Abstract
Prader–Willi syndrome (PWS) in infants is characterized by hypotonia and poor sucking with feeding difficulties. Two autopsy cases of sudden unexpected death during sleep after tube feeding are described herein. For one, gastric aspiration caused by the possible milk regurgitation was suspected. Immunohistochemical examination of lung sections was performed using three antibodies to human α-lactalbumin, human gross cystic disease fluid protein 15, and cow whey β-lactoglobulin. Five cases of sudden unexpected infant death occurring earlier than at 6 months old were selected as controls. Marked immune-staining for infant formula in one PWS subject was evident within terminal bronchioles and alveoli with granular and amorphous features. However, no positive staining was apparent in the other subject, who exhibited contrasting features in milk distribution. Among control cases, one showed mild staining in the bronchiole, but the others did not. The antibody to β-lactoglobulin reacted specifically with formula, with no nonspecific background. Gastric contents in the airway can be a difficult issue because of the consequent terminal gasping. However, because of an episode of antemortem symptoms of potential regurgitation, and from findings at autopsy such as petechiae, we inferred that fatal regurgitation occurred in this PWS infant after tube feeding. Several clinical reports have described milk aspiration, but this pathological report is the first related to aspiration in PWS during tube feeding.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
An infant with Prader–Willi syndrome (PWS) is characterized by neonatal muscular hypotonia and poor sucking with feeding difficulties that require nasogastric tube placement for nutrition: a so-called floppy infant. After failure to thrive in early infancy, affected individuals develop an insatiable appetite, hyperphagia, and obesity during childhood [1]. Growth hormone treatment has improved patients’ body composition and prognosis, but many PWS patients die suddenly of respiratory and cardiovascular causes at all ages [2,3,4]. This genetic disorder, PWS, is caused by the lack of expression of paternal genes on the long arm of chromosome 15, either because of paternal chromosomal deletion or maternal disomy [5].
Milk and oral secretions are the most frequent foreign materials causing pulmonary aspiration in children [6]. Particularly, impairment of weak or uncoordinated swallowing increases risk of respiratory complications in PWS patients [7, 8]. Symptoms of aspiration are usually subtle. The affected patients might display increased noisy breathing during or after feeding [9]. Sudden unexpected death occurs during infancy. For such cases, suspected milk aspiration has been recorded in several reports [10,11,12,13].
However, in a PWS infant case with tube feeding, aspiration from regurgitation of gastric contents is expected to be more plausible than direct aspiration from impaired oral swallowing [14]. The inhalation of vomitus including acid, gastric particles, and the mixture of stomach contents engenders acute lung injury, clinically designated as acute respiratory distress syndrome, in addition to obstruction of the narrow airway [15].
Postmortem evaluation of gastric aspiration is not easy for a couple of reasons. Gastric contents in the airway can be a consequence of terminal gasping during physiological gastroesophageal reflux [16]. Alex et al. showed that aspiration of agonal or postmortem artifact was observed only to a mild degree among sudden infant death syndrome (SIDS) cases, suggesting that the features have limited relevance to the mechanism of suffocation [17]. Another reason is that such gastric aspiration might be enhanced during artificial cardiopulmonary resuscitation (CPR) attempts at the scene and hospital [18]. However, almost every infant is transferred to an emergency hospital, even in a situation of cardiac arrest, where the infant subsequently receives CPR [17]. Forensic pathologists often encounter the dilemma of antemortem fatal aspiration or a secondary terminal event.
As described herein, we examined two cases of sudden unexpected infant death (SUID) of PWS. The infants of these cases, who were found unconscious during sleep after tube feeding, exhibited quite contrasting features in postmortem examinations. To investigate the possibility of gastric aspiration, we attempted to detect milk components using immunohistochemical analysis. After presenting the results of our investigation, we can discuss their meanings.
Case presentation
Two cases of SUID occurring in PWS infants are described below. Major findings of postmortem examinations are presented in Table 1.
Case PWS#1: A 5-month-old male infant had been born by caesarean section at the full term of gestation, with birth weight of 2,144 g. He had respiratory distress, hypotonia, and feeding failure after birth, for which he was admitted to the neonatal intensive care unit (NICU) for 2 months. PWS was diagnosed based on genetic testing as deletion in the SNRPN-containing region of the father’s chromosome 15. His mother fed him formula through a nasogastric tube at home. Two days before his death, she noticed hard breathing and noisy throat in the baby. She took him to a pediatrician. Two days later, in the early morning, he was found unresponsive in the supine position at home. Then, he was taken to an emergency hospital. He was in cardiopulmonary arrest on arrival. He died 30 min after arrival at the hospital while undergoing CPR.
Case PWS#2: A 3-month-old female infant, who had been delivered vaginally at 34 weeks, with birth weight of 2690 g. PWS was suspected from a low Apgar score, feeding failure, and hypotonia and was subsequently diagnosed from genetic testing as having paternal deletion. After hospitalization in the NICU for 6 weeks, she had a seizure at home. However, an electroencephalogram yielded no abnormal finding. One day around noon, 3 h after feeding her baby through a gastric tube, the mother found the baby to be unresponsive during sleep. She rushed her baby to the emergency room. The baby was in cardiopulmonary arrest on arrival, but CPR treatment restored her heartbeat temporarily. Finally, she died 5 h after arriving at the hospital.
As control subjects, we selected five cases of SUID involving infants younger than 6 months old who were compatible to the two subjects. The PWS and control subjects described herein were autopsied in our department within 2 days after death.
Findings at autopsy and in histopathology
The series of postmortem examinations was performed extensively as reported earlier [19]. Briefly, full autopsy was conducted with tissue examination of formalin-fixed organs using hematoxylin–eosin (H&E) staining under microscopy, including special staining, such as Gram staining. Biochemical laboratory tests were applied for a couple of markers that are stable at postmortem. However, only a few test items such as C reactive protein (CRP) were examined because of the limited amount of serum samples obtained from autopsy. Viral titer including specific antibody was measured in the serum for adenovirus types 3 and 7, influenza A and B viruses, respiratory syncytial virus, coxsackievirus B1–B6, and cytomegalovirus IgG and IgM [20]. Blood culture was also performed for screening for pathogenic bacteria. Furthermore, we routinely check genome sequence for major genes regarded as causes of cardiovascular diseases such as QT prolongation, Brugada syndrome, arrhythmogenic right ventricular cardiomyopathy, and hypertrophic cardiomyopathy in the Ion PGM system (Thermo Fisher Scientific Inc.) [21].
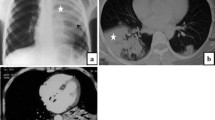
Case PWS#1: Gross findings at autopsy included many petechiae in the thymus, epicardium, and pleura. Coagulated milk was found in the lower part of the esophagus, as well as in the stomach (Fig. 1). Small whitish aggregates were present in the lung bronchi. Tonsils and cervical lymph nodes were swollen. Histological examination under a microscope showed increased lymphatic follicles in the tonsils and hemophagocytic features in the cervical lymph nodes. Neutrophil infiltration was observed in the spleen. The level of CRP was 0.2 mg/dl. All examined viruses were negative. Genetic testing detected no predictable pathogenic mutation for cardiovascular diseases.
Case PWS#2: At autopsy, no marked change was found except for congestion in the organs. No apparent petechiae were observed in the thymus, epicardium, or pleura. The stomach was empty. No milk aggregate was present in the esophagus. The lymph nodes were not swollen. Inflammatory cell proliferation in the spleen was not observed, CRP was 0.3 mg/dl. Titers were below the reference level for all viruses. No pathogenetic mutation was detected in candidate genes of cardiovascular diseases.
Immunohistochemical analysis
In an earlier study, we detected breast milk and infant formula components using specific antibodies [22]. Immunohistochemical analysis was applied to lung tissues. At least four specimens were obtained, including one each from the upper left, lower left, upper right, and lower right lobe. Deparaffinized sections of 3-µm thickness were prepared. Immunological reactions were tested using the following antibodies to human α-lactalbumin (Dako, Agilent Corp., USA), human gross cystic disease fluid protein 15 (GCDFP15) (Dako), and cow whey protein β-lactoglobulin (Morinaga Milk Industry Co. Ltd., Yokohama, Japan). Iwadate et al. [23] recommended the antibody for human α-lactalbumin for immunohistochemical analysis to detect milk. Immunostaining was conducted in the EnVision Flex system (Dako) using diaminobenzidine as the substrate for peroxidase, as described earlier [24].
Immunohistochemical analysis was applied to sections of the lungs. The staining features were compatible among the three antibodies used. Antibodies to α-lactalbumin and GCDFP15 antibodies reacted with both breast milk and infant formula, accompanied by cross-reactive positive staining like mucus cells, and mild background. By contrast, anti-β-lactoglobulin antibody reacted only with formula, but the reaction was specific with no background in the specimens. For milk, particularly formula, β-lactoglobulin yielded the best result.
As presented in Fig. 2, marked immunostaining for the feeding component was evident within terminal bronchiole and alveolar spaces with granular and amorphous patterns for case PWS#1. Foreign materials were identified with eosinophilic granules in serial sections by H&E staining. By contrast, no apparent positive staining was observed in the lung sections associated with case PWS#2.
H&E staining (A–D) and immunohistochemistry using anti-β-lactoglobulin antibody (E–H) to the lung sections for the PWS and control subjects: A and E, bronchiole in PWS#1 (× 100); B and F, alveoli in PWS#1 (× 100); C and G, PWS#2 (× 40); and D and H, control #1 (× 100). The left H&E staining and the other right immunostaining are serial sections
For the five control cases, one showed positive, with mild staining only in the bronchiole of lungs; the others did not. In three cases including the positive one, small volumes of milk aggregates were present in the stomach, but not in their esophagus. The degrees of major findings are presented in Table 1.
Discussion
Because gastric aspiration was suspected for one of the two SUID cases of PWS, the presence of milk in the airway was examined using immunohistochemical analysis with antibodies specific to the components. The infant in case PWS#1 was found to have a large amount of formula in the bronchioles and alveoli of the lungs, but none was found for case PWS#2.
For case PWS#1, formula components were detected widely in bronchioles and in alveoli. The distribution was much deeper than that of the positive case C#1 in the control group. Therefore, the features in PWS case were unlikely to be an artifact caused by agonal gastroesophageal reflux. The mother might have noticed regurgitation sounds in the infant’s throat with a visit to hospital. The infant should suffer from abnormal swallowing or regurgitation in tube feeding. There was also extreme petechial bleeding. Based on the observations described above, it can be inferred that muscular hypotonia was associated with the lethal event. Mohammed et al. reported that nasogastric intubation led to aspiration pneumonia as a result of gastric dilatation [25]. We inferred that regurgitation of gastric contents occurred in this PWS#1 infant under tube feeding.
In general, gastric contents are inhaled into the lungs of 12–40% of infants whose deaths are attributed to SIDS or SUID [14, 26]. This spontaneous aspiration caused by gastroesophageal reflux, including steps of relaxation of esophageal sphincters, suppression of upper airway protective reflexes, and terminal inspiratory efforts, occurs even among non-resuscitated corpses as a terminal event [27]. Terminal gasping and autoresuscitation are thought to generate aspiration from the gastroesophageal route [14, 28, 29]. It is noteworthy that the presence of gastric contents in the airway does not invariably imply the occurrence of antemortem fatal aspiration [26].
The presence of milk in the lungs was also observed in one among the five control SUID cases. In this C#1 case, because a tiny volume of milk was present in the stomach, secondary gastric aspiration can be expected to have occurred in the agonal or postmortem phase. In accordance with the evidence obtained in a previous series of SIDS autopsy cases [17, 26], the degree of aspiration was limited to a mild degree. The sleeping position at the scene reportedly was supine for the two cases of milk aspiration: PWS#1 and C#1. However, no apparent difference of the risk of gastric aspiration into the upper airway and lungs has been confirmed by placing infants in either a supine or prone sleeping position [30].
Formula could not be confirmed in bronchioles or pulmonary alveoli of the PWS#2 infant. Resuscitation was performed for 5 h. Judging whether milk aspiration occurred at the time of death is difficult. Krous et al. [15] pointed out that the aspiration observation is not apparent in infants that had achieved resuscitation for over 48 h. However, it is unlikely that milk would become undetectable within about 5 h. Therefore, the case is judged as sudden death that had occurred by a mechanism other than aspiration. Central apnea has been known to occur in PWS [31, 32], but confirming central apnea is difficult based solely on postmortem examinations. Since no other cause was identifiable, SIDS is suspected to have occurred in this PWS#2 case.
Identifying ingested milk coagulates in the airway solely based on morphology in H&E-stained sections was not easy. For immunohistochemical analysis of milk, the use of α-lactoalbumin antibody has been recommended [22]. Maiese et al. [33] verified milk aspiration from results of immunohistochemistry. However, this anti-α-lactoalbumin antibody was unavailable. The manufacturer discontinued the product. This study used anti-GCDFP15. This antibody also reacted with both breast milk and infant formula, producing comparable staining patterns. In clinical practice, the breast cancer marker GCDFP15 antibody is used routinely with reaction conditions that have been already set by laboratories. These antibodies are useful for milk screening, but an important shortcoming related to their use is that they also react with, for example, goblet cells that secrete mucin.
Our earlier study demonstrated that β-lactoglobulin antibody is effective for detecting infant formula [22]. The reaction is specific. Moreover, it has the least non-specific background in these immunohistochemical examinations. However, it is a shortcoming that this antibody reacts only with formula, but not with human breast milk [34]. Results of this study also support the usefulness of this antibody for the detection of infant formula.
In conclusion, two contrasting cases of PWS have been presented herein. In one case, gastric aspiration was confirmed from immunohistochemical examination using antibodies to milk and formula components, which were effective to prove their presence in the airway. Results indicate that gastric milk can be aspirated into the airway during and after tube feeding in PWS infants.
References
Cassidy SB, Schwartz S, Miller JL, Driscoll DJ (2012) Prader-Willi syndrome. Genet Med 14:10–26. https://doi.org/10.1038/gim.0b013e31822bead0
Butler MG, Manzardo AM, Heinemann J, Loker C, Loker J (2017) Causes of death in Prader-Willi syndrome: Prader-Willi syndrome association (USA) 40-year mortality survey. Genet Med 19:635–642. https://doi.org/10.1038/gim.2016.178
Pacoricona Alfaro DL, Lemoine P, Ehlinger V, Molinas C, Diene G, Valette M, Pinto G, Coupaye M, Poitou-Bernert C, Thuilleaux D, Arnaud C, Tauber M (2019) Causes of death in Prader-Willi syndrome: lessons from 11 years’ experience of a national reference center. Orphanet J Rare Dis 14:238. https://doi.org/10.1186/s13023-019-1214-2
Pomara C, D’Errico S, Riezzo I, de Cillis GP, Fineschi V (2005) Sudden cardiac death in a child affected by Prader-Willi syndrome. Int J Legal Med 119:153–157. https://doi.org/10.1007/s00414-004-0513-9
Sahoo T, del Gaudio D, German JR, Shinawi M, Peters SU, Person RE, Garnica A, Cheung SW, Beaudet AL (2008) Prader-Willi phenotype caused by paternal deficiency for the HBII-85 C/D box small nucleolar RNA cluster. Nat Genet 40:719–721. https://doi.org/10.1038/ng.158
Karim RM, Momin IA, Lalani II, Merchant SS, Sewani AA, Hassan BS, Mahmood N (1999) Aspiration pneumonia in pediatric age group: etiology, predisposing factors and clinical outcome. J Pak Med Assoc 49:105–108
Hurren BJ, Flack NA (2016) Prader-Willi syndrome: a spectrum of anatomical and clinical features. Clin Anat 29:590–605. https://doi.org/10.1002/ca.22686
Salehi P, Stafford HJ, Glass RP, Leavitt A, Beck AE, McAfee A, Ambartsumyan L, Chen M (2017) Silent aspiration in infants with Prader-Willi syndrome identified by videofluoroscopic swallow study. Medicine (Baltimore) 96:e9256. https://doi.org/10.1097/MD.0000000000009256
Tan HL, Urquhart DS (2017) Respiratory complications in children with Prader Willi syndrome. Paediatr Respir Rev 22:52–59. https://doi.org/10.1016/j.prrv.2016.08.002
Van Vliet G, Deal CL, Crock PA, Robitaille Y, Oligny LL (2004) Sudden death in growth hormone-treated children with Prader-Willi syndrome. J Pediatr 144:129–131. https://doi.org/10.1016/j.jpeds.2003.09.049
Schrander-Stumpel CT, Curfs LM, Sastrowijoto P, Cassidy SB, Schrander JJ, Fryns JP (2004) Prader-Willi syndrome: causes of death in an international series of 27 cases. Am J Med Genet A 124A:333–338. https://doi.org/10.1002/ajmg.a.20371
Nagai T, Obata K, Tonoki H, Temma S, Murakami N, Katada Y, Yoshino A, Sakazume S, Takahashi E, Sakuta R, Niikawa N (2005) Cause of sudden, unexpected death of Prader-Willi syndrome patients with or without growth hormone treatment. Am J Med Genet A 136:45–48. https://doi.org/10.1002/ajmg.a.30777
Eiholzer U (2005) Deaths in children with Prader-Willi syndrome. A contribution to the debate about the safety of growth hormone treatment in children with PWS. Horm Res 63:33–39. https://doi.org/10.1159/000082745
Sloan TB, Kaye CI (1991) Rumination risk of aspiration of gastric contents in the Prader-Willi syndrome. Anesth Analg 73:492–495. https://doi.org/10.1213/00000539-199110000-00023
Raghavendran K, Nemzek J, Napolitano LM, Knight PR (2011) Aspiration-induced lung injury. Crit Care Med 39:818–826. https://doi.org/10.1097/CCM.0b013e31820a856b
Krous HF, Masoumi H, Haas EA, Chadwick AE, Stanley C, Thach BT (2007) Aspiration of gastric contents in sudden infant death syndrome without cardiopulmonary resuscitation. J Pediatr 150:241–246. https://doi.org/10.1016/j.jpeds.2006.12.012
Alex N, Thompson JM, Becroft DM, Mitchell EA (2005) Pulmonary aspiration of gastric contents and the sudden infant death syndrome. J Paediatr Child Health 41:428–431. https://doi.org/10.1111/j.1440-1754.2005.00660.x
Krous HF, Haas EA, Chadwick AE, Masoumi H, Mhoyan A, Stanley C (2008) Delayed death in sudden infant death syndrome: a San Diego SIDS/SUDC Research Project 15-year population-based report. Forensic Sci Int 176:209–216. https://doi.org/10.1016/j.forsciint.2007.09.009
Osawa M, Ueno Y, Ikeda N, Ikematsu K, Yamamoto T, Irie W, Kozawa S, Kotani H, Hamayasu H, Murase T, Shingu K, Sugimoto M, Nagao R, Kakimoto Y (2020) Circumstances and factors of sleep-related sudden infancy deaths in Japan. PLoS One 15:e0233253. https://doi.org/10.1371/journal.pone.0233253
Osawa M, Nagao R, Kakimoto Y, Kakiuchi Y, Satoh F (2019) Sudden infant death after vaccination: survey of forensic autopsy files. Am J Forensic Med Pathol 40:232–237. https://doi.org/10.1097/PAF.0000000000000494
Osawa M, Inaoka Y, Ochiai E, Hasegawa I, Satoh F (2014) Variable single C repetitive tract in KCNQ1: postmortem molecular testing in a sudden and unexpected death. Exp Clin Cardiol 20:927–930
Kakimoto Y, Matsushima Y, Tanaka M, Hayashi H, Wang T, Yokoyama K, Ochiai E, Osawa M (2020) MicroRNA profiling of gastric content from breast-fed and formula-fed infants to estimate last feeding: a pilot study. Int J Legal Med 134:903–909. https://doi.org/10.1007/s00414-019-02226-7
Iwadate K, Doy M, Nishimaki Y, Liang F, Takatori T, Hasekura H (2000) Immunohistochemical examination of the lungs in infant death cases using antibodies against milk components. Forensic Sci Int 110:19–28. https://doi.org/10.1016/s0379-0738(99)00229-7
Satoh F, Tachibana H, Hasegawa I, Osawa M (2010) Sudden death caused by chronic Chagas disease in a non-endemic country: autopsy report. Pathol Int 60:225–240. https://doi.org/10.1111/j.1440-1827.2009.02503.x
Mohammed AM, Dennis RJ (2016) Use of a venting PEG tube in the management of recurrent acute gastric dilatation associated with Prader-Willi syndrome. J Surg Case Rep 1:1–2. https://doi.org/10.1093/jscr/rjv174
Iwadate K, Doy M, Ito Y (2001) Screening of milk aspiration in 105 infant death cases by immunostaining with anti-human alpha-lactalbumin antibody. Forensic Sci Int 122:95–100. https://doi.org/10.1016/s0379-0738(01)00469-8
Thach BT (2000) Sudden infant death syndrome: can gastroesophageal reflux cause sudden infant death? Am J Med 108(Suppl 4a):144S-148S. https://doi.org/10.1016/s0002-9343(99)00354-x
Al-Adnani M, Cohen MC, Scheimberg I (2011) Gastroesophageal reflux disease and sudden infant death: mechanisms behind an under-recognized association. Pediatr Dev Pathol 14:53–56. https://doi.org/10.2350/09-11-0748-CR.1
Hornby L, Dhanani S, Shemie SD (2018) Update of a systematic review of autoresuscitation after cardiac arrest. Crit Care Med 46:e268–e272. https://doi.org/10.1097/CCM.0000000000002920
Byard RW, Beal SM (2000) Gastric aspiration and sleeping position in infancy and early childhood. J Paediatr Child Health 36:403–405. https://doi.org/10.1046/j.1440-1754.2000.00503.x
Urquhart DS, Gulliver T, Williams G, Harris MA, Nyunt O, Suresh S (2013) Central sleep-disordered breathing and the effects of oxygen therapy in infants with Prader-Willi syndrome. Arch Dis Child 98:592–595. https://doi.org/10.1136/archdischild-2012-303441
Cohen M, Hamilton J, Narang I (2014) Clinically important age-related differences in sleep related disordered breathing in infants and children with Prader-Willi Syndrome. PLoS One 9:e101012. https://doi.org/10.1371/journal.pone.0101012
Maiese A, La Russa R, Arcangeli M, Volonnino G, De Matteis A, Frati P, Fineschi V (2020) Multidisciplinary approach to suspected sudden unexpected infant death caused by milk-aspiration: a case report. World J Clin Cases 8:4128–4134. https://doi.org/10.12998/wjcc.v8.i18.4128
Mäkinen-Kiljunen S, Sorva R (1993) Bovine beta-lactoglobulin levels in hydrolysed protein formulas for infant feeding. Clin Exp Allergy 23:287–291. https://doi.org/10.1111/j.1365-2222.1993
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Research involving human participants and/or animals
Not applicable.
Ethics approval
This project was approved as a retrospective clinical study by the Ethics Committee of Tokai University School of Medicine.
Informed consent
Available for research. The study protocol is disclosed on our web site as a project of opting out. According to the guidance of the ethical committee, publication is possible with protection of personal information.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Osawa, M., Ikeda, H., Ueda, A. et al. Gastric aspiration in sudden unexpected infant death of Prader–Willi syndrome: immunohistochemical detection of feeding components. Int J Legal Med 136, 1883–1888 (2022). https://doi.org/10.1007/s00414-022-02883-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00414-022-02883-1