Abstract
Purpose
To determine the negative conversion regularity of high-risk human papillomavirus (HR-HPV) and to evaluate the prognostic implications of HR-HPV testing in patients with cervical cancer after treatment.
Methods
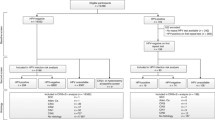
A retrospective post-treatment analysis of 173 patients with cervical cancer was performed from January 2011 to December 2012. Patients who had HR-HPV infection before treatment were included. Clinical and pathological characteristics, as well as follow-up information, were reviewed.
Results
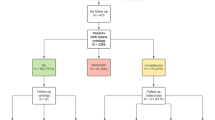
The negative conversion rate of HR-HPV reached 68.9 % within half a year and increased most rapidly within the first 2 years after treatment. Univariate and multivariate analyses suggested that the negative conversion rate of HR-HPV was significantly correlated with clinical stage, treatment regimens, and HR-HPV type (P < 0.05). In our analysis of 173 patients, we found that HR-HPV status was predictive of 3-year survival rate and disease recurrence (P < 0.05). Pelvic recurrence, but not distant metastasis, was influenced by HR-HPV status (P < 0.05). Through 2 × 2 table analysis, we found that HR-HPV was more sensitive (71.43 %) and specific (94.20 %) than cervical cytology (sensitivity 62.86 % and specificity 78.26 %).
Conclusions
The negative conversion rate of HR-HPV increased most rapidly within the first 2 years of cervical cancer surveillance. Persistent HPV infection was associated with a poor prognosis and had an impact on recurrence sites. Further large and multi-center prospective studies should be performed, but these results of this study suggested that HR-HPV monitoring is necessary to be used as a means of cervical cancer surveillance.

Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136:E359–E386
Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J (2016) Cancer statistics in China, 2015. CA Cancer J Clin 66:115–132
Siegel RL, Miller KD, Jemal A (2016) Cancer statistics, 2016. CA Cancer J Clin 66:7–30
Khalil J, El Kacemi H, Afif M, Kebdani T, Benjaafar N (2015) Five years’ experience treating locally advanced cervical cancer with concurrent chemoradiotherapy: results from a single institution. Arch Gynecol Obstet 292:1091–1099
Siegel R, Naishadham D, Jemal A (2013) Cancer statistics, 2013. CA Cancer J Clin 63:11–30
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65:87–108
Bernard HU, Burk RD, Chen Z, van Doorslaer K, zur Hausen H, de Villiers EM (2010) Classification of papillomaviruses (PVs) based on 189 PV types and proposal of taxonomic amendments. Virology 401:70–79
Muñoz N, Bosch FX, de Sanjosé S, Herrero R, Castellsagué X, Shah KV, Snijders PJ, Meijer CJ (2003) International Agency for Research on Cancer Multicenter Cervical Cancer Study Group. Epidemiologic classification of human papillomavirus types associated with cervical cancer. N Engl J Med 348:518–527
Kamal EM, El Sayed GA, El Behery MM, El Shennawy GA (2014) HPV detection in a self-collected vaginal swab combined with VIA for cervical cancer screening with correlation to histologically confirmed CIN. Arch Gynecol Obstet 290:1207–1213
Denny L, Boa R, Williamson AL, Allan B, Hardie D, Stan R, Myer L (2008) Human papillomavirus infection and cervical disease in human immunodeficiency virus-1-infected women. Obstet Gynecol 111:1380–1387
Bae JH, Lee SJ, Kim CJ, Hur SY, Park YG, Lee WC, Kim YT, Ng TL, Bock HL, Park JS (2008) Human papillomavirus (HPV) type distribution in Korean women: a meta-analysis. J Microbiol Biotechnol 18:788–794
Das P, Thomas A, Mahantshetty U, Shrivastava SK, Deodhar K, Mulherkar R (2012) HPV genotyping and site of viral integration in cervical cancers in Indian women. PLoS One 7:e41012
Gizzo S, Ancona E, Patrelli TS, Saccardi C, Anis O, Donato D, Nardelli GB (2013) Fertility preservation in young women with cervical cancer: an oncologic dilemma or a new conception of fertility sparing surgery? Cancer Invest 31:189
Das P, Thomas A, Kannan S, Deodhar K, Shrivastava SK, Mahantshetty U, Mulherkar R (2015) Human papillomavirus (HPV) genome status and cervical cancer outcome—a retrospective study. Indian J Med Res 142:525–532
Agapova M, Duignan A, Smith A, O’Neill C, Basu A (2015) Long-term costs of introducing HPV-DNA post-treatment surveillance to national cervical cancer screening in Ireland. Expert Rev Pharmacoecon Outcomes Res 15:999–1005
Smith MA, Gertig D, Hall M, Simms K, Lew JB, Malloy M, Saville M, Canfell K (2016) Transitioning from cytology-based screening to HPV-based screening at longer intervals: implications for resource use. BMC Health Serv Res 16:147
Hillemanns P (2016) The paradigm shift in cervical cancer screening in Germany. Arch Gynecol Obstet 293(1):3–4
Elit L, Kennedy EB, Fyles A, Metser U (2016) Follow-up for cervical cancer: a Program in Evidence-Based Care systematic review and clinical practice guideline update. Curr Oncol 23:109–118
Salani R, Backes FJ, Fung MF, Holschneider CH, Parker LP, Bristow RE, Goff BA (2011) Posttreatment surveillance and diagnosis of recurrence in women with gynecologic malignancies: society of Gynecologic Oncologists recommendations. Am J Obstet Gynecol 204:466–478
Intharaburan S, Tanapat Y, Tatanan K, Sangkhavasi K, Komolpis S, Buranawit K, Jarruwale P (2012) Human papillomavirus infection following radiation therapy or concurrent chemoradiation for invasive cervical cancer. J Med Assoc Thai 95:S38–S41
Brand RA (2009) Standards of reporting: the CONSORT, QUORUM, and STROBE guidelines. Clin Orthop Relat Res 467:1393–1394
Boehmer G, Wang L, Iftner A, Holz B, Haedicke J, von Wasielewski R, Martus P, Iftner T (2014) A population-based observational study comparing Cervista and Hybrid Capture 2 methods: improved relative specificity of the Cervista assay by increasing its cut-off. BMC Infect Dis 14:674
Silva J, Cerqueira F, Medeiros R (2014) Chlamydia trachomatis infection: implications for HPV status and cervical cancer. Arch Gynecol Obstet 289:715–723
Plummer M, Schiffman M, Castle PE, Maucort-Boulch D, Wheeler CM, ALTS Group (2007) A 2-year prospective study of human papillomavirus persistence among women with a cytological diagnosis of atypical squamous cells of undetermined significance or low-grade squamous intraepithelial lesion. J Infect Dis 195:1582–1589
Ryu A, Nam K, Kwak J, Kim J, Jeon S (2012) Early human papillomavirus testing predicts residual/recurrent disease after LEEP. J Gynecol Oncol 23:217–225
Kim CJ, Lee YS, Kwack HS, Yoon WS, Park TC, Park JS (2010) Specific human papillomavirus types and other factors on the risk of cervical intraepithelial neoplasia: a case-control study in Korea. Int J Gynecol Cancer 20:1067–1073
Nam K, Chung S, Kim J, Jeon S, Bae D (2009) Factors associated with HPV persistence after conization in patients with negative margins. J Gynecol Oncol 20:91–95
Hu D, Zhou J, Wang F, Shi H, Li Y, Li B (2015) HPV-16 E6/E7 promotes cell migration and invasion in cervical cancer via regulating cadherin switch in vitro and in vivo. Arch Gynecol Obstet 292:1345–1354
Lagos M, Van De Wyngard V, Poggi H, Cook P, Viviani P, Barriga MI, Pruyas M, Ferreccio C (2015) HPV16/18 genotyping for the triage of HPV positive women in primary cervical cancer screening in Chile. Infect Agent Cancer 10:43
Yu MC, Austin RM, Lin J, Beck T, Beriwal S, Comerci JT, Edwards RP, Sukumvanich P, Kelley J, Olawaiye AB (2015) The role of high-risk human Papilloma virus testing in the surveillance of cervical cancer after treatment. Arch Pathol Lab Med 139:1437–1440
Costa S, Venturoli S, Origoni M, Preti M, Mariani L, Cristoforoni P, Sandri MT (2015) Performance of HPV DNA testing in the follow-up after treatment of high-grade cervical lesions, adenocarcinoma in situ (AIS) and microinvasive carcinoma. Ecancermedicalscience 9:528
Park JY, Bae J, Lim MC, Lim SY, Lee DO, Kang S, Park SY, Nam BH, Seo SS (2009) Role of high risk-human papilloma virus test in the follow-up of patients who underwent conization of the cervix for cervical intraepithelial neoplasia. J Gynecol Oncol 20:86–90
Song YJ, Kim JY, Lee SK, Lim HS, Lim MC, Seo SS, Kang S, Lee DO, Park SY (2011) Persistent human papillomavirus DNA is associated with local recurrence after radiotherapy of uterine cervical cancer. Int J Cancer 129:896–902
Origoni M, Cristoforoni P, Costa S, Mariani L, Scirpa P, Lorincz A, Sideri M (2012) HPV-DNA testing for cervical cancer precursors: from evidence to clinical practice. Ecancermedicalscience 6:258
Noventa M, Ancona E, Cosmi E, Saccardi C, Litta P, D’Antona D, Nardelli GB, Gizzo S (2014) Usefulness, methods and rationale of lymph nodes HPV-DNA investigation in estimating risk of early stage cervical cancer recurrence: a systematic literature review. Clin Exp Metastasis 31:853–867
Deng T, Feng Y, Zheng J, Huang Q, Liu J (2015) Low initial human papillomavirus viral load may indicate worse prognosis in patients with cervical carcinoma treated with surgery. J Gynecol Oncol 26:111–117
Kocken M, Helmerhorst TJ, Berkhof J, Louwers JA, Nobbenhuis MA, Bais AG, Hogewoning CJ, Zaal A, Verheijen RH, Snijders PJ, Meijer CJ (2011) Risk of recurrent high-grade cervical intraepithelial neoplasia after successful treatment: a long-term multi-cohort study. Lancet Oncol 12:441–450
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no potential conflicts of interest.
Ethical approval
For this type of study, formal consent is not required, and this article does not contain any studies with animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Song, D., Kong, Wm., Zhang, Tq. et al. The negative conversion of high-risk human papillomavirus and its performance in surveillance of cervical cancer after treatment: a retrospective study. Arch Gynecol Obstet 295, 197–203 (2017). https://doi.org/10.1007/s00404-016-4197-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-016-4197-4