Abstract
Objective
Recently, the 7 Tesla (7 T) Epilepsy Task Force published recommendations for 7 T magnetic resonance imaging (MRI) in patients with pharmaco-resistant focal epilepsy in pre-surgical evaluation. The objective of this study was to implement and evaluate this consensus protocol with respect to both its practicability and its diagnostic value/potential lesion delineation surplus effect over 3 T MRI in the pre-surgical work-up of patients with pharmaco-resistant focal onset epilepsy.
Methods
The 7 T MRI protocol consisted of T1-weighted, T2-weighted, high-resolution-coronal T2-weighted, fluid-suppressed, fluid-and-white-matter-suppressed, and susceptibility-weighted imaging, with an overall duration of 50 min. Two neuroradiologists independently evaluated the ability of lesion identification, the detection confidence for these identified lesions, and the lesion border delineation at 7 T compared to 3 T MRI.
Results
Of 41 recruited patients > 12 years of age, 38 were successfully measured and analyzed. Mean detection confidence scores were non-significantly higher at 7 T (1.95 ± 0.84 out of 3 versus 1.64 ± 1.19 out of 3 at 3 T, p = 0.050). In 50% of epilepsy patients measured at 7 T, additional findings compared to 3 T MRI were observed. Furthermore, we found improved border delineation at 7 T in 88% of patients with 3 T-visible lesions. In 19% of 3 T MR-negative cases a new potential epileptogenic lesion was detected at 7 T.
Conclusions
The diagnostic yield was beneficial, but with 19% new 7 T over 3 T findings, not major. Our evaluation revealed epilepsy outcomes worse than ILAE Class 1 in two out of the four operated cases with new 7 T findings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Focal epilepsy
Epilepsy affects over 50 million people worldwide, making the treatment of epilepsy a global challenge for neurology [1]. Focal epilepsies as defined by the International League Against Epilepsy (ILAE) [2], not only causes seizures, but also cognitive, psychological, and social impairments [3]. Up to 40% of these patients do not respond to treatment with two or more anti-seizure medications (ASMs) and are thus termed “pharmaco-resistant,” or “refractory” [4]. In these cases, surgery is recommended [5] and can be curative, provided a complete resection/disconnection of the epileptogenic zone (the area of cortex indispensable for the generation of clinical seizures [6]) can be achieved. Pre-surgical evaluation uses a variety of diagnostic tools, such as an analysis of seizure semiology, (non-) invasive electrophysiological (EEG) recordings, neuropsychological testing, and structural as well as functional neuroimaging. These try to define the location and boundaries of the epileptogenic zone as well as eloquent brain areas. Furthermore, in MRI negative cases even invasive procedures such as implantation of intracranial electrodes for stereo EEG prove to be indispensable [7].
The main pathological entities responsible for focal pharmaco-resistant epilepsies are focal cortical dysplasia (FCD), epilepsy-associated tumors (LEATs, e.g., dysembryoplastic neuroepithelial tumor (DNET) or ganglioglioma (GG)), hippocampal sclerosis (HCS), or vascular malformations [8,9,10]. Complete resection/disconnection of these lesions may result in seizure freedom in up to 60% of cases [5]. Successful surgery is, however, dependent on the precise localization of epileptogenic foci [6]. The current imaging standard, 3 T MRI [9], does not yield findings in one of three cases (MR-negative, MRN) [10]. In addition to MRN cases, some suspected lesions, including mild cortical dysplasia or epilepsy-associated tumors, are not precisely delineated or optimally characterized on 3 T MRI, and need further invasive EEG exploration. MRN cases have reduced odds for postsurgical freedom from seizures [11], but surgery is still often attempted to reduce seizure frequency, and to prevent the consequences of long-term refractory epilepsies, i.e., cognitive deficits and sudden unexpected death in epilepsy [9].
7 T MRI for epilepsy
The higher signal-to-noise ratio and altered contrast behavior of 7 T MRI [12] can increase detection sensitivity and delineation of potential epileptogenic lesions. Consequently, 7 T studies were found to improve FCD detection [13] and better identification of subtle vascular malformations due to increased SWI contrast, as well as delineation of cavernomas [14] and hippocampal scleroses, especially subfield neuronal rarefication correlating with epilepsy outcome [15]. Recent reviews determined that 7 T identified new lesions in 31% of MRN cases [16] and have shown an average detection rate for epileptic foci of 65% compared to 22% at lower fields [17].
Recently, the 7 T Epilepsy Task Force published a consensus protocol recommendation [10] that encompasses the use of sub-millimeter 3D T1-weighted MRI (T1w-MRI), T2-weighted MRI (T2w-MRI), fluid attenuated inversion recovery (FLAIR), and T2*-weighted MRI (T2*w-MRI) sequences of the whole brain. Additional recommended scans are high-resolution coronal T2w-MRI of the hippocampus and white matter (WM)-suppressed imaging (WMS) for the detection of gray matter aberrations.
Purpose
The purpose of this study was to evaluate the practicality of the 7 T Epilepsy Task Force consensus recommendation in clinical practice and to investigate the advantage of this 7 T protocol compared to a dedicated 3 T MRI routine protocol at a busy epilepsy surgical center. Additionally, we correlated our 7 T images with the sum of clinical diagnostic data, and contextualized to the current state of research on morphological 7 T MRI in epilepsy [10, 16, 17].
Methods
Hypothesis
Our hypothesis was that the 7 T Epilepsy Task Force recommendation offers improved rates and confidence for lesion detection, as well as better focal delineation than a dedicated 3 T MRI epilepsy protocol. The null hypothesis accordingly stated that the protocol does not offer any quantifiable benefits over the current clinical gold standard.
Cohort recruitment
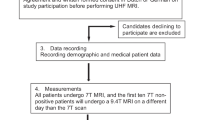
After institutional review board approval (EK 1039/2020) for this prospective study, consecutive patients with pharmaco-resistant focal epilepsy were recruited in 2020–2022 during the pre-surgical evaluation process (according to ILAE standards [6, 18, 19]) at the Medical University of Vienna (MUV) according to the presented inclusion and exclusion criteria by the Department of Neurology, Department of Neurosurgery, and Department of Pediatrics and Adolescent Medicine (Fig. 1).
Inclusion criteria: Age ≥ 12 years; informed consent by the subject, and, if applicable, their legal guardian; pharmaco-resistant focal epilepsy with either suboptimal delineation of the epileptogenic lesion on 3 T MRI or MRN despite the congruency of other pre-surgical investigations (seizure semiology, ictal/interictal video-EEG, neuropsychological testing, PET imaging); and pre-surgical evaluation and/or epilepsy surgery at our centers.
Exclusion criteria: Age < 12 years; weight < 30 kg; claustrophobia; pregnancy; breastfeeding; and ferromagnetic implants for the 7 T scan.
7 T Protocol
We conducted all scans on the MUV High-Field MR Centre’s 7 T MR scanner (7 T Magnetom retrofitted to Magnetom.plus, VE12U software, Siemens Healthineers, Erlangen) using a 32-channel head receive / 1-channel transmit coil array (Nova Medical). As a first-generation 7 T scanner, the system is not certified for clinical routine use. Therefore, we were not able to make resection decisions based on 7 T data but compared the data postoperatively.
The morphological MRI protocol consisted of the following sequences according to the ILAE consensus protocol (more detailed in Suppl. Table 1): 3D T1w (MP2RAGE, TR = 5000 ms, TE = 4.13 ms, Tacq = 8:02 min, resolution = 0.75 × 0.75 × 0.75mm3); 3D T2w (TSE, TR = 4000 ms, TE = 118 ms, Tacq = 7:02 min, resolution = 0.7 × 0.7 × 0.7mm3); coronal hippocampal T2w (TSE, TR = 5900 ms, TE = 49 ms, Tacq = 6:07 min, resolution = 0.5 × 0.4 × 2.0mm3); 3D T2w fluid-suppressed (FLAIR, TR = 8000 ms, TE = 305 ms, Tacq = 12:18 min, resolution = 0.9 × 0.9 × 0.9mm3); 3D T2w fluid- and WM-suppressed (TSE, TR = 8000 ms, TE = 321 ms, Tacq = 3:54 min, resolution = 0.9 × 0.8 × 1.0mm3); and transversal SWI (GRE, TR = 21 ms, TE = 14 ms, Tacq = 7:29 min, resolution = 0.25 × 0.25 × 1.5mm3). Including scan setup times and pre-scans, this imaging protocol required at least 50 min of measurement time. The average image quality is demonstrated in Fig. 2.
Example of the 7 T epilepsy consensus protocol performance acquired for all modalities in patient #22 with a histologically confirmed dysembryoplastic neuroepithelial tumor (DNET). While 7 T contrast and resolution benefits are quite noticeable, so are radio frequency excitation (B1+-)inhomogeneities in particular. Arrows highlight regions of interest for pathology or artifacts. T1w T1-weighted, T2w T2-weighted, FLAIR Fluid-attenuated inversion recovery, WMS white matter suppressed, 7 T 7 Tesla, SWI susceptibility-weighted imaging, HR high resolution, R right side, L left side
3 T clinical protocol and other diagnostics
Routine 3 T MRI data were collected as the reference standard from the hospital imaging system for every patient. All 3 Tesla examinations were performed following the Harmonized Neuroimaging of Epilepsy Structural Sequences (HARNESS-MRI) protocol recommended by the ILAE neuroimaging task force [9, 20], with isotropic, millimetric 3D T1w-MRI, FLAIR, and high-resolution 2D sub-millimetric T2w-MRI (detailed in Suppl. Table 1).
3 T MRI results together with Video-EEG monitoring (VEM; semiology at clinical seizure onset, ictal and interictal EEG) and [18F]FDG and MET PET [5, 6] estimated the extent of the epileptogenic zone as the gold standard for further evaluation. Furthermore, if available, resected areas with histological diagnoses as ground-truth, as well as the ILAE outcome classification [21], were collected. All of these data are presented in Table 1.
Comparison of 7 T MRI to clinical routine data
All MRI data were evaluated independently by two board-certified neuroradiologists specialized in epilepsy neuroimaging (rater 1: G.K., 16 years of neuroradiological experience in epilepsy neuroimaging; rater 2: L.H., two years of experience in epilepsy neuroimaging). First, they were blinded to clinical data and separately evaluated the 7 T data patient-wise, with a focal location hypothesis based on semiology made available to the raters only afterward. After a cool-down period of at least two weeks, the same process was repeated with clinical 3 T MRI. In a final round, 3 T and 7 T MRI were compared directly.
The evaluated parameters for 3 T and 7 T MRI were (1.) the presence of lesions (yes/no), (2.) the radiologist’s confidence in the identification of a present (scaled 1–3, with 3 being the highest), and (3.) its delineation (descriptive, detailed in Supp. Table 2). Imaging features denoted were blurred gray matter/white matter interface, transmantle signs, local signs of brain atrophy, abnormalities of the hippocampus and temporal lobe in general, as well as neoplasms and vascular abnormalities.
All completed ratings were aggregated by G.H., averaging confidence scores (with a 0 used in case a rater did not identify any lesion in a subject; rounded up) and summarizing 7 T delineation benefits compared to 3 T and to the clinical gold standard. For the presence of lesions, the detection rates for 3 T and 7 T, as well as inter-rater kappa (κ) for 3 T and 7 T, were calculated. Identification confidence between 3 and 7 T was tested using a paired Wilcoxon Signed-Rank tests.
We defined the final study outcome for 7 T MRI as percentages for:
-
(1)
Lesion identification as either “3 T MR-negative with 7 T lesion finding “ (also known in the literature as diagnostic gain), “7 T findings without plausible epileptogenicity”, or “both 3 T and 7 T MR-negative”.
-
(2)
Improvements in delineation in 3 T-positive cases as “7 T improved lesion delineation over 3 T”, “MR-positive at 3 T, but MR-negative at 7 T”, or “equivalent delineation at 3 T and 7 T”.
-
(3)
“Overall cases with benefits of 7 T MRI over 3 T MRI”, i.e., the sum of improved lesion identification and delineation over the whole cohort.
-
(4)
“Potentially positive treatment impact by 7 T”, i.e., the sum of improved lesion identification and delineation over the whole cohort, or “no new diagnostic information by 7 T” compared to the above-defined clinical gold standard. In patients that received surgery and had a follow up evaluation after three months, we identified patients that did not achieve seizure freedom as “surgical outcomes worse than ILAE class 1 with additional 7 T findings”.
Results
Cohort measurement
Forty-one patients were recruited, of whom 38 completed the 7 T protocol. One subject was excluded due to a previously undisclosed 7 T-unsafe cosmetic implant (after consent, but prior to scanning) and two subjects (one adolescent) aborted the 7 T scan due to stress regarding the enclosed space inside the scanner. In addition, only the expected transient, sensory side-effects of 7 T scans [22] (vertigo, discomfort with scan noises and enclosure, electro-gustatory perceptions) were reported. Figure 1 visualizes the participation process. The 38 remaining patients (age range 14–49 years, 20 females) are characterized in detail in Table 1. No data from the three dropout cases were processed further. The remaining subjects included mainly clinical 3 T MRN cases (n = 21), as well as those with insufficient clinical delineation (n = 17) necessary for surgical planning.
At the end of evaluation, 16 of the 38 patients had received surgery and pathohistological lesion evaluation (Table 1) while surgical outcome data (ILAE Outcome Classification) was available for 14 of these 16 patients (median of 22 months after surgery). In 12 of 16 histological classified patients, there was a previous 7 T finding. In 6/10 cases at 3 T and 7/11 cases at 7 T histological diagnosis was correctly predicted based on MRI alone.
7 T versus 3 T evaluation
When considering only 3 T MR imaging, rater #1 reported a positive detection rate of 45%, with an inter-rater κ of 0.66. In contrast, at 7 T, rater #1 had a detection rate of 55%, with a κ of 0.79. Considering all 22 patients with either 3 T or 7 T findings, mean confidence scores for 3 T (1.64 ± 0.84) were lower than for 7 T MRI (1.95 ± 1.19) (p = 0.050). These findings are displayed in detail in Table 2. Mean scores were higher for 7 T in nine patients, equal in ten, and higher for 3 T in three. At 7 T, relevant motion artifacts were found in two cases and B1+-inhomogeneities, especially of the right-side temporal lobe, were visible in all patients. As patient #6, who had only 3 T findings, did not receive surgery during data analysis, we could not clarify whether this signified worse performance at 7 T or a 3 T false-positive result.
7 T delineation benefits
We observed specific improvements offered by the 7 T Epilepsy Task Force consensus protocol over clinical 3 T MRI. The new findings in 3 T MRN patients included three cases of structural hippocampal abnormalities and one of FCD located in the temporal lobe. Figure 3 shows one of these cases, where 7 T MRI identified an abnormal right hippocampus. In 15 of 17 cases (88%) with 3 T MRI findings, the 7 T protocol added new information. This included better delineation of lesion extent based on better visibility of abnormalities, i.e., the transmantle sign (TMS), cortical dysplasia, or microgyria (Fig. 4). In particular, 7 T SWI performed better in identifying blood vessel abnormalities that are sometimes associated with cortical malformations, as seen in Fig. 5. Overall, the raters considered T2w coronal MRI most important due to its high resolution. These findings are all summarized in Table 2, with the full rater-wise evaluation available in Supp. Table 2.
In this case, patient #32, 7 T MRI could identify hippocampal asymmetry regarding size and structure of the right hippocampus whereas 3 T MRI remained negative. On T2w 7 T MRI, the right hippocampus appeared larger during neuroradiological evaluation than the left side. Arrows highlight regions of interest for pathology or artifacts. T2w T2-weighted, FLAIR Fluid-attenuated inversion recovery, 7 T 7 Tesla, 3 T 3 Tesla, HR high resolution, B1+ radio frequency excitation, R right side, L left side
In patients such as #24, the 7 T epilepsy consensus protocol adds visible details that could be useful for neurosurgical planning, such as the enhanced delineation of an focal cortical dysplasia (FCD) (A) vs. B)) or the accompanying trans-mantle sign (TMS) in image (C) vs. D)) of this example. On T1w imaging (E vs F), the FCD is clearly visible at 7 T, while inconspicuous at 3 T. Arrows highlight regions of interest for pathology or artifacts. T1w T1-weighted, T2w T2-weighted, 7 T 7 Tesla, 3 T 3 Tesla, B1+ radio frequency excitation, R right side, L left side
Example of the ability of 7 T susceptibility-weighted imaging (SWI) to enhance the imaging of vessels compared to 3 T in skull-stripped transversal images of patient #27. There is a clear increase of detail in the delineation of the developmental venous anomaly surrounding the cavernoma. 7 T 7 Tesla, 3 T 3 Tesla, R right side, L left side
Overall 7 T MRI benefits within the cohort
Of the twenty-one 3 T MRN patients, four (19%) patients were “3 T MR-negative with 7 T lesion finding “, one (5%) patient had a “7 T finding without plausible epileptogenicity”, and the remaining 16 (76%) patients remained “both 3 T and 7 T MR-negative”.
Of the 17 patients with 3 T findings, 15 (88%) patients had “7 T improved lesion delineation”, “equivalent confidence at 3 T and 7 T” in one case, and “MR-positive at 3 T, but MR-negative at 7 T” in one case (6%).
In summary, 19 of 38 cases (50%), were rated as “overall cases with benefits of 7 T MRI over 3 T MRI” compared to 3 T MRI and with a “potentially positive treatment impact by 7 T” compared to the clinical gold standard. In the remaining 50%, we found “no new diagnostic information by 7 T”. We found “surgical outcomes worse than ILAE Class 1 with additional 7 T findings” in two out of four cases. These results are summarized in Table 3.
Discussion
Our study clearly demonstrates evidence that the newly recommended 7 T Epilepsy Task Force consensus protocol provided potential radiological benefits in 50% of our patient cohort. We were able to identify previously undetected lesions in 19% of 3 T MRN patients and found relevant additional lesion information in 88% of 3 T-visible abnormalities at 7 Tesla imaging with a higher but non-significant radiological confidence than at 3 T. This indicates that the addition of the 7 T MRI consensus protocol to routine clinical practice in presurgical epilepsy investigation using powerful 3 T MRI protocols could be primarily beneficial to the characterization and delineation of relevant pathological entities. Moreover, the 50-min protocol implemented during our study was feasible with only two aborted scan sessions.
Comparison to the state of research
Compared to the expectations set by the consensus protocol publication [10], i.e., better delineation but unlikely new identification of FCDs, more apparent hippocampal abnormalities, better LEAT characterization, and the importance of SWI for vascular malformations, we found high agreement within our cohort. This also includes the identification of B1 + inhomogeneity and subject motion as negative impacts on the utility of 7 T images. Van Lanen et al. [16], who pooled 16 studies that included 275 patients of sufficient design quality, found diagnostic gains defined as 8% to 67% percent of subjects in individual studies who were 7 T-positive, but lower-field-negative. Their reported diagnostic gain of 31% over all studies was higher than the 19% in our single-center study, but that gain reflects larger patient numbers and included clinical 1.5 T MRI as well as differing 7 T protocols.
A second recently published review [17] has described a lesion detection rate of 22% for 3 T MRI and 65% for 7 T MRI in a subset of analyzed studies. Comparing these observations to our different overall rates of findings of 45% for 3 T and 55% for 7 T also demonstrates the importance of a well-defined patient population. Feldman et al., for example, found a higher rate of 7 T lesion detection in MRN cases (25 of 37)[23], but 24 of these patients had clinical 1.5 T scans as a reference in a study cohort size similar to our own, producing higher 7 T benefits than a comparison to state-of-the-art clinical 3 T MRI. De Ciantis et al. [24] investigated a mixed cohort of 1.5 T/3 T MRN cases and found lesions on 7 T MRI in 29% of patients. Verseema et al. [25] observed epileptogenic lesions on 7 T MRI in 23% of 40 mostly 3 T MRN patients. Similar results were seen by Wang et al. [26] with a 22% lesion detection rate in sixty-seven 3 T MRN patients, which is very close to our findings. Interestingly, they improved this result to 43% by the application of morphometric analysis software. Colon et al. [27] investigated 19 3 T MRN cases with 7 T lesion detection in three (16%). Using aditional guidance by magnetoencephalography, they could also identify lesions in another three cases (16%).
Van Lanen et al. published their intent to conduct a more standardized 7 T MRI study [28], and even plan to go beyond the acquisition of T1/T2/T2*-contrast images by including diffusion tensor imaging (DTI) and arterial spin labeling (ASL) in their 7 T protocols.
Future research could include advanced 7 T metabolic imaging modalities, such as CEST [29] or MRSI [30] to yield new insights into MRN epilepsy. But while metabolic changes in other brain pathologies, such as gliomas, can easily be correlated to clinical imaging [31], verification of findings in MRN epilepsy requires significant analytic work and clinical confirmation.
We conclude that the availablility of clinical 3 T MRI versus 1.5 T MRI is the primary driving factor in determining the percieved benefits of 7 T MRI and should, therefore, be discussed when comparing 7 T epilepsy imaging studies. Our study was performed in the setting of a busy tertiary care epilepsy surgery program with experienced epilepsy imaging specialists as readers. This is important, as many existing studies in the field have used general radiological or clinical neurological readers as a reference and their results may have been changed after thorough analysis by an epilepsy imaging expert [32]. As the variety of findings from hippocampal abnormalities to FCDs in our results show, the strength of the consensus protocol lies in the coverage of multiple possible etiologies when other tools of preclinical evaluation remain inconclusive.
Limitations
As our current 7 T scanner is without clinical certification, we could not use our findings in order to modify neurosurgical planning. Thus, no comment on the benefit of 7 T MRI regarding surgical outcome can be made. Whether the additional 7 T findings in two of four surgical failures would have influenced results remains speculative. Our cohort size was small and heterogeneous, making conclusions challenging, especially considering that histological confirmation as the ground-truth was available only for 16 patients and surgical outcome regarding seizure status only in 14. We maintain that our cohort reflects the clinical reality faced in many tertiary epilepsy centers. Exploring 7 T MRI in epilepsy further will require multi-center studies that combine large cohorts with homogeneous inclusion criteria, standardized 7 T approaches such as the one tested in our study, and most importantly, a clear definition of clinical diagnostic gold standards, including histology and surgical outcome.
Our 7 T images show the need for parallel transmit technologies [12, 33] to address field inhomogeneities and reliably measure the temporal lobe, which is highly relevant for epilepsy diagnostics. Furthermore, motion artifacts in multiple scans did negatively affect image quality and should be avoided with motion detection and correction methods in the future [34].
Conclusion
We demonstrated a successful implementation of the 7 T Epilepsy Task Force consensus recommendation for pre-surgical evaluation in a cohort of 38 pharmaco-resistant patients with refractory focal epilepsies. Using the new consensus protocol, our results should be readily pool-able with future studies that follow the same standards. We, therefore, see our presented work as pilot data for future multi-center studies with standardized evaluation.
Ultimately, we demonstrated the utility of the 7 T consensus protocol in a clinically relevant setting that reflects realizable clinical benefits and found potentially epileptogenic lesions in 19% of 3 T MRN cases, and offered more detailed information than 3 T MRI in 88% of the other cases. Therefore, morphological 7 T imaging for epilepsy should not be overemphasized. Nonetheless, in 50% of cases where surgery achieved a worse ILAE classification than class 1, only 7 T MRI detected any abnormalities. In total, 7 T MRI had a potentially beneficial effect over 3 T MRI and other clinical diagnostics in half the study cohort.
Data availability
Pseudonymized 7 T image data from this study cohort will be made available by request from any qualified investigator after approval of the Medical University of Vienna data clearing commission.
Change history
05 April 2024
A Correction to this paper has been published: https://doi.org/10.1007/s00415-024-12257-9
Abbreviations
- 3 T:
-
3 Tesla
- 7 T:
-
7 Tesla
- B1+ :
-
Radio frequency excitation
- DNET:
-
Dysembryoplastic neuroepithelial tumor
- FCD:
-
Focal cortical dysplasia
- FDG:
-
Fluorodeoxyglucose
- FLAIR:
-
Fluid-attenuated inversion recovery
- GG:
-
Ganglioglioma
- HCS:
-
Hippocampal sclerosis
- HR:
-
High resolution
- ILAE:
-
International league against epilepsy
- L:
-
Left side
- LEAT:
-
Epilepsy-associated tumor
- MET:
-
Methionine
- MRI:
-
Magnetic resonance imaging
- MRN:
-
MR-negative
- MUV:
-
Medical University of Vienna
- R:
-
Right side
- PET:
-
Positron emission tomography
- SWI:
-
Susceptibility-weighted imaging
- T1w:
-
T1-weighted
- T2w:
-
T2-weighted
- T2*w:
-
T2*-weighted
- TMS:
-
Transmantle sign
- WM:
-
White matter
- WMS:
-
White matter suppressed
References
Ngugi AK, Bottomley C, Kleinschmidt I et al (2010) Estimation of the burden of active and life-time epilepsy: a meta-analytic approach. Epilepsia 51:883–890. https://doi.org/10.1111/j.1528-1167.2009.02481.x
Scheffer IE, Berkovic S, Capovilla G et al (2017) ILAE classification of the epilepsies: position paper of the ILAE commission for classification and terminology. Epilepsia 58:512–521. https://doi.org/10.1111/epi.13709
Holmes GL (2015) Cognitive impairment in epilepsy: the role of network abnormalities. Epileptic Disord 17:101–116. https://doi.org/10.1684/epd.2015.0739
Ryvlin P, Cross JH, Rheims S (2014) Epilepsy surgery in children and adults. Lancet Neurol 13:1114–1126. https://doi.org/10.1016/S1474-4422(14)70156-5
Baumgartner C, Koren JP, Britto-Arias M, et al (2019) Presurgical epilepsy evaluation and epilepsy surgery. F1000Res 8:1818. https://doi.org/10.12688/f1000research.17714.1
Rosenow F (2001) Presurgical evaluation of epilepsy. Brain 124:1683–1700. https://doi.org/10.1093/brain/124.9.1683
Cossu M, Cardinale F, Castana L, et al (2005) Stereoelectroencephalography in the presurgical evaluation of focal epilepsy: a retrospective analysis of 215 procedures. Neurosurgery 57:706–18; discussion 706–18
Blümcke I, Thom M, Aronica E et al (2013) International consensus classification of hippocampal sclerosis in temporal lobe epilepsy: a task force report from the ILAE commission on diagnostic methods. Epilepsia 54:1315–1329. https://doi.org/10.1111/epi.12220
Bernasconi A, Cendes F, Theodore WH et al (2019) Recommendations for the use of structural magnetic resonance imaging in the care of patients with epilepsy: a consensus report from the International League Against Epilepsy Neuroimaging Task Force. Epilepsia 60:15612. https://doi.org/10.1111/epi.15612
Opheim G, van der Kolk A, Bloch KM et al (2021) 7T epilepsy task force consensus recommendations on the use of 7T in clinical practice. Neurology. https://doi.org/10.1212/WNL.0000000000011413
Téllez-Zenteno JF, Ronquillo LH, Moien-Afshari F, Wiebe S (2010) Surgical outcomes in lesional and non-lesional epilepsy: A systematic review and meta-analysis. Epilepsy Res 89:310–318. https://doi.org/10.1016/j.eplepsyres.2010.02.007
Trattnig S, Springer E, Bogner W et al (2018) Key clinical benefits of neuroimaging at 7 T. Neuroimage 168:477–489. https://doi.org/10.1016/j.neuroimage.2016.11.031
Colon AJ, van Osch MJP, Buijs M et al (2016) Detection superiority of 7 T MRI protocol in patients with epilepsy and suspected focal cortical dysplasia. Acta Neurol Belg 116:259–269. https://doi.org/10.1007/s13760-016-0662-x
Frischer JM, Göd S, Gruber A et al (2012) Susceptibility-weighted imaging at 7 T: improved diagnosis of cerebral cavernous malformations and associated developmental venous anomalies. Neuroimage Clin 1:116–120. https://doi.org/10.1016/j.nicl.2012.09.005
Stefanits H, Springer E, Pataraia E et al (2017) Seven-tesla MRI of hippocampal sclerosis: an in vivo feasibility study with histological Correlations. Invest Radiol. https://doi.org/10.1097/RLI.0000000000000388
van Lanen RHGJ, Colon AJ, Wiggins CJ et al (2021) Ultra-high field magnetic resonance imaging in human epilepsy: a systematic review. Neuroimage Clin 30:102602. https://doi.org/10.1016/j.nicl.2021.102602
Park JE, Cheong E-N, Jung DE et al (2021) Utility of 7 tesla magnetic resonance imaging in patients with epilepsy: a systematic review and meta-analysis. Front Neurol. https://doi.org/10.3389/fneur.2021.621936
Blumcke I, Cross JH, Spreafico R (2013) The international consensus classification for hippocampal sclerosis: an important step towards accurate prognosis. Lancet Neurol 12:844–846. https://doi.org/10.1016/S1474-4422(13)70175-3
Rosenow F, Bast T, Czech T et al (2016) Revised version of quality guidelines for presurgical epilepsy evaluation and surgical epilepsy therapy issued by the Austrian, German, and Swiss working group on presurgical epilepsy diagnosis and operative epilepsy treatment. Epilepsia 57:1215–1220. https://doi.org/10.1111/epi.13449
Wang I, Bernasconi A, Bernhardt B et al (2020) MRI essentials in epileptology: a review from the ILAE imaging taskforce. Epileptic Disord 22:421–437. https://doi.org/10.1684/epd.2020.1174
Wieser HG, Blume WT, Fish D et al (2001) ILAE commission report. proposal for a new classification of outcome with respect to epileptic seizures following epilepsy surgery. Epilepsia 42:282–286
Roberts DC, Marcelli V, Gillen JS et al (2011) MRI magnetic field stimulates rotational sensors of the brain. Curr Biol 21:1635–1640. https://doi.org/10.1016/j.cub.2011.08.029
Feldman RE, Delman BN, Pawha PS et al (2019) 7T MRI in epilepsy patients with previously normal clinical MRI exams compared against healthy controls. PLoS One. https://doi.org/10.1371/journal.pone.0213642
De Ciantis A, Barba C, Tassi L et al (2016) 7T MRI in focal epilepsy with unrevealing conventional field strength imaging. Epilepsia 57:445–454. https://doi.org/10.1111/epi.13313
Veersema TJ, Ferrier CH, van Eijsden P et al (2017) Seven tesla MRI improves detection of focal cortical dysplasia in patients with refractory focal epilepsy. Epilepsia Open 2:162–171. https://doi.org/10.1002/epi4.12041
Wang I, Oh S, Blümcke I et al (2020) Value of 7T MRI and post-processing in patients with nonlesional 3T MRI undergoing epilepsy presurgical evaluation. Epilepsia. https://doi.org/10.1111/epi.16682
Colon AJ, van Osch MJP, Buijs M et al (2018) MEG-guided analysis of 7T-MRI in patients with epilepsy. Seizure. https://doi.org/10.1016/j.seizure.2018.05.019
van Lanen RHGJ, Wiggins CJ, Colon AJ et al (2022) Value of ultra-high field MRI in patients with suspected focal epilepsy and negative 3 T MRI (EpiUltraStudy): protocol for a prospective, longitudinal therapeutic study. Neuroradiology 64:753–764. https://doi.org/10.1007/s00234-021-02884-8
Neal A, Moffat BA, Stein JM et al (2019) Glutamate weighted imaging contrast in gliomas with 7 Tesla magnetic resonance imaging. Neuroimage Clin 22:101694. https://doi.org/10.1016/j.nicl.2019.101694
Hingerl L, Strasser B, Moser P et al (2020) Clinical high-resolution 3D-MR spectroscopic imaging of the human brain at 7 T. Invest Radiol 55:239–248. https://doi.org/10.1097/RLI.0000000000000626
Hangel G, Cadrien C, Lazen P et al (2020) High-resolution metabolic imaging of high-grade gliomas using 7T-CRT-FID-MRSI. Neuroimage Clin 28:102433. https://doi.org/10.1016/j.nicl.2020.102433
Wehner T, Weckesser P, Schulz S et al (2021) Factors influencing the detection of treatable epileptogenic lesions on MRI. A randomized prospective study. Neurol Res Pract 3:41. https://doi.org/10.1186/s42466-021-00142-z
Deniz CM (2019) Parallel transmission for ultrahigh field MRI. Top Magn Reson Imag 28:159–171. https://doi.org/10.1097/RMR.0000000000000204
Zaitsev M, Dold C, Sakas G et al (2006) Magnetic resonance imaging of freely moving objects: prospective real-time motion correction using an external optical motion tracking system. Neuroimage 31:1038–1050. https://doi.org/10.1016/j.neuroimage.2006.01.039
Acknowledgements
This research was supported by Neuroscience Cluster Seed Grants 2021 of the Medical University of Vienna, the Austrian Science Fund (FWF) KLI 1121 grant and the Medical-Scientific Fund of the Mayor of the Federal Capital Vienna (Project Number 21186).
Funding
Open access funding provided by Medical University of Vienna.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None of the authors has any conflict of interest to disclose.
Additional information
The original online version of this article was revised: Duplicated table 1 has been removed and correct table 2 has been updated.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hangel, G., Kasprian, G., Chambers, S. et al. Implementation of a 7T Epilepsy Task Force consensus imaging protocol for routine presurgical epilepsy work-up: effect on diagnostic yield and lesion delineation. J Neurol 271, 804–818 (2024). https://doi.org/10.1007/s00415-023-11988-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-023-11988-5