Abstract
Since widespread vaccination against COVID-19, the development of effective antiviral drugs, and the decreasing number of patients with COVID-19 in intensive care, the risk from SARS-CoV-2 infection appears less threatening. However, studies show that a significant number of patients suffer from long-term sequelae, even months after SARS-CoV-2 infection. The so-called post-COVID syndrome (PCS) often presents a diagnostic and treatment challenge for physicians. This study protocol describes the “All Eyes on PCS” study, which aims to investigate the retinal microvasculature in PCS patients and COVID-19-recovered patients to provide new insights into the pathophysiology of PCS. “All Eyes on PCS” is a prospective, case–control study with the primary objective of detecting endothelial dysfunction (ED) in patients with PCS. Therefore, we intend to recruit patients with PCS, fully SARS-CoV-2-infection-recovered (CR) participants, and SARS-CoV-2-infection-naïve (CN) participants. Baseline measurements will include: (1) patient-specific characteristics, (2) biochemistry, (3) retinal vessel analysis (RVA), (4) survey questionnaires as patient-reported outcomes measurements (PROMs), (5) optical coherence tomography (OCT), OCT angiography (OCTA), and adaptive optics (AO), (6) blood pressure recordings, (7) handgrip strength test. After 6 months, baseline measurements will be repeated in the PCS cohort, and after 1 year, a telephone query will be conducted to assess residual symptoms and treatment needs. The aim of this study is to gain insight into the pathophysiology of PCS and to provide an objective biomarker for diagnosis and treatment, while also creating a comprehensive clinical database of PCS patients.
ClinicalTrials.gov Identifier: NCT05635552; Date: 2.12.2022.
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
Background and rationale
Introduction
COVID-19 is a systemic disease that may result in hyperinflammation caused by infection with the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). In the acute phase of severe disease, hyperinflammation is associated with thromboembolism, myocarditis, disseminated intravascular coagulation, and subsequent respiratory distress syndrome [1]. Although the lungs are primarily affected in this stage, SARS-CoV-2 can also cause systemic endothelial dysfunction and inflammation [2].
Endothelial dysfunction (ED) in acute SARS-CoV-2 infection is characterized by elevated markers of endothelial damage and coagulopathy, and SARS-CoV-2-RNA has been directly detected in the endothelium of deceased patients [3,4,5,6]. The angiotensin-converting enzyme-2 (ACE-2) receptor, which serves as the entry point of SARS-CoV-2 into cells, is expressed not only in various tissues but also in endothelial cells [7, 8]. There is also evidence that the ACE-2 receptor and transmembrane protease serine subtype 2 (TMPRSS2) are expressed in retinal cells, which may facilitate viral internalization into the cell [9, 10]. Studies have shown that infection with the viral S1 subunit leads to endothelial damage, cytokine release, and thrombosis [11, 12]. Internalization of SARS-CoV-2 into the cell and retention in the endosome may lead to Toll-like receptor (TLR)-dependent activation of the NF-κB pathway, resulting in changes in the expression of various genes that could contribute to ED [13]. Whether ED is directly caused by the virus or only indirectly through systemic inflammation remains unclear [14].
While most of SARS-CoV-2-infected individuals fully recover after several weeks, symptoms such as shortness of breath, chest pain, cognitive dysfunction, fatigue, and palpitations persist for months in several patients, as do psychosocial symptoms such as depression, sleep disturbances, and agitation [15]. This is commonly referred to as post-COVID syndrome (PCS, also known as Long-COVID, post-COVID condition), which the WHO defines as a complex of complaints following SARS-CoV-2 infection, usually occurring 3 months after the onset of COVID-19 with symptoms lasting at least 2 months and which cannot be explained by any other diagnosis [16]. In most cases, developing after a mild to moderate primary infection, PCS has an estimated prevalence of around 12–35%. It is important to distinguish from post-intensive care syndrome (PICS), which refers to long-term physical, cognitive, and psychological symptoms that can occur after treatment of severe SARS-CoV-2 infection in intensive care unit (ICU) [17,18,19].
The pathophysiology and mechanisms leading to PCS are still unclear. One possible explanation is that ED persists longer in patients than the acute infection and contributes to chronic, ongoing post-infectious symptoms [20,21,22]. One common symptom in patients with PCS is persistent fatigue, which often fulfills diagnostic criteria of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) [23, 24]. Patients with ME/CFS also show ED which affects both small and large vessels [25]. Immunological abnormalities due to chronic virus persistence may lead to inflammation, and therefore to a prolonged disturbance of the fine interplay between endothelium, immune cells, and platelets [26, 27].
Although some patients with PCS exhibit organ-related involvement, such as myocarditis detected by MRI, elevated troponin T levels, or reduced pulmonary diffusion capacity, there is currently no established routine parameter for characterizing PCS [28]. Endothelial function seems to improve after acute SARS-CoV-2 infection; however, it is still detectable via flow-mediated dilation (FMD) after 6 months [29]. There is evidence suggesting that persistent ED is an independent marker of developing PCS and preliminary findings suggest that the retinal microcirculation may also be affected following severe SARS-CoV-2 infection [30, 31]. However, to our knowledge, until now retinal vessel analysis (RVA) has not been used in PCS patients to characterize ED.
Our hypothesis is that patients with PCS suffer from a prolonged ED which leads to typical PCS symptoms such as fatigue, dyspnea, and cognitive dysfunctions. To examine the responsiveness of retinal vessels to flickering light as a surrogate for ED, we will use RVA and to further characterize the retinal microcirculation, we will include OCT angiography (OCTA) and adaptive optics (AO) measurements. With the study “All Eyes on PCS”, we want to bring new insights into the pathophysiology and possible risk factors of prolonged ED in PCS patients. An impaired retinal microcirculation might prove as a strong and independent predictor of PCS, and therefore serve as an objective biomarker in diagnosis and therapy monitoring. Our objective is to establish a comprehensive biobank comprising individuals with PCS and those who have recovered from COVID-19, with the aim of investigating the pathophysiological mechanisms underlying PCS. The biobank will serve as a valuable resource aimed at understanding the molecular, cellular, and immunological aspects of PCS and help identify potential targets for intervention.
Objectives
Research hypothesis
Patients with PCS exhibit prolonged ED when compared with a SARS-CoV-2-infection-recovered (CR) cohort and SARS-CoV-2-infection-naïve (CN) cohort. This prolonged ED is accompanied by a range of PCS-specific complaints, including fatigue, palpitations, and cognitive impairment. We hypothesize that the symptom burden of PCS is associated with an impaired retinal vessel responsiveness and changes in vessel morphology.
The primary objective of our study is to quantify retinal vessel responsiveness and vessel morphology using the Retinal Vessel Analyzer (RVA) in patients with PCS and compare these parameters to those of the CR and CN cohort.
Secondary objectives are:
-
(1)
To collect patient-reported symptoms (PROMs), assess PCS symptom severity, and then correlate these findings with RVA parameters at baseline.
-
(2)
To monitor the development of symptom severity of PCS and endothelial function after 6 months in the PCS cohort.
-
(3)
To conduct an in-depth study of the retina microcirculation using OCTA and AO.
-
(4)
To classify patients based on the different organ systems affected by PCS and compare RVA parameters between these groups.
-
(5)
To quantify fatigue in patients using the handgrip strength test as a measure of excessive fatiguability.
-
(6)
To use cell culture experiments on retinal epithelial and endothelial cells to explain a possible phenotype of ED in PCS patients.
-
(7)
To characterize the immune phenotype of T cell and monocyte subpopulations in PCS patients.
-
(8)
To investigate the potential contribution of virus reactivation, specifically Epstein–Barr virus (EBV), to ED in PCS patients.
-
(9)
To establish a possible link between ED and the development of autonomic dysfunction by measuring basal cortisol levels in PCS patients.
Trial design
Methods: participants, measurements, and outcomes
Data collection will take place in the University Hospital of the Technical University (TU) Munich, Klinikum rechts der Isar, Germany. Three departments are involved in data collection: Department of Nephrology (lead), Department of Neurology, and Department of Ophthalmology. Recruitment of patients will take place in the post-COVID outpatient clinic at the University Hospital of the Ludwigs-Maximilians-University (LMU) Munich, Campus Großhadern, in general practitioners and via social media.
Inclusion criteria:
-
i.
Patients with PCS (positive PCR or positive rapid antibody test ≥3 months) with a currently existing, PCS-typical complaint complex, ongoing for at least 2 months that cannot be explained by an alternative diagnosis.
-
ii.
Control group (CR): participants recovered from SARS-CoV-2 infection (positive PCR or positive rapid antibody test ≥ 3 months) without any residual symptoms.
-
iii.
Healthy cohort (CN): no history of SARS-CoV-2 infection (exclusion via measurement of disease specific antibodies).
Exclusion criteria:
-
i.
Missing or incomplete consent form.
-
ii.
Age < 18 years.
-
iii.
Pregnancy.
-
iv.
Malignancy.
-
v.
Diseases associated with a significant change in life expectancy.
-
vi.
Autoimmune diseases of the rheumatological type.
-
vii.
Cataract.
-
viii.
Epilepsy.
-
ix.
Glaucoma.
Measurements are carried out by experienced examiners in the departments of nephrology, neurology and ophthalmology. Informed consent is obtained by a clinical study investigator prior to inclusion. A structured medical history and the entry criteria are re-evaluated. A written consent form approved by the ethics committee of the Technical University of Munich, School of Medicine, is used to obtain informed consent.
To establish a PCS biobank, we ask patients to donate their biospecimens. For this purpose, we use an information sheet approved by the ethics committee of the Technical University of Munich, School of Medicine.
Outcomes
Primary outcome
PCS patients show an impaired retinal vessel responsiveness and microcirculation when compared to a SARS-CoV-2-infection-recovered cohort. To investigate ED, both parameters (vMax and aMax) are important [32]. In a recent publication, we could show that vMax is a predictor for all-cause mortality in end-stage renal disease (ESRD) [33]. In addition, we will compare parameters of retinal microcirculation. To analyze microvascular function, central retinal artery equivalent (CRAE), central retinal vein equivalent (CRVE), and arteriolar–venular ratio (AVR) are important variables. Narrower CRAE and wider CRVE have been shown to be predictors for cardiovascular mortality [34]. Therefore, our primary endpoint will focus on differences in both dynamic retinal vessel analysis (DVA, vMax and aMax) and static retinal vessel analysis (SVA, CRAE, CRVE, and AVR) between our PCS cohort and fully SARS-CoV-2-infection-recovered patients.
Secondary outcomes:
-
(1)
To determine whether PCS patients have an impaired retinal vessel responsiveness at baseline compared to the CN cohort. DVA parameters, including aMax and vMax, will be measured at baseline, and mean or median values will be calculated for each cohort, respectively.
-
(2)
To assess whether SVA parameters of retinal vessel analysis (including CRAE, CRVE and AVR) are altered in the PCS cohort compared with CN cohorts. The baseline, mean or median values of these parameters will be calculated for each cohort, respectively.
-
(3)
To investigate whether PCS patients show an improved retinal vessel responsiveness and static parameters after 6 months compared to baseline parameters. The static and dynamic parameters will be measured at baseline and follow-up and mean or median values will be calculated for each cohort, respectively.
-
(4)
To assess whether symptom severity of PCS correlates with impaired retinal vessel responsiveness and static parameters of retinal vessel analysis. The symptom severity measured with patient-reported outcome measures (PROMs), along with the baseline static and dynamic parameters, will be assessed, and mean or median values will be calculated.
-
(5)
To examine whether patients with PCS show a chronic immune activation and a changed amount of circulating angiogenic cells (CAC) and circulating endothelial progenitor cells (CEC) in fluorescence-activated cell sorting (FACS) analysis compared with the CR and CN cohort. At baseline, percentage of CAC and CEC will be measured and mean or median values will be calculated for each cohort, respectively.
-
(6)
To assess whether epithelial and endothelial cells show a change in surface markers and inflammation markers when incubated with recombinant SARS-CoV-2 S1 subunit protein and/or patient serum from the PCS cohort, in cell culture experiments.
-
(7)
To determine whether PCS patients show elevated markers of ED (sICAM, sVCAM, Thrombomodulin, P-Selektin, ADMA, SADMA, Endothelin-1, ACE-1, ACE-2, ANG-2, Pentraxin-3, GDF-15) compared to the CR and CN cohort. At baseline, mean or median values of these markers will be calculated for each cohort, respectively.
-
(8)
To investigate whether PCS patients show elevated markers of chronic inflammation (INFß, TNFα, IFNy, Il-8, Il-6, Il-1ß, Mcp1, Il-10) compared to the CR and CN cohort. At baseline, mean or median values of these markers will be calculated for each cohort, respectively.
-
(9)
To examine whether PCS patients show a reactivation of Epstein–Barr virus (EBV). At baseline, PCR of EBV DNA will be measured and mean or median values will be calculated for each cohort, respectively.
-
(10)
To assess whether patients with PCS show changes of the retinal vasculature as measured by OCT angiography. At baseline, mean values of retinal vessel densities of the superficial and deep vascular complex as well as size of the foveal avascular zone in both eyes will be measured for each cohort, respectively.
-
(11)
To investigate whether PCS patients with impaired retinal vessel responsiveness show an autonomic dysfunction characterized by low cortisol levels. At baseline, mean or median values of cortisol levels will be measured for each cohort, respectively.
-
(12)
To explore whether PCS patients with fatigue fulfill diagnostic criteria of ME/CFS. At baseline, the Canadian Criteria of CFS score and the handgrip strength will be measured and mean or median will be calculated)
Participant timeline
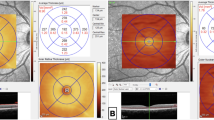
Recruitment for our study is scheduled to begin in October 2022, with the first patients expected to be measured by the end of 2022 or early 2023. In addition to measuring the PCS cohort, CR participants will be recruited and measured, with completion of baseline measurements (T0) planned by June 2023. In the PCS cohort, measurements will be repeated after 6 months (T1), with completion planned by the end of September 2023. One year after the initial measurements, we will conduct a telephone survey to assess any residual symptoms and treatment needs. Data analysis and especially data quality control will continuously happen after measurement of the first patient. Cell culture experiments are ongoing starting with incubation of retinal endothelial cells with SARS-CoV-2 spike antigen (S1) followed by incubation with patient serum (Fig. 1).
Participant timeline and overview of conducted measurements. Figure shows baseline (T0) and follow-up (T1) measurements that are conducted in the “All Eyes on PCS” study. The figure provides a visual representation of the planned measurements and includes sample images of key assessment tools, including adaptive optics (AO), dynamic and static retinal vessel analysis (DVA, SVA) and OCT angiography (OCTA), and patient reported outcome measures (PROMs)
For a detailed list of the variables to be measured, the departments involved in the study, and the relevant hypotheses, please refer to Table 1.
Sample size
In the PCS and CR cohort, we plan to recruit 100 participants. The SARS-CoV-2-infection-naïve cohort is defined as no history of SARS-CoV-2 infection (exclusion via measurement of specific antibodies) and consists of an already established, pre-pandemic healthy cohort and a cohort recruited during the pandemic.
Primarily, a comparison of the group means regarding the dilation of the retinal vessels will be performed using an analysis of variance (ANOVA) (significance level α = 5%).
To estimate the required sample size, we will assume a standard deviation within groups of 2%, based on the largest normative sample published to date, which is consistent with previous healthy control studies in the “diseased vs. healthy controls” group comparison. [39]. A biologically relevant mean difference of aMax between groups is assumed to be 1%—specifically, 2.5 vs. 3.5% dilation for PCS vs. CR. Similar differences of this magnitude have been observed in comparable groups with other disease entities, such as heart failure [40]. With these assumptions, we estimate that 78 evaluable patients per group will be needed to reject the null hypothesis (equality of all group means) with a power of 90%. To account possible dropouts or measurement errors, 100 patients per group should be included in the study. If the global null hypothesis is rejected based on ANOVA, pairwise group comparisons will be made at a significance level of α = 5% as a final test procedure. Power analysis was done by BH using nQuery (version 9.1.0.0).
Recruitment
To achieve an adequate recruitment number, we cooperate with the post-COVID outpatient clinics of the LMU University Hospital (KA, HS). Via social media of the Klinikum rechts der Isar, flyers will be additionally posted online and distributed. To provide adequate information for participants and patients, we are creating a website with further information (https://www.mri.tum.de/alleyesonpcs). Patients recruited via social media who wish to participate in the study must complete a screening form to ensure that only those with PCS are enrolled. Potential study participants will be discussed using the screening form in a weekly meeting with the chief investigator, PI, and doctoral students and will be included or excluded based on a majority vote. In particular, the temporal relationship between acute SARS-CoV-2 infection and the onset of PCS symptoms but also alternative diagnoses are discussed. Enrollment of participants will be conducted by a physician from the Department of Nephrology.
Data collection and management
We have written “standard operating procedures (SOPs)” to ensure standardization of processes. SOPs were translated and can be found in the supplements. Study instruments include:
Questionnaires: Only standardized and established questionnaires are used to assess patient-reported outcomes (“PROMs”).
Laboratory tests: Routine parameters are measured by our in-house clinical chemistry department.
DVA and SVA analysis: Data acquisition will be performed by following the SOPs (Online Resource 1). All examiners will be trained by a single experienced supervisor and must reach high image accuracy and quality in at least ten volunteers. Examiners do not perform data analyses and are only responsible for data acquisition. Data analysis will be performed by independent scientists and will be compared afterward. One rater will perform a quality evaluation and exclude insufficient measurements. Vessel response curves will be evaluated using the cumulative scoring method. The score ranges from 0 to 5. Retinal images with insufficient quality (< 2.5) will be discussed with a second rater and then excluded.
OCT and OCT angiography: Measurements and data analysis are performed by the Department of Neurology (Online Resource 2).
AO: Measurements and data analysis are undertaken by the Department of Ophthalmology (Online Resource 3).
Handgrip strength test: Data acquisition is done by following the SOP “Handgrip strength test” (Online Resource 4).
To promote participant retention, we contact the participants 2 days before their follow-up appointment to ensure that they remember the follow-up visit. Detailed information of PCS patients which were lost to follow-up will be provided.
Data management and confidentiality
Data entry only takes place in the University Hospital of the TU Munich on an assigned key-locked study computer. Excel sheets used for data collection are key-locked and standardized. All collected data will be verified by an independent second scientist (double data verification) and checked for plausibility (e.g., range checks for clinical data) (Online Resource 3). Data which are collected analog e.g., questionnaires and clarification sheets, are stored in a locked room in the University Hospital of the TU Munich. We have developed a checklist for each admitted participant to ensure all necessary information is collected, and in the event of missing data, we have established a protocol for conducting a standardized evaluation (Online Resource 3). Clinical data will be stored on a dedicated study server and in non-pseudonymized form on hard drives (accessible only to study physicians and persons directly involved in the study). Blood samples are labeled with the patient ID and will be stored in the routine nephrology laboratory and in the nephrology research laboratory in locked rooms.
Pseudonymization
For further analysis, all patient data are undergoing double pseudonymization. Patient-related data such as age and gender will be stored in a list, with a sequential study number assigned to each patient. The clinical data are exclusively assigned to the study numbers in a separate list. Both lists are password protected. The list containing the patient-related data and study numbers is kept separately from all other lists by the study director in a password-protected computer. Clinical data and informed consent forms are kept for 10 years, respectively.
Upon revocation of informed consent, all data will be deleted and printed records and collected samples will be destroyed.
Storage of biological specimens for genetic or molecular analysis in this trial
After recruitment, 40 ml of blood is initially collected from patients and sent directly to the nephrology laboratory. There, a portion of the blood is sent for analysis by clinical chemistry, and another portion is further processed. Peripheral blood mononuclear cells (PBMCs) are isolated from the processed blood according to protocol and stored at – 80 °C. In addition, samples of serum and whole blood are also frozen at -80 °C for future analysis; however, are not stored longer than 10 years.
Statistical methods
All statistical analysis will be carried out using R in the current version implemented in RStudio and will be supervised by BH. Used R Packages and their version numbers will be reported in the peer-reviewed manuscripts.
We plan to match our PCS cohort with the CR recovered and CN cohort at least 1:1 based on age and gender. The success of the matching is checked by the matchbalance function and results will be published in the supplements. To create a baseline description before and after matching, Table1 function is used. Depending on the distribution of the values aMax and vMax, a parametric (t test) or non-parametric test (Mann–Whitney test) is used to compare the means or medians, respectively. Boxplots will be plotted with the ggplot2 function. To evaluate a possible function as a biomarker in PCS, receiver operating characteristic (ROC) curves and the area under the curve (AUC) will be calculated using the plotROC function. Correlations will be visualized with the corrplot function. In general, Spearman or Pearson coefficients are calculated depending on the relationship between the two variables. To check for confounding, we will use linear regression models. Interim analyses are planed after completion of baseline recruitment. All participant scientists will have access to the evaluated data. Data will be published after baseline evaluation and after completed follow-up. To perform subgroup analyses, a test for interaction is performed using ANOVA and then visualized using the interaction.plot function. Information and baseline characteristics of patients lost to follow-up will be compared with those who completed the study using non-parametric or parametric tests, respectively. Upon reasonable request, access to the raw data and statistical codes used will be granted to the reviewers in the peer-review process when needed. Another sharing is not intended.
Oversight and monitoring
Chief investigator: Weekly meeting via Zoom with the study base team and PIs. Responsible for overseeing the trial in general.
Principal investigator (PI): Responsible for organizing and coordination of different involved departments and recruitment process in PCS ambulances. Responsible for data management and storage and final data analysis.
RVA-team: Monthly meeting of the three supervisors of RVA (RG, KK, TK). Quality control, data management,and updating on newest developments in the trial. Maintenance of the RVA machine.
Head of biobank: Processing and storage of bio samples. Maintenance of databank.
Study base team: Responsible to carry out technical measurements and blood sample drawing. Arrangement of study appointments. Not primarily involved in data analysis.
Data management is carried out by the PI who directly reports to the chief investigator. Data verification is done by the head of biobank which also carries out data collection and maintenance.
However, in case of RVA parameters, data analysis will be carried out by three independent principal investigators (RG, KK, TK). Data are checked frequently for validity and quality. If abnormalities occur, they are discussed between these three PIs and report to the chief investigator.
If we plan to amend the study protocol (e.g., new measurement of certain parameters), we will consult the ethical review board of TU Munich to discuss the feasibility of implementation. If the amendment is feasible, we will notify all participating researchers of the change. If the change affects study participants, we will inform them of the possible change and obtain new written informed consent. There are no publication restrictions, and we will publish the results in an appropriate peer-reviewed journal.
Discussion
The “All Eyes on PCS” study aims to investigate the potential pathophysiological mechanisms underlying a prolonged endothelial dysfunction (ED) in patients with PCS. The study will use RVA to measure the responsiveness of retinal vessels to flickering light and the retinal microcirculation as a surrogate for ED, as well as OCT and AO measurements to further characterize microvascular function. The design of this study is primarily focused on characterizing ED in PCS patients. In addition, the study aims to establish a comprehensive biobank of PCS and CR cohorts to answer questions regarding the pathophysiology of PCS.
Limitations
To conduct a full-scale characterization of PCS patients, we anticipate challenges in coordinating between the involved departments and scientists, particularly due to the need to perform three technical measurements that require specific expertise and quality standards. To ensure a smooth process, we conducted trial runs, developed SOPs before the start of recruitment, and are using digital study calendars. As participation in the study is voluntary and no immediate benefits are expected for the participants, we anticipate a certain drop-out rate. To avoid recruiting only mildly ill PCS patients, we are collaborating with the PCS ambulance of the LMU University Hospital. However, this could introduce bias toward recruiting only severely affected PCS patients, as mildly affected patients may not make the effort to get an appointment at the ambulance. Therefore, we are adopting a broad recruitment approach that includes involvement of general practitioners and self-help groups to overcome this potential bias.
Conclusion
This paper describes the study protocol for analyzing retinal microvasculature in patients with PCS. Using a multidisciplinary approach that involves comprehensive clinical assessments, retinal microcirculation imaging, blood and tissue sampling, a basic science approach, and patient-reported outcomes, we aim to identify potential risk factors for PCS and gain a better understanding of its pathophysiology. We anticipate that the study will lead to the identification of possible biomarkers and could help improve the management and treatment of PCS.
Trial status
Protocol version number 2. 28.03.23. Date of recruitment start: 19.10.2022. Date of the expected end of the study: Baseline recruitment (T1): End of May 2023. Follow-up: End of September 2023.
Availability of data and materials
All investigators involved in the planning and implementation of the study will have access to the final trial dataset.
Abbreviations
- ACE-2:
-
Angiotensin-converting enzyme-2
- ANOVA:
-
Analysis of variance
- AO:
-
Adaptive optics
- AUC:
-
Area under the curve
- AVR:
-
Arteriolar–venular ratio
- CN:
-
SARS-CoV-2-infection-naïve
- COVID-19:
-
Coronavirus disease 2019
- CR:
-
SARS-CoV-2-infection-recovered
- CRAE:
-
Central retinal arteriolar equivalent
- CRVE:
-
Central retinal venular equivalent
- C19-YRS:
-
COVID-19 Yorkshire Rehabilitation Scale
- DVA:
-
Dynamic retinal vessel analysis
- DVC:
-
Deep vascular complex
- DC:
-
Deceleration capacity
- ED:
-
Endothelial dysfunction
- EBV:
-
Epstein–Barr-virus
- FACS:
-
Fluorescence activated cell sorting
- FSS:
-
Fatigue severity scale
- FAZ:
-
Foveolar avascular zone
- FMD:
-
Flow-mediated dilation
- GAD-7:
-
Generalized anxiety disorder scale-7
- ICU:
-
Intensive care unit
- ME/CFS:
-
Myalgic encephalomyelitis/chronic fatigue syndrome
- OCT:
-
Optical coherence tomography
- PBMC:
-
Peripheral blood mononuclear cells
- PCR:
-
Polymerase chain reaction
- PCS:
-
Post-COVID-19 syndrome
- PHQ-9:
-
Patient Health Questionnaire
- PI:
-
Principal investigator
- PICS:
-
Post-intensive care syndrome
- PROM:
-
Patient-reported outcome measurement
- ROC:
-
Receiver operating characteristics
- RVA:
-
Retinal vessel analysis
- SARS-CoV-2:
-
Severe acute respiratory syndrome coronavirus type 2
- SOP:
-
Standard operating procedures
- SVA:
-
Static vessel analysis
- SVC:
-
Superficial vascular complex
- TMPRSS2:
-
Transmembrane protease serine subtype 2
- TLR:
-
Toll-like receptor
- WHO:
-
World Health Organization
References
Tang D, Comish P, Kang R (2020) The hallmarks of COVID-19 disease. PLoS Pathog 16(5):e1008536
Evans PC et al (2020) Endothelial dysfunction in COVID-19: a position paper of the ESC Working Group for Atherosclerosis and Vascular Biology, and the ESC Council of Basic Cardiovascular Science. Cardiovasc Res 116(14):2177–2184
Montiel V et al (2022) Oxidative stress-induced endothelial dysfunction and decreased vascular nitric oxide in COVID-19 patients. EBioMedicine 77:103893
Wichmann D et al (2020) Autopsy Findings and Venous Thromboembolism in Patients With COVID-19: A Prospective Cohort Study. Ann Intern Med 173(4):268–277
Varga Z et al (2020) Endothelial cell infection and endotheliitis in COVID-19. Lancet 395(10234):1417–1418
Ma Z et al (2022) Endothelial contribution to COVID-19: an update on mechanisms and therapeutic implications. J Mol Cell Cardiol 164:69–82
Hamming I et al (2004) Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol 203(2):631–637
Li M-Y et al (2020) Expression of the SARS-CoV-2 cell receptor gene ACE2 in a wide variety of human tissues. Infect Dis Poverty 9(1):45
Zhou L et al (2021) Expression of the SARS-CoV-2 Receptor ACE2 in Human Retina and Diabetes-Implications for Retinopathy. Invest Ophthalmol Vis Sci 62(7):6
Araujo-Silva CA et al (2021) Presumed SARS-CoV-2 Viral Particles in the Human Retina of Patients With COVID-19. JAMA Ophthalmol 139(9):1015–1021
Nuovo GJ et al (2021) Endothelial cell damage is the central part of COVID-19 and a mouse model induced by injection of the S1 subunit of the spike protein. Ann Diagn Pathol 51:151682
Lei Y et al (2021) SARS-CoV-2 Spike Protein Impairs Endothelial Function via Downregulation of ACE 2. Circ Res 128(9):1323–1326
Urata R et al (2022) Senescent endothelial cells are predisposed to SARS-CoV-2 infection and subsequent endothelial dysfunction. Sci Rep 12(1):11855
Flaumenhaft R, Enjyoji K, Schmaier AA (2022) Vasculopathy in COVID-19. Blood 140(3):222–235
Nalbandian A et al (2021) Post-acute COVID-19 syndrome. Nat Med 27(4):601–615
Soriano JB et al (2022) A clinical case definition of post-COVID-19 condition by a Delphi consensus. Lancet Infect Dis 22(4):e102–e107
Ballering AV et al (2022) Persistence of somatic symptoms after COVID-19 in the Netherlands: an observational cohort study. The Lancet 400(10350):452–461
Zayet S et al (2021) Post-COVID-19 Syndrome: Nine Months after SARS-CoV-2 Infection in a Cohort of 354 Patients: Data from the First Wave of COVID-19 in Nord Franche-Comté Hospital, France. Microorganisms 9(8):1719
Rousseau A-F et al (2021) Post-intensive care syndrome after a critical COVID-19: cohort study from a Belgian follow-up clinic. Ann Intensive Care 11(1):118
Fogarty H et al (2021) Persistent endotheliopathy in the pathogenesis of long COVID syndrome. J Thromb Haemost 19(10):2546–2553
Sollini M et al (2021) Vasculitis changes in COVID-19 survivors with persistent symptoms: an [(18)F]FDG-PET/CT study. Eur J Nucl Med Mol Imaging 48(5):1460–1466
Wang C et al (2022) Long COVID: the nature of thrombotic sequelae determines the necessity of early anticoagulation. Front Cell Infect Microbiol 12:861703
Kedor C et al (2022) A prospective observational study of post-COVID-19 chronic fatigue syndrome following the first pandemic wave in Germany and biomarkers associated with symptom severity. Nat Commun 13(1):5104
Haffke M et al (2022) Endothelial dysfunction and altered endothelial biomarkers in patients with post-COVID-19 syndrome and chronic fatigue syndrome (ME/CFS). J Transl Med 20(1):138
Sørland K et al (2021) Reduced endothelial function in myalgic encephalomyelitis/chronic fatigue syndrome-results from open-label cyclophosphamide intervention study. Front Med 8:642710
Merad M et al (2022) The immunology and immunopathology of COVID-19. Science 375(6585):1122–1127
Castanares-Zapatero D et al (2022) Pathophysiology and mechanism of long COVID: a comprehensive review. Ann Med 54(1):1473–1487
Del Rio C, Collins LF, Malani P (2020) Long-term Health Consequences of COVID-19. JAMA 324(17):1723–1724
Oikonomou E et al (2022) Endothelial dysfunction in acute and long standing COVID-19: A prospective cohort study. Vascul Pharmacol 144:106975
Hohberger B et al (2021) Retinal Microcirculation as a Correlate of a Systemic Capillary Impairment After Severe Acute Respiratory Syndrome Coronavirus 2 Infection. Front Med (Lausanne) 8:676554
Charfeddine S et al (2021) Long COVID 19 Syndrome: Is It Related to Microcirculation and Endothelial Dysfunction? Insights From TUN-EndCOV Study. Front Cardiovasc Med 8:745758
Lim M et al (2013) Systemic associations of dynamic retinal vessel analysis: a review of current literature. Microcirculation 20(3):257–268
Günthner R et al (2019) Impaired Retinal Vessel Dilation Predicts Mortality in End-Stage Renal Disease. Circ Res 124:1796
Seidelmann SB et al (2016) Retinal Vessel Calibers in Predicting Long-Term Cardiovascular Outcomes: The Atherosclerosis Risk in Communities Study. Circulation 134(18):1328–1338
Krupp LB et al (1988) Fatigue in Multiple Sclerosis. Arch Neurol 45(4):435–437
Spitzer RL et al (2006) A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med 166(10):1092–1097
Carruthers BM et al (2003) Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. J Chronic Fatigue Syndrome 11(1):7–115
Kroenke K, Spitzer RL, Williams JB (2001) The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med 16(9):606–613
Zampiccoli E et al (2022) Eyes on amyloidosis: microvascular retinal dysfunction in cardiac amyloidosis. ESC Heart Fail 9(2):1186–1194
Nägele MP et al (2018) Retinal microvascular dysfunction in heart failure. Eur Heart J 39(1):47–56
Funding
Open Access funding enabled and organized by Projekt DEAL. This study is funded by the Bavarian State Ministry of Science and Art, as part of its Special Funding Program for the Scientific Coronavirus Research Projects of the Technical University of Munich School of Medicine at the University Medical Center Klinikum Rechts der Isar (Funding Identification Number H.4001.1.7-53/7).
Author information
Authors and Affiliations
Contributions
CS is chief investigator; he conceived the study and led the proposal and protocol development. TK contributed to study design and developed the proposal and protocol. RH wrote the first version of the proposal and contributed to study design. RG and KK are the technical supervisors and lead methodologists regarding the RVA measurements. MB contributed to study design. RW and BK are the lead methodologists regarding the OCT measurements. NB is the lead methodologists regarding the AO measurement. ML and AR are lead methodologists regarding cell culture, FACS experiments, and the basic science approach in general. All authors contributed to study design. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare that they have no financial or non-financial interests that are directly or indirectly related to the work submitted for publication.
Ethical approval
Ethical Board of the Technical University Munich (Approval number: 2022-317-S-SR). Written, informed consent to participate will be obtained from all participants. Copies of the original ethical approval document and an English translation are in the supplements (Online Resource 4). Ethical Board of the LMU Munich (Approval number: 21-1165).
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kuchler, T., Hausinger, R., Braunisch, M.C. et al. All eyes on PCS: analysis of the retinal microvasculature in patients with post-COVID syndrome—study protocol of a 1 year prospective case–control study. Eur Arch Psychiatry Clin Neurosci (2023). https://doi.org/10.1007/s00406-023-01724-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00406-023-01724-5