Abstract
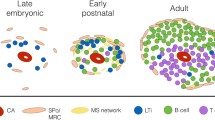
Immunohistological analysis of 31 human spleens from the 11th week of gestation to the early postnatal period suggested that fetal organ development may be preliminarily divided into four stages. At stage 0 the organ anlage contained erythrocyte precursors, few macrophages and almost no lymphocytes. Fetal spleens of stage I exhibited arterial vascular lobules and lymphocytes just began colonizing the organ. At stage II, B and T lymphocytes formed periarteriolar clusters. B cell clusters predominated, because B cells aggregated around the more peripheral branches of splenic arterioles, while T cells occupied the more centrally located parts of the vessels. The vascular lobules of stage I and II consisted of central arterioles surrounded by B cells, capillaries and peripheral venules. The lobular architecture slowly dissolved at late stage II when sinuses grew out from the peripheral venules into the centre of the lobule. Interestingly, the B cell accumulations around peripheral arterioles did not represent the precursors of follicles, but apparently persisted as periarteriolar B cell clusters in the adult splenic red pulp, while follicles containing FDCs developed at late stage II from B cells in direct contact to T cell clusters around larger arterial vessels. At stage III before birth the lobular architecture was no longer recognized. The chemokine CXCL13 was already present in vascular smooth muscle and adjacent stromal cells at stage I before B cells immigrated. CCL21, on the contrary, was only demonstrated in fibroblast-like cells supporting T cell clusters from stage II onwards.




Similar content being viewed by others
Abbreviations
- ABC:
-
Avidin-biotinylated peroxidase complex
- AP:
-
Alkaline phosphatase
- DAB:
-
Diaminobenzidine
- FDC:
-
Follicular dendritic cell
- MZ:
-
Marginal zone
- SMA:
-
Smooth muscle alpha actin
References
Ansel KM, Ngo VN, Hyman PL, Luther S, Förster R, Sedgwick JD, Browning JL, Lipp M, Cyster JG (2000) A chemokine-driven positive feedback loop organizes lymphoid follicles. Nature 406:309–314
Cupedo T, Lund FE, Ngo VN, Randall TD, Jansen W, Greuter MJ, de Waal-Malefyt R, Kraal G, Cyster JG, Mebius RE (2004) Initiation of cellular organization in lymph nodes is regulated by non-B cell-derived signals and is not dependent on CXC chemokine ligand 13. J Immunol 173:4889–4896
Dammers PM, Lodewijk ME, Zandvoort A, Kroese FGM (2002) Marginal zone B cells in neonatal rats express intermediate levels of CD90 (Thy-1). Dev Immunol 9:187–195
Dijksta CD, Döpp EA (1983) Ontogenetic development of T- and B-lymphocytes and non-lymphoid cells in the white pulp of the rat spleen. Cell Tissue Res 229:351–363
Dijkstra CD, Döpp EA, Joling P, Kraal G (1985) The heterogeneity of mononuclear phagocytes in lymphoid organs: distinct macrophage subpopulations in the rat recognized by monoclonal antibodies. Immunology 54:589–599
Eikelenboom P, Levenbach MGE, van den Brink HR, Streefkerk JG (1979) Development of T and B cell areas in peripheral lymphoid organs of the rat. Anat Rec 194:523–538
Friedberg SH, Weissman IL (1974) Lymphoid tissue architecture II. Ontogeny of peripheral T and B cells in mice: evidence against Peyer´s Patches as the site of generation of B cells. J Immunol 113:1477–1492
Fütterer A, Mink K, Luz A, Kosco-Vilbois MH, Pfeffer K (1998) The lymphotoxin β receptor controls organogenesis and affinity maturation in peripheral lymphoid tissues. Immunity 9:59–70
Kraal G (1992) Cells in the marginal zone of the spleen. Int Rev Cytol 132:31–74
Mall FP (1889) The lobule of the spleen. J Hopkins Hosp Bull 9:218–219
Martin R (1951) Plastische Rekonstruktion des bindegewebigen Gerüsts der Milz eines Neugeborenen. Z Anat Entwicklungsgesch 116:96–104
Mebius RE (2003) Organogenesis of lymphoid tissues. Nat Rev Immunol 3:292–303
Namikawa R, Mizuno T, Matsuoka H, Fukami H, Ueda R, Itoh G, Matsuyama M, Takahashi T (1986) Ontogenic development of T and B cells and non-lymphoid cells in the white pulp of human spleen. Immunology 57:61–69
Ngo VN, Cornall RJ, Cyster JG (2001) Splenic T zone development is B cell dependent. J Exp Med 194:1649–1660
Sasaki K, Matsumura G (1988) Spleen lymphocytes and haemopoiesis in the mouse embryo. J Anat 160:27–37
Steiniger B, Barth P (2000) Microanatomy and function of the spleen. Adv Anat Embryol 151:1–100
Steiniger B, Barth P, Herbst B, Hartnell A, Crocker PR (1997) The species-specific structure of microanatomical compartments in the human spleen: strongly sialoadhesin-positive macrophages occur in the perifollicular zone, but not in the marginal zone. Immunology 92:307–316
Steiniger B, Hellinger A, Barth P (2001) The perifollicular and marginal zones of the human splenic white pulp: do fibroblasts guide lymphocyte immigration? Am J Pathol 159:501–512
Steiniger B, Rüttinger L, Barth PJ (2003) The three-dimensional structure of human splenic white pulp compartments. J Histochem Cytochem 51:655–663
Steiniger B, Timphus EM, Jacob R, Barth PJ (2005) CD27+ B cells in human lymphatic organs: re-evaluating the splenic marginal zone. Immunology 116:429–442
Steiniger B, Timphus EM, Barth PJ (2006) The splenic marginal zone in humans and rodents—an enigmatic compartment and its inhabitants. Histochem Cell Biol 126:641–648
Timens W, Poppema S (1985) Lymphocyte compartments in human spleen. An immunohistologic study in normal spleens and noninvolved spleens in Hodgkin´s disease. Am J Pathol 120:443–454
Timens W, Rozeboom T, Poppema S (1987) Fetal and neonatal development of human spleen: an immunohistological study. Immunology 60:603–609
Timens W, Boes A, Rozeboom-Uiterwijk T, Poppema S (1988) Immuno-architecture of human fetal lymphoid tissues. Virchows Arch Pathol Anat 413:563–571
Tumanov AV, Kuprash DV, Lagarkova MA, Grivennikov SI, Abe K, Shakhov AN, Drutskaya LN, Stewart CL, Chervonsky AV, Nedospasov SA (2002) Distict role of surface lymphotoxin expressed by B cells in the organization of secondary lymphoid tissues. Immunity 17:239–250
Vellguth S, von Gaudecker B, Müller-Hermelink H-K (1985): The development of the human spleen. Ultrastructural studies from the 14th to the 24th week of gestation. Cell Tissue Res 242:579–592
Wilkins BS, Green A, Wild AE, Jones DB (1994) Extramedullary haematopoiesis in fetal and adult human spleen: a quantitative immunohistological study. Histopathology 24:241–247
Wolf BC, Luevano E, Neiman RS (1983) Evidence to suggest that the human fetal spleen is not a hematopoietic organ. Am J Clin Pathol 80:140–144
Acknowledgments
We thank Dr. E. W. Herbst, Institute of Pathology, Klinikum Neubrandenburg, D-17022 Neubrandenburg, for contributing several specimens. Katrin Lampp and Anja Seiler provided expert technical assistance. Reiner Westermann, Gerwin Santo and J. Jäkel helped with image processing. Ilse Feisel skilfully drew Fig. 4. This work was supported by grant Ste 360/10-1 of the Deutsche Forschungsgemeinschaft.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Steiniger, B., Ulfig, N., Riße, M. et al. Fetal and early post-natal development of the human spleen: from primordial arterial B cell lobules to a non-segmented organ. Histochem Cell Biol 128, 205–215 (2007). https://doi.org/10.1007/s00418-007-0296-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-007-0296-4