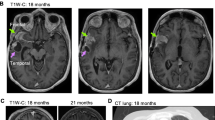
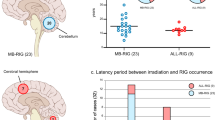
Abstract
Despite great advances in understanding of molecular pathogenesis and achievement of a high cure rate in medulloblastoma, recurrent medulloblastomas are still dismal. Additionally, misidentification of secondary malignancies due to histological ambiguity leads to misdiagnosis and eventually to inappropriate treatment. Nevertheless, the genomic characteristics of recurrent medulloblastomas are poorly understood, largely due to a lack of matched primary and recurrent tumor tissues. We performed a genomic analysis of recurrent tumors from 17 pediatric medulloblastoma patients. Whole transcriptome sequencing revealed that a subset of recurrent tumors initially diagnosed as locally recurrent medulloblastomas are secondary glioblastomas after radiotherapy, showing high similarity to the non-G-CIMP proneural subtype of glioblastoma. Further analysis, including whole exome sequencing, revealed missense mutations or complex gene fusion events in PDGFRA with augmented expression in the secondary glioblastomas after radiotherapy, implicating PDGFRA as a putative driver in the development of secondary glioblastomas after treatment exposure. This result provides insight into the possible application of PDGFRA-targeted therapy in these second malignancies. Furthermore, genomic alterations of TP53 including 17p loss or germline/somatic mutations were also found in most of the secondary glioblastomas after radiotherapy, indicating a crucial role of TP53 alteration in the process. On the other hand, analysis of recurrent medulloblastomas revealed that the most prevalent alterations are the loss of 17p region including TP53 and gain of 7q region containing EZH2 which already exist in primary tumors. The 7q gain events are frequently accompanied by high expression levels of EZH2 in both primary and recurrent medulloblastomas, which provides a clue to a new therapeutic target to prevent recurrence. Considering the fact that it is often challenging to differentiate between recurrent medulloblastomas and secondary glioblastomas after radiotherapy, our findings have major clinical implications both for correct diagnosis and for potential therapeutic interventions in these devastating diseases.






Similar content being viewed by others
References
Behjati S, Gundem G, Wedge DC, Roberts ND, Tarpey PS, Cooke SL, Van Loo P, Alexandrov LB, Ramakrishna M, Davies H et al (2016) Mutational signatures of ionizing radiation in second malignancies. Nat Commun 7:12605. https://doi.org/10.1038/ncomms12605
Boeva V, Popova T, Bleakley K, Chiche P, Cappo J, Schleiermacher G, Janoueix-Lerosey I, Delattre O, Barillot E (2012) Control-FREEC: a tool for assessing copy number and allelic content using next-generation sequencing data. Bioinformatics 28:423–425. https://doi.org/10.1093/bioinformatics/btr670
Brastianos PK, Taylor-Weiner A, Manley PE, Jones RT, Dias-Santagata D, Thorner AR, Lawrence MS, Rodriguez FJ, Bernardo LA, Schubert L et al (2014) Exome sequencing identifies BRAF mutations in papillary craniopharyngiomas. Nat Genet 46:161–165. https://doi.org/10.1038/ng.2868
Brennan CW, Verhaak RG, McKenna A, Campos B, Noushmehr H, Salama SR, Zheng S, Chakravarty D, Sanborn JZ, Berman SH et al (2013) The somatic genomic landscape of glioblastoma. Cell 155:462–477. https://doi.org/10.1016/j.cell.2013.09.034
Christopherson KM, Rotondo RL, Bradley JA, Pincus DW, Wynn TT, Fort JA, Morris CG, Mendenhall NP, Marcus RB Jr, Indelicato DJ (2014) Late toxicity following craniospinal radiation for early-stage medulloblastoma. Acta Oncol 53:471–480. https://doi.org/10.3109/0284186X.2013.862596
Cibulskis K, Lawrence MS, Carter SL, Sivachenko A, Jaffe D, Sougnez C, Gabriel S, Meyerson M, Lander ES, Getz G (2013) Sensitive detection of somatic point mutations in impure and heterogeneous cancer samples. Nat Biotechnol 31:213–219. https://doi.org/10.1038/nbt.2514
Dobin A, Davis CA, Schlesinger F, Drenkow J, Zaleski C, Jha S, Batut P, Chaisson M, Gingeras TR (2013) STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29:15–21. https://doi.org/10.1093/bioinformatics/bts635
Ellison DW, Onilude OE, Lindsey JC, Lusher ME, Weston CL, Taylor RE, Pearson AD, Clifford SC, United Kingdom Children’s Cancer Study Group Brain Tumour Consortium (2005) beta-Catenin status predicts a favorable outcome in childhood medulloblastoma: the United Kingdom Children’s Cancer Study Group Brain Tumour Committee. Journal of clinical oncology : official journal of the American Society of Clinical Oncology 23:7951–7957. https://doi.org/10.1200/jco.2005.01.5479
Elsamadicy AA, Babu R, Kirkpatrick JP, Adamson DC (2015) Radiation-induced malignant gliomas: a current review. World Neurosurg 83:530–542. https://doi.org/10.1016/j.wneu.2014.12.009
Forbes SA, Tang G, Bindal N, Bamford S, Dawson E, Cole C, Kok CY, Jia M, Ewing R, Menzies A et al (2010) COSMIC (the Catalogue of Somatic Mutations in Cancer): a resource to investigate acquired mutations in human cancer. Nucleic Acids Res 38:D652–D657. https://doi.org/10.1093/nar/gkp995
Ge H, Liu K, Juan T, Fang F, Newman M, Hoeck W (2011) FusionMap: detecting fusion genes from next-generation sequencing data at base-pair resolution. Bioinformatics 27:1922–1928. https://doi.org/10.1093/bioinformatics/btr310
Gessi M, Giangaspero F, Pietsch T (2003) Atypical teratoid/rhabdoid tumors and choroid plexus tumors: when genetics “surprise” pathology. Brain Pathol 13:409–414
Hill RM, Kuijper S, Lindsey JC, Petrie K, Schwalbe EC, Barker K, Boult JK, Williamson D, Ahmad Z, Hallsworth A et al (2015) Combined MYC and P53 defects emerge at medulloblastoma relapse and define rapidly progressive, therapeutically targetable disease. Cancer Cell 27:72–84. https://doi.org/10.1016/j.ccell.2014.11.002
Iyer MK, Chinnaiyan AM, Maher CA (2011) ChimeraScan: a tool for identifying chimeric transcription in sequencing data. Bioinformatics 27:2903–2904. https://doi.org/10.1093/bioinformatics/btr467
Jakacki RI, Burger PC, Zhou T, Holmes EJ, Kocak M, Onar A, Goldwein J, Mehta M, Packer RJ, Tarbell N et al (2012) Outcome of children with metastatic medulloblastoma treated with carboplatin during craniospinal radiotherapy: a Children’s Oncology Group Phase I/II study. J Clin Oncol 30:2648–2653. https://doi.org/10.1200/JCO.2011.40.2792
Korshunov A, Benner A, Remke M, Lichter P, von Deimling A, Pfister S (2008) Accumulation of genomic aberrations during clinical progression of medulloblastoma. Acta Neuropathol 116:383–390. https://doi.org/10.1007/s00401-008-0422-y
Korshunov A, Chavez L, Northcott PA, Sharma T, Ryzhova M, Jones DTW, von Deimling A, Pfister SM, Kool M (2017) DNA-methylation profiling discloses significant advantages over NanoString method for molecular classification of medulloblastoma. Acta Neuropathol 134:965–967. https://doi.org/10.1007/s00401-017-1776-9
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25:1754–1760. https://doi.org/10.1093/bioinformatics/btp324
MacDonald TJ, Aguilera D, Kramm CM (2011) Treatment of high-grade glioma in children and adolescents. Neuro-oncology 13:1049–1058. https://doi.org/10.1093/neuonc/nor092
Martin M (2011) Cutadapt removes adapter sequences from highthroughput sequencing reads. EMBnet J 17:10–12
Massimino M, Gandola L, Spreafico F, Biassoni V, Luksch R, Collini P, Solero CN, Simonetti F, Pignoli E, Cefalo G et al (2009) No salvage using high-dose chemotherapy plus/minus reirradiation for relapsing previously irradiated medulloblastoma. Int J Radiat Oncol Biol Phys 73:1358–1363. https://doi.org/10.1016/j.ijrobp.2008.06.1930
McPherson A, Hormozdiari F, Zayed A, Giuliany R, Ha G, Sun MG, Griffith M, Heravi Moussavi A, Senz J, Melnyk N et al (2011) deFuse: an algorithm for gene fusion discovery in tumor RNA-Seq data. PLoS Comput Biol 7:e1001138. https://doi.org/10.1371/journal.pcbi.1001138
Michaud K, de Tayrac M, D’Astous M, Duval C, Paquet C, Samassekou O, Gould PV, Saikali S (2016) Contribution of 1p, 19q, 9p and 10q automated analysis by FISH to the diagnosis and prognosis of oligodendroglial tumors according to WHO 2016 guidelines. PLoS One 11:e0168728. https://doi.org/10.1371/journal.pone.0168728
Morrissy AS, Garzia L, Shih DJ, Zuyderduyn S, Huang X, Skowron P, Remke M, Cavalli FM, Ramaswamy V, Lindsay PE et al (2016) Divergent clonal selection dominates medulloblastoma at recurrence. Nature 529:351–357. https://doi.org/10.1038/nature16478
Ng AK, Kenney LB, Gilbert ES, Travis LB (2010) Secondary malignancies across the age spectrum. Semin Radiat Oncol 20:67–78. https://doi.org/10.1016/j.semradonc.2009.09.002
Ning MS, Perkins SM, Dewees T, Shinohara ET (2015) Evidence of high mortality in long term survivors of childhood medulloblastoma. J Neurooncol 122:321–327. https://doi.org/10.1007/s11060-014-1712-y
Northcott PA, Dubuc AM, Pfister S, Taylor MD (2012) Molecular subgroups of medulloblastoma. Expert Rev Neurother 12:871–884. https://doi.org/10.1586/ern.12.66
Northcott PA, Jones DT, Kool M, Robinson GW, Gilbertson RJ, Cho YJ, Pomeroy SL, Korshunov A, Lichter P, Taylor MD et al (2012) Medulloblastomics: the end of the beginning. Nat Rev Cancer 12:818–834. https://doi.org/10.1038/nrc3410
Northcott PA, Shih DJ, Peacock J, Garzia L, Morrissy AS, Zichner T, Stutz AM, Korshunov A, Reimand J, Schumacher SE et al (2012) Subgroup-specific structural variation across 1,000 medulloblastoma genomes. Nature 488:49–56. https://doi.org/10.1038/nature11327
Northcott PA, Shih DJ, Remke M, Cho YJ, Kool M, Hawkins C, Eberhart CG, Dubuc A, Guettouche T, Cardentey Y et al (2012) Rapid, reliable, and reproducible molecular sub-grouping of clinical medulloblastoma samples. Acta Neuropathol 123:615–626. https://doi.org/10.1007/s00401-011-0899-7
Noushmehr H, Weisenberger DJ, Diefes K, Phillips HS, Pujara K, Berman BP, Pan F, Pelloski CE, Sulman EP, Bhat KP et al (2010) Identification of a CpG island methylator phenotype that defines a distinct subgroup of glioma. Cancer Cell 17:510–522. https://doi.org/10.1016/j.ccr.2010.03.017
Oh YT, Cho HJ, Kim J, Lee JH, Rho K, Seo YJ, Choi YS, Jung HJ, Song HS, Kong DS et al (2014) Translational validation of personalized treatment strategy based on genetic characteristics of glioblastoma. PLoS One 9:e103327. https://doi.org/10.1371/journal.pone.0103327
Park AK, Lee SJ, Phi JH, Wang KC, Kim DG, Cho BK, Haberler C, Fattet S, Dufour C, Puget S et al (2012) Prognostic classification of pediatric medulloblastoma based on chromosome 17p loss, expression of MYCC and MYCN, and Wnt pathway activation. Neuro-oncology 14:203–214. https://doi.org/10.1093/neuonc/nor196
Paugh BS, Qu C, Jones C, Liu Z, Adamowicz-Brice M, Zhang J, Bax DA, Coyle B, Barrow J, Hargrave D et al (2010) Integrated molecular genetic profiling of pediatric high-grade gliomas reveals key differences with the adult disease. J Clin Oncol 28:3061–3068. https://doi.org/10.1200/JCO.2009.26.7252
Paugh BS, Zhu X, Qu C, Endersby R, Diaz AK, Zhang J, Bax DA, Carvalho D, Reis RM, Onar-Thomas A et al (2013) Novel oncogenic PDGFRA mutations in pediatric high-grade gliomas. Cancer Res 73:6219–6229. https://doi.org/10.1158/0008-5472.CAN-13-1491
Phillips JJ, Aranda D, Ellison DW, Judkins AR, Croul SE, Brat DJ, Ligon KL, Horbinski C, Venneti S, Zadeh G et al (2013) PDGFRA amplification is common in pediatric and adult high-grade astrocytomas and identifies a poor prognostic group in IDH1 mutant glioblastoma. Brain Pathol 23:565–573. https://doi.org/10.1111/bpa.12043
Pietsch T, Schmidt R, Remke M, Korshunov A, Hovestadt V, Jones DT, Felsberg J, Kaulich K, Goschzik T, Kool M et al (2014) Prognostic significance of clinical, histopathological, and molecular characteristics of medulloblastomas in the prospective HIT2000 multicenter clinical trial cohort. Acta Neuropathol 128:137–149. https://doi.org/10.1007/s00401-014-1276-0
Pugh TJ, Weeraratne SD, Archer TC, Pomeranz Krummel DA, Auclair D, Bochicchio J, Carneiro MO, Carter SL, Cibulskis K, Erlich RL et al (2012) Medulloblastoma exome sequencing uncovers subtype-specific somatic mutations. Nature 488:106–110. https://doi.org/10.1038/nature11329
Ramaswamy V, Remke M, Bouffet E, Bailey S, Clifford SC, Doz F, Kool M, Dufour C, Vassal G, Milde T et al (2016) Risk stratification of childhood medulloblastoma in the molecular era: the current consensus. Acta Neuropathol 131:821–831. https://doi.org/10.1007/s00401-016-1569-6
Ramaswamy V, Remke M, Bouffet E, Faria CC, Perreault S, Cho YJ, Shih DJ, Luu B, Dubuc AM, Northcott PA et al (2013) Recurrence patterns across medulloblastoma subgroups: an integrated clinical and molecular analysis. Lancet Oncol 14:1200–1207. https://doi.org/10.1016/S1470-2045(13)70449-2
Robinson G, Parker M, Kranenburg TA, Lu C, Chen X, Ding L, Phoenix TN, Hedlund E, Wei L, Zhu X et al (2012) Novel mutations target distinct subgroups of medulloblastoma. Nature 488:43–48. https://doi.org/10.1038/nature11213
Robinson GW, Orr BA, Wu G, Gururangan S, Lin T, Qaddoumi I, Packer RJ, Goldman S, Prados MD, Desjardins A et al (2015) vismodegib exerts targeted efficacy against recurrent sonic Hedgehog-subgroup medulloblastoma: results from phase II pediatric brain tumor consortium studies PBTC-025B and PBTC-032. J Clin Oncol 33:2646–2654. https://doi.org/10.1200/JCO.2014.60.1591
Rudin CM, Hann CL, Laterra J, Yauch RL, Callahan CA, Fu L, Holcomb T, Stinson J, Gould SE, Coleman B et al (2009) Treatment of medulloblastoma with hedgehog pathway inhibitor GDC-0449. N Engl J Med 361:1173–1178. https://doi.org/10.1056/NEJMoa0902903
Sandmann T, Bourgon R, Garcia J, Li C, Cloughesy T, Chinot OL, Wick W, Nishikawa R, Mason W, Henriksson R et al (2015) Patients with proneural glioblastoma may derive overall survival benefit from the addition of bevacizumab to first-line radiotherapy and temozolomide: retrospective analysis of the AVAglio Trial. J Clin Oncol 33:2735–2744. https://doi.org/10.1200/JCO.2015.61.5005
Spence T, Sin-Chan P, Picard D, Barszczyk M, Hoss K, Lu M, Kim SK, Ra YS, Nakamura H, Fangusaro J et al (2014) CNS-PNETs with C19MC amplification and/or LIN28 expression comprise a distinct histogenetic diagnostic and therapeutic entity. Acta Neuropathol 128:291–303. https://doi.org/10.1007/s00401-014-1291-1
Suzuki K, Ojima M, Kodama S, Watanabe M (2003) Radiation-induced DNA damage and delayed induced genomic instability. Oncogene 22:6988–6993. https://doi.org/10.1038/sj.onc.1206881
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, van Baren MJ, Salzberg SL, Wold BJ, Pachter L (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28:511–515. https://doi.org/10.1038/nbt.1621
Verhaak RG, Hoadley KA, Purdom E, Wang V, Qi Y, Wilkerson MD, Miller CR, Ding L, Golub T, Mesirov JP et al (2010) Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell 17:98–110. https://doi.org/10.1016/j.ccr.2009.12.020
Worst BC, van Tilburg CM, Balasubramanian GP, Fiesel P, Witt R, Freitag A, Boudalil M, Previti C, Wolf S, Schmidt S et al (2016) Next-generation personalised medicine for high-risk paediatric cancer patients—the INFORM pilot study. Eur J Cancer 65:91–101. https://doi.org/10.1016/j.ejca.2016.06.009
Yamanaka R, Hayano A, Kanayama T (2016) Radiation-induced gliomas: a comprehensive review and meta-analysis. Neurosurg Rev. https://doi.org/10.1007/s10143-016-0786-8
Acknowledgements
This study was supported by a grant from the National R&D Program for Cancer Control, Ministry for Health and Welfare, Republic of Korea (1420020). This work was also supported by the 2016 Research Fund (1.160052.01) of Ulsan National Institute of Science and Technology (UNIST) and the Collaborative Genome Program for Fostering New Post-Genome Industry of the National Research Foundation (NRF) funded by the Ministry of Science and ICT (MSIT) (NRF-2017M3C9A5031004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no potential conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Phi, J.H., Park, A.K., Lee, S. et al. Genomic analysis reveals secondary glioblastoma after radiotherapy in a subset of recurrent medulloblastomas. Acta Neuropathol 135, 939–953 (2018). https://doi.org/10.1007/s00401-018-1845-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-018-1845-8