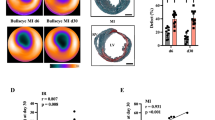
Abstract
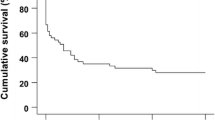
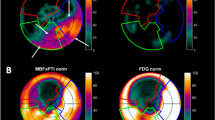
Nonrevascularizable coronary artery disease is a frequent cause of hibernating myocardium leading to heart failure (HF). Currently, there is a paucity of therapeutic options for patients with this condition. There is a lack of animal models resembling clinical features of hibernating myocardium. Here we present a large animal model of hibernating myocardium characterized by serial multimodality imaging. Yucatan minipigs underwent a surgical casein ameroid implant around the proximal left anterior descending coronary artery (LAD), resulting in a progressive obstruction of the vessel. Pigs underwent serial multimodality imaging including invasive coronary angiography, cardiac magnetic resonance (CMR), and hybrid 18F-Fluorodeoxyglucose positron emission tomography-computed tomography (FDG-PET/CT). A total of 43 pigs were operated on and were followed for 120 ± 37 days with monthly multimodality imaging. 24 pigs (56%) died during the follow-up. Severe LAD luminal stenosis was documented in all survivors. In the group of 19 long-term survivors, 17 (90%) developed left ventricular systolic dysfunction [median LVEF of 35% (IQR 32.5–40.5%)]. In 17/17, at-risk territory was viable on CMR and 14 showed an increased glucose uptake in the at-risk myocardium on 18FDG-PET/CT. The present pig model resembles most of the human hibernated myocardium characteristics and associated heart failure (systolic dysfunction, viable myocardium, and metabolic switch to glucose). This human-like model might be used to test novel interventions for nonrevascularizable coronary artery disease and ischemia heart failure as a previous stage to clinical trials.








Similar content being viewed by others
References
Alvino VV, Fernandez-Jimenez R, Rodriguez-Arabaolaza I, Slater S, Mangialardi G, Avolio E, Spencer H, Culliford L, Hassan S, Sueiro Ballesteros L, Herman A, Ayaon-Albarran A, Galan-Arriola C, Sanchez-Gonzalez J, Hennessey H, Delmege C, Ascione R, Emanueli C, Angelini GD, Ibanez B, Madeddu P (2018) Transplantation of allogeneic pericytes improves myocardial vascularization and reduces interstitial fibrosis in a swine model of reperfused acute myocardial infarction. J AmHeart Assoc. https://doi.org/10.1161/JAHA.117.006727
Bito V, Heinzel FR, Weidemann F, Dommke C, van der Velden J, Verbeken E, Claus P, Bijnens B, De Scheerder I, Stienen GJ, Sutherland GR, Sipido KR (2004) Cellular mechanisms of contractile dysfunction in hibernating myocardium. Circ Res 94:794–801. https://doi.org/10.1161/01.RES.0000124934.84048.DF
Bito V, van der Velden J, Claus P, Dommke C, Van Lommel A, Mortelmans L, Verbeken E, Bijnens B, Stienen G, Sipido KR (2007) Reduced force generating capacity in myocytes from chronically ischemic, hibernating myocardium. Circ Res 100:229–237. https://doi.org/10.1161/01.RES.0000257829.07721.57
Bobi J, Solanes N, Fernandez-Jimenez R, Galan-Arriola C, Dantas AP, Fernandez-Friera L, Galvez-Monton C, Rigol-Monzo E, Aguero J, Ramirez J, Roque M, Bayes-Genis A, Sanchez-Gonzalez J, Garcia-Alvarez A, Sabate M, Roura S, Ibanez B, Rigol M (2017) Intracoronary administration of allogeneic adipose tissue-derived mesenchymal stem cells improves myocardial perfusion but not left ventricle function, in a translational model of acute myocardial infarction. J Am Heart Assoc. https://doi.org/10.1161/JAHA.117.005771
Botker HE, Hausenloy D, Andreadou I, Antonucci S, Boengler K, Davidson SM, Deshwal S, Devaux Y, Di Lisa F, Di Sante M, Efentakis P, Femmino S, Garcia-Dorado D, Giricz Z, Ibanez B, Iliodromitis E, Kaludercic N, Kleinbongard P, Neuhauser M, Ovize M, Pagliaro P, Rahbek-Schmidt M, Ruiz-Meana M, Schluter KD, Schulz R, Skyschally A, Wilder C, Yellon DM, Ferdinandy P, Heusch G (2018) Practical guidelines for rigor and reproducibility in preclinical and clinical studies on cardioprotection. Basic Res Cardiol 113:39. https://doi.org/10.1007/s00395-018-0696-8
Braunwald E (2013) Research advances in heart failure: a compendium. Circ Res 113:633–645. https://doi.org/10.1161/CIRCRESAHA.113.302254
Buckley NM, Gootman PM, Yellin EL, Brazeau P (1979) Age-related cardiovascular effects of catecholamines in anesthetized piglets. Circ Res 45:282–292. https://doi.org/10.1161/01.res.45.2.282
Cabrera JA, Butterick TA, Long EK, Ziemba EA, Anderson LB, Duffy CM, Sluiter W, Duncker DJ, Zhang J, Chen Y, Ward HB, Kelly RF, McFalls EO (2013) Reduced expression of mitochondrial electron transport chain proteins from hibernating hearts relative to ischemic preconditioned hearts in the second window of protection. J Mol Cell Cardiol 60:90–96. https://doi.org/10.1016/j.yjmcc.2013.03.018
Caillaud D, Calderon J, Reant P, Lafitte S, Dos Santos P, Couffinhal T, Roques X, Barandon L (2010) Echocardiographic analysis with a two-dimensional strain of chronic myocardial ischemia induced with ameroid constrictor in the pig. Interact Cardiovasc Thorac Surg 10:689–693. https://doi.org/10.1510/icvts.2010.232819
Canty JM Jr, Klocke FJ (1987) Reductions in regional myocardial function at rest in conscious dogs with chronically reduced regional coronary artery pressure. Circ Res 61:II107–116
Fallavollita JA, Logue M, Canty JM Jr (2001) Stability of hibernating myocardium in pigs with a chronic left anterior descending coronary artery stenosis: absence of progressive fibrosis in the setting of stable reductions in flow, function and coronary flow reserve. J Am Coll Cardiol 37:1989–1995. https://doi.org/10.1016/s0735-1097(01)01250-5
Gedik N, Kruger M, Thielmann M, Kottenberg E, Skyschally A, Frey UH, Cario E, Peters J, Jakob H, Heusch G, Kleinbongard P (2017) Proteomics/phosphoproteomics of left ventricular biopsies from patients with surgical coronary revascularization and pigs with coronary occlusion/reperfusion: remote ischemic preconditioning. Sci Rep 7:7629. https://doi.org/10.1038/s41598-017-07883-5
Gheorghiade M, Sopko G, De Luca L, Velazquez EJ, Parker JD, Binkley PF, Sadowski Z, Golba KS, Prior DL, Rouleau JL, Bonow RO (2006) Navigating the crossroads of coronary artery disease and heart failure. Circulation 114:1202–1213. https://doi.org/10.1161/CIRCULATIONAHA.106.623199
Heusch G, Libby P, Gersh B, Yellon D, Bohm M, Lopaschuk G, Opie L (2014) Cardiovascular remodelling in coronary artery disease and heart failure. Lancet 383:1933–1943. https://doi.org/10.1016/S0140-6736(14)60107-0
Heusch G, Schulz R (1996) Hibernating myocardium: a review. J Mol Cell Cardiol 28:2359–2372. https://doi.org/10.1006/jmcc.1996.0229
Heusch G, Schulz R, Rahimtoola SH (2005) Myocardial hibernation: a delicate balance. Am J Physiol Heart Circ Physiol 288:H984–999. https://doi.org/10.1152/ajpheart.01109.2004
Heusch G, Skyschally A, Schulz R (2011) The in-situ pig heart with regional ischemia/reperfusion—ready for translation. J Mol Cell Cardiol 50:951–963. https://doi.org/10.1016/j.yjmcc.2011.02.016
Holley CT, Long EK, Butterick TA, Duffy CM, Lindsey ME, Stone LH, McFalls EO, Kelly RF (2015) Mitochondrial fusion proteins in revascularized hibernating hearts. J Surg Res 195:29–36. https://doi.org/10.1016/j.jss.2014.12.052
Hughes GC, Post MJ, Simons M (1985) Annex BH (2003) Translational physiology: porcine models of human coronary artery disease: implications for preclinical trials of therapeutic angiogenesis. J Appl Physiol 94:1689–1701. https://doi.org/10.1152/japplphysiol.00465.2002
Ibanez B, Aletras AH, Arai AE, Arheden H, Bax J, Berry C, Bucciarelli-Ducci C, Croisille P, Dall'Armellina E, Dharmakumar R, Eitel I, Fernandez-Jimenez R, Friedrich MG, Garcia-Dorado D, Hausenloy DJ, Kim RJ, Kozerke S, Kramer CM, Salerno M, Sanchez-Gonzalez J, Sanz J, Fuster V (2019) Cardiac MRI endpoints in myocardial infarction experimental and clinical trials: JACC scientific expert panel. J Am Coll Cardiol 74:238–256. https://doi.org/10.1016/j.jacc.2019.05.024
Inou T, Tomoike H, Watanabe K, Kikuchi Y, Mizukami M, Kurozumi T, Nakamura M (1980) A newly developed X-ray transparent ameroid constrictor for study on progression of gradual coronary stenosis. Basic Res Cardiol 75:537–543. https://doi.org/10.1007/bf01907835
Keeran KJ, Jeffries KR, Zetts AD, Taylor J, Kozlov S, Hunt TJ (2017) A chronic cardiac ischemia model in swine using an ameroid constrictor. J Vis Exp. https://doi.org/10.3791/56190
Khatibzadeh S, Farzadfar F, Oliver J, Ezzati M, Moran A (2013) Worldwide risk factors for heart failure: a systematic review and pooled analysis. Int J Cardiol 168:1186–1194. https://doi.org/10.1016/j.ijcard.2012.11.065
Lindsey ML, Bolli R, Canty JM Jr, Du XJ, Frangogiannis NG, Frantz S, Gourdie RG, Holmes JW, Jones SP, Kloner RA, Lefer DJ, Liao R, Murphy E, Ping P, Przyklenk K, Recchia FA, Schwartz Longacre L, Ripplinger CM, Van Eyk JE, Heusch G (2018) Guidelines for experimental models of myocardial ischemia and infarction. Am J Physiol Heart Circ Physiol 314:H812–H838. https://doi.org/10.1152/ajpheart.00335.2017
Lozano I, Capin E, de la Hera JM, Llosa JC, Carro A, Lopez-Palop R (2015) Diffuse coronary artery disease not amenable to revascularization: long-term prognosis. Rev Esp Cardiol (Engl Ed) 68:631–633. https://doi.org/10.1016/j.rec.2015.02.013
McFalls EO, Kelly RF, Hu Q, Mansoor A, Lee J, Kuskowski M, Sikora J, Ward HB, Zhang J (2007) The energetic state within hibernating myocardium is normal during dobutamine despite inhibition of ATP-dependent potassium channel opening with glibenclamide. Am J Physiol Heart Circ Physiol 293:H2945–2951. https://doi.org/10.1152/ajpheart.00012.2007
McFalls EO, Murad B, Haspel HC, Marx D, Sikora J, Ward HB (2003) Myocardial glucose uptake after dobutamine stress in chronic hibernating swine myocardium. J Nucl Cardiol 10:385–394. https://doi.org/10.1016/s1071-3581(03)00431-8
Millard RW (1981) Induction of functional coronary collaterals in the swine heart. Basic Res Cardiol 76:468–473. https://doi.org/10.1007/bf01908345
Neubauer S (2007) The failing heart–an engine out of fuel. N Engl J Med 356:1140–1151. https://doi.org/10.1056/NEJMra063052
Patterson RE, Kirk ES (1983) Analysis of coronary collateral structure, function, and ischemic border zones in pigs. Am J Physiol 244:H23–31. https://doi.org/10.1152/ajpheart.1983.244.1.H23
Peterson LR, Gropler RJ (2010) Radionuclide imaging of myocardial metabolism. Circ Cardiovasc Imaging 3:211–222. https://doi.org/10.1161/CIRCIMAGING.109.860593
Roth DM, Maruoka Y, Rogers J, White FC, Longhurst JC, Bloor CM (1987) Development of coronary collateral circulation in left circumflex Ameroid-occluded swine myocardium. Am J Physiol 253:H1279–1288. https://doi.org/10.1152/ajpheart.1987.253.5.H1279
Sanchez-Gonzalez J, Fernandez-Jimenez R, Nothnagel ND, Lopez-Martin G, Fuster V, Ibanez B (2015) Optimization of dual-saturation single bolus acquisition for quantitative cardiac perfusion and myocardial blood flow maps. J Cardiovasc Magn Reson 17:21. https://doi.org/10.1186/s12968-015-0116-2
Sayago-Silva I, Garcia-Lopez F, Segovia-Cubero J (2013) Epidemiology of heart failure in Spain over the last 20 years. Rev Esp Cardiol (Engl Ed) 66:649–656. https://doi.org/10.1016/j.rec.2013.03.012
Schulz R, Guth BD, Pieper K, Martin C, Heusch G (1992) Recruitment of an inotropic reserve in moderately ischemic myocardium at the expense of metabolic recovery. A model of short-term hibernation. Circ Res 70:1282–1295. https://doi.org/10.1161/01.res.70.6.1282
Schulz R, Miyazaki S, Miller M, Thaulow E, Heusch G, Ross J Jr, Guth BD (1989) Consequences of regional inotropic stimulation of ischemic myocardium on regional myocardial blood flow and function in anesthetized swine. Circ Res 64:1116–1126. https://doi.org/10.1161/01.res.64.6.1116
St Louis JD, Hughes GC, Kypson AP, DeGrado TR, Donovan CL, Coleman RE, Yin B, Steenbergen C, Landolfo KP, Lowe JE (2000) An experimental model of chronic myocardial hibernation. Ann Thorac Surg 69:1351–1357. https://doi.org/10.1016/s0003-4975(00)01130-9
Tarkia M, Stark C, Haavisto M, Kentala R, Vahasilta T, Savunen T, Strandberg M, Hynninen VV, Saunavaara V, Tolvanen T, Teras M, Rokka J, Pietila M, Saukko P, Roivainen A, Saraste A, Knuuti J (2015) Cardiac remodeling in a new pig model of chronic heart failure: assessment of left ventricular functional, metabolic, and structural changes using PET, CT, and echocardiography. J Nucl Cardiol 22:655–665. https://doi.org/10.1007/s12350-015-0068-9
Tsang HG, Rashdan NA, Whitelaw CB, Corcoran BM, Summers KM, MacRae VE (2016) Large animal models of cardiovascular disease. Cell Biochem Funct 34:113–132. https://doi.org/10.1002/cbf.3173
Unger EF (2001) Experimental evaluation of coronary collateral development. Cardiovasc Res 49:497–506. https://doi.org/10.1016/s0008-6363(00)00285-6
Wai T, Garcia-Prieto J, Baker MJ, Merkwirth C, Benit P, Rustin P, Ruperez FJ, Barbas C, Ibanez B, Langer T (2015) Imbalanced OPA1 processing and mitochondrial fragmentation cause heart failure in mice. Science 350:aad016. https://doi.org/10.1126/science.aad0116
Acknowledgements
This study has been partially funded by the Horizon 2020 European Research Area Network on Cardiovascular Diseases (ERA-CVD) Joint Transnational Call “AC16/00021: FAT4HEART,” by the Spanish Society of Cardiology through a “Translational Research grant 2019,” and by the Instituto de Salud Carlos III (ISCIII) and the European Regional Development Fund (ERDF) through a FIS grant (Ref # PI16/02110). Imaging phenotyping was partially supported by the Comunidad de Madrid (S2017/BMD-3867 RENIM-CM) and cofunded with European structural and investment funds. The CNIC is supported by the ISCIII, the Ministerio de Ciencia e Innovación and the Pro CNIC Foundation, and is a Severo Ochoa Center of Excellence (SEV-2015-0505).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Javier Sanchez-González is a Philips employee. The rest of authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Martínez-Milla, J., Galán-Arriola, C., Carnero, M. et al. Translational large animal model of hibernating myocardium: characterization by serial multimodal imaging. Basic Res Cardiol 115, 33 (2020). https://doi.org/10.1007/s00395-020-0788-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-020-0788-0