Abstract
Purpose
This study aims to aggregate and analyze existing clinical evidence to compare the efficacy and adverse effects of unilateral or bilateral botulinum toxin injections for the treatment of adductor spasmodic dysphonia (ADSD).
Methods
Reports from non-randomized controlled trials and cohort studies pertaining to the efficacy and adverse effects of unilateral and bilateral botulinum toxin injections for ADSD were identified and retrieved from four electronic databases from inception to July 2023. The meta-analysis employed fixed or random effects models to assess pooled relative risks (RR), mean differences (MDs), and standard mean differences (SMDs) with their corresponding 95% confidence intervals (CIs).
Results
We included two non-randomized controlled trials and seven cohort studies comprising 854 total patients. Meta-analysis of the included studies showed that bilateral botulinum toxin injections associated with a longer duration of vocal improvement (MD = − 2.89, 95% CI − 3.13 to − 2.65, I2 = 0%, P < 0.00001). However, bilateral botulinum toxin injections associated with an increase in adverse effects, including a longer duration of breathy voice quality (SMD = − 0.51, 95% CI − 0.79 to − 0.22, I2 = 35%, P = 0.0005) and a higher occurrence of swallowing difficulties (RR = 0.46, 95% CI 0.35 to 0.11, I2 = 0%, P < 0.00001).
Conclusion
Bilateral botulinum toxin injections for ADSD showed a longer duration of vocal improvement, a longer breathy voice duration and a higher dysphagia occurrence and duration than unilateral injections.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Spasmodic dysphonia (SD), also known as laryngeal dystonia (LD), is a focal dystonia primarily characterized by irregular and uncontrollable interruptions in phonation that are commonly accompanied by vocal effort [1, 2]. The reported prevalence of SD varies between 0.9 and 13.7 per 100,000 individuals worldwide [3]. Due to diagnostic challenges and the lack of a global epidemiological investigation, the true prevalence remains underestimated.
SD can be further classified based on the affected muscle groups as either adductor spasmodic dysphonia (ADSD), abductor spasmodic dysphonia (ABSD), or mixed-type dysphonia, with ADSD being the most common subtype (accounting for 90–95% of cases) [4]. The irregular voice breaks caused by laryngeal muscle spasms have been shown to significantly impair patient quality of life, leading to decreased work attendance and performance [5, 6]. Furthermore, anxiety and depression coexist in 7.1–62% of ADSD patients [7], and approximately one-fifth of ADSD patients experience suicidal ideation [8].
Injecting botulinum toxin into the affected muscles for treating SD has a history of over 40 years. Blitzer et al. [9] demonstrated the effectiveness of local botulinum toxin injection for ADSD in 1986. The 2018 update to the Clinical Practice Guideline: Hoarseness (Dysphonia) [10] explicitly recommended botulinum toxin injection as a treatment for patients with voice disorders caused by SD. It is estimated that nearly 85% of SD patients are treated with botulinum toxin injections in modern clinical practice [11].
Following botulinum toxin injection, SD patients usually experience three stages of vocal improvement: first, a “weak breathy voice,” progression to a “normal strong voice,” and finally reversion to “spasmodic phonation” [12] during which the therapeutic effects of botulinum toxin often coexist with adverse effects. Previous studies have demonstrated that duration of vocal improvement after botulinum toxin injection typically ranges from 8.0 to 15.1 weeks, and is accompanied by a variety of adverse effects which may be influenced by individual differences (e.g., breathy voice, dysphagia/aspiration, coughing, and complete loss of voice) [13,14,15]. Clinicians often tailor personalized treatment plans based on the severity of the vocal disorder, diverse patient needs for voice recovery, botulinum toxin treatment efficacy, and patient feedback on adverse effects.
Therefore, a key question remains: is there a way to optimize treatment efficacy and minimize adverse effects for SD at the lowest economic cost? Numerous studies have explored different injection approaches, including comparing the efficacy and adverse effects of unilateral and bilateral botulinum toxin injections for ADSD. However, due to discrepancies in study design, outcome measures, and research findings, there is currently no unified consensus on whether unilateral or bilateral botulinum toxin injection is superior in terms of efficacy and adverse effect profile. Thus, the present study aims to aggregate and analyze existing evidence regarding the efficacy and adverse effects of unilateral and bilateral botulinum toxin injections for ADSD. These findings may provide insight needed to optimize the clinical approach to botulinum toxin injection treatment for ADSD.
Methods
This study adhered to the PRISMA 2020 recommendations [16] and employed a pre-established protocol and a clear, replicable literature search strategy for the systematic review and meta-analysis of existing evidence. The study has been registered with PROSPERO (Registration Number: CRD42023451772).
Study selection
The inclusion criteria were formulated according to the Population, Intervention, Comparison, Outcomes and Study (PICOS) principle:
-
(1)
Population: adult patients diagnosed with ADSD (with or without tremor).
-
(2)
Intervention: unilateral botulinum toxin injections (regardless of side).
-
(3)
Comparison: bilateral botulinum toxin injections.
-
(4)
Outcomes: both efficacy evaluation and adverse effect assessment were considered. Efficacy evaluation included the duration of vocal improvement, injection interval, and relevant severity scores for vocal disorders. Adverse effect assessment encompassed the duration or occurrence rate of symptoms such as complete loss of voice, breathy voice, swallowing difficulties, and coughing.
-
(5)
Study: non-randomized controlled trials and cohort studies.
Exclusion criteria consisted of duplicates, unclear study design, articles without access to original data or full texts, animal studies, case reports, reviews, and systematic reviews.
Search and screening strategy
The search was conducted using four medical electronic databases (PubMed, Embase, Web of Science, and Cochrane Library), scanning for all articles published up to July 2023. Language, date, and geographical restrictions were not applied in the literature search. A combination of subject terms and free-text terms were used to construct the search; the complete search strategy is provided in the appendix. References included within relevant articles were additionally scrutinized, and experts in the field were contacted to confirm any ongoing but unpublished relevant research.
The literature screening process involved the following steps: initially, two reviewers (LYY and CD) independently screened the titles and abstracts of relevant literature from each major electronic database. The results of their screenings were then compared. Studies that overlapped (i.e., articles identified as relevant by both reviewers) proceeded to the next screening step directly. For non-overlapping studies, a discussion was held to reach a consensus on whether or not they should transition to the next screening step. If discrepancies persisted, a third team member (ZJ) was consulted to reach a consensus. Subsequently, a comprehensive full-text screening of the preliminarily screened literature was conducted. LYY and CD independently read the full texts and determined inclusion or exclusion based on pre-defined criteria. The final inclusion of articles was determined through a process of comparison and discussion, as outlined previously.
Data extraction and critical appraisal
LYY and CD independently used a standardized data extraction template to isolate relevant information and data from the included literature. The data extracted by the two reviewers were examined and compared by an independent third party. In cases of discordant data extraction, consensus was achieved through discussion. A quality assessment of the bias present in the included studies was conducted using the ROBINS-I tool [17], which categorizes literature quality into one of five levels: low risk of bias, moderate risk of bias, high risk of bias, critical risk of bias, or no information.
Statistical analysis
A meta-analysis was subsequently performed on the collated data derived from the included literature. Descriptive summaries were provided for studies with sample sizes > 10. For studies with two or more research papers focusing on a specific outcome, Review Manager software (version 5.4) was used to aggregate effect sizes. Continuous variables were reported using mean differences (MDs) or standardized mean differences (SMDs) along with the associated 95% confidence interval (CI). Binary variables were reported using relative risk (RR) and the associated 95% CI. Heterogeneity was evaluated using the I-squared (I2) statistic, which describes the degree of between-study variability. If the heterogeneity was low (I2 < 50%), the fixed-effect model was adopted for analysis. If heterogeneity was high (I2 > 50%), we performed a sensitivity analysis to observe the study with the greatest heterogeneity and excluded this study from the analysis. Additionally, we conducted a subgroup analysis according to the actual situation of the study to reduce heterogeneity.
Results
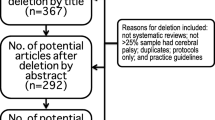
A total of 2182 records were retrieved from various electronic databases as follows: PubMed = 408; Cochrane Library = 44; Web of Science = 215; Embase = 1515. After excluding 939 duplicate records and reviewing titles and abstracts, a final set of 9 articles were included for this study. These articles consisted of seven cohort studies and two non-randomized controlled trials, with a collective sample size of n = 854 cases. The study selection process is detailed in a PRISMA diagram (Fig. 1).
PRISMA flow diagram. *PubMed = 408; Cochrane = 44; Web of Science = 215; Embase = 1515. **Abstracts and titles screened for relevance (n = 1185); conference papers, reports, and review articles (n = 30). Others (protocols, letters, technical notes, animal studies, non-English language; n = 2). N-rct, non-randomized controlled trials; Ccs, cohort studies. From: Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:n71. https://doi.org/10.1136/bmj.n71
Study characteristics and risk-of-bias assessment
The main characteristics of the literature included in this study are shown in Table 1.
The quality and bias risk assessments for the nine included studies were conducted using the ROBINS-I tool. First, a reference target randomized controlled trial (RCT) was established with the following specific parameters: the trial type was RCT, the study population consisted of ADSD patients, the intervention in the experimental group received unilateral botulinum toxin injections, and the control group received bilateral botulinum toxin injections. Outcome measures were categorized based on the design of each individual study, which could include both intention-to-treat analysis results and as-treated analysis results. These outcome measures were defined as the duration of vocal improvement, injection interval, duration of breathy voice, duration and frequency of swallowing difficulties, duration of coughing, and duration of complete voice loss.
Following this, the identification of potential confounding factors was facilitated through literature review and integration of specialized knowledge. The severity of vocal disorders in ADSD patients was identified as a crucial confounding domain, as it could influence the decision-making process of patients and physicians when choosing between unilateral or bilateral botulinum toxin injections for treatment. Additionally, consideration was given to the presence of any external interventions (e.g., speech training), which could have affected the studies.
Ultimately, by addressing key questions, the risk of bias was assessed for each study across different domains, leading to an overall evaluation of bias-associated risk after aggregation (Table 2).
Efficacy
Duration of vocal improvement
Results regarding the duration of improved voice were reported in two retrospective cohort studies [18, 23] and one non-randomized controlled study [15].The mixed duration of improved voice demonstrated significant difference, but showed significant heterogeneity (I2 = 65%, P = 0.06), which was completely reduced by excluding the study by Amy et al. from the analysis [18] (I2 = 0%, P = 0.32), and the result remained that unilateral injections resulted in a significantly shorter duration of improved voice compared with bilateral injections (MD = − 2.89, 95% CI − 3.13 to − 2.65, P < 0.00001; Fig. 2) [15, 23].
Treatment interval
The results pertaining to the treatment interval were reported in five retrospective cohort studies [12, 19,20,21, 23] and one prospective cohort study [22]. Among these, one study [12] did not explicitly provide averages (i.e., mean ± SD) for injection interval times in each group. Regardless, a meta-analysis was not performed upon these data due to evidence of high heterogeneity (I2 = 96%, P < 0.00001; Fig. 3), and we could not reduce the heterogeneity using the leave-one-out statistical method.
Patient self-assessment of voice score
Two studies, conducted by Upile [22] and Behrad [21], explored the injection interval and also quantified the degree of vocal improvement. Patients in these studies provided a self-assessment on the extent of vocal improvement following botulinum toxin injections using the following six-point scale: 0—no improvement; 1—very slight improvement; 2—slight improvement; 3—moderate improvement; 4—marked improvement; 5—extreme improvement/near normal. Upile’s findings [22] demonstrated that the unilateral injection group had a higher vocal score (4.24) compared to the bilateral injection group (3.93). Similarly, Behrad [21] found that the proportion of patients in the unilateral injection group achieving a vocal score of 5 (near normal) was higher (54.2%) than in the bilateral injection group (46.1%).
Adverse effects
Duration of breathy voice
The length of time that patients experienced a breathy voice quality was reported in four retrospective cohort studies [12, 18,19,20], one prospective cohort study [22], and one non-randomized controlled study [15].The mixed duration of breathy voice demonstrated significant difference, but showed significant heterogeneity (I2 = 59%, P = 0.03), which was completely reduced by excluding the study by Upile et al. from the analysis [22] (I2 = 35%, P = 0.19), and the result remained that unilateral injections resulted in a significantly shorter duration of breathy voice compared with bilateral injections (SMD = − 0.51, 95% CI − 0.79 to − 0.22, P = 0.0005; Fig. 4) [12, 15, 18,19,20].
Dysphagia occurrence and duration
The occurrence and duration of dysphagia were reported in one retrospective cohort study [21], one prospective cohort study [22], and one non-randomized controlled study [15]. No significant difference in the duration of dysphagia was found between the two groups (SMD = − 0.31, 95% CI − 1.78 to 1.16, P = 0.68, I2 = 0; Fig. 5).
The rate of occurrence of dysphagia was reported in one retrospective cohort study [18], one prospective cohort study [22], and two non-randomized controlled studies [15, 24]. The mixed rate of occurrence of dysphagia demonstrated significant difference, but showed significant heterogeneity (I2 = 51%, P = 0.11), which was completely reduced by excluding the study by Upile et al. from the analysis [22] (I2 = 0%, P = 0.53), and the result remained that unilateral injections resulted in a significantly lower rate of occurrence of dysphagia compared with bilateral injections (RR = 0.46, 95% CI 0.35 to 0.61, P < 0.00001; Fig. 6) [15, 18, 24].
Duration of coughing
The duration of coughing following treatment was reported in one retrospective cohort study [21] and one prospective cohort study [22].The pooled mean difference showed no significant difference (MD = − 1.28, 95% CI − 4.26 to 1.70, P = 0.40; Fig. 7) in the duration of coughing. However, the result was significantly heterogeneous (I2 = 73%, P = 0.06), and we could not reduce the heterogeneity using the leave-one-out statistical method.
Duration of total voice loss
The duration of total voice loss was reported in one retrospective cohort study [21] and one prospective cohort study [22]. However, due to the absence of cases with complete voice loss in the unilateral injection group in one study [22], it was not possible to pool the results. Nevertheless, the evident trend in both studies associates a longer duration of complete voice loss with bilateral botulinum toxin injections.
The collective study findings presented above, along with the GRADE evaluation, are summarized in Table 3.
Discussion
The objective of this study was to compare the efficacy and adverse effects of unilateral and bilateral botulinum toxin injections for treating ADSD. The systematic review was based upon evidence derived from seven cohort studies and two non-randomized controlled trials. The results were pooled and evaluated through meta-analysis to gain insight into the effect of laterality on overall treatment efficacy and side effect profile.
The efficacy outcome measures in the studies investigating single versus bilateral injections included both subjective patient self-assessments (e.g., duration of voice improvement, patient-determined injection interval, maximum efficacy, and voice score), as well as objective assessments [24] made based upon acoustic analyses. However, since ADSD has a profound effect on patient experience and quality of life, most studies used subjective assessments as the primary outcome measures.
A previous meta-analysis that is comparable to the present report [25] pooled data from 22 studies, using pre- and post-treatment efficacy of different injection approaches (unilateral/bilateral) as the primary outcome. Data derived from 134 patients were evaluated, and no significant difference was identified between the two injection approaches. In this report, evidence was obtained from seven cohort studies and two non-randomized controlled trials, representing data from a total of 854 patients. The duration of voice improvement post injection and the injection interval were used as outcome measures to assess the impact of injection approach on efficacy.
After sensitivity analysis was used to exclude articles with the greatest heterogeneity, the pooled results showed that bilateral Botox injection was associated with a longer duration of voice improvement than unilateral injection. In terms of injection interval, the high heterogeneity among various studies could not be reduced, which was considered to be due to the fact that none of the included studies placed timing restrictions on retreatment schedules following a failure of patient improvement, leading to significant bias from variability in patient circumstance. Therefore, data could not be pooled or analyzed for this outcome measure. To address this issue, Steven et al. [26] categorized the reinjection interval after symptom recurrence as either “ < 2 weeks” or “ > 2 months” and compared the injection interval of unilateral versus bilateral injections. They found no significant difference between unilateral and bilateral injections for patients reinjected at < 2 weeks or > 2 months after recurrence.
Notably, the severity of voice impairment in patients with ADSD significantly impacts the treatment efficacy, side effects, risk of bias, and quality assessment reported by studies. Nonetheless, only two of the studies included in this report [21, 22] considered the severity of voice impairment in the evaluation of treatment efficacy, and these reported findings using a simple and subjective six-point rating scale. There is a significant need for more scientific, objective, and ADSD-specific measures of disease severity. Since the data types of the final outcomes were inconsistent between these two studies and original data were inaccessible, the results could not be pooled.
Three studies were excluded from the meta-analysis due to inconsistencies in the reported outcome measures. Two retrospective cohort studies [26, 27] used maximum efficacy (efficacy of > 3 months duration) and minimum side effects (side effects of < 2 weeks duration) as the evaluation criteria. Steven et al. [26] assessed the optimal effect and side effect duration in 45 patients receiving the same botulinum toxin dose. This was accomplished by comparing the number of injections resulting in an efficacy duration beyond 3 months (i.e., optimal effect) versus under 3 months, and duration of side effects under 2 weeks (i.e., optimal side effects) versus beyond 2 weeks, as well as the number of injections with or without optimal effect and side effects. The results suggested unilateral injection was more frequently associated with both optimal effect and side effects.
Recognizing that the small sample sizes and shortened timelines of the previous studies could not adequately reflect dynamic changes in injections arising from individual patient differences, Ishaan et al. [27] analyzed data from 4023 injections in 272 ADSD patients treated with botulinum toxin injections between 1994 and 2018. In contrast with the previous study [26], this longitudinal retrospective analysis found that bilateral injections were more frequently associated with optimal effect and side effects.
One non-randomized controlled study [24] employed an objective acoustic assessment of unilateral and bilateral treatment efficacy by comparing voice acoustic parameters at three time points (pre-injection, 1 week post injection, and 4 weeks post injection). They found that all voice acoustic parameters except signal-to-noise ratio improved 1 week after unilateral injection, whereas only voice break factor significantly improved after bilateral injection. At 4 weeks post injection, parameters in the unilateral group were closer to normal values compared to the bilateral group. Therefore, unilateral injection resulted in better and more rapid improvement in acoustic parameters.
The efficacy of botulinum toxin injections for ADSD must be weighed against side effects including breathiness, dysphagia, and coughing. Accordingly, most of the studies included in this report discussed efficacy in light of the side effect profile. After sensitivity analysis was used to exclude articles with the greatest heterogeneity, pooled results showed that compared to unilateral injections, bilateral botulinum toxin injections was associated with longer duration of breathiness and higher incidence of dysphagia. There was insufficient evidence for the reporting of cough and complete voice loss due to the small number of studies and low sample sizes.
Notably, although the results of this report can inform dynamic adjustment of injection protocols during the treatment cycle, they do not present a simple solution to the choice of unilateral versus bilateral approach for the initial injection, since the included studies did not concurrently correlate efficacy and side effects with injection approaches. Surveys indicate that 87% of U.S. physicians prefer bilateral injections for patients as the initial treatment [28], mainly due to patient demand. Ishaan [20] and Steven et al. [26] categorized treatments by the number of injections and correlated the optimal efficacy (> 3 months) with optimal side effects (< 2 weeks) to compare the number of unilateral versus bilateral injections that concurrently resulted in an optimal effect/side effect combination. This analysis informs the choice of initial injection approach; however, the results of these two studies were inconsistent, warranting further investigation into the analytical approach. Additionally, Zwirner et al. [24] evaluated aerodynamic parameters at three timepoints and found significantly higher mean air flow rate 1 week after bilateral compared to unilateral injections, which is consistent with the higher incidence of dysphagia observed following bilateral injections.
Our study has some limitations. First, the included studies contained patients with both ADSD and comorbid essential vocal tremor [18]. ADSD and essential tremor both manifest as impaired neuromuscular control of the larynx. Steven et al. [26] found no significant difference in the desirable profile of maximum efficacy with minimum side effects between unilateral versus bilateral injections in ADSD patients with essential tremor. Therefore, studies containing patients with both conditions were not excluded, although efficacy and side effects specifically for ADSD cannot be determined. Second, the included non-randomized controlled studies and cohort studies were found to have a moderate-to-high risk of bias based on quality and bias assessment. Third, there was high heterogeneity among the studies, including botulinum toxin type, injection doses, and treatment cycles, and the number of studies was small after sensitivity analysis. Additionally, subgroup analysis was not possible due to the small number of studies and lack of subgroup data.
The goal of this study was to investigate which injection approach results in better efficacy and fewer adverse effects for ADSD. Due to the currently limited and low-quality evidence, the results evaluated here can only provide a reference for dynamic adjustment of injection protocols during serial botulinum toxin treatment cycles. Suggestions for improving future research in this field are provided based on limitations of the included studies as follows:
First, a large degree of heterogeneity in injection doses existed across studies. Selection of initial doses were varied and not justified in most studies. During serial botulinum toxin treatments, earlier studies used fixed doses for comparison, while later studies dynamically adjusted doses based on physician experience and patient responses, introducing heterogeneity. We suggest reporting injected doses as low/medium/high dose groups to enable future subgroup meta-analyses by dose.
Second, there was a notable lack of ADSD-specific outcome measures. Efficacy assessments relied primarily on subjective measures such as duration of voice improvement and injection interval, with few studies incorporating objective acoustic analyses. Severity of voice impairment in particular was largely overlooked. Based on proposed core outcome measurement sets (patient-reported outcome measures, perceptual analysis, acoustic analyses, visual analyses and aerodynamic measurements) for common voice disorders by the European Laryngological Society (ELS) [29], core measurement tools for ADSD include: a PROM (the VHI) and perceptual rating measurements (voice breaks + the GRBAS: grade, roughness, breathiness, and strain, with less importance placed on asthenia). Acoustic measures were added as optional due to conflicting evidence. Future studies should combine subjective and objective assessments.
Third, there were numerous inconsistencies in study duration and design; follow-up periods ranged from 3 months for a single injection, to many years administering more than 20 injections. Lee et al. [19] noted that the difference in injection intervals between unilateral and bilateral groups became minimized over time, supporting the need proposed herein to analyze short-versus long-term studies. Given the predominance of retrospective cohort studies, which are prone to bias, short-term randomized controlled trails and prospective cohort studies with long-term follow-up should be considered, as well as nonrandomized self-controlled before-after studies using the cyclic nature of botulinum toxin injections.
Finally, there was a general lack of subgroup analysis in the injection approach, which is problematic as the unilateral/bilateral dichotomy is overly simplistic. Some studies used alternating unilateral injections [12, 18, 19] versus consistent unilateral injections, warranting comparison. Other variations, including dose equality for bilateral injections [18], should also be considered for future subgroup analyses using more granular groupings.
Conclusion
Our meta-analysis and critical review of this field showed that bilateral botulinum toxin injections for ADSD showed a longer duration of vocal improvement, a longer breathy voice duration and a higher dysphagia occurrence and duration than unilateral injections. However, the insufficient evidence and significant heterogeneity among studies suggests that a variety of methodological improvements are required in this field, including increasing the number of studies and sample size, refining subgroup analyses (e.g., dose, approach, and duration), diversifying study design, and utilizing ADSD-specific outcome measures to her quality evidence.
Data availability
Not applicable.
References
Hintze JM, Ludlow CL, Bansberg SF, Adler CH, Lott DG (2017) Spasmodic dysphonia: a review. Part 1: pathogenic factors. Otolaryngol Head Neck Surg 157(4):551–557. https://doi.org/10.1177/0194599817728521
Simonyan K, Barkmeier-Kraemer J, Blitzer A, Hallett M, Houde JF, Jacobson Kimberley T, Ozelius LJ, Pitman MJ, Richardson RM, Sharma N, Tanner K (2021) The NIH/NIDCD workshop on research priorities in spasmodic dysphonia/laryngeal dystonia. Laryngeal dystonia: multidisciplinary update on terminology, pathophysiology, and research priorities. Neurology 96(21):989–1001. https://doi.org/10.1212/WNL.0000000000011922
Hyodo M, Hisa Y, Nishizawa N, Omori K, Shiromoto O, Yumoto E, Sanuki T, Nagao A, Hirose K, Kobayashi T, Asano K, Sakaguchi M (2021) The prevalence and clinical features of spasmodic dysphonia: a review of epidemiological surveys conducted in Japan. Auris Nasus Larynx. https://doi.org/10.1016/j.anl.2020.08.013
Hyodo M, Hirose K, Nagao A, Nakahira M, Kobayashi T (2022) Botulinum toxin therapy for spasmodic dysphonia in Japan: the history and an update. Toxins (Basel) 14(7):451. https://doi.org/10.3390/toxins14070451
Meyer TK, Hu A, Hillel AD (2013) Voice disorders in the workplace: productivity in spasmodic dysphonia and the impact of botulinum toxin. Laryngoscope 123(Suppl 6):S1-14. https://doi.org/10.1002/lary.24292
Meyer TK, Spiekerman C, Kaye R, Blitzer A, Kamizi RS, Jiang L, Weaver EM (2021) Association of laryngeal botulinum neurotoxin injection with work productivity for patients with spasmodic dysphonia. JAMA Otolaryngol Head Neck Surg 147(9):804–810. https://doi.org/10.1001/jamaoto.2021.1745
Hu A, Hillel A, Zhao W, Meyer T (2018) Anxiety and depression in spasmodic dysphonia patients. World J Otorhinolaryngol Head Neck Surg 4(2):110–116. https://doi.org/10.1016/j.wjorl.2018.04.004
Worthley A, Simonyan K (2021) Suicidal ideations and attempts in patients with isolated dystonia. Neurology 96(11):e1551–e1560. https://doi.org/10.1212/WNL.0000000000011596
Blitzer A, Brin MF, Fahn S, Lange D, Lovelace RE (1986) Botulinum toxin (BOTOX) for the treatment of “spastic dysphonia” as part of a trial of toxin injections for the treatment of other cranial dystonias. Laryngoscope 96(11):1300–1301
Stachler RJ, Francis DO, Schwartz SR et al (2018) Clinical practice guideline: Hoarseness (Dysphonia) (update). Otolaryngol Head Neck Surg 158(1_suppl):S1–S42. https://doi.org/10.1177/0194599817751030
Dressler D, Altenmüller E, Giess R, Krauss JK, Adib Saberi F (2022) The epidemiology of dystonia: the Hannover epidemiology study. J Neurol 269(12):6483–6493. https://doi.org/10.1007/s00415-022-11310-9
Koriwchak MJ, Netterville JL, Snowden T, Courey M, Ossoff RH (1996) Alternating unilateral botulinum toxin type A (BOTOX) injections for spasmodic dysphonia. Laryngoscope 106(12 Pt 1):1476–1481. https://doi.org/10.1097/00005537-199612000-00006
Patel PN, Kabagambe EK, Starkweather JC, Keller M, Gamsarian V, Lee J, Kulkarni V, Garrett CG, Francis DO (2018) Outcomes of onabotulinum toxin a treatment for adductor spasmodic dysphonia and laryngeal tremor. JAMA Otolaryngol Head Neck Surg 144(4):293–299. https://doi.org/10.1001/jamaoto.2017.3088
Blitzer A (2010) Spasmodic dysphonia and botulinum toxin: experience from the largest treatment series. Eur J Neurol 17(Suppl 1):28–30. https://doi.org/10.1111/j.1468-1331.2010.03047.x
Langeveld TP, Drost HA, Baatenburg de Jong RJ (1998) Unilateral versus bilateral botulinum toxin injections in adductor spasmodic dysphonia. Ann Otol Rhinol Laryngol 107(4):280–284. https://doi.org/10.1177/000348949810700403
Page MJ, McKenzie JE, Bossuyt PM et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71. https://doi.org/10.1136/bmj.n71
Sterne JA, Hernán MA, Reeves BC et al (2016) ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355:i4919
Stone A, Powell ME, Hamers K, Fletcher KC, Francis DO, Courey MS, Netterville JL, Garrett CG (2022) Optimizing Botox regimens in patients with adductor spasmodic dysphonia and essential tremor of voice: a 31-year experience. Laryngoscope Investig Otolaryngol 7(5):1499–1505. https://doi.org/10.1002/lio2.915
Lee SJ, Kang MS, Choi HS, Lim JY (2020) Alternating unilateral versus bilateral injections of botulinum toxin for the treatment of adductor spasmodic dysphonia. Otolaryngol Head Neck Surg 164(4):815–820. https://doi.org/10.1177/0194599820957608
French G, Bosch JD, Randall DR (2020) Retrospective review of dosing trends in botulinum toxin injections for the treatment of adductor spasmodic dysphonia in a long-term cohort. J Otolaryngol Head Neck Surg 49(1):4. https://doi.org/10.1186/s40463-020-0401-4
Elmiyeh B, Prasad VM, Upile T, Saunders N, Youl BD, Epstein R, Rubin JS (2010) A single-centre retrospective review of unilateral and bilateral Dysport injections in adductor spasmodic dysphonia. Logoped Phoniatr Vocol 35(1):39–44. https://doi.org/10.3109/14015431003604804
Upile T, Elmiyeh B, Jerjes W, Prasad V, Kafas P, Abiola J, Youl B, Epstein R, Hopper C, Sudhoff H, Rubin J (2009) Unilateral versus bilateral thyroarytenoid Botulinum toxin injections in adductor spasmodic dysphonia: a prospective study. Head Face Med 5:20. https://doi.org/10.1186/1746-160X-5-20
Inagi K, Ford CN, Bless DM, Heisey D (1996) Analysis of factors affecting botulinum toxin results in spasmodic dysphonia. J Voice 10(3):306–313. https://doi.org/10.1016/s0892-1997(96)80012-9
Zwirner P, Murry T, Woodson GE (1993) A comparison of bilateral and unilateral botulinum toxin treatments for spasmodic dysphonia. Eur Arch Otorhinolaryngol 250(5):271–276. https://doi.org/10.1007/BF00186224
Whurr R, Nye C, Lorch M (1998) Meta-analysis of botulinum toxin treatment of spasmodic dysphonia: a review of 22 studies. Int J Lang Commun Disord 33(Suppl):327–329. https://doi.org/10.3109/13682829809179445
Bielamowicz S, Stager SV, Badillo A, Godlewski A (2002) Unilateral versus bilateral injections of botulinum toxin in patients with adductor spasmodic dysphonia. J Voice 16(1):117–123. https://doi.org/10.1016/s0892-1997(02)00080-2
Dharia I, Bielamowicz S (2020) Unilateral versus bilateral botulinum toxin injections in adductor spasmodic dysphonia in a large cohort. Laryngoscope 130(11):2659–2662. https://doi.org/10.1002/lary.28457. (Epub 2019 Dec 14)
Shoffel-Havakuk H, Rosow DE, Lava CX, Hapner ER, Johns MM 3rd (2019) Common practices in botulinum toxin injection for spasmodic dysphonia treatment: a national survey. Laryngoscope 129(7):1650–1656. https://doi.org/10.1002/lary.27696
Schuering JHC, van Hof KS, Heijnen BJ, van Benthem PPG, Sjögren EV, Langeveld APM (2021) Proposal for a core outcome set of measurement instruments to assess quality of voice in adductor spasmodic dysphonia based on a literature review. J Voice 35(6):933.e7-933.e21. https://doi.org/10.1016/j.jvoice.2020.02.010
Acknowledgements
We would like to acknowledge LetPub (https://www.letpub.com/editorial-certicate) for their help in English language editing.
Funding
This study is jointly supported by funding from (1) Xinglin Scholars Scientific Research Promotion Plan of Chengdu University of Traditional Chinese Medicine-Innovation team of Traditional Chinese Medicine Otorhinolaryngology Discipline, Natural Science (No. XKTD2021003); (2) Scientific Project of Hospital of Chengdu University of Traditional Chinese Medicine (No.21ZL01); (3) Sichuan Provincial Administration of Traditional Chinese Medicine Research Project (2023MS300).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by [YL], [FC], [FL], [DC] and [JZ]. The first draft of the manuscript was written by [YL], [LZ], [RH], [CW], [XX] and [LT]. The final manuscript was revised by [LZ] and [LT]. All authors commented on previous versions of the manuscript and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There is no declaration of any conflict of interests among the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Feifei Chen and Fangqi Liang are the co-first authors.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, Y., Chen, F., Liang, F. et al. Comparison of the efficacy and adverse effects of unilateral or bilateral botulinum toxin injections for adductor spasmodic dysphonia: a systematic review and meta-analysis. Eur Arch Otorhinolaryngol 281, 1357–1369 (2024). https://doi.org/10.1007/s00405-023-08366-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-023-08366-2