Abstract
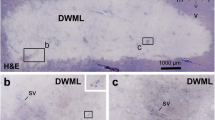
Clinical studies have identified white matter (WM) lesions as hyperintensive regions in the MRI images of elderly patients. Since a cerebrovascular origin was attributed to such lesions, the present analysis set out to define the microvascular histopathologic changes in the periventricular WM in the aged. Post-mortem samples of the frontal, parietal, and occipital periventricular WM of 40–90-year-old subjects were prepared for quantitative light and electron microscopy. Light microscopic examination revealed microvascular fibrohyalinosis as the most common type of microvascular damage in the elderly. Ultrastructural analysis identified the microvascular thickening as collagen deposits affecting the basement membrane. The vascular density did not correlate with the age. The basement membrane pathology significantly increased, while the number of intact microvessels gradually decreased, with advancing age in the frontal and occipital WM. Finally, peripheral atherosclerosis coincided with massive microvascular fibrosis, particularly in the frontal WM. Our results demonstrate an age-related microvascular degeneration in the periventricular WM, which may contribute to the development of WM lesions by hindering a sufficient supply of nutrients to the affected WM sites. Furthermore, the data accord with previous observations identifying the frontal lobe as the site at which WM vulnerability is most pronounced. Finally, atherosclerosis in large, peripheral vessels is considered to be a predictive marker of microvascular pathology in the WM.



Similar content being viewed by others
References
Akiguchi I, Tomimoto H, Wakita H, Kawamoto Y, Matsuo A, Ohnishi K, Watanabe T, Budka H (2004) Topographical and cytopathological lesion analysis of the white matter in Binswanger’s disease brains. Acta Neuropathol (Berl) 107:563–570
Barber R, Gholkar A, Scheltens P, Ballard C, McKeith IG, O’Brien JT (2000) MRI volumetric correlates of white matter lesions in dementia with Lewy bodies and Alzheimer’s disease. Int J Geriatr Psychiatry 15:911–916
Barkhof F, Scheltens P (2002) Imaging of white matter lesions. Cerebrovasc Dis 13(Suppl 2):21–30
Bartzokis G, Cummings JL, Sultzer D, Henderson VW, Nuechterlein KH, Mintz J (2003) White matter structural integrity in healthy aging adults and patients with Alzheimer disease: a magnetic resonance imaging study. Arch Neurol 60:393–398
Bronge L (2002) Magnetic resonance imaging in dementia. A study of brain white matter changes. Acta Radiol Suppl 428:1–32
Brown WR, Moody DM, Challa VR, Thore CR, Anstrom JA (2002) Venous collagenosis and arteriolar tortuosity in leukoaraiosis. J Neurol Sci 203–204:159–163
Brown WR, Moody DM, Thore CR, Challa VR (2000) Apoptosis in leukoaraiosis. AJNR Am J Neuroradiol 21:79–82
Challa VR, Thore CR, Moody DM, Anstrom JA, Brown WR (2004) Increase of white matter string vessels in Alzheimer’s disease. J Alzheimers Dis 6:379–383; discussion 443–449
De Jong GI, Farkas E, Stienstra CM, Plass JR, Keijser JN, de la Torre JC, Luiten PGM (1999) Cerebral hypoperfusion yields capillary damage in the hippocampal CA1 area that correlates with spatial memory impairment. Neuroscience 91:203–210
de Groot JC, de Leeuw FE, Oudkerk M, Hofman A, Jolles J, Breteler MM (2000) Cerebral white matter lesions and depressive symptoms in elderly adults. Arch Gen Psychiatry 57:1071–1076
de Groot JC, de Leeuw FE, Oudkerk M, van Gijn J, Hofman A, Jolles J, Breteler MM (2000) Cerebral white matter lesions and cognitive function: the Rotterdam Scan Study. Ann Neurol 47:145–151
de Leeuw FE, de Groot JC, Achten E, Oudkerk M, Ramos LM, Heijboer R, Hofman A, Jolles J, van Gijn J, Breteler MM (2001) Prevalence of cerebral white matter lesions in elderly people: a population based magnetic resonance imaging study. The Rotterdam Scan Study. J Neurol Neurosurg Psychiatry 70:9–14
de Leeuw FE, de Groot JC, Oudkerk M, Witteman JC, Hofman A, van Gijn J, Breteler MM (2002) Hypertension and cerebral white matter lesions in a prospective cohort study. Brain 125:765–772
de Reuck J (1971) The human periventricular arterial blood supply and the anatomy of cerebral infarctions. Eur Neurol 5:321–334
Desmond DW (2002) Cognition and white matter lesions. Cerebrovasc Dis 13(Suppl 2):53–57
Englund E (2002) Neuropathology of white matter lesions in vascular cognitive impairment. Cerebrovasc Dis 13(Suppl 2):11–15
Erkinjuntti T (2002) Subcortical vascular dementia. Cerebrovasc Dis 13(Suppl 2):58–60
Farkas E, Annaházi A, Institóris Á, Mihály A, Luiten PGM, Bari F (2005) Diazoxide and dimethyl sulphoxide alleviate experimental cerebral hypoperfusion-iduced white matter injury in the rat brain. Neurosci Lett 373:195–199
Farkas E, De Jong GI, de Vos RAI, Jansen Steur EN, Luiten PGM (2000) Pathological features of cerebral cortical capillaries are doubled in Alzheimer’s disease and Parkinson’s disease. Acta Neuropathol (Berl) 100:395–402
Farkas E, Donka G, de Vos RAI, Mihály A, Bari F, Luiten PGM (2004) Experimental cerebral hypoperfusion imposes white matter injury and microglial activation in the rat brain. Acta Neuropathol (Berl) 108:57–64
Farkas E, Luiten PGM (2001) Cerebral microvascular pathology in aging and Alzheimer’s disease. Prog Neurobiol 64:575–611
Fazekas F, Ropele S, Enzinger C, Gorani F, Seewann A, Petrovic K, Schmidt R (2005) MTI of white matter hyperintensities. Brain 128:2926–2932
Fazekas F, Schmidt R, Kleinert R, Kapeller P, Roob G, Flooh E (1998) The spectrum of age-associated brain abnormalities: their measurement and histopathological correlates. J Neural Transm Suppl 53:31–39
Fazekas F, Schmidt R, Scheltens P (1998) Pathophysiologic mechanisms in the development of age-related white matter changes of the brain. Dement Geriatr Cogn Disord 9(Suppl 1):2–5
Ferro JM, Madureira S (2002) Age-related white matter changes and cognitive impairment. J Neurol Sci 203–204:221–225
Firbank MJ, Lloyd AJ, Ferrier N, O’Brien JT (2004) A volumetric study of MRI signal hyperintensities in late-life depression. Am J Geriatr Psychiatry 12:606–612
Goldberg MP, Ransom BR (2003) New light on white matter. Stroke 34:330–332
Gootjes L, Teipel SJ, Zebuhr Y, Schwarz R, Leinsinger G, Scheltens P, Moller HJ, Hampel H (2004) Regional distribution of white matter hyperintensities in vascular dementia, Alzheimer’s disease and healthy aging. Dement Geriatr Cogn Disord 18:180–188
Head D, Buckner RL, Shimony JS, Williams LE, Akbudak E, Conturo TE, McAvoy M, Morris JC, Snyder AZ (2004) Differential vulnerability of anterior white matter in non-demented aging with minimal acceleration in dementia of the Alzheimer type: evidence from diffusion tensor imaging. Cereb Cortex 14:410–423
Kawamura J, Meyer JS, Terayama Y, Weathers S (1992) Leuko-araiosis and cerebral hypoperfusion compared in elderly normals and Alzheimer’s dementia. J Am Geriatr Soc 40:375–380
Ketonen LM (1998) Neuroimaging of the aging brain. Neurol Clin 16:581–598
Kobari M, Meyer JS, Ichijo M, Oravez WT (1990) Leukoaraiosis: correlation of MR and CT findings with blood flow, atrophy, and cognition. AJNR Am J Neuroradiol 11:273–281
Kobayashi K, Hayashi M, Nakano H, Fukutani Y, Sasaki K, Shimazaki M, Koshino Y (2002) Apoptosis of astrocytes with enhanced lysosomal activity and oligodendrocytes in white matter lesions in Alzheimer’s disease. Neuropathol Appl Neurobiol 28:238–251
Kuo HK, Lipsitz LA (2004) Cerebral white matter changes and geriatric syndromes: is there a link? J Gerontol A Biol Sci Med Sci 59:818–826
Lopez OL, Becker JT, Jungreis CA, Rezek D, Estol C, Boller F, DeKosky ST (1995) Computed tomography—but not magnetic resonance imaging—identified periventricular white-matter lesions predict symptomatic cerebrovascular disease in probable Alzheimer’s disease. Arch Neurol 52:659–664
Manolio TA, Burke GL, O’Leary DH, Evans G, Beauchamp N, Knepper L, Ward B (1999). Relationships of cerebral MRI findings to ultrasonographic carotid atherosclerosis in older adults: the cardiovascular health study. CHS Collaborative research group. Arterioscler Thromb Vasc Biol 19:356–365
Marstrand JR, Garde E, Rostrup E, Ring P, Rosenbaum S, Mortensen EL, Larsson HB (2002) Cerebral perfusion and cerebrovascular reactivity are reduced in white matter hyperintensities. Stroke 33:972–976
Moody DM, Bell MA, Challa VR (1990) Features of the cerebral vascular pattern that predict vulnerability to perfusion or oxygenation deficiency: an anatomic study. AJNR Am J Neuroradiol 11:431–439
Moody DM, Brown WR, Challa VR, Ghazi-Birry HS, Reboussin DM (1997) Cerebral microvascular alterations in aging, leukoaraiosis, and Alzheimer’s disease. Ann NY Acad Sci 826:103–116
Moody DM, Thore CR, Anstrom JA, Challa VR, Langefeld CD, Brown WR (2004) Quantification of afferent vessels shows reduced brain vascular density in subjects with leukoaraiosis. Radiology 233:883–890
O’Brien J, Desmond P, Ames D, Schweitzer I, Harrigan S, Tress B (1996) A magnetic resonance imaging study of white matter lesions in depression and Alzheimer’s disease. Br J Psychiatry 168:477–485
Ohtani R, Tomimoto H, Kawasaki T, Yagi H, Akiguchi I, Shibasaki H (2003) Cerebral vasomotor reactivity to postural change is impaired in patients with cerebrovascular white matter lesions. J Neurol 250:412–417
Pantoni L, Garcia JH (1997) Pathogenesis of leukoaraiosis. Stroke 28:652–659
Pugh KG, Lipsitz LA (2002) The microvascular frontal-subcortical syndrome of aging. Neurobiol Aging 23:421–431
Roman GC (2002) On the history of lacunes, etat crible, and the white matter lesions of vascular dementia. Cerebrovasc Dis 13(Suppl 2):1–6
Scheltens P, Barkhof F, Leys D, Wolters EC, Ravid R, Kamphorst W (1995) Histopathologic correlates of white matter changes on MRI in Alzheimer’s disease and normal aging. Neurology 45:883–888
Schmidt R, Schmidt H, Kapeller P, Enzinger C, Ropele S, Saurugg R, Fazekas F (2002) The natural course of MRI white matter hyperintensities. J Neurol Sci 203–204:253–257
Takashima S, Tanaka K (1978) Development of cerebrovascular architecture and its relationship to periventricular leukomalacia. Arch Neurol 35:11–16
Tang Y, Nyengaard JR, Pakkenberg B, Gundersen HJ (1997) Age-induced white matter changes in the human brain: a stereological investigation. Neurobiol Aging 18:609–615
Thomas AJ, Perry R, Barber R, Kalaria RN, O’Brien JT (2002) Pathologies and pathological mechanisms for white matter hyperintensities in depression. Ann NY Acad Sci 977:333–339
Tomimoto H, Akiguchi I, Wakita H, Suenaga T, Nakamura S, Kimura J (1997) Regressive changes of astroglia in white matter lesions in cerebrovascular disease and Alzheimer’s disease patients. Acta Neuropathol (Berl) 94:146–152
Ueno M, Tomimoto H, Akiguchi I, Wakita H, Sakamoto H (2002) Blood-brain barrier disruption in white matter lesions in a rat model of chronic cerebral hypoperfusion. J Cereb Blood Flow Metab 22:97–104
Verny M, Duyckaerts C, Pierot L, Hauw JJ (1991) Leuko-araiosis. Dev Neurosci 13:245–250
Wakita H, Tomimoto H, Akiguchi I, Kimura J (1994) Glial activation and white matter changes in the rat brain induced by chronic cerebral hypoperfusion: an immunohistochemical study. Acta Neuropathol (Berl) 87:484–492
Wakita H, Tomimoto H, Akiguchi I, Matsuo A, Lin JX, Ihara M, McGeer PL (2002) Axonal damage and demyelination in the white matter after chronic cerebral hypoperfusion in the rat. Brain Res 924:63–70
Ylikoski R, Ylikoski A, Erkinjuntti T, Sulkava R, Raininko R, Tilvis R (1993) White matter changes in healthy elderly persons correlate with attention and speed of mental processing. Arch Neurol 50:818–824
Acknowledgments
The study was supported by the Lewy Body Project Foundation, The Netherlands; the Hungarian Research Fund (OTKA) grant F042803; and a Bolyai János Research Scholarship of the Hungarian Academy of Sciences to E.F.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Farkas, E., de Vos, R.A.I., Donka, G. et al. Age-related microvascular degeneration in the human cerebral periventricular white matter. Acta Neuropathol 111, 150–157 (2006). https://doi.org/10.1007/s00401-005-0007-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-005-0007-y