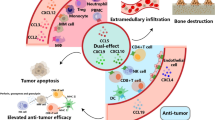
Abstract
Anti-programmed cell death protein 1 (PD-1) antibody drugs, nivolumab and pembrolizumab, are regarded as first-line therapies for advanced malignant melanoma. Anti-PD-1 therapy suppresses tumor immunity, and the therapeutic effect is frequently correlated with the number of tumor-infiltrating lymphocytes (TIL) and tumor mutation burden (TMB). However, sampling tumor tissues from the metastatic sites to examine the number of TILs and TMB level is often challenging. Herein, we focused on chemokines in blood to determine whether they can predict the therapeutic effect of anti-PD-1 (nivolumab) therapy. First, we measured 44 types of chemokines and cytokines in the blood of 8 advanced malignant melanomas before anti-PD-1 (nivolumab) treatment and examined the relationship between the levels of these proteins and therapeutic effect of the drug treatment, which suggested that C–C motif chemokine 5 (CCL5) and C–X–C motif chemokine ligand 12 (CXCL12) were candidates for biomarkers to predict the therapeutic effect of anti-PD-1 therapy. Next, we measured the blood levels of CCL5 and CXCL12 in 22 patients with advanced malignant melanomas before the administration of anti-PD-1 antibody. We evaluated tumor infiltration of CD8-positive T cells by immunostaining in nine patients in whom the metastatic site could be sampled at the beginning of the treatment. The patients with lower than average levels of CCL5 and CXCL12 had a large number of TILs (P = 0.04) and good disease-specific survival rate (P = 0.04). Therefore, CCL5 and CXCL12 could likely be used as biomarkers to predict the therapeutic effect of anti-PD-1 (nivolumab) therapy.



Similar content being viewed by others
Availability of data and material
The datasets used or analyzed during the current study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
Hodi FS, O’Day SJ, McDermott DF et al (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363:711–723
Topalian SL, Hodi FS, Brahmer JR et al (2012) Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med 366:2443–2454
Hamid O, Robert C, Daud A et al (2013) Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med 369:134–144
Ansell SM, Lesokhin AM, Borrello I et al (2015) PD-1 blockade with nivolumab in relapsed or refractory Hodgkin’s lymphoma. N Engl J Med 372:311–319
Garon EB, Rizvi NA, Hui R et al (2015) Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med 372:2018–2028
Freeman GJ, Long AJ, Iwai Y et al (2000) Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med 192:1027–1034
Jiang X, Wang J, Deng X et al (2019) Role of the tumor microenvironment in PD-L1/PD-1-mediated tumor immune escape. Mol Cancer 18:10
Topalian SL, Sznol M, McDermott DF et al (2014) Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. Topalian SL J Clin Oncol 32:1020–1030
Robert C, Long GV, Brady B et al (2015) Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med 372:320–330
Robert C, Schachter J, Long GV et al (2015) Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med 372:2521–2532
Wolchok JD, Kluger H, Callahanet MK et al (2013) Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med 369:122–133
Postow MA, Chesney J, Pavlick AC et al (2015) Nivolumab and ipilimumab versus ipilimumab in untreated melanoma. N Engl J Med 372:2006–2017
De Velasco G, Je Y, Bossé D et al (2017) Comprehensive meta-analysis of key immune-related adverse events from CTLA-4 and PD-1/PD-L1 inhibitors in cancer patients. Cancer Immunol Res 5:312–318
Tajmir-Riahi A, Bergmann T, Schmid M et al (2018) Life-threatening autoimmune cardiomyopathy reproducibly induced in a patient by checkpoint inhibitor therapy. J Immunother 41:35–38
Herbst RS, Soria JC, Kowanetz M et al (2014) Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 515:563–567
Taube JM, Klein A, Brahmer JR et al (2014) Association of PD-1, PD-1 ligands, and other features of the tumor immune microenvironment with response to anti-PD-1 therapy. Clin Cancer Res 20:5064–5074
Alexandrov LB, Nik-Zainal S, Wedge DC et al (2013) Signatures of mutational processes in human cancer. Nature 500:415–421
RutledgeWC KJ, Gao J et al (2013) Tumor infiltrating lymphocytes in glioblastoma are associated with specific genomic alterations and related to transcriptional class. Clin Cancer Res 19:4951–4960
Rooney MS, Shukla SA, Wu CJ et al (2015) Molecular and genetic properties of tumors associated with local immune cytolytic activity. Cell 160:48–61
Brown SD, Warren RL, Gibb EA et al (2014) Neo-antigens predicted by tumor genome meta-analysis correlate with increased patient survival. Genome Res 24:743–750
Schumacher TN, Schreiber RD (2015) Neoantigens in cancer immunotherapy. Science 348:69–74
Rizvi NA, Hellmann MD, Snyder A et al (2015) Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 348:124–128
Le DT, Uram JN, Wang H et al (2015) PD-1 blockade in tumors with mismatch-repair deficiency. N Engl J Med 372:2509–2520
Rizk EM, Seffens AM, Trager MH et al (2020) Biomarkers predictive of survival and response to immune checkpoint inhibitors in melanoma. Am J Clin Dermatol 21:1–11
Mantovani A, Allavena P, Sica A et al (2008) Cancer-related inflammation. Nature 454:436–444
Fujimura T, Sato Y, Tanita K et al (2019) Association of baseline serum levels of CXCL5 with the efficacy of nivolumab in advanced melanoma. Front Med 6:86
Edge SB, Compton CC, Fritz AG, Greene FL, Trotti A (eds) (2010) AJCC cancer staging manual, 7th edn. Springer, New York
Schwartz LH, Litière S, de Vries E et al (2016) RECIST 1.1-Update and clarification: From the RECIST committee. Eur J Cancer 62:132–137
Fujimoto Y, Inoue N, Morimoto K et al (2020) Significant association between high serum CCL5 levels and better disease-free survival of patients with early breast cancer. Cancer Sci 111:209–218
Rusetska N, Kowalski K, Zalewski K et al (2021) CXCR4/ACKR3/CXCL12 axis in the lymphatic metastasis of vulvar squamous cell carcinoma. J Clin Pathol
Schall TJ, Bacon K, Toy KJ et al (1990) Selective attraction of monocytes and T lymphocytes of the memory phenotype by cytokine RANTES. Nature 347:669–671
Balkwill FR (2012) The chemokine system and cancer. J Pathol 226:148–157
Oppermann M (2004) Chemokine receptor CCR5: insights into structure, function, and regulation. Cell Signal 16:1201–1210
González-Martín A, Gómez L, Lustgarten J et al (2011) Maximal T cell-mediated antitumor responses rely upon CCR5 expression in both CD4(+) and CD8(+) T cells. Cancer Res 71:5455–5466
Lapteva N, Huang XF (2010) CCL5 as an adjuvant for cancer immunotherapy. Expert Opin Biol Ther 10:725–733
González-Martin A, Mira E, Mañes S (2012) CCR5 as a potential target in cancer therapy: inhibition or stimulation? Anticancer Agents Med Chem 12:1045–1057
Aldinucci D, Colombatti A (2014) The inflammatory chemokine CCL5 and cancer progression. Mediators Inflamm:292376
Weitzenfeld P, Ben-Baruch A (2014) The chemokine system, and its CCR5 and CXCR4 receptors, as potential targets for personalized therapy in cancer. Cancer Lett 352:36–53
Chang LY, Lin YC, Mahalingam J et al (2012) Tumor-derived chemokine CCL5 enhances TGF-beta-mediated killing of CD8(+) T cells in colon cancer by T-regulatory cells. Cancer Res 72:1092–1102
Sugasawa H, Ichikura T, Kinoshita M, Ono S et al (2008) Gastric cancer cells exploit CD4+ cell-derived CCL5 for their growth and prevention of CD8+ cell-involved tumor elimination. Int J Cancer 122:2535–2541
Sharma M, Afrin F, Satija N et al (2011) Stromal-derived factor-1/CXCR4 signaling: indispensable role in homing and engraftment of hematopoietic stem cells in bone marrow. Stem Cells Dev 20:933–946
Zboralski D, Hoehlig K, Eulberg D et al (2017) Increasing tumor-infiltrating T Cells through inhibition of CXCL12 with NOX-A12 synergizes with PD-1 blockade. Cancer Immunol Res 11:950–956
Meng W, Xue S, Chen Y (2018) The role of CXCL12 in tumor microenvironment. Gene 641:105–110
Teicher BA, Fricker SP (2010) CXCL12 (SDF-1)/CXCR4 pathway in cancer. Clin Cancer Res 16:2927–2931
Ochoa-Callejero L, Perez-Martinez L, Rubio-Mediavilla S et al (2013) Maraviroc, a CCR5 antagonist, prevents development of hepatocellular carcinoma in a mouse model. PLoS ONE 8:e53992
D’Alterio C, Barbieri A, Portella L et al (2012) Inhibition of stromal CXCR4 impairs development of lung metastases. Cancer Immunol Immun 61:1713–1720
Cashen AF (2009) Plerixafor hydrochloride: a novel agent for the mobilization of peripheral blood stem cells. Drug Today 45:497–505
Liang Z, Zhan W, Zhu A et al (2012) Development of a unique small molecule modulator of CXCR4. PLoS ONE 7:e34038
Parameswaran R, Yu M, Lim M et al (2011) Combination of drug therapy in acute lymphoblastic leukemia with a CXCR4 antagonist. Leukemia 25:1314–1323
Kuhne MR, Mulvey T, Belanger B et al (2013) BMS-936564/MDX-1338: a fully human anti-CXCR4 antibody induces apoptosis in vitro and shows antitumor activity in vivo in hematologic malignancies. Clin Cancer Res 19:357–366
Tamamura H, Hori A, Kanzaki N et al (2003) T140 analogs as CXCR4 antagonists identified as anti-metastatic agents in the treatment of breast cancer. FEBS Lett 550:79–83
Ma LX, Qiao HQ, He CJ et al (2012) Modulating the interaction of CXCR4 and CXCL12 by low-molecular-weight heparin inhibits hepatic metastasis of colon cancer. Invest New Drug 30:508–517
Osella-Abate S, Vignale C, Annaratone L et al (2021) Microenvironment in cutaneous melanomas: a gene expression profile study may explain the role of histological regression. J Eur Acad Dermatol Venereol 35:e35–e38
Gabellini C, Gómez-Abenza E, Ibáñez-Molero S et al (2018) Interleukin 8 mediates bcl-xL-induced enhancement of human melanoma cell dissemination and angiogenesis in a zebrafish xenograft model. Int J Cancer 142:584–596
Dhawan P, Richmond A (2002) Role of CXCL1 in tumorigenesis of melanoma. J Leukoc Biol 72:9–18
Fujisawa Y, Yoshino K, Otsuka A et al (2018) Baseline neutrophil to lymphocyte ratio combined with serum lactate dehydrogenase level associated with outcome of nivolumab immunotherapy in a Japanese advanced melanoma population. Br J Dermatol 179:213–215
Rosner S, Kwong E, Shoushtari AN et al (2018) Peripheral blood clinical laboratory variables associated with outcomes following combination nivolumab and ipilimumab immunotherapy in melanoma. Cancer Med 7:690–697
Kuk D, Shoushtari AN, Barker CA et al (2016) Prognosis of mucosal, uveal, acral, nonacral cutaneous, and unknown primary melanoma from the time of first metastasis. Oncologist 21:848–854
Capasso A, Lang J, Pitts TM et al (2018) Characterization of immune responses to anti-PD-1 mono and combination immunotherapy in hematopoietic humanized mice implanted with tumor xenografts. J Immunother Cancer 7:37
Acknowledgements
We thank Ms. Aya Uchiyama for the technical assistance.
Funding
None.
Author information
Authors and Affiliations
Contributions
KN conceived, designed, and performed the experiments; analyzed the data; and wrote the manuscript. AA, YK, and RO conceived and designed the experiments and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests.
Ethics approval
The protocol for this human study was approved by the ethics committee of Shinshu University Graduate School of Medicine, Matsumoto, Japan (Permit No: 569).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nakamura, K., Ashida, A., Kiniwa, Y. et al. Chemokine level predicts the therapeutic effect of anti-PD-1 antibody (nivolumab) therapy for malignant melanoma. Arch Dermatol Res 314, 887–895 (2022). https://doi.org/10.1007/s00403-021-02305-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00403-021-02305-z