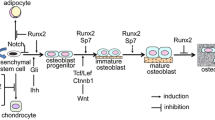
Abstract
Transcription factors play important roles in the regulation of cartilage development by controlling the expression of chondrogenic genes. Genetic studies have revealed that Sox9/Sox5/Sox6, Runx2/Runx3 and Osterix in particular are essential for the sequential steps of cartilage development. Importantly, these transcription factors form network systems that are also required for appropriate cartilage development. Molecular cloning approaches have largely contributed to the identification of several transcriptional partners for Sox9 and Runx2 during cartilage development. Although the importance of a negative-feedback loop between Indian hedgehog (Ihh) and parathyroid hormone-related protein (PTHrP) in chondrocyte hypertrophy has been well established, recent studies indicate that several transcription factors interact with the Ihh–PTHrP loop and demonstrated that Ihh has multiple functions in the regulation of cartilage development. The most common cartilage disorder, osteoarthritis, has been reported to result from the pathological action of several transcription factors, including Runx2, C/EBPβ and HIF-2α. On the other hand, NFAT family members appear to play roles in the protection of cartilage from osteoarthritis. It is also becoming important to understand the homeostasis and regulation of articular chondrocytes, because they have different cellular and molecular features from chondrocytes of the growth plate. This review summarizes the regulation and roles of transcriptional network systems in cartilage development and their pathological roles in osteoarthritis.




Similar content being viewed by others
References
Akiyama H, Lefebvre V (2011) Unraveling the transcriptional regulatory machinery in chondrogenesis. J Bone Miner Metab 29(4):390–395. https://doi.org/10.1007/s00774-011-0273-9
Akiyama H, Chaboissier MC, Martin JF, Schedl A, de Crombrugghe B (2002) The transcription factor Sox9 has essential roles in successive steps of the chondrocyte differentiation pathway and is required for expression of Sox5 and Sox6. Genes Dev 16(21):2813–2828. https://doi.org/10.1101/gad.1017802
Amano K, Ichida F, Sugita A, Hata K, Wada M, Takigawa Y, Nakanishi M, Kogo M, Nishimura R, Yoneda T (2008) MSX2 stimulates chondrocyte maturation by controlling Ihh expression. J Biol Chem 283(43):29513–29521. https://doi.org/10.1074/jbc.M803681200
Amano K, Hata K, Sugita A, Takigawa Y, Ono K, Wakabayashi M, Kogo M, Nishimura R, Yoneda T (2009) Sox9 family members negatively regulate maturation and calcification of chondrocytes through up-regulation of parathyroid hormone-related protein. Mol Biol Cell 20(21):4541–4551. https://doi.org/10.1091/mbc.E09-03-0227
Amano K, Hata K, Muramatsu S, Wakabayashi M, Takigawa Y, Ono K, Nakanishi M, Takashima R, Kogo M, Matsuda A, Nishimura R, Yoneda T (2011) Arid5a cooperates with Sox9 to stimulate chondrocyte-specific transcription. Mol Biol Cell 22(8):1300–1311. https://doi.org/10.1091/mbc.E10-07-0566
Araldi E, Khatri R, Giaccia AJ, Simon MC, Schipani E (2011) Lack of HIF-2alpha in limb bud mesenchyme causes a modest and transient delay of endochondral bone development. Nat Med 17(1):25–26. https://doi.org/10.1038/nm0111-25 (author reply 27–29)
Arnold MA, Kim Y, Czubryt MP, Phan D, McAnally J, Qi X, Shelton JM, Richardson JA, Bassel-Duby R, Olson EN (2007) MEF2C transcription factor controls chondrocyte hypertrophy and bone development. Dev Cell 12(3):377–389. https://doi.org/10.1016/j.devcel.2007.02.004
Chen W, Ma J, Zhu G, Jules J, Wu M, McConnell M, Tian F, Paulson C, Zhou X, Wang L, Li YP (2014) Cbfbeta deletion in mice recapitulates cleidocranial dysplasia and reveals multiple functions of Cbfbeta required for skeletal development. Proc Natl Acad Sci USA 111(23):8482–8487. https://doi.org/10.1073/pnas.1310617111
Dy P, Wang W, Bhattaram P, Wang Q, Wang L, Ballock RT, Lefebvre V (2012) Sox9 directs hypertrophic maturation and blocks osteoblast differentiation of growth plate chondrocytes. Dev Cell 22(3):597–609. https://doi.org/10.1016/j.devcel.2011.12.024
Feldmann M, Brennan FM, Elliott MJ, Williams RO, Maini RN (1995) TNF alpha is an effective therapeutic target for rheumatoid arthritis. Ann N Y Acad Sci 766:272–278
Foster JW, Dominguez-Steglich MA, Guioli S, Kwok C, Weller PA, Stevanovic M, Weissenbach J, Mansour S, Young ID, Goodfellow PN et al (1994) Campomelic dysplasia and autosomal sex reversal caused by mutations in an SRY-related gene. Nature 372(6506):525–530. https://doi.org/10.1038/372525a0
Greenblatt MB, Ritter SY, Wright J, Tsang K, Hu D, Glimcher LH, Aliprantis AO (2013) NFATc1 and NFATc2 repress spontaneous osteoarthritis. Proc Natl Acad Sci USA 110(49):19914–19919. https://doi.org/10.1073/pnas.1320036110
Hata K, Nishimura R, Ueda M, Ikeda F, Matsubara T, Ichida F, Hisada K, Nokubi T, Yamaguchi A, Yoneda T (2005) A CCAAT/enhancer binding protein beta isoform, liver-enriched inhibitory protein, regulates commitment of osteoblasts and adipocytes. Mol Cell Biol 25(5):1971–1979. https://doi.org/10.1128/mcb.25.5.1971-1979.2005
Hata K, Nishimura R, Muramatsu S, Matsuda A, Matsubara T, Amano K, Ikeda F, Harley VR, Yoneda T (2008) Paraspeckle protein p54nrb links Sox9-mediated transcription with RNA processing during chondrogenesis in mice. J Clin Investig 118(9):3098–3108. https://doi.org/10.1172/jci31373
Hata K, Takashima R, Amano K, Ono K, Nakanishi M, Yoshida M, Wakabayashi M, Matsuda A, Maeda Y, Suzuki Y, Sugano S, Whitson RH, Nishimura R, Yoneda T (2013) Arid5b facilitates chondrogenesis by recruiting the histone demethylase Phf2 to Sox9-regulated genes. Nat Commun 4:2850. https://doi.org/10.1038/ncomms3850
Hattori T, Coustry F, Stephens S, Eberspaecher H, Takigawa M, Yasuda H, de Crombrugghe B (2008) Transcriptional regulation of chondrogenesis by coactivator Tip60 via chromatin association with Sox9 and Sox5. Nucleic Acids Res 36(9):3011–3024. https://doi.org/10.1093/nar/gkn150
Hattori T, Muller C, Gebhard S, Bauer E, Pausch F, Schlund B, Bosl MR, Hess A, Surmann-Schmitt C, von der Mark H, de Crombrugghe B, von der Mark K (2010) SOX9 is a major negative regulator of cartilage vascularization, bone marrow formation and endochondral ossification. Development (Cambridge England) 137(6):901–911. https://doi.org/10.1242/dev.045203
Hayashida M, Okazaki K, Fukushi J, Sakamoto A, Iwamoto Y (2009) CCAAT/enhancer binding protein beta mediates expression of matrix metalloproteinase 13 in human articular chondrocytes in inflammatory arthritis. Arthritis Rheum 60(3):708–716. https://doi.org/10.1002/art.24332
He X, Ohba S, Hojo H, McMahon AP (2016) AP-1 family members act with Sox9 to promote chondrocyte hypertrophy. Development (Cambridge England) 143(16):3012–3023. https://doi.org/10.1242/dev.134502
Hino K, Saito A, Kido M, Kanemoto S, Asada R, Takai T, Cui M, Cui X, Imaizumi K (2014) Master regulator for chondrogenesis, Sox9, regulates transcriptional activation of the endoplasmic reticulum stress transducer BBF2H7/CREB3L2 in chondrocytes. J Biol Chem 289(20):13810–13820. https://doi.org/10.1074/jbc.M113.543322
Hirata M, Kugimiya F, Fukai A, Saito T, Yano F, Ikeda T, Mabuchi A, Sapkota BR, Akune T, Nishida N, Yoshimura N, Nakagawa T, Tokunaga K, Nakamura K, Chung UI, Kawaguchi H (2012) C/EBPbeta and RUNX2 cooperate to degrade cartilage with MMP-13 as the target and HIF-2alpha as the inducer in chondrocytes. Hum Mol Genet 21(5):1111–1123. https://doi.org/10.1093/hmg/ddr540
Hosaka Y, Saito T, Sugita S, Hikata T, Kobayashi H, Fukai A, Taniguchi Y, Hirata M, Akiyama H, Chung UI, Kawaguchi H (2013) Notch signaling in chondrocytes modulates endochondral ossification and osteoarthritis development. Proc Natl Acad Sci USA 110(5):1875–1880. https://doi.org/10.1073/pnas.1207458110
Huang W, Zhou X, Lefebvre V, de Crombrugghe B (2000) Phosphorylation of SOX9 by cyclic AMP-dependent protein kinase A enhances SOX9’s ability to transactivate a Col2a1 chondrocyte-specific enhancer. Mol Cell Biol 20(11):4149–4158
Huang W, Chung UI, Kronenberg HM, de Crombrugghe B (2001) The chondrogenic transcription factor Sox9 is a target of signaling by the parathyroid hormone-related peptide in the growth plate of endochondral bones. Proc Natl Acad Sci USA 98(1):160–165. https://doi.org/10.1073/pnas.011393998
Ikeda T, Kamekura S, Mabuchi A, Kou I, Seki S, Takato T, Nakamura K, Kawaguchi H, Ikegawa S, Chung UI (2004) The combination of SOX5, SOX6, and SOX9 (the SOX trio) provides signals sufficient for induction of permanent cartilage. Arthritis Rheum 50(11):3561–3573. https://doi.org/10.1002/art.20611
Ikegami D, Akiyama H, Suzuki A, Nakamura T, Nakano T, Yoshikawa H, Tsumaki N (2011) Sox9 sustains chondrocyte survival and hypertrophy in part through Pik3ca-Akt pathways. Development (Cambridge England) 138(8):1507–1519. https://doi.org/10.1242/dev.057802
Jones SA, Scheller J, Rose-John S (2011) Therapeutic strategies for the clinical blockade of IL-6/gp130 signaling. J Clin Investig 121(9):3375–3383. https://doi.org/10.1172/jci57158
Kamekura S, Kawasaki Y, Hoshi K, Shimoaka T, Chikuda H, Maruyama Z, Komori T, Sato S, Takeda S, Karsenty G, Nakamura K, Chung UI, Kawaguchi H (2006) Contribution of runt-related transcription factor 2 to the pathogenesis of osteoarthritis in mice after induction of knee joint instability. Arthritis Rheum 54(8):2462–2470. https://doi.org/10.1002/art.22041
Kanatani N, Fujita T, Fukuyama R, Liu W, Yoshida CA, Moriishi T, Yamana K, Miyazaki T, Toyosawa S, Komori T (2006) Cbf beta regulates Runx2 function isoform-dependently in postnatal bone development. Dev Biol 296(1):48–61. https://doi.org/10.1016/j.ydbio.2006.03.039
Kawakami Y, Tsuda M, Takahashi S, Taniguchi N, Esteban CR, Zemmyo M, Furumatsu T, Lotz M, Izpisua Belmonte JC, Asahara H (2005) Transcriptional coactivator PGC-1alpha regulates chondrogenesis via association with Sox9. Proc Natl Acad Sci USA 102(7):2414–2419. https://doi.org/10.1073/pnas.0407510102
Kawane T, Komori H, Liu W, Moriishi T, Miyazaki T, Mori M, Matsuo Y, Takada Y, Izumi S, Jiang Q, Nishimura R, Kawai Y, Komori T (2014) Dlx5 and mef2 regulate a novel runx2 enhancer for osteoblast-specific expression. J Bone Miner Res 29(9):1960–1969. https://doi.org/10.1002/jbmr.2240
Kobayashi T, Chung UI, Schipani E, Starbuck M, Karsenty G, Katagiri T, Goad DL, Lanske B, Kronenberg HM (2002) PTHrP and Indian hedgehog control differentiation of growth plate chondrocytes at multiple steps. Development (Cambridge England) 129(12):2977–2986
Kobayashi T, Soegiarto DW, Yang Y, Lanske B, Schipani E, McMahon AP, Kronenberg HM (2005) Indian hedgehog stimulates periarticular chondrocyte differentiation to regulate growth plate length independently of PTHrP. J Clin Investig 115(7):1734–1742. https://doi.org/10.1172/jci24397
Komori T (2011) Signaling networks in RUNX2-dependent bone development. J Cell Biochem 112(3):750–755. https://doi.org/10.1002/jcb.22994
Komori T, Yagi H, Nomura S, Yamaguchi A, Sasaki K, Deguchi K, Shimizu Y, Bronson RT, Gao YH, Inada M, Sato M, Okamoto R, Kitamura Y, Yoshiki S, Kishimoto T (1997) Targeted disruption of Cbfa1 results in a complete lack of bone formation owing to maturational arrest of osteoblasts. Cell 89(5):755–764
Kronenberg HM (2007) The role of the perichondrium in fetal bone development. Ann N Y Acad Sci 1116:59–64. https://doi.org/10.1196/annals.1402.059
Lanske B, Karaplis AC, Lee K, Luz A, Vortkamp A, Pirro A, Karperien M, Defize LH, Ho C, Mulligan RC, Abou-Samra AB, Juppner H, Segre GV, Kronenberg HM (1996) PTH/PTHrP receptor in early development and Indian hedgehog-regulated bone growth. Science 273(5275):663–666
Lauder SN, Carty SM, Carpenter CE, Hill RJ, Talamas F, Bondeson J, Brennan P, Williams AS (2007) Interleukin-1beta induced activation of nuclear factor-kappab can be inhibited by novel pharmacological agents in osteoarthritis. Rheumatology 46(5):752–758. https://doi.org/10.1093/rheumatology/kel419
Masuda K, Ripley B, Nishimura R, Mino T, Takeuchi O, Shioi G, Kiyonari H, Kishimoto T (2013) Arid5a controls IL-6 mRNA stability, which contributes to elevation of IL-6 level in vivo. Proc Natl Acad Sci USA 110(23):9409–9414. https://doi.org/10.1073/pnas.1307419110
Matsubara T, Kida K, Yamaguchi A, Hata K, Ichida F, Meguro H, Aburatani H, Nishimura R, Yoneda T (2008) BMP2 regulates Osterix through Msx2 and Runx2 during osteoblast differentiation. J Biol Chem 283(43):29119–29125. https://doi.org/10.1074/jbc.M801774200
Miyaki S, Nakasa T, Otsuki S, Grogan SP, Higashiyama R, Inoue A, Kato Y, Sato T, Lotz MK, Asahara H (2009) MicroRNA-140 is expressed in differentiated human articular chondrocytes and modulates interleukin-1 responses. Arthritis Rheum 60(9):2723–2730. https://doi.org/10.1002/art.24745
Miyaki S, Sato T, Inoue A, Otsuki S, Ito Y, Yokoyama S, Kato Y, Takemoto F, Nakasa T, Yamashita S, Takada S, Lotz MK, Ueno-Kudo H, Asahara H (2010) MicroRNA-140 plays dual roles in both cartilage development and homeostasis. Genes Dev 24(11):1173–1185. https://doi.org/10.1101/gad.1915510
Muramatsu S, Wakabayashi M, Ohno T, Amano K, Ooishi R, Sugahara T, Shiojiri S, Tashiro K, Suzuki Y, Nishimura R, Kuhara S, Sugano S, Yoneda T, Matsuda A (2007) Functional gene screening system identified TRPV4 as a regulator of chondrogenic differentiation. J Biol Chem 282(44):32158–32167. https://doi.org/10.1074/jbc.M706158200
Nakajima M, Shi D, Dai J, Tsezou A, Zheng M, Norman PE, Takahashi A, Ikegawa S, Jiang Q (2011) Replication studies in various ethnic populations do not support the association of the HIF-2alpha SNP rs17039192 with knee osteoarthritis. Nat Med 17(1):26–27. https://doi.org/10.1038/nm0111-26 (author reply 27–29)
Nakamura Y, Yamamoto K, He X, Otsuki B, Kim Y, Murao H, Soeda T, Tsumaki N, Deng JM, Zhang Z, Behringer RR, Crombrugghe B, Postlethwait JH, Warman ML, Nakamura T, Akiyama H (2011) Wwp2 is essential for palatogenesis mediated by the interaction between Sox9 and mediator subunit 25. Nat Commun 2:251. https://doi.org/10.1038/ncomms1242
Nakashima K, Zhou X, Kunkel G, Zhang Z, Deng JM, Behringer RR, de Crombrugghe B (2002) The novel zinc finger-containing transcription factor osterix is required for osteoblast differentiation and bone formation. Cell 108(1):17–29
Nishimura R, Hata K, Ikeda F, Ichida F, Shimoyama A, Matsubara T, Wada M, Amano K, Yoneda T (2008) Signal transduction and transcriptional regulation during mesenchymal cell differentiation. J Bone Miner Metab 26(3):203–212. https://doi.org/10.1007/s00774-007-0824-2
Nishimura R, Hata K, Matsubara T, Wakabayashi M, Yoneda T (2012a) Regulation of bone and cartilage development by network between BMP signalling and transcription factors. J Biochem 151(3):247–254. https://doi.org/10.1093/jb/mvs004
Nishimura R, Wakabayashi M, Hata K, Matsubara T, Honma S, Wakisaka S, Kiyonari H, Shioi G, Yamaguchi A, Tsumaki N, Akiyama H, Yoneda T (2012b) Osterix regulates calcification and degradation of chondrogenic matrices through matrix metalloproteinase 13 (MMP13) expression in association with transcription factor Runx2 during endochondral ossification. J Biol Chem 287(40):33179–33190. https://doi.org/10.1074/jbc.M111.337063
Nishimura R, Hata K, Takahata Y, Murakami T, Nakamura E, Yagi H (2017) Regulation of cartilage development and diseases by transcription factors. J Bone Metab 24(3):147–153. https://doi.org/10.11005/jbm.2017.24.3.147
Oh JH, Park SY, de Crombrugghe B, Kim JE (2012) Chondrocyte-specific ablation of Osterix leads to impaired endochondral ossification. Biochem Biophys Res Commun 418(4):634–640. https://doi.org/10.1016/j.bbrc.2012.01.064
Ornitz DM, Legeai-Mallet L (2017) Achondroplasia: development, pathogenesis, and therapy. Dev Dyn 246(4):291–309. https://doi.org/10.1002/dvdy.24479
Qin X, Jiang Q, Matsuo Y, Kawane T, Komori H, Moriishi T, Taniuchi I, Ito K, Kawai Y, Rokutanda S, Izumi S, Komori T (2015) Cbfb regulates bone development by stabilizing Runx family proteins. J Bone Miner Res 30(4):706–714. https://doi.org/10.1002/jbmr.2379
Ranger AM, Gerstenfeld LC, Wang J, Kon T, Bae H, Gravallese EM, Glimcher MJ, Glimcher LH (2000) The nuclear factor of activated T cells (NFAT) transcription factor NFATp (NFATc2) is a repressor of chondrogenesis. J Exp Med 191(1):9–22
Rock MJ, Prenen J, Funari VA, Funari TL, Merriman B, Nelson SF, Lachman RS, Wilcox WR, Reyno S, Quadrelli R, Vaglio A, Owsianik G, Janssens A, Voets T, Ikegawa S, Nagai T, Rimoin DL, Nilius B, Cohn DH (2008) Gain-of-function mutations in TRPV4 cause autosomal dominant brachyolmia. Nat Genet 40(8):999–1003. https://doi.org/10.1038/ng.166
Rodova M, Lu Q, Li Y, Woodbury BG, Crist JD, Gardner BM, Yost JG, Zhong XB, Anderson HC, Wang J (2011) Nfat1 regulates adult articular chondrocyte function through its age-dependent expression mediated by epigenetic histone methylation. J Bone Miner Res 26(8):1974–1986. https://doi.org/10.1002/jbmr.397
Saito T, Ikeda T, Nakamura K, Chung UI, Kawaguchi H (2007) S100A1 and S100B, transcriptional targets of SOX trio, inhibit terminal differentiation of chondrocytes. EMBO Rep 8(5):504–509. https://doi.org/10.1038/sj.embor.7400934
Saito A, Hino S, Murakami T, Kanemoto S, Kondo S, Saitoh M, Nishimura R, Yoneda T, Furuichi T, Ikegawa S, Ikawa M, Okabe M, Imaizumi K (2009) Regulation of endoplasmic reticulum stress response by a BBF2H7-mediated Sec23a pathway is essential for chondrogenesis. Nat Cell Biol 11(10):1197–1204. https://doi.org/10.1038/ncb1962
Saito T, Fukai A, Mabuchi A, Ikeda T, Yano F, Ohba S, Nishida N, Akune T, Yoshimura N, Nakagawa T, Nakamura K, Tokunaga K, Chung UI, Kawaguchi H (2010) Transcriptional regulation of endochondral ossification by HIF-2alpha during skeletal growth and osteoarthritis development. Nat Med 16(6):678–686. https://doi.org/10.1038/nm.2146
Saito A, Kanemoto S, Zhang Y, Asada R, Hino K, Imaizumi K (2014) Chondrocyte proliferation regulated by secreted luminal domain of ER stress transducer BBF2H7/CREB3L2. Mol Cell 53(1):127–139. https://doi.org/10.1016/j.molcel.2013.11.008
Shimoyama A, Wada M, Ikeda F, Hata K, Matsubara T, Nifuji A, Noda M, Amano K, Yamaguchi A, Nishimura R, Yoneda T (2007) Ihh/Gli2 signaling promotes osteoblast differentiation by regulating Runx2 expression and function. Mol Biol Cell 18(7):2411–2418. https://doi.org/10.1091/mbc.E06-08-0743
Shirakawa K, Maeda S, Gotoh T, Hayashi M, Shinomiya K, Ehata S, Nishimura R, Mori M, Onozaki K, Hayashi H, Uematsu S, Akira S, Ogata E, Miyazono K, Imamura T (2006) CCAAT/enhancer-binding protein homologous protein (CHOP) regulates osteoblast differentiation. Mol Cell Biol 26(16):6105–6116. https://doi.org/10.1128/mcb.02429-05
Sinningen K, Tsourdi E, Rauner M, Rachner TD, Hamann C, Hofbauer LC (2012) Skeletal and extraskeletal actions of denosumab. Endocrine 42(1):52–62. https://doi.org/10.1007/s12020-012-9696-x
Smits P, Li P, Mandel J, Zhang Z, Deng JM, Behringer RR, de Crombrugghe B, Lefebvre V (2001) The transcription factors L-Sox5 and Sox6 are essential for cartilage formation. Dev Cell 1(2):277–290
Smits P, Dy P, Mitra S, Lefebvre V (2004) Sox5 and Sox6 are needed to develop and maintain source, columnar, and hypertrophic chondrocytes in the cartilage growth plate. J Cell Biol 164(5):747–758. https://doi.org/10.1083/jcb.200312045
Sugita S, Hosaka Y, Okada K, Mori D, Yano F, Kobayashi H, Taniguchi Y, Mori Y, Okuma T, Chang SH, Kawata M, Taketomi S, Chikuda H, Akiyama H, Kageyama R, Chung UI, Tanaka S, Kawaguchi H, Ohba S, Saito T (2015) Transcription factor Hes1 modulates osteoarthritis development in cooperation with calcium/calmodulin-dependent protein kinase 2. Proc Natl Acad Sci USA 112(10):3080–3085. https://doi.org/10.1073/pnas.1419699112
Takigawa Y, Hata K, Muramatsu S, Amano K, Ono K, Wakabayashi M, Matsuda A, Takada K, Nishimura R, Yoneda T (2010) The transcription factor Znf219 regulates chondrocyte differentiation by assembling a transcription factory with Sox9. J Cell Sci 123(Pt 21):3780–3788. https://doi.org/10.1242/jcs.071373
Tanaka T, Yoshida N, Kishimoto T, Akira S (1997) Defective adipocyte differentiation in mice lacking the C/EBPbeta and/or C/EBPdelta gene. EMBO J 16(24):7432–7443. https://doi.org/10.1093/emboj/16.24.7432
Taniguchi Y, Kawata M, Ho Chang S, Mori D, Okada K, Kobayashi H, Sugita S, Hosaka Y, Inui H, Taketomi S, Yano F, Ikeda T, Akiyama H, Mills AA, Chung UI, Tanaka S, Kawaguchi H, Saito T (2017) Regulation of chondrocyte survival in mouse articular cartilage by p63. Arthritis Rheumatol (Hoboken NJ) 69(3):598–609. https://doi.org/10.1002/art.39976
Tardif G, Pelletier JP, Fahmi H, Hum D, Zhang Y, Kapoor M, Martel-Pelletier J (2013) NFAT3 and TGF-beta/SMAD3 regulate the expression of miR-140 in osteoarthritis. Arthritis Res Ther 15(6):R197. https://doi.org/10.1186/ar4387
Taylor PC, Keystone EC, van der Heijde D, Weinblatt ME, Del Carmen Morales L, Reyes Gonzaga J, Yakushin S, Ishii T, Emoto K, Beattie S, Arora V, Gaich C, Rooney T, Schlichting D, Macias WL, de Bono S, Tanaka Y (2017) Baricitinib versus placebo or adalimumab in rheumatoid arthritis. N Engl J Med 376(7):652–662. https://doi.org/10.1056/NEJMoa1608345
Tominaga H, Maeda S, Hayashi M, Takeda S, Akira S, Komiya S, Nakamura T, Akiyama H, Imamura T (2008) CCAAT/enhancer-binding protein beta promotes osteoblast differentiation by enhancing Runx2 activity with ATF4. Mol Biol Cell 19(12):5373–5386. https://doi.org/10.1091/mbc.E08-03-0329
Tsuda M, Takahashi S, Takahashi Y, Asahara H (2003) Transcriptional co-activators CREB-binding protein and p300 regulate chondrocyte-specific gene expression via association with Sox9. J Biol Chem 278(29):27224–27229. https://doi.org/10.1074/jbc.M303471200
Tsushima H, Okazaki K, Hayashida M, Ushijima T, Iwamoto Y (2012) CCAAT/enhancer binding protein beta regulates expression of matrix metalloproteinase-3 in arthritis. Ann Rheum Dis 71(1):99–107. https://doi.org/10.1136/annrheumdis-2011-200061
Ushijima T, Okazaki K, Tsushima H, Iwamoto Y (2014) CCAAT/enhancer-binding protein beta regulates the repression of type II collagen expression during the differentiation from proliferative to hypertrophic chondrocytes. J Biol Chem 289(5):2852–2863. https://doi.org/10.1074/jbc.M113.492843
Vortkamp A, Lee K, Lanske B, Segre GV, Kronenberg HM, Tabin CJ (1996) Regulation of rate of cartilage differentiation by Indian hedgehog and PTH-related protein. Science 273(5275):613–622
Wagner T, Wirth J, Meyer J, Zabel B, Held M, Zimmer J, Pasantes J, Bricarelli FD, Keutel J, Hustert E, Wolf U, Tommerup N, Schempp W, Scherer G (1994) Autosomal sex reversal and campomelic dysplasia are caused by mutations in and around the SRY-related gene SOX9. Cell 79(6):1111–1120
Wang Y, Sul HS (2009) Pref-1 regulates mesenchymal cell commitment and differentiation through Sox9. Cell Metab 9(3):287–302. https://doi.org/10.1016/j.cmet.2009.01.013
Wang J, Gardner BM, Lu Q, Rodova M, Woodbury BG, Yost JG, Roby KF, Pinson DM, Tawfik O, Anderson HC (2009a) Transcription factor Nfat1 deficiency causes osteoarthritis through dysfunction of adult articular chondrocytes. J Pathol 219(2):163–172. https://doi.org/10.1002/path.2578
Wang W, Lian N, Li L, Moss HE, Wang W, Perrien DS, Elefteriou F, Yang X (2009b) Atf4 regulates chondrocyte proliferation and differentiation during endochondral ossification by activating Ihh transcription. Development (Cambridge England) 136(24):4143–4153. https://doi.org/10.1242/dev.043281
Yamashita S, Miyaki S, Kato Y, Yokoyama S, Sato T, Barrionuevo F, Akiyama H, Scherer G, Takada S, Asahara H (2012) L-Sox5 and Sox6 proteins enhance chondrogenic miR-140 microRNA expression by strengthening dimeric Sox9 activity. J Biol Chem 287(26):22206–22215. https://doi.org/10.1074/jbc.M112.343194
Yang S, Kim J, Ryu JH, Oh H, Chun CH, Kim BJ, Min BH, Chun JS (2010) Hypoxia-inducible factor-2alpha is a catabolic regulator of osteoarthritic cartilage destruction. Nat Med 16(6):687–693. https://doi.org/10.1038/nm.2153
Yoshida E, Noshiro M, Kawamoto T, Tsutsumi S, Kuruta Y, Kato Y (2001) Direct inhibition of Indian hedgehog expression by parathyroid hormone (PTH)/PTH-related peptide and up-regulation by retinoic acid in growth plate chondrocyte cultures. Exp Cell Res 265(1):64–72. https://doi.org/10.1006/excr.2001.5161
Yoshida CA, Yamamoto H, Fujita T, Furuichi T, Ito K, Inoue K, Yamana K, Zanma A, Takada K, Ito Y, Komori T (2004) Runx2 and Runx3 are essential for chondrocyte maturation, and Runx2 regulates limb growth through induction of Indian hedgehog. Genes Dev 18(8):952–963. https://doi.org/10.1101/gad.1174704
Yoshida M, Hata K, Takashima R, Ono K, Nakamura E, Takahata Y, Murakami T, Iseki S, Takano-Yamamoto T, Nishimura R, Yoneda T (2015) The transcription factor Foxc1 is necessary for Ihh-Gli2-regulated endochondral ossification. Nat Commun 6:6653. https://doi.org/10.1038/ncomms7653
Zhang C, Tang W, Li Y (2012) Matrix metalloproteinase 13 (MMP13) is a direct target of osteoblast-specific transcription factor osterix (Osx) in osteoblasts. PloS One 7(11):e50525. https://doi.org/10.1371/journal.pone.0050525
Zou W, Chen X, Shim JH, Huang Z, Brady N, Hu D, Drapp R, Sigrist K, Glimcher LH, Jones D (2011) The E3 ubiquitin ligase Wwp2 regulates craniofacial development through mono-ubiquitylation of goosecoid. Nat Cell Biol 13(1):59–65. https://doi.org/10.1038/ncb2134
Funding
This work was partly supported by JSPS KAKENHI Grant number 16H06393.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Rights and permissions
About this article
Cite this article
Nishimura, R., Hata, K., Nakamura, E. et al. Transcriptional network systems in cartilage development and disease. Histochem Cell Biol 149, 353–363 (2018). https://doi.org/10.1007/s00418-017-1628-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-017-1628-7