Abstract
Purpose
To analyze the mechanisms involved in the fetal heart rate (FHR) abnormalities after the epidural analgesia in labor.
Methods
A prospective unblinded single-center observational study on 55 term singleton pregnant women with spontaneous labor. All women recruited underwent serial bedside measurements of the main hemodynamic parameters using a non-invasive ultrasound system (USCOM-1A). Total vascular resistances (TVR), heart rate (HR), stroke volume (SV), cardiac output (CO) and arterial blood pressure were measured before epidural administration (T0), after 5 min 5 (T1) from epidural bolus and at the end of the first stage of labor (T2). FHR was continuously recorded through computerized cardiotocography before and after the procedure.
Results
The starting CO was significantly higher in a subgroup of women with low TVR than in women with high-TVR group. After the bolus of epidural analgesia in the low-TVR group there was a significant reduction in CO and then increased again at the end of the first stage, in the high-TVR group the CO increased insignificantly after the anesthesia bolus, while it increased significantly in the remaining part of the first stage of labor. On the other hand, CO was inversely correlated with the number of decelerations detected on cCTG in the 1 hour after the epidural bolus while the short-term variation was significantly lower in the group with high-TVR.
Conclusion
Maternal hemodynamic status at the onset of labor can make a difference in fetal response to the administration of epidural analgesia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Since the beginning of its history, analgesia in labor has always had controversial aspects and doubts about the maternal and fetus/neonatal consequences, thus safety remains a challenge to pursue. Transient abnormalities in fetal heart rate (FHR) have been described in up to 15% of the cases after the use of analgesia during labor [1] complicating the interpretation of fetal CTG and the prediction of a fetal acidemia at birth. Decelerations of FHR and bradycardia have been reported for all types of labor analgesia (epidural, spinal, combined spinal-epidural and intravenous opioids) [2]. The clinical significance of these changes is not entirely clear, however, there is a common consensus on maternal and fetal oxygenative and vascular pathophysiology [3]. It has been reported that the fetal oxygenation is altered with the dose dependent administration of epidural analgesia [4], as well has been proposed the hypothesis of uterine hyperactivity due to the reduction of catecholamines [5] or maternal hypotension due to an imbalance of adrenalin/noradrenalin ratio [6]. In this context several studies have described uteroplacental and fetal hemodynamics after labor analgesia with differences in clinical characteristics (antenatal, induction of labor, high risk or low risk pregnancies), vascular district evaluated, type of anesthesia (continuous infusion, single dose, self-controlled) and drugs used [7]. In the majority of studies, FHR changes is not associated with an increased incidence of cesarean section and did not appear to have an immediate effect on neonatal status as determined by Apgar scores [8]. Based on recent evidence of a maladaptive cardiovascular response to pregnancy complicated by placental syndromes [9,10,11,12], maternal hemodynamic assessment it has become an interesting way to evaluate maternal–fetal interactions from a different point of view. Labor and delivery are events that have a great impact on maternal general hemodynamics the change in maternal position from supine to lateral alone may produce an increase in cardiac output (+ 21.7%), decreased heart rate (− 5.6%), and increased maternal stroke volume (+ 26.5%) [13]. Anxiety, pain and exertion increases both heart rate and stroke volume, just as the utero-placental consequences of the reduced venous return to the heart due to caval compression from the supine position are well known. An increment in basal cardiac output of 12% has been reported in a group of women during labor [14].
The objective of the present study is to analyze the hemodynamic pattern of women during labor before and after epidural analgesia and its relationship with FHR.
Patients and methods
This was a prospective unblinded single-center observational study carried out at Salesi Maternal-Neonatal University Hospital in Ancona (Italy), between March 2018 and June 2019. The center treats 1800 parturients per year, with an epidural analgesia rate in labor of 40% and a cesarean delivery rate of approximately 24%.
Fifty five low-risk pregnant women in active labor with normal FHR trace submitted to epidural analgesia were recruited. Inclusion criteria were: healthy single pregnancy after the 37th week of gestation, spontaneous active labor (cervical dilation of at least 3 cm), age 18–40 years, height 155–180 cm, body mass index < 35 kg/m2, normal FHR pattern at admission. Exclusion criteria were: history of hypotensive episodes, pre-existing or actual hypertensive or metabolic disorders, psychiatric or somatic disease, fetal/neonatal malformations, other contraindications for epidural analgesia. Informed consent was obtained from all individual participants included in the study.
Epidural analgesia (EA)
After venous cannulation and survey of maternal parameters an epidural catheter was inserted at the L2-3 or L3-4 space. A bolus of 20 mL levobupivacaine and 10 μg of sufentanyl was subsequently administered, followed by a continuous infusion of a 10 mL/hour solution of either 0.0625% levobupivacaine with sufentanyl 0.5 μg/mL.
Hemodynamic evaluation
Hemodynamic pattern was assessed using a non-invasive ultrasonic monitor (USCOM®, USCOM Ltd, NSW, Australia), used for the cardiovascular evaluation in pregnancy and validated versus echocardiography [15]. A transducer was placed on the suprasternal notch to measure transaortic or transpulmonary blood flow, respectively. At least three consecutive cycles were registered for each scan, by two trained researchers, to obtain the main cardiac parameters including total vascular resistances (TVR), heart rate (HR), stroke volume (SV), cardiac output (CO), arterial blood pressure. These measurements were obtained before (T0) and after 5 min 5 (T1) from epidural bolus, and at the end of the first stage of labor (T2).
Computerized cardiotocography (cCTG)
The cCTG was performed for 1 h after epidural bolus by Sonicaid Oxford 8002 System (Manor Way, Old Woking, Surrey, England). Short-term variation (STV) was calculated as the average of sequential 1y16 minute pulse interval differences by Dawes-Redman software-based algorithm.
The protocol of this prospective study was approved by the ethics committee of our center and written informed consent was obtained from each patient.
Statistical analysis
Comparisons were performed using Pearson chi-squared test for proportions, and using independent samples t-test or the Kruskal–Wallis test for continuous data. Descriptive data were analyzed using IBM SPSS Statistics for Windows, Version 22.0 (IBM Corp Armonk, NY, USA). A P value < 0.05 was considered statistically significant. This study was performed in line with the principles of the Declaration of Helsinki. This is an observational study. The internal academic Research Ethics Committee has confirmed that no ethical approval is required.
Results
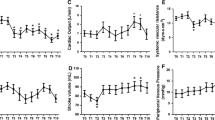
The 55 Patients recruited were divided in two subgroups Low-TVR and High-TVR using the reported cut-off 1200 dyne/sec/cm−5 [16, 17]. Characteristics of the study population are resumed in the Table 1. No significant differences were found in the characteristics of the two groups, not even in the rate of cesarean sections and in neonatal outcomes. Hemodynamics and cCTG records are summarized in the Tables 2 and 3. In the whole population Cardiac Output (CO) underwent a slight increase after epidural analgesia (EA) and a significant increase for the remainder of the first stage of labor (Fig. 1). Analyzing the CO trend by dividing the two subgroups we noticed that in the Low-TVR group the starting CO was significantly higher than in the High-TVR group (5.52 ± 0.52 vs 3.60 ± 0.88 L/min) (Fig. 2). After the bolus of epidural analgesia in the Low-TVR group there was a significant reduction in CO and then increased again at the end of the first stage, in the High-TVR group the CO increased insignificantly after the anesthesia bolus, while it increased significantly in the remaining part of the first stage of labor (Fig. 3). On the other hand, CO was inversely related with the number of decelerations detected on cCTG in the 1 hour after the epidural bolus (R = − 0.1685; p < 0.0001) (Fig. 4) while the Short-term variation was significantly lower in the group with High-TVR (Fig. 4).
Discussion
The correlated effects of epidural analgesia during labor have been extensively studied, nevertheless few studies have evaluated the phenomenon from the point of view of maternal hemodynamics. The main finding of this study was that if patients are selected on the basis of total vascular resistance, the hemodynamic attitude during labor and the response to epidural analgesia change significantly. We have shown that low vascular resistances are associated with higher levels of cardiac output and that this seems to guarantee better utero-placental and fetal performance during labor. Cardiac Output is calculated from stroke volume multiplied by heart rate, it increases throughout pregnancy as early as in the 5 week of pregnancy reaching in the three trimester, about 30–50% above that in the nonpregnant state [18]. Echocardiography is most commonly used for hemodynamics in pregnancy, invasive techniques are seldom used. An insufficient increasing of cardiac output during pregnancy has been associated to neonatal complications [19]. The influence of labor on hemodynamic values has been controversial, according to some authors there would be an increase in resting CO up to 50% [20,21,22] according to others there would be no changes [23]. According with previous evidences that CO may be linked to a fetal distress [16, 17], the hypothesis is that a lower CO, and thus a lower cardiac index, can affect fetal well-being as an expression of reduced cardiac performance and therefore of a reduced utero-placental perfusion.
Despite a physiological progressive reduction of vascular resistance by action of pregnancy mediators (nitric oxide, progesterone, prostaglandins,) and to the development of a low resistance circuit to the placenta, Doppler studies have associated the high resistance in uterine arteries to high peripheral vascular resistance and low maternal cardiac output [24].
The uterine fraction of maternal CO has been reported to be about 12% at term [25] on the other hand It has been calculated that utero/placental perfusion can be reduced by at least 60% during a uterine contraction in labor [26]. In the time of a contraction, most fetuses resist a period of short hypoxia, while fetuses with lower hypoxic tolerance limits show signs of compensation detected by the CTG in the form of decelerations and reduced variability. Short term variation (STV) is affirmed as a good predictor for fetal acid–base status during pregnancy and despite there are demonstrations of a significant increase in short and long term variation in peripartum period [27] in our series, in women with high-TVR, STV after an epidural bolus was significantly lower than in women with low-TVR, on the other hand women with low TVR and higher levels of CO have an improuved fetal response to maternal hypotension induced by epidural analgesic drugs as demonstrated by the reduced number of decelerations and higher short term variation.
Although with the limit of a relatively small sample size, the present study confirms a close link between maternal hemodynamics and uterus placental pathophysiology and studies the practice of epidural analgesia from a different point of view that allows to recognize differences substantial among women in labor. Childbear outcomes may be closely related to maternal low cardiac reserves, selecting a cohort of women in whom epidural analgesia can further worsen the hemodynamic stress of labor.
References
Van de Velde M, Teunkens A, Hanssens M, Vandermeersch E, Verhaeghe J (2004) Intrathecal sufentanil and fetal heart rate abnormalities a double-blind, double placebo controlled trial comparing two forms of combined spinal epidural analgesia with epidural analgesia in labor. Anesth Analg. https://doi.org/10.1213/01.ANE.0000101980.34587.66
Reynolds F (2011) Labour analgesia and the baby: good news is no news. Int J Obstet Anesth 20:38–50. https://doi.org/10.1016/j.ijoa.2010.08.004
Gude P, Kaci CSB, Sieker M, Vogelsang H, Bellgardt M, Herzog-Niescery J, Weber TP, Weber J, Teubner S, Kern P (2021) The influence of labor epidural analgesia on maternal, uteroplacental and fetoplacental hemodynamics in normotensive parturients: a prospective observational study. Int J Obstet Anesth 45:83–89. https://doi.org/10.1016/j.ijoa.2020.10.011
East CE, Colditz PB (2002) Effect of maternal epidural analgesia on fetal intrapartum oxygen saturation. Am J Perinatol 19:119–126. https://doi.org/10.1055/s-2002-25312
Abrao KC, Francisco RP, Miyadahira S, Cicarelli DD, Zugaib M (2009) Elevation of uterine basal tone and fetal heart rate abnormalities after labor analgesia: a randomized controlled trial. Obstet Gynecol 113:41–47. https://doi.org/10.1097/AOG.0b013e31818f5eb6
Arnaout L, Ghiglione S, Figueireido S, Mignon A (2008) Effects of maternal analgesia and anesthesia on the fetus and newborn. J Obstet Gynecol Biol Reprod. https://doi.org/10.1016/j.jgyn.2007.11.010
Hattler J, Klimek M, Rossaint R, Heesen M (2016) The effect of combined spinal-epidural versus epidural analgesia in laboring women on nonreassuring fetal heart rate tracings: systematic review and meta-analysis. Anesth Analg 123:955–964. https://doi.org/10.1213/ANE.0000000000001412
Anim-Somuah M, Smyth RM, Cyna AM, Cuthbert A (2018) Epidural versus non epidural or no analgesia for pain management in labour. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD000331.pub4
Thilaganathan B, Kalafat E (2019) Cardiovascular system in preeclampsia and beyond. Hypertension 73:522–531. https://doi.org/10.1161/HYPERTENSIONAHA.118.11191
Pisani I, Tiralongo GM, Lo Presti D, Gagliardi G, Farsetti D, Vasapollo B, Novelli GP, Andreoli A, Valensise H (2017) Correlation between maternal body composition and haemodynamic changes in pregnancy: different profiles for different hypertensive disorders. Pregnancy Hypertens 10:131–134. https://doi.org/10.1016/j.preghy.2017.07.149
Valensise H, Farsetti D, Lo Presti D, Pisani I, Tiralongo GM, Gagliardi G, Vasapollo B, Novelli GP (2016) Preterm delivery and elevated maternal total vascular resistance: signs of suboptimal cardiovascular adaptation to pregnancy? Ultrasound Obstet Gynecol 48:491–495. https://doi.org/10.1002/uog.15910
Giannubilo SR, Pasculli A, Tidu E, Biagini A, Boscarato V, Ciavattini A (2017) Relationship between maternal hemodynamics and plasma natriuretic peptide concentrations during pregnancy complicated by preeclampsia and fetal growth restriction. J Perinatol 37:484–487. https://doi.org/10.1038/jp.2016.264
Lee W, Rokey R, Miller J, Cotton DB (1989) Maternal hemodynamic effects of uterine contractions by M-mode and pulsed-Doppler echocardiography. Am J Obstet Gynecol 161:974–977. https://doi.org/10.1016/0002-9378(89)90765-5
Hunter S, Robson SC (1992) Adaptation of the maternal heart in pregnancy. Brit Heart J 68:540–543. https://doi.org/10.1136/hrt.68.12.540
Vinayagam D, Patey O, Thilaganathan B, Khalil A (2017) Cardiac output assessment in pregnancy: comparison of two automated monitors with echocardiography. Ultrasound Obstet Gynecol 49:32–38. https://doi.org/10.1002/uog.15915
Tiralongo GM, Lo Presti D, Pisani I, Gagliardi G, Scala RL, Novelli GP, Vasapollo B, Andreoli A, Valensise H (2015) Assessment of total vascular resistance and total body water in normotensive women during the first trimester of pregnancy. a key for the prevention of preeclampsia. Pregnancy Hypertens 5:193–197. https://doi.org/10.1016/j.preghy.2015.02.001
Valensise H, Tiralongo GM, Pisani I, Farsetti D, Lo Presti D, Gagliardi G, Basile MR, Novelli GP, Vasapollo B (2018) Maternal hemodynamics early in labor: a possible link with obstetric risk? Ultrasound Obstet Gynecol 51:509–513. https://doi.org/10.1002/uog.17447
Flo K, Wilsgaard T, Vårtun A, Acharya G (2010) A longitudinal study of the relationship between maternal cardiac output measured by impedance cardiography and uterine artery blood flow in the second half of pregnancy. BJOG 117:837–844. https://doi.org/10.1111/j.1471-0528.2010.02548.x
Wald RM, Silversides CK, Kingdom J, Toi A, Lau CS, Mason J, Colman JM, Sermer M, Siu SC (2015) Maternal cardiac output and fetal Doppler predict adverse neonatal outcomes in pregnant women with heart disease. J Am Heart Assoc 4:e002414. https://doi.org/10.1161/JAHA.115.002414
Kuhn JC, Sørum Falk R, Langesæter S (2017) Haemodynamic changes during labour: continuous minimally invasive monitoring in 20 healthy parturients. Int J Obstet Anesth 31:74–83. https://doi.org/10.1016/j.ijoa.2017.03.003
Tamás P, Szilágyi A, Jeges S, Vizer M, Csermely T, Ifi Z, Bálint A, Szabó I (2007) Effects of maternal central hemodynamics on fetal heart rate patterns. Acta Obstet Gynecol Scand 86:711–714. https://doi.org/10.1080/00016340701252217
Valensise H, Lo Presti D, Tiralongo GM, Pisani I, Gagliardi G, Vasapollo B, Frigo MG (2016) Foetal heart rate deceleration with combined spinal-epidural analgesia during labour: a maternal haemodynamic cardiac study. J Matern Fetal Neonatal Med 29:1980–1986. https://doi.org/10.3109/14767058.2015.1072156
Lees MM, Scott DB, Kerr MG (1970) Haemodynamic changes associated with labour. J Obstet Gynaecol Br Commonw 77:29–36. https://doi.org/10.1111/j.1471-0528.1970.tb03404.x
Tay J, Masini G, McEniery CM, Giussani DA, Shaw CJ, Wilkinson IB, Bennett PR, Lees CC (2019) Uterine and fetal placental Doppler indices are associated with maternal cardiovascular function. Am J Obstet Gynecol 220:96.e1-96.e8. https://doi.org/10.1016/j.ajog.2018.09.017
Thaler I, Manor D, Itskovitz J, Rottem S, Levit N, Timor-Tritsch I, Brandes JM (1990) Changes in uterine blood flow during human pregnancy. Am J Obstet Gynecol 162:121–125. https://doi.org/10.1016/0002-9378(90)90834-t
Janbu T, Nesheim BI (1987) Uterine artery blood velocities during contractions in pregnancy and labour related to intrauterine pressure. Br J Obstet Gynaecol 94:1150–1155. https://doi.org/10.1111/j.1471-0528.1987.tb02314.x
Kapaya H, Jacques R, Almond T, Rosser MH, Anumba D (2020) Is short-term-variation of fetal-heart-rate a better predictor of fetal acidaemia in labour? A Feasibility Study. PLoS ONE 15:e0236982. https://doi.org/10.1371/journal.pone.0236982
Funding
Open access funding provided by Università Politecnica delle Marche within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Stefano Raffaele Giannubilo, Mirco Amici and Simone Pizzi. The first draft of the manuscript was written by Stefano Raffaele Giannubilo and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Giannubilo, S.R., Amici, M., Pizzi, S. et al. Maternal hemodynamics and computerized cardiotocography during labor with epidural analgesia. Arch Gynecol Obstet 307, 1789–1794 (2023). https://doi.org/10.1007/s00404-022-06658-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-022-06658-2