Abstract
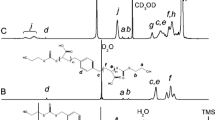
A poly(l-lactide) diol was obtained through ring opening polymerization of l-lactide, using 1,6 hexanediol and tin(II) 2 ethylhexanoate as a catalyst. In the second step, the poly(l-lactide) macromer (mLA) was obtained by the reaction of poly(l-lactide) diol with methacrylic anhydride. The effective incorporation of the polymerizable end groups was assessed by Fourier transform infrared spectroscopy and nuclear magnetic resonance (1H NMR). Besides, poly(l-lactide) networks (pmLA) were prepared by photopolymerization of mLA. Further, the macromer was copolymerized with 2-hydroxyethyl acrylate seeking to tailor the hydrophilicity of the system. A set of hydrophilic copolymer networks were obtained. The phase microstructure of the new system and the network architecture was investigated by differential scanning calorimetry, infrared spectroscopy, dynamic mechanical spectroscopy, thermogravimetry, and water sorption studies.











Similar content being viewed by others
References
Langer R, Vacanti JP (1993) Science 260:920
Peppas NA, Langer R (2004) Aiche J 50:536
Jagur-Grodzinski J (2006) Polym Adv Technol 17:395
Lavik E, Langer R (2004) Appl Microbiol Biotechnol 65:1
Cao Y, Carol TI (2005) Aust J Chem 58:691
Wang YK, Yong T, Ramakrishna S (2005) Aust J Chem 58:704
Grijpma DW, Pennings AJ (1994) Macromol Chem Phys 195:1633
Holland SJ, Tighe B, Gould PLJ (1986) J Control Release 4:155
Vert M, Li SM, Spenlehaur G, Guerin P (1992) J Mater Sci Mater Med 3:432
Vert M, Schwarch G, Coudane J (1995) J Macromol Sci Pure Appl Chem A32:787
Kharas GB, Kamenetsky M, Simantirakis J, Beinlich KC, Rizzo AMT, Caywood GA, Watson K (1997) J Appl Polym Sci 66:1123
Peter SJ, Kim P, Yasko AW, Yaszemski MJ, Mikos AG (1999) J Biomed Mater Res 44:314
Domb AJ, Manor N, Elmalak O (1996) Biomaterials 17:411
Kim BS, Hrkach JS, Langer R (2000) J Polym Sci Part A: Polym Chem 38:1277
Burdick JA, Mason MN, Anseth KS (2001) J Biomater Sci Polym Ed 12:1253
Burdick JA, Padera RF, Huang JV, Anseth KS (2002) J Biomed Mater Res 63:484
Horbett TA, Schway MB, Ratner BD (1985) J Colloid Interface Sci 104:28
Horbett TA, Schway MB (1988) J Biomed Mater Res 22:763
Chinn JA, Horbett TA, Ratner BD, Schway MB (1989) J Colloid Interface Sci 127:67
Ertel SI, Ratner BD, Horbett TA (1990) J Biomed Mater Res 24:1637
Muller M, Oehr C (1999) Surf Coatings Technol 116:802
Klee D, Villari RV, Hocker H, Dekker B, Mittermayer C (1995) J Mater Sci Mater Med 5:592
Fang YE, Lu XB, Wang SZ, Zhao X, Fang F (1996) J Appl Polym Sci 62:2209
Ajayaghosh A, Das S (1992) J Appl Polym Sci 45:1617
Geuskens G, Etoc A, Michele PD (2000) Eur Polym J 36:265
Kang IK, Kwon BK, Lee JH, Lee HB (1993) Biomaterials 14:787
Lee JS, Kaibara M, Iwaki M, Sasabe H, Suzuki Y, Kusakabe M (1993) Biomaterials 14:958
Sato H, Tsuji H, Ikeda S, Ikemoto N, Ishikawa J, Nishimoto S (1999) J Biomed Mater Res 44:22
Park TG, Cohen S, Langer R (1992) Macromolecules 25:116
Barakat I, Dubois PH, Grandfils CH, Jérôme R (1999) J Polym Sci Part A: Polym Chem 37:2401
Grijpma DW, Pennings AJ (1991) Polym Bull 25:335
Grijpma DW, Zondervan GJ, Pennings AJ (1991) Polym Bull 25:327
Albertsson AC, Gruvegard M (1995) Polymer 36:1009
Jamshidi K, Hoyn SH, Ikada Y (1988) Polymer 29:2229
Hoyn SH, Jamshidi K, Ikada Y (1997) Biomaterials 18:1503
Escobar Ivirico JL, Salmerón Sánchez M, Sabater i Serra R, Meseguer Dueñas JM, Gómez Ribelles JL, Monleón Pradas M (2006) Macromol Chem Phys 207:2195
Escobar Ivirico JL, Costa Martínez E, Salmerón Sánchez M, Criado Muñoz I, Gómez Ribelles JL, Monleón Pradas M (2007) J Biomed Mater Res. Part B Appl Biomat 83B:266
Tutusaus O, Delfosse S, Simal F, Demonceau A, Noels AF, Núñez R, Viñas C, Teixidor F (2002) Inorg Chem Commun 5:941
Kanaoka S, Sawamoto M, Higashimura T (1991) Macromolecules 24:2309
Salmerón Sánchez M, Gómez Ribelles JL, Hernández Sánchez F, Mano JF (2005) Thermochim Acta 430:201
López-Rodríguez N, López-Arraiza A, Meaurio E, Sarasua JR (2006) Polym Eng Sci 9:1299
Flory PJ (1953) Principles of polymer chemistry. Cornell University Press, Ithaca
Arima Y, Iwata H (2007) Biomaterials 28:3074
Marchin KL, Berrie CL (2003) Langmuir 19:9883
Pelham JR, Wang YL (1997) Proc Natl Acad Sci U S A 94:13661
Gray DS, Tien J, Chen CS (2003) J Biomed Mater Res 66A:605
Genes NG, Rowley JA, Mooney DJ, Bonassar LJ (2004) Arch Biochem Biophys 422:161
Acknowledgments
The support of the Spanish Ministry of Science through project No. MAT2006-08120 (including the FEDER financial support) is kindly acknowledged. JLEI acknowledges Generalitat Valenciana for the support through the predoctoral grant CTBPRB/2005/075.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Short text for the table of contents
A new set of copolymer networks based on poly(l-lactide) macromer p(mLA) was synthesized, whose water sorption behavior can be modulated by copolymerization with a hydrophilic monomer. The new block copolymer network is able to tailor the water sorption capacity, keeping the p(mLA) properties. The figure shows the ideal structure for the hydrophilized p(l-lactide) networks.

Rights and permissions
About this article
Cite this article
Escobar Ivirico, J.L., Salmerón-Sánchez, M., Gómez Ribelles, J.L. et al. Poly(l-lactide) networks with tailored water sorption. Colloid Polym Sci 287, 671–681 (2009). https://doi.org/10.1007/s00396-009-2026-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-009-2026-z