Abstract
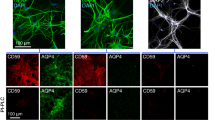
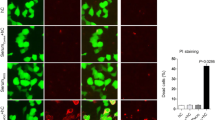
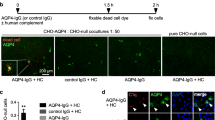
Neuromyelitis optica spectrum disorder (herein called NMO) is an autoimmune inflammatory disease of the central nervous system in which immunoglobulin G antibodies against astrocyte water channel aquaporin-4 (AQP4-IgG) cause demyelination and neurological deficit. Injury to oligodendrocytes, which do not express AQP4, links the initiating pathogenic event of AQP4-IgG binding to astrocyte AQP4 to demyelination. Here, we report evidence for a complement ‘bystander mechanism’ to account for early oligodendrocyte injury in NMO in which activated, soluble complement proteins following AQP4-IgG binding to astrocyte AQP4 result in deposition of the complement membrane attack complex (MAC) on nearby oligodendrocytes. Primary cocultures of rat astrocytes and mature oligodendrocytes exposed to AQP4-IgG and complement showed early death of oligodendrocytes in close contact with astrocytes, which was not seen in pure oligodendrocyte cultures, in cocultures exposed to AQP4-IgG and C6-depleted serum, or when astrocytes were damaged by a complement-independent mechanism. Astrocyte-oligodendrocyte cocultures exposed to AQP4-IgG and complement showed prominent MAC deposition on oligodendrocytes in contact with astrocytes, whereas C1q, the initiating protein in the classical complement pathway, and C3d, a component of the alternative complement pathway, were deposited only on astrocytes. Early oligodendrocyte injury with MAC deposition was also found in rat brain following intracerebral injection of AQP4-IgG, complement and a fixable dead-cell stain. These results support a novel complement bystander mechanism for early oligodendrocyte injury and demyelination in NMO.






Similar content being viewed by others
References
Asavapanumas N, Ratelade J, Verkman AS (2014) Unique neuromyelitis optica pathology produced in naive rats by intracerebral administration of NMO-IgG. Acta Neuropathol 127:539–551
Asgari N, Khorooshi R, Lillevang ST, Owens T (2013) Complement-dependent pathogenicity of brain-specific antibodies in cerebrospinal fluid. J Neuroimmunol 254:76–82
Bennett JL, Lam C, Kalluri SR, Saikali P, Bautista K, Dupree C, Glogowska M, Case D, Antel JP, Owens GP, Gilden D, Nessler S, Stadelmann C, Hemmer B (2009) Intrathecal pathogenic anti–aquaporin-4 antibodies in early neuromyelitis optica. Ann Neurol 66:617–629
Bradl M, Misu T, Takahashi T, Watanabe M, Mader S, Reindl M, Adzemovic M, Bauer J, Berger T, Fujihara K, Itoyama Y, Lassmann H (2009) Neuromyelitis optica: pathogenicity of patient immunoglobulin in vivo. Ann Neurol 66:630–643
Brown DR, Kretzschmar HA (1998) The glio-toxic mechanism of α-aminoadipic acid on cultured astrocytes. J Neurocytol 27:109–118
Crane JM, Lam C, Rossi A, Gupta T, Bennett JL, Verkman AS (2011) Binding affinity and specificity of neuromyelitis optica autoantibodies to aquaporin-4 M1/M23 isoforms and orthogonal arrays. J Biol Chem 286:16516–16524
Dincman TA, Beare JE, Ohri SS, Whittemore SR (2012) Isolation of cortical mouse oligodendrocyte precursor cells. J Neurosci Methods 209:219–226
Gotze O, Muller-Eberhard HJ (1970) Lysis of erythrocytes by complement in the absence of antibody. J Exp Med 132:898–915
Hinson SR, Roemer SF, Lucchinetti CF, Fryer JP, Kryzer TJ, Chamberlain JL, Howe CL, Pittock SJ, Lennon VA (2008) Aquaporin-4-binding autoantibodies in patients with neuromyelitis optica impair glutamate transport by down-regulating EAAT2. J Exp Med 205:2473–2481
Hinson SR, Romero MF, Popescu BF, Lucchinetti CF, Fryer JP, Wolburg H, Fallier-Becker P, Noell S, Lennon VA (2012) Molecular outcomes of neuromyelitis optica (NMO)-IgG binding to aquaporin-4 in astrocytes. Proc Natl Acad Sci USA 109:1245–1250
Huck S, Grass F, Hortnagl H (1984) The glutamate analog α-aminoadipic acid is taken up by astrocytes before exerting its gliotoxic effect in vitro. J Neurosci 4:2650–2657
Jarius S, Wildemann B (2010) AQP4 antibodies in neuromyelitis optica: diagnostic and pathogenetic relevance. Nat Rev Neurol 6:383–392
Lennon VA, Kryzer TJ, Pittock SJ, Verkman AS, Hinson SR (2005) IgG marker of optic-spinal multiple sclerosis binds to the aquaporin-4 water channel. J Exp Med 202:473–477
Lucchinetti CF, Mandler RN, McGavern D, Bruck W, Gleich G, Ransohoff RM, Trebst C, Weinshenker B, Wingerchuk D, Parisi JE, Lassmann H (2002) A role for humoral mechanisms in the pathogenesis of Devic’s neuromyelitis optica. Brain 125:1450–1461
Marignier R, Nicolle A, Watrin C, Touret M, Cavagna S, Varrin-Doyer M, Cavillon G, Rogemond V, Confavreux C, Honnorat J, Giraudon P (2010) Oligodendrocytes are damages by neuromyelitis optic immunoglobulin G via astrocyte injury. Brain 133:2578–2591
Misu T, Fujihara K, Kakita A, Konno H, Nakamura M, Watanabe S, Takahashi T, Nakashima I, Takahashi H, Itoyama Y (2007) Loss of aquaporin 4 in lesions of neuromyelitis optica: distinction from multiple sclerosis. Brain 130:1224–1234
Papadopoulos MC, Verkman AS (2012) Aquaporin 4 and neuromyelitis optica. Lancet Neurol 11:535–544
Park CC, Shin ML, Simard JM (1997) The complement membrane attack complex and the bystander effect in cerebral vasospasm. J Neurosurg 87:294–300
Parratt JD, Prineas JW (2010) Neuromyelitis optica: a demyelinating disease characterized by acute destruction and regeneration of a perivascular astrocytes. Mult Scler 16:1156–1172
Phuan PW, Ratelade J, Rossi A, Tradtrantip L, Verkman AS (2012) Complement-dependent cytotoxicity in neuromyelitis optica requires aquaporin-4 protein assembly in orthogonal arrays. J Biol Chem 287:13829–13839
Piddlesden SJ, Morgan BP (1993) Killing of rat glial cells by complement: deficiency of the rat analogue of CD59 is the cause of oligodendrocyte susceptibility to lysis. J Neuroimmunol 48:169–175
Ratelade J, Asavapanumas N, Ritchie AM, Wemlinger S, Bennett JL, Verkman AS (2013) Involvement of antibody-dependent cell-mediated cytotoxicity in inflammatory demyelination in a mouse model of neuromyelitis optica. Acta Neuropathol 126:699–709
Ratelade J, Bennett JL, Verkman AS (2011) Evidence against cellular internalization in vivo of NMO-IgG, aquaporin-4, and excitatory amino acid transporter 2 in neuromyelitis optica. J Biol Chem 286:45156–45164
Roemer SF, Parisi JE, Lennon VA, Benarroch EE, Lassmann H, Bruck W, Mandler RN, Weinshenker BG, Pittock SJ, Wingerchuk DM, Lucchinetti CF (2007) Pattern-specific loss of aquaporin-4 immunoreactivity distinguishes neuromyelitis optica from multiple sclerosis. Brain 130:1194–1205
Rossi A, Ratelade J, Papadopoulos MC, Bennett JL, Verkman AS (2012) Neuromyelitis optica IgG does not alter aquaporin-4 water permeability, plasma membrane M1/M23 isoform content or supramolecular assembly. Glia 60:2027–2039
Saadoun S, Waters P, Bell BA, Vincent A, Verkman AS, Papadopoulos MC (2010) Intra-cerebral injection of neuromyelitis optica immunoglobulin G and human complement produces neuromyelitis optica lesions in mice. Brain 133:349–361
Sagan SA, Winger RC, Cruz-Herranz A, Nelson PA, Hagberg S, Miller CN, Spencer CM, Ho PP, Bennette JL, Levy M, Levin MH, Verkman AS, Stienman L, Green AJ, Anderson MS, Sobel RA, Zamvil SS (2016) Tolerance checkpoint bypass permits emergence of pathogenic T cells to neuromyelitis optica autoantigen aquaporin-4. Proc Natl Acad Sci USA 113:14781–14786
Scolding NJ, Morgan BP, Compston DA (1998) The expression of complement regulatory proteins by adult human oligodendrocytes. J Neuroimmunol 84:69–75
Smith AJ, Jin BJ, Ratelade J, Verkman AS (2014) Aggregation state determines the localization and function of M1- and M23-aquaporin-4 in astrocytes. J Cell Biol 204:559–573
Smith AJ, Verkman AS (2015) Superresolution imaging of aquaporin-4 cluster size in antibody-stained paraffin brain sections. Biophys J 109:2511–2522
Tradtrantip L, Asavapanumas N, Verkman AS (2013) Therapeutic cleavage of anti-aquaporin-4 autoantibody in neuromyelitis optica by an IgG-selective proteinase. Mol Pharmacol 83:1268–1275
Tradtrantip L, Ratelade J, Zhang H, Verkman AS (2013) Enzymatic deglycosylation converts pathogenic neuromyelitis optica anti-aquaporin-4 immunoglobulin G into therapeutic antibody. Ann Neurol 73:77–85
Tradtrantip L, Zhang H, Saadoun S, Phuan PW, Lam C, Papadopoulos MC, Bennett JL, Verkman AS (2012) Anti–aquaporin-4 monoclonal antibody blocker therapy for neuromyelitis optica. Ann Neurol 71:314–322
Tradtrantip L, Zhang H, Anderson MO, Saadoun S, Phuan PW, Papadopoulos MC, Bennett JL, Verkman AS (2012) Small-molecule inhibitors of NMO-IgG binding to aquaporin-4 reduce astrocyte cytotoxicity in neuromyelitis optica. FASEB J 26:2197–2208
Waxman SG, Black JA (1984) Freeze-fracture ultrastructure of the perinodal astrocyte and associated glial junctions. Brain Res 308:77–87
Whitney KD, McNamara JO (2000) GluR3 autoantibodies destroy neural cells in a complement dependent manner modulated by complement regulatory proteins. J Neurosci 20:7307–7316
Wing MG, Zajicek J, Seilly DJ, Compston DA, Lachmann PJ (1992) Oligodendrocytes lack glycolipid anchored proteins which protect them against complement lysis. Restoration of resistance to lysis by incorporation of CD59. Immunology 76:140–145
Wolburg H, Wolburg-Buchholz K, Fallier-Becker P, Noell S, Mack AF (2011) Structure and functions of aquaporin-4-based orthogonal arrays of particles. Int Rev Cell Mol Biol 287:1–41
Wrzos C, Winkler A, Metz I, Kayser DM, Thal DR, Wegner C, Bruck W, Nessler S, Bennett JL, Stadelmann C (2014) Early loss of oligodendrocytes in human and experimental neuromyelitis optic lesions. Acta Neuropathol 127:523–538
Yao X, Su T, Verkman AS (2016) Clobetasol promotes remyelination in a mouse model of neuromyelitis optica. Acta Neuropathol Commun 4:42
Zipfel PF, Skerka C (2009) Complement regulators and inhibitory proteins. Nat Rev Immunol 9:729–740
Acknowledgements
This work was supported by Grants EY13574, EB00415, DK35124, and DK72517 from the National Institutes of Health, and a Grant from the Guthy-Jackson Charitable Foundation. We thank Dr. Jeffrey Bennett (University of Colorado Denver, Aurora, CO) for providing recombinant monoclonal NMO antibody rAb-53.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors has competing interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tradtrantip, L., Yao, X., Su, T. et al. Bystander mechanism for complement-initiated early oligodendrocyte injury in neuromyelitis optica. Acta Neuropathol 134, 35–44 (2017). https://doi.org/10.1007/s00401-017-1734-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-017-1734-6