter plot and data from Del Vecchio et al. (2019), with permission
Motor unit changes following strength training. a Whilst the more invasive fine-wire/needle electromyography is still considered the ‘gold standard’ for discerning the activity of single motor unit action potentials, recent advances in technology have allowed decomposition (line 1 in orange) of the interference electromyogram (the summated motor unit activity) from surface recordings (i.e., high-density EMG). Inferring changes in the nervous system from the global surface EMG is limited due to amplitude cancellation and the non-linear relationship between the size of action potentials and recruitment threshold; however, decomposition of the signal into individual motor unit spike trains infers activity of single motoneurons due to one-to-one relationship between axonal (left) and motor unit (right) action potentials by the muscle unit. From Del Vecchio et al. (2020), with permission. b A raster plot of decomposed motor unit spike trains from high-density EMG during a trapezoidal contraction at 35% of maximal force before and after short-term resistance training (intervention) or no change in physical activity (control). Short-term resistance training decreased motor unit recruitment thresholds (note the dark blue boxes), whereas derecruitment thresholds remained unchanged.
It is important to note that motoneurons receive two types of inputs; ionotropic, which depolarise and hyperpolarise motoneurons, sub-serving specific motor commands and reflexes (Heckman and Enoka 2012), and neuromodulatory, which involve binding of second-messenger systems (e.g., serotonin, noradrenaline) released by the axons of the brainstem raphe nuclei that bind on G-proteins and activate voltage-dependent channels on the motoneuron dendrites (Fig. 1H, I; Heckman and Enoka 2012). The latter allows the generation of strong persistent inward currents, which can increase responsiveness of motoneurons to ionotropic inputs (Heckmann et al. 2005). It is possible to estimate the strength of persistent inward currents in humans with the so-called paired motor unit technique during voluntary contractions with a prescribed trajectory of force increase and decrease (Gorassini et al. 2002; Afsharipour et al. 2020). Specifically, during the ascending phase of the contraction a relatively low-threshold (control) motor unit increases its firing frequency whilst a second, higher-threshold (test) motor unit is recruited, which then continues firing during the descending phase of the contraction at lower levels of synaptic input required to recruit it in the first instance. The strength of the persistent inward currents is then estimated as motor unit recruitment hysteresis, which is quantified as the difference between the instantaneous firing frequency of the control unit at test unit recruitment and derecruitment. Alternatively, motor unit saturation has also been suggested as a potential estimate of persistent inward current strength (Johnson et al. 2017) as it appears to be inherently linked to neuromodulatory input (Hyngstrom et al. 2008; Revill and Fuglevand 2017). Motoneuron afterhyperpolarisation duration has been shown to decrease following short-term resistance training (Christie and Kamen 2010), which might indicate increased flow of positive charged ions onto the motoneurons and thus increased probability of action potential generation, possibly as a result of increased monoaminergic drive. Furthermore, in the study by Del Vecchio et al. (2019) recruitment threshold of motor units were found to be decreased, but no changes were noted in derecruitment threshold relative to force produced, suggesting the hysteresis of motor unit recruitment had changed as a result of resistance training (Kim et al. 2019a). However, the lack of changes in the motoneuron input–output relationship (the relationship between motor unit discharge rate and force production) cast doubt that increased neuromodulatory input contributed to increased force production following short-term resistance training (Del Vecchio et al. 2019). Nevertheless, a more direct investigation into the role of neuromodulatory inputs to motoneurons following resistance training is warranted, particularly since data on rodents suggest that alterations in ionic conductance of motoneurons and augmented electrophysiological properties of both slow and fast-type motoneurons are evident after resistance training (Gardiner et al. 2006; Krutki et al. 2017).
Due to lack of changes in the motoneuron input–output relationship following resistance training, decreased motor unit recruitment and augmented discharge rate are likely of supraspinal origin (Del Vecchio et al. 2019). However, as already discussed, data from stimulation studies is inconclusive concerning the role of the motor cortex in the adaptations to resistance training and is limited insofar as it does not provide information about volitional muscle activity. Cortical activity underpinning volitional muscle activity can be assessed using electroencephalography (EEG), which measures postsynaptic brain activity with high temporal resolution. The negative excitatory post-synaptic potentials in EEG around the time of voluntary movement, known as movement-related cortical potentials, have been shown to display attenuated amplitude at several scalp sites during the same relative force levels following resistance training (Falvo et al. 2010). Furthermore, recent data in non-human primates has indicated a supraspinal contribution to resistance training adaptations (Glover and Baker 2020). These findings imply that the motor cortical demand is reduced with increased force production as a result of resistance training. However, it is unclear whether motor cortical demand is solely reduced, or whether it reflects changes in reticulospinal and/or intraneuronal networks projecting to spinal motoneurons. Pairing EEG with HDsEMG recordings and analysing the coherence between cortical and motoneuronal signals in specific frequency domains (Gallego et al. 2015; Holobar et al. 2018) may facilitate such understanding.
Finally, it is important to highlight that presently available data suggest motor unit adaptations following resistance training are not threshold-specific (Van Cutsem et al. 1998; Del Vecchio et al. 2019). Following short-term resistance training, motor unit conduction velocity has been shown to increase selectively for high-threshold motor units (Casolo et al. 2019); however, this likely reflects changes in electrophysiological properties of muscle fibres (e.g., alterations in the capacity and transport activity of NA+-K+ pump), rather than alterations in neural synaptic input. The uniform increase in discharge rate across the motor pool is also inconsistent with the idea of augmented reticulospinal input following resistance training (Glover and Baker 2020), since this tract seem to preferentially recruit higher-threshold, larger motoneurons (Ziemann et al. 1999). However, different inputs could be augmented concurrently with resistance training; for example, reticulospinal input that may have a bias towards higher-threshold motoneurons, neuromodulatory input that is longer-lasting in low-threshold motoneurons (Lee and Heckman 1998), with possible additional inputs from interneural networks in the motor cortex. Future research should thus consider concomitant contribution from different sources of input that are likely responsible for uniform changes in motoneuron discharge rate across the entire motor pool.
Further considerations and conclusions
The present review has principally discussed neural adaptations to resistance training based on recordings of the agonist muscle(s). Indeed, the literature has predominantly focused on neurophysiological changes in the agonist muscles, with relatively little regard for antagonist and synergists. However, increased force production of the agonist muscle following resistance training may occur due to upregulation of activity within the agonist itself, as well as suppression of the antagonist and/or facilitation of synergist muscles. Early studies investigating interference EMG amplitude suggest reduced antagonist activation following resistance training (Carolan and Cafarelli 1992), though conflicting evidence also exists (Holtermann et al. 2005). Notably, muscles are not controlled by distinct territories within the motor cortex, but are overlapped and intertwined, and more likely interconnected by intrinsic collaterals involved in the integrated control of muscle synergies (Devanne et al. 2006; Capaday et al. 2013). Thus, it is conceivable that focusing on recordings of a single, typically the agonist muscle neglects the possibility of changes in intermuscular coordination as a result of resistance training. Whilst coordination is conceptually difficult to measure with stimulation techniques such as TMS, the distribution of different inputs to the motoneuron pool between synergists has been investigated previously (Laine et al. 2015), but not in the context of resistance training. Future studies should consider concomitant recordings of synergists and antagonists to provide a broader understanding of neural adaptations to resistance training within the whole motor pool.
In conclusion, there is considerable evidence consistent with the notion that the early increases in force production following resistance training are underpinned by neural adaptations. However, despite the proliferation of studies in the field in the last two decades, the precise site of putative neural adaptations remains unclear. Based on the available evidence, it is likely that neural adaptations are underpinned by alterations in the cortical and/or subcortical structures, with changes in inhibitory cortical interneurons and reticular formation being the most potent candidates. The advances in decomposition of neural signals (i.e., HDsEMG), coupled with the use of existing methods (e.g., EEG), as well as indirect probing of reticular formation in humans (e.g., via auditory startle stimuli), have considerable potential to contribute to completing the puzzle regarding the site of neural adaptations to resistance training in the coming decade.
Abbreviations
- EEG:
-
Electroencephalogram
- EMG:
-
Electromyogram
- HDsEMG:
-
High-density surface electromyogram
- H-reflex:
-
The Hoffman reflex
- TMS:
-
Transcranial magnetic stimulation
References
Aagaard P, Simonsen EB, Andersen JL et al (2002) Neural adaptation to resistance training: changes in evoked V-wave and H-reflex responses. J Appl Physiol 92:2309–2318. https://doi.org/10.1152/japplphysiol.01185.2001
Adrian ED, Bronk DW (1928) The discharge of impulses in motor nerve fibres: Part I. Impulses in single fibres of the phrenic nerve. J Physiol 66:81–101. https://doi.org/10.1113/jphysiol.1928.sp002509
Afsharipour B, Manzur N, Duchcherer J et al (2020) Estimation of self-sustained activity produced by persistent inward currents using firing rate profiles of multiple motor units in humans. J Neurophysiol 124:63–85. https://doi.org/10.1152/jn.00194.2020
Ansdell P, Brownstein CG, Škarabot J et al (2020) Task-specific strength increases after lower-limb compound resistance training occurred in the absence of corticospinal changes in vastus lateralis. Exp Physiol 105:1132–1150. https://doi.org/10.1113/EP088629
Avela J, Gruber M (2011) Transcranial magnetic stimulation as a tool to study the role of motor cortex in human muscle function. In: Komi P (ed) Neuromuscular aspects of sport performance. Wiley-Blackwell, Hoboken, pp 115–134. https://doi.org/10.1002/9781444324822.ch8
Bachtiar V, Stagg C (2014) The role of inhibition in human motor cortical plasticity. Neuroscience 278:93–104. https://doi.org/10.1016/j.neuroscience.2014.07.059
Baker SN, Perez MA (2017) Reticulospinal contributions to gross hand function after human spinal cord injury. J Neurosci 37:9778–9784. https://doi.org/10.1523/JNEUROSCI.3368-16.2017
Barker AT, Jalinous R, Freeston IL (1985) Non-invasive magnetic stimulation of human motor cortex. Lancet (London, England) 1:1106–1107. https://doi.org/10.1016/s0140-6736(85)92413-4
Beck S, Taube W, Gruber M et al (2007) Task-specific changes in motor evoked potentials of lower limb muscles after different training interventions. Brain Res 1179:51–60. https://doi.org/10.1016/j.brainres.2007.08.048
Bestmann S, Krakauer JW (2015) The uses and interpretations of the motor-evoked potential for understanding behaviour. Exp Brain Res 233:679–689. https://doi.org/10.1007/s00221-014-4183-7
Bostock H, Grafe P (1985) Activity-dependent excitability changes in normal and demyelinated rat spinal root axons. J Physiol 365:239–257. https://doi.org/10.1113/jphysiol.1985.sp015769
Brownstein CG, Ansdell P, Škarabot J et al (2018) Motor cortical and corticospinal function differ during an isometric squat compared to isometric knee extension. Exp Physiol 103:1251–1263. https://doi.org/10.1113/EP086982
Burke D, Gandevia SC (1999) Properties of human peripheral nerves: implications for studies of human motor control. Prog Brain Res 123:427–435. https://doi.org/10.1016/s0079-6123(08)62878-2
Burke D, Gandevia SC, McKeon B (1984) Monosynaptic and oligosynaptic contributions to human ankle jerk and H-reflex. J Neurophysiol 52:435–448. https://doi.org/10.1152/jn.1984.52.3.435
Capaday C, Ethier C, Van Vreeswijk C, Darling WG (2013) On the functional organization and operational principles of the motor cortex. Front Neural Circuits 7:66. https://doi.org/10.3389/fncir.2013.00066
Carolan B, Cafarelli E (1992) Adaptations in coactivation after isometric resistance training. J Appl Physiol 73:911–917. https://doi.org/10.1152/jappl.1992.73.3.911
Carroll TJ, Riek S, Carson RG (2001) Neural adaptations to resistance training: implications for movement control. Sports Med 31:829–840. https://doi.org/10.2165/00007256-200131120-00001
Carroll TJ, Riek S, Carson RG (2002) The sites of neural adaptation induced by resistance training in humans. J Physiol 544:641–652. https://doi.org/10.1113/jphysiol.2002.024463
Carroll TJ, Barton J, Hsu M, Lee M (2009) The effect of strength training on the force of twitches evoked by corticospinal stimulation in humans. Acta Physiol (Oxf) 197:161–173. https://doi.org/10.1111/j.1748-1716.2009.01992.x
Carroll TJ, Selvanayagam VS, Riek S, Semmler JG (2011) Neural adaptations to strength training: moving beyond transcranial magnetic stimulation and reflex studies. Acta Physiol (Oxf) 202:119–140. https://doi.org/10.1111/j.1748-1716.2011.02271.x
Casolo A, Farina D, Falla D et al (2019) Strength training increases conduction velocity of high-threshold motor units. Med Sci Sport Exerc 52:955–967. https://doi.org/10.1249/MSS.0000000000002196
Christie A, Kamen G (2010) Short-term training adaptations in maximal motor unit firing rates and afterhyperpolarization duration. Muscle Nerve 41:651–660. https://doi.org/10.1002/mus.21539
Christie A, Kamen G (2014) Cortical inhibition is reduced following short-term training in young and older adults. Age (Dordr) 36:749–758. https://doi.org/10.1007/s11357-013-9577-0
Cisek P, Kalaska JF (2010) Neural Mechanisms for Interacting with a World Full of Action Choices. Annu Rev Neurosci 33:269–298. https://doi.org/10.1146/annurev.neuro.051508.135409
Colomer-Poveda D, Romero-Arenas S, Lundbye-Jensen J et al (2019) Contraction intensity-dependent variations in the responses to brain and corticospinal tract stimulation after a single session of resistance training in men. J Appl Physiol 127:1128–1139. https://doi.org/10.1152/japplphysiol.01106.2018
Coombs TA, Frazer AK, Horvath DM et al (2016) Cross-education of wrist extensor strength is not influenced by non-dominant training in right-handers. Eur J Appl Physiol 116:1757–1769. https://doi.org/10.1007/s00421-016-3436-5
Del Vecchio A, Farina D (2020) Interfacing the neural output of the spinal cord: robust and reliable longitudinal identification of motor neurons in humans. J Neural Eng 17:016003. https://doi.org/10.1088/1741-2552/ab4d05
Del Vecchio A, Negro F, Felici F, Farina D (2017) Associations between motor unit action potential parameters and surface EMG features. J Appl Physiol 123:835–843. https://doi.org/10.1152/japplphysiol.00482.2017
Del Vecchio A, Casolo A, Negro F et al (2019) The increase in muscle force after 4 weeks of strength training is mediated by adaptations in motor unit recruitment and rate coding. J Physiol 597:1873–1887. https://doi.org/10.1113/JP277250
Del Vecchio A, Holobar A, Falla D et al (2020) Tutorial: analysis of motor unit discharge characteristics from high-density surface EMG signals. J Electromyogr Kinesiol 53:102426. https://doi.org/10.1016/j.jelekin.2020.102426
Devanne H, Cassim F, Ethier C et al (2006) The comparable size and overlapping nature of upper limb distal and proximal muscle representations in the human motor cortex. Eur J Neurosci 23:2467–2476. https://doi.org/10.1111/j.1460-9568.2006.04760.x
Di Lazzaro V, Pilato F, Dileone M et al (2007) Segregating two inhibitory circuits in human motor cortex at the level of GABAA receptor subtypes: a TMS study. Clin Neurophysiol 118:2207–2214. https://doi.org/10.1016/j.clinph.2007.07.005
Duchateau J, Enoka RM (2011) Human motor unit recordings: origins and insight into the integrated motor system. Brain Res 1409:42–61. https://doi.org/10.1016/j.brainres.2011.06.011
Duclay J, Martin A, Robbe A, Pousson M (2008) Spinal reflex plasticity during maximal dynamic contractions after eccentric training. Med Sci Sports Exerc 40:722–734. https://doi.org/10.1249/MSS.0b013e31816184dc
Durbaba R, Cassidy A, Budini F, Macaluso A (2013) The effects of isometric resistance training on stretch reflex induced tremor in the knee extensor muscles. J Appl Physiol 114:1647–1656. https://doi.org/10.1152/japplphysiol.00917.2011
El-Sayes J, Turco CV, Skelly LE et al (2019) The effects of biological sex and ovarian hormones on exercise-induced neuroplasticity. Neuroscience 410:29–40. https://doi.org/10.1016/j.neuroscience.2019.04.054
Enoka RM (1988) Muscle strength and its development: new perspectives . Sport Med An Int J Appl Med Sci Sport Exerc 6:146–168. https://doi.org/10.2165/00007256-198806030-00003
Enoka RM, Duchateau J (2015) Inappropriate interpretation of surface EMG signals and muscle fiber characteristics impedes progress on understanding the control of neuromuscular function. J Appl Physiol. https://doi.org/10.1152/japplphysiol.00280.2015
Falvo MJ, Sirevaag EJ, Rohrbaugh JW, Earhart GM (2010) Resistance training induces supraspinal adaptations: evidence from movement-related cortical potentials. Eur J Appl Physiol 109:923–933. https://doi.org/10.1007/s00421-010-1432-8
Farina D, Merletti R, Enoka RM (2014) The extraction of neural strategies from the surface EMG: an update. J Appl Physiol 117:1215–1230. https://doi.org/10.1152/japplphysiol.00162.2014
Farina D, Negro F, Muceli S, Enoka RM (2016) Principles of motor unit physiology evolve with advances in technology. Physiology (Bethesda) 31:83–94. https://doi.org/10.1152/physiol.00040.2015
Fimland MS, Helgerud J, Gruber M et al (2009) Functional maximal strength training induces neural transfer to single-joint tasks. Eur J Appl Physiol 107:21–29. https://doi.org/10.1007/s00421-009-1096-4
Folland J, Williams A (2007) The adaptations to strength training: morphological and neurological contributions to increased strength. Sports Med 37:145–168. https://doi.org/10.2165/00007256-200737020-00004
Furubayashi T, Ugawa Y, Terao Y et al (2000) The human hand motor area is transiently suppressed by an unexpected auditory stimulus. Clin Neurophysiol 111:178–183. https://doi.org/10.1016/S1388-2457(99)00200-X
Gallego JA, Dideriksen JL, Holobar A et al (2015) Influence of common synaptic input to motor neurons on the neural drive to muscle in essential tremor. J Neurophysiol 113:182–191. https://doi.org/10.1152/jn.00531.2014
Gardiner P, Dai Y, Heckman CJ (2006) Effects of exercise training on α-motoneurons. J Appl Physiol 101:1228–1236. https://doi.org/10.1152/japplphysiol.00482.2006
Giboin L-S, Weiss B, Thomas F, Gruber M (2018) Neuroplasticity following short-term strength training occurs at supraspinal level and is specific for the trained task. Acta Physiol 222:e12998. https://doi.org/10.1111/apha.12998
Glover I, Baker S (2020) Cortical, corticospinal and reticulospinal contributions to strength training. J Neurosci 40:5820–5832. https://doi.org/10.1523/JNEUROSCI.1923-19.2020
Goodwill AM, Pearce AJ, Kidgell DJ (2012) Corticomotor plasticity following unilateral strength training. Muscle Nerve 46:384–393. https://doi.org/10.1002/mus.23316
Gorassini M, Yang JF, Siu M, Bennett DJ (2002) Intrinsic activation of human motoneurons: possible contribution to motor unit excitation. J Neurophysiol 87:1850–1858. https://doi.org/10.1152/jn.00024.2001
Griffin L, Cafarelli E (2007) Transcranial magnetic stimulation during resistance training of the tibialis anterior muscle. J Electromyogr Kinesiol 17:446–452. https://doi.org/10.1016/j.jelekin.2006.05.001
Häkkinen K, Kallinen M, Izquierdo M et al (1998) Changes in agonist-antagonist EMG, muscle CSA, and force during strength training in middle-aged and older people. J Appl Physiol 84:1341–1349. https://doi.org/10.1152/jappl.1998.84.4.1341
Heckman CJ, Enoka RM (2012) Motor unit. Compr Physiol 2:2629–2682. https://doi.org/10.1002/cphy.c100087
Heckmann CJ, Gorassini MA, Bennett DJ (2005) Persistent inward currents in motoneuron dendrites: implications for motor output. Muscle Nerve 31:135–156. https://doi.org/10.1002/mus.20261
Holobar A, Zazula D (2007) Multichannel blind source separation using convolution Kernel compensation. IEEE Trans Signal Process 55:4487–4496. https://doi.org/10.1109/TSP.2007.896108
Holobar A, Gallego JA, Kranjec J et al (2018) Motor unit-driven identification of pathological tremor in electroencephalograms. Front Neurol 9:879. https://doi.org/10.3389/fneur.2018.00879
Holtermann A, Roeleveld K, Vereijken B, Ettema G (2005) Changes in agonist EMG activation level during MVC cannot explain early strength improvement. Eur J Appl Physiol 94:593–601. https://doi.org/10.1007/s00421-005-1365-9
Hyngstrom AS, Johnson MD, Heckman CJ (2008) Summation of excitatory and inhibitory synaptic inputs by motoneurons with highly active dendrites. J Neurophysiol 99:1643–1652. https://doi.org/10.1152/jn.01253.2007
Jankowska E, Hammar I, Slawinska U et al (2003) Neuronal basis of crossed actions from the reticular formation on feline hindlimb motoneurons. J Neurosci 23:1867–1878. https://doi.org/10.1523/jneurosci.23-05-01867.2003
Johnson MD, Thompson CK, Tysseling VM et al (2017) The potential for understanding the synaptic organization of human motor commands via the firing patterns of motoneurons. J Neurophysiol 118:520–531. https://doi.org/10.1152/jn.00018.2017
Kalmar JM (2018) On task: considerations and future directions for studies of corticospinal excitability in exercise neuroscience and related disciplines. Appl Physiol Nutr Metab 43:1113–1121. https://doi.org/10.1139/apnm-2018-0123
Kamen G, Knight CA (2004) Training-related adaptations in motor unit discharge rate in young and older adults. J Gerontol Ser A Biol Sci Med Sci 59:1334–1338. https://doi.org/10.1093/gerona/59.12.1334
Kidgell DJ, Pearce AJ (2011) What has transcranial magnetic stimulation taught us about neural adaptations to strength training? A brief review. J Strength Cond Res 25:3208–3217. https://doi.org/10.1519/JSC.0b013e318212de69
Kidgell DJ, Bonanno DR, Frazer AK et al (2017) Corticospinal responses following strength training: a systematic review and meta-analysis. Eur J Neurosci 46:2648–2661. https://doi.org/10.1111/ejn.13710
Kim EH, Hassan AS, Heckman CJ (2019a) Changes in motor unit discharge patterns following strength training. J Physiol 597:3509–3510. https://doi.org/10.1113/JP278137
Kim Y, Lai B, Mehta T et al (2019b) Exercise training guidelines for multiple sclerosis, stroke, and parkinson disease: rapid review and synthesis. Am J Phys Med Rehabil 98:613–621. https://doi.org/10.1097/PHM.0000000000001174
Knikou M (2008) The H-reflex as a probe: pathways and pitfalls. J Neurosci Methods 171:1–12. https://doi.org/10.1016/j.jneumeth.2008.02.012
Krutki P, Mrówczyński W, Baczyk M et al (2017) Adaptations of motoneuron properties after weight-lifting training in rats. J Appl Physiol 123:664–673. https://doi.org/10.1152/japplphysiol.00121.2017
Laine CM, Martinez-Valdes E, Falla D et al (2015) Motor neuron pools of synergistic thigh muscles share most of their synaptic input. J Neurosci 35:12207–12216. https://doi.org/10.1523/JNEUROSCI.0240-15.2015
Lawrence DG, Kuypers HGJM (1968) The functional organization of the motor system in the monkey: I. The effects of bilateral pyramidal lesions. Brain 91:1–14. https://doi.org/10.1093/brain/91.1.1
Lee RH, Heckman CJ (1998) Bistability in spinal motoneurons in vivo: systematic variations in persistent inward currents. J Neurophysiol 80:583–593. https://doi.org/10.1152/jn.1998.80.2.583
Lee M, Gandevia SC, Carroll TJ (2009) Unilateral strength training increases voluntary activation of the opposite untrained limb. Clin Neurophysiol 120:802–808. https://doi.org/10.1016/j.clinph.2009.01.002
Leung M, Rantalainen T, Teo W-P, Kidgell D (2017) The corticospinal responses of metronome-paced, but not self-paced strength training are similar to motor skill training. Eur J Appl Physiol 117:2479–2492. https://doi.org/10.1007/s00421-017-3736-4
Liddell E, Sherrington C (1925) Recruitment and some other features of reflex inhibition. Proc R Soc London B B97:488–518
Manca A, Hortobágyi T, Rothwell J, Deriu F (2018) Neurophysiological adaptations in the untrained side in conjunction with cross-education of muscle strength: a systematic review and meta-analysis. J Appl Physiol 124:1502–1518. https://doi.org/10.1152/japplphysiol.01016.2017
Marchand-Pauvert V, Nicolas G, Burke D, Pierrot-Deseilligny E (2002) Suppression of the H reflex in humans by disynaptic autogenetic inhibitory pathways activated by the test volley. J Physiol 542:963–976. https://doi.org/10.1113/jphysiol.2002.021683
Martin PG, Butler JE, Gandevia SC, Taylor JL (2008) Noninvasive stimulation of human corticospinal axons innervating leg muscles. J Neurophysiol 100:1080–1086. https://doi.org/10.1152/jn.90380.2008
Martinez-Valdes E, Negro F, Laine CM et al (2017) Tracking motor units longitudinally across experimental sessions with high-density surface electromyography. J Physiol 595:1479–1496. https://doi.org/10.1113/JP273662
McNeil CJ, Butler JE, Taylor JL, Gandevia SC (2013) Testing the excitability of human motoneurons. Front Hum Neurosci 7:152. https://doi.org/10.3389/fnhum.2013.00152
Moritani T, DeVries HA (1979) Neural factors versus hypertrophy in the time course of muscle strength gain. Am J Phys Med 58:115–130. https://pubmed.ncbi.nlm.nih.gov/453338/
Nuzzo J, Barry B, Jones M et al (2017) Effects of four weeks of strength training on the corticomotoneuronal pathway. Med Sci Sport Exerc 49:2286–2296. https://doi.org/10.1249/MSS.0000000000001367
Orth M, Snijders AH, Rothwell JC (2003) The variability of intracortical inhibition and facilitation. Clin Neurophysiol 114:2362–2369. https://doi.org/10.1016/s1388-2457(03)00243-8
Peterson BW, Maunz RA, Pitts NG, Mackel RG (1975) Patterns of projection and branching of reticulospinal neurons. Exp Brain Res 23:333–351. https://doi.org/10.1007/BF00238019
Revill AL, Fuglevand AJ (2017) Inhibition linearizes firing rate responses in human motor units: implications for the role of persistent inward currents. J Physiol 595:179–191. https://doi.org/10.1113/JP272823
Riddle CN, Edgley SA, Baker SN (2009) Direct and indirect connections with upper limb motoneurons from the primate reticulospinal tract. J Neurosci 29:4993–4999. https://doi.org/10.1523/jneurosci.3720-08.2009
Rossini PM, Burke D, Chen R et al (2015) Non-invasive electrical and magnetic stimulation of the brain, spinal cord, roots and peripheral nerves: basic principles and procedures for routine clinical and research application. An updated report from an I.F.C.N. Committee. Clin Neurophysiol 126:1071–1107. https://doi.org/10.1016/j.clinph.2015.02.001
Sale DG (1988) Neural adaptation to resistance training. Med Sci Sports Exerc 20:S135-145. https://doi.org/10.1249/00005768-198810001-00009
Sale DG, MacDougall JD, Upton AR, McComas AJ (1983) Effect of strength training upon motoneuron excitability in man. Med Sci Sports Exerc 15:57–62. https://pubmed.ncbi.nlm.nih.gov/6843320/
Scaglioni G, Ferri A, Minetti AE et al (2002) Plantar flexor activation capacity and H reflex in older adults: adaptations to strength training. J Appl Physiol 92:2292–2302. https://doi.org/10.1152/japplphysiol.00367.2001
Schubert M, Beck S, Taube W et al (2008) Balance training and ballistic strength training are associated with task-specific corticospinal adaptations. Eur J Neurosci 27:2007–2018. https://doi.org/10.1111/j.1460-9568.2008.06186.x
Siddique U, Rahman S, Frazer AK et al (2020) Determining the sites of neural adaptations to resistance training: a systematic review and meta-analysis. Sport Med 50:1107–1128. https://doi.org/10.1007/s40279-020-01258-z
Singh AM, Staines WR (2015) The effects of acute aerobic exercise on the primary motor cortex. J Mot Behav 47:328–339. https://doi.org/10.1080/00222895.2014.983450
Škarabot J, Ansdell P, Brownstein CG et al (2019a) Electrical stimulation of human corticospinal axons at the level of the lumbar spinal segments. Eur J Neurosci 49:1254–1267. https://doi.org/10.1111/ejn.14321
Škarabot J, Mesquita RNO, Brownstein CG, Ansdell P (2019b) Myths and methodologies: how loud is the story told by the transcranial magnetic stimulation-evoked silent period? Exp Physiol 104:635–642. https://doi.org/10.1113/EP087557
Tanji J, Evarts EV (1976) Anticipatory activity of motor cortex neurons in relation to direction of an intended movement. J Neurophysiol 39:1062–1068. https://doi.org/10.1152/jn.1976.39.5.1062
Taylor JL, Gandevia SC (2004) Noninvasive stimulation of the human corticospinal tract. J Appl Physiol 96:1496–1503. https://doi.org/10.1152/japplphysiol.01116.2003
Tazoe T, Perez MA (2017) Cortical and reticular contributions to human precision and power grip. J Physiol 595:2715–2730. https://doi.org/10.1113/JP273679
Van Cutsem M, Duchateau J, Hainaut K (1998) Changes in single motor unit behaviour contribute to the increase in contraction speed after dynamic training in humans. J Physiol 513:295–305. https://doi.org/10.1111/j.1469-7793.1998.295by.x
Vila-Chã C, Falla D, Farina D (2010) Motor unit behavior during submaximal contractions following six weeks of either endurance or strength training. J Appl Physiol 109:1455–1466. https://doi.org/10.1152/japplphysiol.01213.2009
Weier AT, Pearce AJ, Kidgell DJ (2012) Strength training reduces intracortical inhibition. Acta Physiol 206:109–119. https://doi.org/10.1111/j.1748-1716.2012.02454.x
Wood SJ, Slater CJ (2001) Safety factor at the neuromuscular junction. Prog Neurobiol 64:393–429. https://doi.org/10.1016/s0301-0082(00)00055-1
Yacyshyn AF, Woo EJ, Price MC, McNeil CJ (2016) Motoneuron responsiveness to corticospinal tract stimulation during the silent period induced by transcranial magnetic stimulation. Exp Brain Res 234:3457–3463. https://doi.org/10.1007/s00221-016-4742-1
Zaaimi B, Edgley SA, Soteropoulos DS, Baker SN (2012) Changes in descending motor pathway connectivity after corticospinal tract lesion in macaque monkey. Brain 135:2277–2289. https://doi.org/10.1093/brain/aws115
Zaaimi B, Dean LR, Baker SN (2018) Different contributions of primary motor cortex, reticular formation, and spinal cord to fractionated muscle activation. J Neurophysiol 119:235–250. https://doi.org/10.1152/jn.00672.2017
Zehr EP (2002) Considerations for use of the Hoffmann reflex in exercise studies. Eur J Appl Physiol 86:455–468. https://doi.org/10.1007/s00421-002-0577-5
Ziemann U, Lönnecker S, Steinhoff BJ, Paulus W (1996) Effects of antiepileptic drugs on motor cortex excitability in humans: a transcranial magnetic stimulation study. Ann Neurol 40:367–378. https://doi.org/10.1002/ana.410400306
Ziemann U, Ishii K, Borgheresi A et al (1999) Dissociation of the pathways mediating ipsilateral and contralateral motor-evoked potentials in human hand and arm muscles. J Physiol 518(Pt 3):895–906. https://doi.org/10.1111/j.1469-7793.1999.0895p.x
Zijdewind I, Toering ST, Bessem B et al (2003) Effects of imagery motor training on torque production of ankle plantar flexor muscles. Muscle Nerve 28:168–173. https://doi.org/10.1002/mus.10406
Funding
No funding was used to assist in the preparation of the article.
Author information
Authors and Affiliations
Contributions
All authors conceived and designed the work; JŠ drafted manuscript; JŠ and ADV prepared figures; All authors reviewed and critically revised the manuscript; All authors gave final approval of the version to be published and agree to be accountable for all aspects of work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest, financial or otherwise.
Additional information
Communicated by Michael Lindinger.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Škarabot, J., Brownstein, C.G., Casolo, A. et al. The knowns and unknowns of neural adaptations to resistance training. Eur J Appl Physiol 121, 675–685 (2021). https://doi.org/10.1007/s00421-020-04567-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-020-04567-3