Abstract
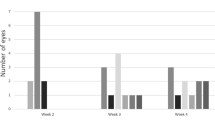
• Background: Chronic hypotony is an important cause of functional failure after proliferative vitreoretinopathy (PVR) surgery even if the retina is successfully reattached. The purpose of this study was to create a relevant model of chronic hypotony in the rabbit. • Methods: Eighteen pigmented rabbits weighing 3–4 kg were used in our experiment. We performed pars plana lensectomy on 14 eyes of 14 rabbits with a fragmatome and a vitreous cutter. At the end of surgery, we injected 0.2 ml of phosphatebuffered saline containing 100000 cultured rabbit dermal fibroblasts over the epiciliary area. As a control, we performed pars plana lensectomy alone on four eyes of four rabbits. The intraocular pressure (IOP) was measured on days 7, 14, 21, 28. Two rabbits were killed on day 7 and prepared for histology. Two additional rabbits were killed on day 14, and the remaining ten rabbits were killed on day 28. • Results: All 14 eyes following lensectomy and fibroblast injection had an IOP less than 5 mm Hg on each follow-up and a mean of 2.5±0.6 mm Hg (mean±SD) on day 28. Four control eyes with lensectomy alone had an IOP of 7.5±2.1 mm Hg on day 28 (P<0.05). On gross examination, we identified a variety of pathologic changes, including the development of a fibrous translucent epiciliary membrane that caused distortion of ciliary processes, iridociliary adhesion and anterior displacement of the peripheral retina on day 28. Microscopic examination of eyes obtained on day 28 showed changes in the ciliary epithelium that included absence or atrophy of the non-pigmented ciliary epithelium, atrophy and cystic vacuolization of the pigmented ciliary epithelium, and interstitital edema of the ciliary body stroma. • Conclusion: We have created a model of chronic hypotony with epiciliary membrane using cultured fibroblasts in the rabbit. This model may be useful to help elucidate the pathophysiology of chronic hypotony and to investigate potential treatments.
Similar content being viewed by others
References
Barr CC, Lai MY, Lean JS, Linton KL, Trese M, Abram G, Ryan SJ, Azen SP (1993) Postoperative intraocular pressure abnormalities in the silicone study. Ophthalmology 100: 1629–1635
Burk LL, Shields MB, Proia AD, McCuen BW (1988) Intraocular pressure following intravitreal silicone oil injection. Ophthalmic Surg 19: 565–569
Chandler DB, Quansah FA, Hida T, Machemer R (1986) A refined experimental model for proliferative vitreoretinopathy. Graefe’s Arch Clin Exp Ophthalmol 224: 86–91
Coleman DJ (1995) Evaluation of ciliary body detachment in hypotony. Retina 15: 312–318
Cox MS, Trese MT, Murphy PL (1986) Silicone oil for advanced proliferative vitreoretinopathy. Ophthalmology 93: 646–650
Dunbar Hoskins H Jr, Kass M (1989) Aqueous humor formation. In: Klein EA (ed) Becker-Shaffer’s diagnosis and therapy of the glaucoma. Mosby, St Louis, pp 18–40
Fastenburg DM, Diddie KP, Dorey K, Ryan SJ (1982) The role of cellular proliferation in an experimental model of massive periretinal proliferation. Am J Ophthalmol 93: 565–572
Han DP, Lewis MT, Kuhn EM, Abrams GW, Mieler WF, Wiliams GA, Aaberg TM (1990) Relaxing retinotomies and retinectomies: surgical results and predictors of visual outcome. Arch Ophthalmol 108: 694–697
Hida T, Chandler DB, Sheta SM (1987) Classification of the stages of proliferative vitreoretinopathy in a refined experimental model in the rabbit eye. Graefe’s Arch Clin Exp Ophthalmol 225: 303–307
Hitchins CA, Grierson I, Hiscott PS (1985) The effects of injections of cultured fibroblasts into the rabbit vitreous. Graefe’s Arch Clin Exp Ophthalmol 223: 237–249
Krupin T, Weiss A, Becker B, Holmberg N, Fritz C (1977) Increased intraocular pressure following topical azide or nitroprusside. Invest Ophthalmol Vis Sci 16: 1002–1007
Lewis H, Aaberg TM (1988) Anterior proliferative vitreoretinopathy. Am J Ophthalmol 105: 277–284
Lewis H, Aaberg TM, Abrams GW, Han DP, Kreiger AE (1987) Management of the lens capsule during pars plana lensectomy. Am J Ophthalmol 103: 109–110
McCuen BW, Landers MB, Machemer R (1985) The use of silicone oil fol lowing failed vitrectomy for retinal detachment with advanced proliferative vitreoretinopathy. Ophthalmology 92: 1029–1034
Morse LS, McCuen BW, Machemer R (1990) Relaxing retinotomies: analysis of anatomic and visual results. Ophthalmology 97: 642–647
Pederson JE (1982) Experimental retinal detachment. Arch Ophthalmol 100: 1814–1816
Pederson JE (1986) Ocular hypotony. Trans Ophthalmol Soc UK 105: 220–226
Pederson JE, Gaasterland DE, MacLellan HM (1979) Experimental ciliochoroidal detachment: effect on intraocular pressure and aqueous humor flow. Arch Ophthalmol 97: 536–541
Pederson JE, MacLellan HM (1982) Medical therapy for experimental hypotony. Arch Ophthalmol 100: 815–817
Shields MB (1992) Aqueous humor dynamics. I. Anatomy and physiology. In: Brown C (ed) Textbook of glaucoma. Williams and Wilkins, Baltimore, pp 5–36
Stallman JB, Meyers SM (1988) Repeated fluid-gas exchange for hypotony after vitreoretinal surgery for proliferative vitreoretinopathy. Am J Ophthalmol 106: 147–153
Suguro K, Toris CB, Pederson JE (1985) Uveoscleral outflow following cyclodialysis in the monkey eye using a fluorescent tracer. Invest Ophthalmol Vis Sci 26: 810–813
Tano Y, Machemer R (1980) Intravitreal autotransplantation of fibroblasts. Am J Ophthalmol 89: 121–130
Toris CB, Pederson JE (1985) Effect of intraocular pressure on uveoscleral outflow following cyclodialysis in the monkey eye. Invest Ophthalmol Vis Sci 26: 1745–1749
Zarbin MA, Michels RG, Green WR (1991) Dissection of epiciliary tissue to treat chronic hypotony after surgery for retinal detachment with proliferative vitreoretinopathy. Retina 11: 208–213
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kim, H.C., Hayashi, A., Shalash, A. et al. A model of chronic hypotony in the rabbit. Graefe’s Arch Clin Exp Ophthalmol 236, 69–74 (1998). https://doi.org/10.1007/s004170050045
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s004170050045