Abstract
Mitochondrial DNA (mtDNA) is of great value in forensics to procure information about a person when a next of kin, personal belongings, or other sources of nuclear DNA (nDNA) are unavailable, or nDNA is lacking in quality and quantity. The quality and reliability of the results depend greatly on ensuring optimal conditions for the given method, for instance, the optimal input of the copy number (CN) in next-generation sequencing (NGS) methods. The unavailability of commercial quantitative PCR (qPCR) methods to determine mtDNA CN creates the necessity to rely on recommendations to infer mtDNA CN from nDNA yield. Because nDNA yield varies between individuals, tissues, parts of the same tissue, and because mtDNA CN varies between tissues, such assumptions must be examined for a specific context, rather than be generalized. This study compares mtDNA CN calculated from nDNA yield and qPCR measured mtDNA CN. Seventy-five femurs from the Second World War victims were used as samples; they were cut below the greater trochanter, surface contaminants were removed by mechanical and chemical cleaning, samples were fully demineralized, and DNA was isolated. PowerQuant® Kit (Promega) was used to analyze DNA yield. An in-house method was used to determine mtDNA CN. Comparison of mtDNA CN from nDNA derived calculations and measured mtDNA CN highlighted vast differences. The results emphasize the need to perform qPCR to assess mtDNA CN before NGS analyses of aged bones’ mitogenomes rather than estimating mtDNA CN from nDNA yield to ensure the quality and reliability of the results of NGS analysis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In both routine practice and research in forensic genetics, samples such as hair shafts without roots or skeletonized human remains are frequently encountered that are deemed difficult to analyze because of the poorly preserved state they are found in. This is reflected in nucleic acid forensic analyses, such as gold standard STR typing, for which nDNA from such samples may be lacking in quantity and quality, resulting in the use of alternatives to at least partially find the desired answers [1,2,3,4]. The most advantageous of these—despite lacking discriminatory power—is mtDNA [5]. The mitogenome has distinct characteristics such as a round, enclosed shape, making it more resilient to breakdown [6]. The specific inheritance pattern of mtDNA—a mother passes on the mitogenome to all her children, both male and female—makes it possible to examine and follow maternal lineages and populations [7,8,9]. A unique characteristic of mtDNA is its rather diverse and strongly tissue-dependent copy number (CN) per cell as opposed to DNA, which is always present in two copies per cell [10,11,12,13,14]. Cases in point include crime and missing person identification investigations [15], ancient DNA studies [16], historical case studies [9, 17], and ancestry studies [5].
When badly preserved DNA samples are analyzed, and sequencing of mtDNA must be performed, a correct assessment of the mtDNA copy number is needed, especially when the next-generation sequencing (NGS) approach is used. For NGS, a precise input of nucleic acids is recommended or even required for ruling out factors such as chimeric read and nuclear mitochondrial segments of nDNA called NUMTs, which could impact results and their interpretation, especially when input DNA is too high [18, 19]. According to Parson (W. Parson, personal correspondence, MtDNA analysis, and interpretation using EMPOP, ISFG pre-congress workshop, Prague, September 9th, 2019), 1000 to 5000 mitogenome copies are optimal for the best performance of NGS analysis. For quantifying mtDNA, there are no commercially available kits; this problem was addressed by researchers developing various in-house qPCR mtDNA kits [20,21,22,23]. It is possible to follow different published multiplex approaches when preparing an in-house qPCR assay, for example, the one published by Alonso et al. [24]. However, for the widespread use in routine forensic casework, the use of commercial kits is preferred due to the validations required [25,26,27]. To overcome the problem of the lack of commercial mtDNA qPCR kits, various manuals for using NGS technology for mtDNA sequencing recommend estimation of the input amount of mtDNA based on the nDNA quantity measured. Such an example is the Application Guide for Precision ID mtDNA NGS panels for both the whole genome and control region [28], for which 0.3 ng of DNA is claimed to correspond to 8700 copies of the mitogenome; even so, it is noted that the sample source and degradation may affect this prediction [28]. Because of the tissue specificity of the mitogenome CN, such predictions may be inaccurate to various degrees even if made for a certain type of tissue—the aforementioned poorly preserved bones being one example—which poses a problem for further analyses. However, the recently published Technical Note on the Optimization of Input DNA for Applied Biosystems™ Precision ID mtDNA Panels [29] included commentary advising mtDNA quantification, but no changes have been made in the guidelines for HID Ion Chef™ Instrument automated library preparations.
Generally, the mitogenome CN is tissue-specific [14], and bone tissue requires special attention when determining the correct mtDNA NGS input quantity if one takes into account the findings on the preservation of DNA in old bones. For aged bones, high nDNA degradation has been observed [30, 31], and there is a great variability in the preservation of DNA between different types of bones [31,32,33,34] within each bone [35,36,37] and between individuals [30, 31]. This represents the extraordinary complexity of old bone tissue, which most likely also reflects the differences in mtDNA preservation and its quantity and quality.
This study compares mtDNA CN calculated from nDNA qPCR results ([Auto] fragment (ng/μl) quantity measured with the PowerQuant™ kit (Promega) [38]) based on an NGS panel application guide recommendations [28] and measured mtDNA CN ([113 bp] fragment (copies/μl) measured with an in-house method based on Alonso et al. [24]) to ascertain a possible prediction ability of mtDNA CN from nDNA, using skeletonized human remains excavated from two separate Slovenian Second World War mass graves as a DNA source.
The Slovenian Medical Ethics Committee approved this study, approval number 0120-22/2017/3.
Materials and methods
Sample selection
Samples from two mass gravesites located near one another and dating back to the Second World War were selected because of their challenging yet relevant characteristics in forensic genetic examinations (skeletonized human remains, long postmortem interval, and DNA degradation). The samples were the diaphyses of femurs, cut below the greater trochanter, from 75 skeletons. Both gravesites are located in Slovenia, and their characteristics have been described in previously published studies [30, 37].
Preventing contamination
The utensils were cleaned with 6% sodium hypochlorite, followed by a bidistilled water rinse, 80% ethanol wash, and sterilization. UV irradiation of utensils, plastic consumables, and reagents was the final step in preventing contamination. Single-use nitrile gloves, gowns, and caps were used by personnel to avoid contamination with contemporary DNA. Extraction-negative controls and negative template controls were used to track any possible contamination events.
Bone sample preparation and automated DNA extraction
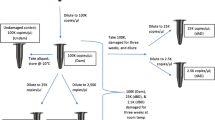
A room designed specifically for treatment of skeletonized human remains was used to prepare the samples; contaminants that were possibly present on the surface were removed with the outermost layer of bone in a fume cupboard (Iskra Pio, Šentjernej, Slovenia) by using a rotary tool (Schick, Schemmerhofen, Germany) with detachable burrs and diamond discs. A 5% Alconox detergent (Sigma-Aldrich, St. Louis, MO, USA) wash followed, proceeded by washing with bidistilled water (Millipore, Darmstadt, Germany) and 80% ethanol (Fisher Scientific, Loughborough, UK). The samples were then dried overnight and broken into smaller pieces that were then pulverized; to prevent DNA damage caused by overheating, the metal jars used for grinding were cooled with liquid nitrogen just before grinding the samples into powder with a Bead Beater MillMix 20 homogenizer (Tehtnica, Domel, Železniki, Slovenia). The oscillation frequency was set to 30 Hz and the time of grinding to 1 min. Ten milliliters of ethylenediaminetetraacetic acid (EDTA; Promega, Madison, WI, USA) was added to 0.5 g of fine bone powder to demineralize it through overnight incubation at 37 °C. A previously published research [39] describes this optimized process in extensive detail. The Biorobot EZ1 (Qiagen, Hilden, Germany) and the EZ1 DNA Investigator Kit (Qiagen) were used with the following settings as suggested by the manufacturer: trace protocol and 50 μl of DNA in a Tris EDTA buffer elution.
DNA quantification
An average of replications was used as a result for each sample, and requantification was performed in samples where more than 2-fold difference between the replicates was observed.
DNA yield
To analyze DNA, the PowerQuant System (Promega) was used. The kit measures the amount of nDNA [Auto] target, the amount of Y-chromosome DNA, and a longer [Deg] target to assess potential degradation (the ratio between the [Auto]/[Deg] targets) and the presence of PCR inhibitors. The two multi-copy targets for assessing degradation differ in length: the shorter 84 bp autosomal [Auto] target and the longer 294 bp degradation [Deg] target. The presence of PCR inhibitors is determined with internal positive control (IPC), which co-amplifies with the sample. The [Auto]/[Deg] ratio threshold was set to 2, and the IPC shift threshold was set to 0.30, as per the manufacturer’s guidelines. The QuantStudio™ 5 Real-Time PCR System (TFS) and Design and Analysis Software v1.5.2 (TFS) were used. Serial dilutions (0.0032 ng/μl, 0.08 ng/μl, 2 ng/μl, and 50 ng/μl) of PowerQuant® Male gDNA (Promega) with PowerQuant® Dilution Buffer (Promega) were used for the standard curve, as per manufacturer’s guidelines. The PowerQuant Analysis Tool (Promega), a Microsoft Excel macro file provided by the manufacturer, was used to view the results, calculate the results averages, and calculate the Auto/Deg ratio. Quantifications were performed in duplicate for all samples, and a negative template control was included in each run. The quantity of DNA was determined from the shorter [Auto] target, and the [Deg] target was used only for calculating the degradation of DNA [38].
mtDNA copy number
A modified in-house qPCR protocol was structured in line with the method published by Alonso et al. (2004), in which a 620 bp fragment (isolated from a human donor DNA) covering the HV1 region of mitogenome was used as a standard, and a single-copy target (113 bp) was used to determine the mitogenome copy number [24]. A spectrophotometric method using Synergy H4 (BioTek) was used to quantify the mitogenome. The copy number was then calculated based on the molecular weight of the fragment. After concentration normalization, serial dilutions (6,000,000 copies/μl, 600,000 copies/μl, 60,000 copies/μl, 6000 copies/μl, 600 copies/μl, and 60 copies/μl) were used to create a standard curve. The primers and fluorescent-labeled TaqMan MGB probes were custom-ordered (Applied Biosystems, Renfrewshire, UK). The QuantStudio™ 5 Real-Time PCR System (TFS) and Design and Analysis Software v1.5.2 (TFS) were used for quantification. TaqMan™ Universal Master Mix II with Uracil-N-glycosylase (TFS) was used instead of 1X TaqMan Universal PCR Master Mix (AB) because the latter has been discontinued. Duplicate quantifications were made for all samples, and a negative template control was included in each run.
Additionally, the copy number of mitogenome was calculated as per manufacturer’s guidelines, where it is stated that 0.3 ng of nDNA corresponds to 8700 copies of mitogenome (or 0.1 ng of nDNA is 2900 copies of mitogenome) [28]. Furthermore, a ratio between measured and calculated copy number of mtDNA has been determined.
Statistical evaluation
IBM SPSS Statistics for Mac, Version 23.0 (IBM Corp., Armonk, NY 2015), and Microsoft Excel for Mac, Version 16.67 (Microsoft Corporation 2022), were used to carry out descriptive statistics.
Results
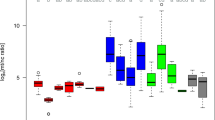
The complete results for 75 femurs excavated from the Second World War mass graves for the nDNA PowerQuant Kit (Promega) qPCR analyses, mtDNA in-house qPCR analyses, and calculations are shown in Supplementary Material 1 (SM1—Table 1, 2, and 3) and in Fig. 1.
Comparison of qPCR results of mtDNA [113 bp] in copies/μl and calculated mtDNA copies/μl based on nDNA [Auto] target (84 bp) (PowerQuant System, Promega) and manufacturer's instructions [28]
qPCR and calculations
The quantity of nDNA [Auto] fragment expressed in ng/μl ranged between 0.0022 ng/μl and 0.3283 ng/μl, and it averaged 0.0846 ng/μl. IPC shifts were between −3 (S59) and 0.76 (S65) (see SM1, Table 1).
The measured CN of the mtDNA 113 bp fragment (copies/μl) ranged from 5547 to 375,685 copies/μl and averaged 115,019.79 copies/μl (see SM1, Table 1).
The calculated mtDNA CN ranged from 64 to 9521, and the ratio between the measured and calculated CN ranged from 3 to 2498 (see SM1, Table 2).
Discussion
Our results derived from 75 femurs from two Second World War mass graves showed that there is a vast difference when calculating mtDNA copy number from nDNA quantity (using the short multi-copy target PowerQuant System (Promega), and as recommended by TFS [28]) and mtDNA quantity measured with qPCR (using short in-house single-copy target from quantification assay published by Alonso et al. [24]). When quantifying nDNA with the PowerQuant System (Promega) and mtDNA with Alonso’s in-house assay, shorter targets are used to determine nDNA and mtDNA yields [24, 38], and therefore, these targets (84 bp for nDNA and 113 bp for mtDNA) were used for calculations and comparisons. The length of the shorter mtDNA target also coincides with the length of the targets in NGS applications; for example, 153 bp is the average amplicon size for the Precision ID mtDNA Control Region Panel, as stated in the Precision ID mtDNA Panels with the HID Ion S5™ / HID Ion GeneStudio™ S5 System Application Guide [28], indicating that, for estimating the appropriate input of mtDNA for NGS analysis, information on the quantity of the shorter 113 bp mtDNA fragment is important.
Even if the same type of skeletal element was used, and the same intra-bone part was sampled, the wide interval between minimum and maximum in both targets for measuring the nDNA yield as well as the mtDNA copy number shows large variability in DNA content in samples even though the remains had the same postmortem interval (~75 years) and had been exposed to similar environmental conditions (both mass graves were located in karst caves), indicating the high complexity of aged bone tissue in the preservation of DNA. Namely, numerous studies have shown that there are differences in nDNA yields between different types of skeletal elements [33, 40, 41] and even between different parts of the same skeletal element [35,36,37]. Furthermore, in all samples measured, mtDNA CN was 3 to 2498-fold larger than from nDNA qPCR results calculated mtDNA CN, which indicates that an underestimation would have been made if mtDNA CN was calculated from nDNA qPCR results instead of being measured using qPCR mtDNA target. This can be attributed to the better resilience [6] and much larger CN of mitogenome in comparison to nDNA [10,11,12,13,14]. Our results conclude that nDNA is much more degraded compared to mtDNA, which certifies that mtDNA has a better preservation ability in aged skeletal remains. Calculating mtDNA CN from nDNA qPCR results as per recommendations [28] therefore portrays much lower mtDNA CN than it would if mtDNA CN is measured with qPCR using mtDNA target. Moreover, variability in DNA content between the samples can also be attributed to inter-individual differences, as seen in our study of Second World War femur diaphyses and other studies performed on aged bones [30, 31].
Successful identification of skeletonized remains often relies upon DNA analyses, frequently focusing on the mid-diaphysis of weight-bearing long bones. In a study that explored intra-bone DNA variability using bovine and porcine femora, along with calcanei and tali, DNA from fresh and short-term environmentally exposed bone was extracted utilizing demineralization and standard lysis buffer protocols. DNA quantity and quality were measured. Overall, femoral epiphyses, metaphyses, and tarsals had more nuclear and mitochondrial DNA than did the femoral diaphyses. DNA loss was much more rapid in buried bones than in surface-exposed bones, while DNA quality differed based on environment but not bone region/element. However, the relation between nDNA yield and mtDNA CN was not explored, bones were of animal origin and either fresh or only short-term exposed to the environment (buried) [42]. To gain insight into specific contexts such as mtDNA copy number in decades-long environmentally exposed human bones, additional analyses must be performed. Because of the high complexity of DNA preservation in old skeletal remains, we recommend performing the mtDNA qPCR analyses before mtDNA NGS, and not assessing it from nDNA qPCR results, to avoid issues that could result from incorrect mtDNA input in high-priced NGS analyses. Low-cost qPCR tests are undemanding and—when working with challenging samples—a necessity.
Conclusion
According to the comparison between calculated mtDNA CN from nDNA quantity (PowerQuant [Auto] target) and qPCR measured mtDNA CN [113 bp target], there would have been an underestimation at assessing mtDNA CN in the 75 Second World War femurs studied. Because of the great complexity of aged bones, there is a need for mtDNA qPCR quantification for NGS analyses to correctly meet the input requirements of such analyses and to generate accurate results. It must be stressed that the association between nDNA and mtDNA examined in this study is only applicable in this way when comparing the results of the specific kit/method combination: a multi-copy target PowerQuant System (Promega) for nDNA and an in-house single-copy target qPCR mtDNA quantification method based on Alonso et al. [24] using short targets of each assay. Comparisons and relations between any other combinations of kits used for determining the nDNA quantity and mtDNA CN must be tested separately.
Data availability
The authors declare that all the data are available.
References
Allen M, Engerström AS, Meyers S, Handt O, Saldeen T, von Haeseler A, Pääbo S, Gyllensten U (1998) Mitochondrial DNA sequencing of shed hairs and saliva on robbery caps: sensitivity and matching probabilities. J Forensic Sci 43(3):453–464
Davis CL (1998) Mitochondrial DNA: State of Tennessee v Paul Ware. Profiles DNA 1:6
Divne AM, Nilsson M, Calloway C, Reynolds R, Erlich H, Allen M (2005) Forensic casework analysis using the HVI / HVII mtDNA linear array assay. J Forensic Sci 50(3):548–554
Dudás E, Susa É, Pamjav H, Szabolcsi Z (2020) Identification of World War II bone remains found in Ukraine using classical anthropological and mitochondrial DNA results. Int J Legal Med 134:487–489. https://doi.org/10.1007/s00414-019-02026-z
Marshall C, Taylor R, Sturk-Andreaggi K, Barritt-Ross S, Berg GE, McMahon TP (2020) Mitochondrial DNA haplogrouping to assist with the identification of unknown service members from the World War II Battle of Tarawa. Forensic Sci Int Genet 47:102291. https://doi.org/10.1016/j.fsigen.2020.102291
Goodwin WH (2016) DNA: Mitochondrial DNA. In: Payne-James J, Byard W (eds) Encyclopedia of forensic and legal medicine, 2nd edn. Elsevier, pp 351–358
Giles RE, Blanc H, Cann HM, Wallace DC (1980) Maternal inheritance of human mitochondrial DNA. Proc Natl Acad Sci USA 77(11):6715–6719
Pipek O, Medgyes-Horváth A, Dobos L, Steger J, Szalai-Gindl J, Visontai D et al (2019) Worldwide human mitochondrial haplogroup distribution from urban sewage. Sci Rep 9:11624. https://doi.org/10.1038/s41598-019-48093-5
Wurst C, Maixner F, Castella V, Cipollini G, Hotz G, Zink A (2022) The lady from Basel’s Barfüsserkirche – molecular confirmation of the mummy’s identity through mitochondrial DNA of living relatives spanning 22 generations. Forensic Sci Int Genet 56:102604. https://doi.org/10.1016/j.fsigen.2021.102604
Brown WM, George M Jr, Wilson AC (1979) Rapid evolution of animal mitochondrial DNA. Proc Natl Acad Sci USA 76:1967–1972
Veltri KI, Espiritu M, Singh G (1990) Distinct genomic copy number in mitochondria of different mammalian organs. J Cell Physiol 143:160–164
Lee YH, Chung U, Park MJ, Yoo JE, Han GR, Shin KJ (2006) Differential distribution of human mitochondrial DNA in somatic tissues and hairs. Ann Hum Genet 70(1):59–65. https://doi.org/10.1111/j.1529-8817.2005.00217.x
Higgins D, Rohrlach AB, Kaidonis J, Townsend G, Austin JJ (2015) Differential nuclear and mitochondrial DNA preservation in post-mortem teeth with implications for forensic and ancient DNA studies. PLoS One 10(5):e0126935. https://doi.org/10.1371/journal.pone.0126935
Wachsmuth M, Hübner A, Li M, Madea B, Stoneking M (2016) Age-related and heteroplasmy-related variation in human mtDNA copy number. PLoS Genet 12(3):e1005939. https://doi.org/10.1371/journal.pgen.1005939
Cuenca D, Battaglia J, Halsing M, Sheehan S (2020) Mitochondrial sequencing of missing persons DNA casework by implementing Thermo Fisher’s precision ID mtDNA whole genome assay. Genes 11:1303. https://doi.org/10.3390/genes11111303
Översti S, Majander K, Salmela E, Salo K, Arppe L, Belskiy S et al (2019) Human mitochondrial DNA lineages in Iron-Age Fennoscandia suggest incipient admixture and eastern introduction of farming-related maternal ancestry. Sci Rep 9:16883. https://doi.org/10.1038/s41598-019-51045-8
Ivanov P, Wadhams M, Roby R, Holland MM, Weedn VW, Parsons TJ (1996) Mitochondrial DNA sequence heteroplasmy in the Grand Duke of Russia Georgij Romanov establishes the authenticity of the remains of Tsar Nicholas II. Nat Genet 12:417–420. https://doi.org/10.1038/ng0496-417
Calvignac S, Konecny L, Malard F, Douady CJ (2011) Preventing the pollution of mitochondrial datasets with nuclear mitochondrial paralogs (numts). Mitochondrion 11:246–254. https://doi.org/10.1016/j.mito.2010.10.004
Churchill Cihlar J, Strobl C, Lagacé R, Muenzler M, Parson W, Budowle B (2020a) Distinguishing mitochondrial DNA and NUMT sequences amplified with the precision ID mtDNA whole genome panel. Mitochondrion 55:122–133
Niederstätter H, Köchl S, Grubwieser P, Pavlic M, Steinlechner M, Parson W (2007) A modular real-time PCR concept for determining the quantity and quality of human nuclear and mitochondrial DNA. Forensic Sci Int Genet 1(1):29–34. https://doi.org/10.1016/j.fsigen.2006.10.007
Andréasson H, Gyllensten U, Allen M (2002) Real-time DNA quantification of nuclear and mitochondrial DNA in forensic analysis. BioTechniques 33(2):402–411. https://doi.org/10.2144/02332rr07
Xavier C, Eduardoff M, Strobl C, Parson W (2019) SD quants – sensitive detection tetraplex-system for nuclear and mitochondrial DNA quantification and degradation inference. Forensic Sci Int Genetics 42:29–44. https://doi.org/10.1016/j.fsigen.2019.06.004
Kavlick MF (2019) Development of a triplex mtDNA qPCR assay to assess quantification, degradation, inhibition, and amplification target copy numbers. Mitochondrion 46:41–50. https://doi.org/10.1016/j.mito.2018.09.007
Alonso A, Martín P, Albarrán C, García P, García O, Fernández de Simón L, García-Hirschfeld J, Sancho M, Dela Rúa C, Fernández-Piqueras J (2004) Real-time PCR designs to estimate nuclear and mitochondrial DNA copy number in forensic and ancient DNA studies. Forensic Sci Int 139(2–3):141–149. https://doi.org/10.1016/j.forsciint.2003.10.008
Didier M, Welti S, Skillman J, Hickey S, Smith J, Peck M, Holt C, Kotkin M, Stephens K (2019) Establishing STR and identity SNP analysis thresholds for reliable interpretation and practical implementation of MPS gDNA casework. Forensic Sci Int Genet Suppl Ser 7(1):363–364. https://doi.org/10.1016/j.fsigss.2019.10.013
Børsting C, Rockenbauer E, Morling N (2009) Validation of a single nucleotide polymorphism (SNP) typing assay with 49 SNPs for forensic genetic testing in a laboratory accredited according to the ISO 17025 standard. Forensic Sci Int Genet 4(1):34–42. https://doi.org/10.1016/j.fsigen.2009.04.004
Buchard A, Kampmann ML, Poulsen L, Børsting C, Morling N (2019) ISO 17025 validation of a next-generation sequencing assay for relationship testing. Electrophoresis 37(21):2822–2831. https://doi.org/10.1002/elps.201600269
Thermo Fisher Scientific (2021) Precision ID mtDNA Panels with the HID Ion S5™/HID Ion GeneStudio™ S5 System application guide, Revision C.0. https://assets.thermofisher.com/TFS-Assets/LSG/manuals/MAN0017770_PrecisionID_mtDNA_Panels_S5_UG.pdf
Thermo Fisher Scientific (2022) Technical Note: The optimization of input DNA for applied biosystems™ precision ID mtDNA panels, revision A. https://assets.thermofisher.com/TFS-Assets/GSD/Technical-Notes/optimization-input-dna-precision-id-mtdna-panel-tech-note.pdf
Zupanič Pajnič I, Gornjak Pogorelc B, Balažic J (2010) Molecular genetic identification of skeletal remains from the Second World War Konfin I mass grave in Slovenia. Int J Legal Med 124:307–317. https://doi.org/10.1007/s00414-010-0431-y
Zupanič Pajnič I, Inkret J, Zupanc T, Podovšovnik E (2021) Comparison of nuclear DNA yield and STR typing success in Second World War petrous bones and metacarpals III. Forensic Sci Int Genet 55:1–6. https://doi.org/10.1016/j.fsigen.2021.102578
Šuligoj A, Mesesnel S, Leskovar T, Podovšivnik E, Zupanič Pajnič I (2022) Comparison of DNA preservation between adult and non-adult ancient skeletons. Int J Legal Med 136(6):1521–1539. https://doi.org/10.1007/s00414-022-02881-3
Zupanc T, Podovšovnik E, Zupanič Pajnič I, Obal M (2021) High DNA yield from metatarsal and metacarpal bones from Slovenian Second World War Skeletal remains. Forensic Sci Int Genet 51:102426. https://doi.org/10.1016/j.fsigen.2020.102426
Božič L, Benedik Bevc E, Podovšovnik E, Zupanc T, Zupanič Pajnič I (2022) Comparison of DNA preservation between ribs and vertebrae. Int J Legal Med 136(5):1247–1253. https://doi.org/10.1007/s00414-022-02860-8
Benedik Bevc T, Božič L, Podovšovnik E, Zupanc T, Zupanič Pajnič I (2021) Intra-bone nuclear DNA variability and STR typing success in Second World War 12th thoracic vertebrae. Forensic Sci Int Genet 55:1–7. https://doi.org/10.1016/j.fsigen.2021.102587
Božič L, Benedik Bevc T, Podovšovnik E, Zupanc T, Zupanič Pajnič I (2021) Intra-bone nuclear DNA variability and STR typing success in Second World War first ribs. Int J Legal Med 135(6):2199–2208. https://doi.org/10.1007/s00414-021-02681-1
Inkret J, Podovšovnik E, Zupanc T, Haring G, Zupanič Pajnič I (2021) Intra-bone nuclear DNA variability in Second World War metatarsal and metacarpal bones. Int J Legal Med 135(4):1245–1256. https://doi.org/10.1007/s00414-021-02528-9
Promega, PowerQuant®System technical manual, instructions for use of products PQ5002 and PQ5008, Revised 8/22. https://worldwide.promega.com/-/media/files/resources/protocols/technical-manuals/tmd/powerquant-system-technical-manual.pdf?rev=eed77410b69d43a2b0ea03fc78c7b69c&sc_lang=en
Zupanič Pajnič I (2016) Extraction of DNA from human skeletal material. In: Goodwin W (ed) Forensic DNA typing protocols, methods in molecular biology, Springer Science & Business Media, vol 1420. LLC, New York, pp 89–108
Geršak ŽM, Zupanič Pajnič I, Črešnar M, Zupanc T (2019) Determination of DNA yields rates in six different skeletal elements in ancient bones. Forensic Sci Int Genet 7(1):120–122. https://doi.org/10.1016/j.fsigss.2019.09.047
Obal M, Zupanič Pajnič I, Gornjak Pogorelc B, Zupanc T (2019) Different skeletal elements as a source of DNA for genetic identification of Second World War victims. Forensic Sci Int Genet Suppl Ser 7(1):27–29. https://doi.org/10.1016/j.fsigss.2019.09.013
Antinick TC, Foran DR (2019) Intra- and inter-element variability in mitochondrial and nuclear DNA from fresh and environmentally exposed skeletal remains. J Forensic Sci 64:88–97. https://doi.org/10.1111/1556-4029.13843
Acknowledgements
The authors would like to thank the Slovenian Government Commission on Concealed Mass Graves for its support in the exhumations of the Second World War victims.
Funding
Financial support for this study was provided by the Slovenian Research and Innovation Agency (project J3-3080 “Inferring ancestry from DNA for human identification”).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Research involves aged skeletons. The Medical Ethics Committee of the Republic of Slovenia approved the research (0120-22/2017/3).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• qPCR for mtDNA and nDNA was performed on 75 femurs from the Second World War victims.
• The relationship between calculated and measured mtDNA CN was explored.
• The comparison showed vast differences between calculated and measured results.
• MtDNA qPCR must be performed to meet the correct NGS input needs.
Supplementary information
ESM 1
(XLSX 23 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Obal, M., Zupanc, T. & Zupanič Pajnič, I. Measure quantity of mitochondrial DNA in aged bones or calculate it from nuclear DNA quantitative PCR results?. Int J Legal Med 137, 1653–1659 (2023). https://doi.org/10.1007/s00414-023-03074-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00414-023-03074-2