Abstract
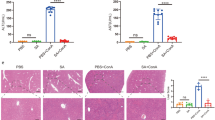
The spleen plays a critical role in post-infarct myocardial remodeling. However, the role of the spleen in exacerbating myocardial infarction (MI) during acute ischemia/reperfusion (I/R) injury is unknown. The present study tests the hypothesis that splenic leukocytes are activated by substances released from ischemic myocardium to subsequently exacerbate myocardial injury during reperfusion. The left coronary artery in C57BL/6 mice underwent various durations of occlusion followed by 60 min of reperfusion (denoted as min/min of I/R) with or without splenectomy prior to I/R injury. Splenectomy significantly decreased myocardial infarct size (IS) in 40′/60′ and 50′/60′ groups (p < 0.05); however, it had no effect on IS in 10′/60′, 20′/60′ and 30′/60′ groups (p = NS). In the 20′/60′ group, infusion of 40-min ischemic heart homogenate (40-IHH) upon reperfusion increased IS by >threefold versus infusion of 10-IHH (p < 0.05). Splenectomy abolished the infarct-exacerbating effect of 40-IHH, which was restored by splenic leukocyte adoptive transfer (SPAT). Furthermore, depletion of HMGB1 in the 40-IHH group abolished its infarct-exacerbating effect (p < 0.05), and 40-IHH failed to increase IS in both RAGE−/− mice and splenectomized wild-type mice with SPAT from RAGE−/− mice. The injection of 40-IHH significantly increased formyl peptide receptor 1 (FPR1) expression in sham spleens when compared to 10-IHH-treated sham and control mice. cFLFLF, a specific FPR1 antagonist, reduced myocardial neutrophil infiltration and abrogated the infarct-exacerbating effect of 40-IHH during reperfusion. A cardio (HMGB1)–splenic (RAGE receptor) signaling axis exists and contributes to myocardial infarct exacerbation during reperfusion after prolonged ischemic insults by activating splenic leukocytes. The FPR1 is a potential therapeutic target for inhibiting the cardio–splenic axis that augments infarct size during post-ischemic reperfusion.







Similar content being viewed by others
References
Aleshin A, Ananthakrishnan R, Li Q, Rosario R, Lu Y, Qu W, Song F, Bakr S, Szabolcs M, D’Agati V, Liu R, Homma S, Schmidt AM, Yan SF, Ramasamy R (2008) RAGE modulates myocardial injury consequent to LAD infarction via impact on JNK and STAT signaling in a murine model. Am J Physiol Heart Circ Physiol 294:H1823–H1832. doi:10.1152/ajpheart.01210.2007
Andrassy M, Volz HC, Igwe JC, Funke B, Eichberger SN, Kaya Z, Buss S, Autschbach F, Pleger ST, Lukic IK, Bea F, Hardt SE, Humpert PM, Bianchi ME, Mairbaurl H, Nawroth PP, Remppis A, Katus HA, Bierhaus A (2008) High-mobility group box-1 in ischemia–reperfusion injury of the heart. Circulation 117:3216–3226. doi:10.1161/CIRCULATIONAHA.108.769331
Appleyard RF, Cohn LH (1993) Myocardial stunning and reperfusion injury in cardiac surgery. J Card Surg 8:316–324
Bagai A, Dangas GD, Stone GW, Granger CB (2014) Reperfusion strategies in acute coronary syndromes. Circ Res 114:1918–1928. doi:10.1161/CIRCRESAHA.114.302744
Bainey KR, Armstrong PW (2014) Clinical perspectives on reperfusion injury in acute myocardial infarction. Am Heart J 167:637–645. doi:10.1016/j.ahj.2014.01.015
Berthelot F, Fattoum L, Casulli S, Gozlan J, Marechal V, Elbim C (2012) The effect of HMGB1, a damage-associated molecular pattern molecule, on polymorphonuclear neutrophil migration depends on its concentration. J Innate Immun 4:41–58. doi:10.1159/000328798
Bucciarelli LG, Kaneko M, Ananthakrishnan R, Harja E, Lee LK, Hwang YC, Lerner S, Bakr S, Li Q, Lu Y, Song F, Qu W, Gomez T, Zou YS, Yan SF, Schmidt AM, Ramasamy R (2006) Receptor for advanced-glycation end products: key modulator of myocardial ischemic injury. Circulation 113:1226–1234. doi:10.1161/CIRCULATIONAHA.105.575993
Cannon CP, Gibson CM, Lambrew CT, Shoultz DA, Levy D, French WJ, Gore JM, Weaver WD, Rogers WJ, Tiefenbrunn AJ (2000) Relationship of symptom-onset-to-balloon time and door-to-balloon time with mortality in patients undergoing angioplasty for acute myocardial infarction. JAMA 283:2941–2947
Daley JM, Thomay AA, Connolly MD, Reichner JS, Albina JE (2008) Use of Ly6G-specific monoclonal antibody to deplete neutrophils in mice. J Leukoc Biol 83:64–70. doi:10.1189/jlb.0407247
Emami H, Singh P, MacNabb M, Vucic E, Lavender Z, Rudd JH, Fayad ZA, Lehrer-Graiwer J, Korsgren M, Figueroa AL, Fredrickson J, Rubin B, Hoffmann U, Truong QA, Min JK, Baruch A, Nasir K, Nahrendorf M, Tawakol A (2015) Splenic metabolic activity predicts risk of future cardiovascular events: demonstration of a cardiosplenic axis in humans. JACC Cardiovasc Imaging 8:121–130. doi:10.1016/j.jcmg.2014.10.009
Frenkel D, Pachori AS, Zhang L, Dembinsky-Vaknin A, Farfara D, Petrovic-Stojkovic S, Dzau VJ, Weiner HL (2009) Nasal vaccination with troponin reduces troponin specific T-cell responses and improves heart function in myocardial ischemia–reperfusion injury. JACC Cardiovasc Imaging 21:817–829. doi:10.1093/intimm/dxp051
Ge L, Zhou X, Ji WJ, Lu RY, Zhang Y, Zhang YD, Ma YQ, Zhao JH, Li YM (2015) Neutrophil extracellular traps in ischemia–reperfusion injury-induced myocardial no-reflow: therapeutic potential of DNase-based reperfusion strategy. Am J Physiol Heart Circ Physiol 308:H500–H509. doi:10.1152/ajpheart.00381.2014
Gigliotti JC, Huang L, Ye H, Bajwa A, Chattrabhuti K, Lee S, Klibanov AL, Kalantari K, Rosin DL, Okusa MD (2013) Ultrasound prevents renal ischemia–reperfusion injury by stimulating the splenic cholinergic anti-inflammatory pathway. J Am Soc Nephrol 24:1451–1460. doi:10.1681/ASN.2013010084
Heusch G (2015) Molecular basis of cardioprotection: signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res 116:674–699. doi:10.1161/CIRCRESAHA.116.305348
Heusch G, Botker HE, Przyklenk K, Redington A, Yellon D (2015) Remote ischemic conditioning. J Am Coll Cardiol 65:177–195. doi:10.1016/j.jacc.2014.10.031
Hiroi T, Wajima T, Negoro T, Ishii M, Nakano Y, Kiuchi Y, Mori Y, Shimizu S (2013) Neutrophil TRPM2 channels are implicated in the exacerbation of myocardial ischaemia/reperfusion injury. Cardiovasc Res 97:271–281. doi:10.1093/cvr/cvs332
Homma T, Kinugawa S, Takahashi M, Sobirin MA, Saito A, Fukushima A, Suga T, Takada S, Kadoguchi T, Masaki Y, Furihata T, Taniguchi M, Nakayama T, Ishimori N, Iwabuchi K, Tsutsui H (2013) Activation of invariant natural killer T cells by alpha-galactosylceramide ameliorates myocardial ischemia/reperfusion injury in mice. J Mol Cell Cardiol 62:179–188. doi:10.1016/j.yjmcc.2013.06.004
Hu X, Fu W, Jiang H (2012) HMGB1: a potential therapeutic target for myocardial ischemia and reperfusion injury. Int J Cardiol 155:489. doi:10.1016/j.ijcard.2011.12.066
Huebener P, Pradere JP, Hernandez C, Gwak GY, Caviglia JM, Mu X, Loike JD, Jenkins RE, Antoine DJ, Schwabe RF (2014) The HMGB1/RAGE axis triggers neutrophil-mediated injury amplification following necrosis. J Clin Invest 125:539–550. doi:10.1172/JCI76887
Ibanez B, Heusch G, Ovize M, Van de Werf F (2015) Evolving therapies for myocardial ischemia/reperfusion injury. J Am Coll Cardiol 65:1454–1471. doi:10.1016/j.jacc.2015.02.032
Ismahil MA, Hamid T, Bansal SS, Patel B, Kingery JR, Prabhu SD (2014) Remodeling of the mononuclear phagocyte network underlies chronic inflammation and disease progression in heart failure: critical importance of the cardiosplenic axis. Circ Res 114:266–282. doi:10.1161/CIRCRESAHA.113.301720
Jiang D, Liang J, Fan J, Yu S, Chen S, Luo Y, Prestwich GD, Mascarenhas MM, Garg HG, Quinn DA, Homer RJ, Goldstein DR, Bucala R, Lee PJ, Medzhitov R, Noble PW (2005) Regulation of lung injury and repair by Toll-like receptors and hyaluronan. Nat Med 11:1173–1179. doi:10.1038/nm1315
Jin C, Cleveland JC, Ao L, Li J, Zeng Q, Fullerton DA, Meng X (2014) Human myocardium releases heat shock protein 27 (HSP27) after global ischemia: the proinflammatory effect of extracellular HSP27 through toll-like receptor (TLR)-2 and TLR4. Mol Med 20:280–289. doi:10.2119/molmed.2014.00058
Kain V, Prabhu SD, Halade GV (2014) Inflammation revisited: inflammation versus resolution of inflammation following myocardial infarction. Basic Res Cardiol 109:444. doi:10.1007/s00395-014-0444-7
Kikuchi K, Tancharoen S, Ito T, Morimoto-Yamashita Y, Miura N, Kawahara K, Maruyama I, Murai Y, Tanaka E (2013) Potential of the angiotensin receptor blockers (ARBs) telmisartan, irbesartan, and candesartan for inhibiting the HMGB1/RAGE axis in prevention and acute treatment of stroke. Int J Mol Sci 14:18899–18924. doi:10.3390/ijms140918899
Leuschner F, Panizzi P, Chico-Calero I, Lee WW, Ueno T, Cortez-Retamozo V, Waterman P, Gorbatov R, Marinelli B, Iwamoto Y, Chudnovskiy A, Figueiredo JL, Sosnovik DE, Pittet MJ, Swirski FK, Weissleder R, Nahrendorf M (2010) Angiotensin-converting enzyme inhibition prevents the release of monocytes from their splenic reservoir in mice with myocardial infarction. Circ Res 107:1364–1373. doi:10.1161/CIRCRESAHA.110.227454
Li Y, Si R, Feng Y, Chen HH, Zou L, Wang E, Zhang M, Warren HS, Sosnovik DE, Chao W (2011) Myocardial ischemia activates an injurious innate immune signaling via cardiac heat shock protein 60 and Toll-like receptor 4. J Biol Chem 286:31308–31319. doi:10.1074/jbc.M111.246124
Mandal P, Novotny M, Hamilton TA (2005) Lipopolysaccharide induces formyl peptide receptor 1 gene expression in macrophages and neutrophils via transcriptional and posttranscriptional mechanisms. J Immunol 175:6085–6091
Matzinger P (2002) The danger model: a renewed sense of self. Science 296:301–305. doi:10.1126/science.1071059
McDonald KA, Huang H, Tohme S, Loughran P, Ferrero K, Billiar T, Tsung A (2014) The TLR4 antagonist eritoran tetrasodium attenuates liver ischemia and reperfusion injury through inhibition of HMGB1 signaling. Mol Med. doi:10.2119/molmed.2014.00076
Murphy SL, Xu J, Kochanek KD (2013) Deaths: final data for 2010. Natl Vital Stat Rep 61:1–117
Nahrendorf M, Swirski FK, Aikawa E, Stangenberg L, Wurdinger T, Figueiredo JL, Libby P, Weissleder R, Pittet MJ (2007) The healing myocardium sequentially mobilizes two monocyte subsets with divergent and complementary functions. J Exp Med 204:3037–3047. doi:10.1084/jem.20070885
Orlova VV, Choi EY, Xie C, Chavakis E, Bierhaus A, Ihanus E, Ballantyne CM, Gahmberg CG, Bianchi ME, Nawroth PP, Chavakis T (2007) A novel pathway of HMGB1-mediated inflammatory cell recruitment that requires Mac-1-integrin. EMBO J 26:1129–1139. doi:10.1038/sj.emboj.7601552
Panaro MA, Acquafredda A, Sisto M, Lisi S, Maffione AB, Mitolo V (2006) Biological role of the N-formyl peptide receptors. Immunopharmacol Immunotoxicol 28:103–127. doi:10.1080/08923970600625975
Rohde D, Schon C, Boerries M, Didrihsone I, Ritterhoff J, Kubatzky KF, Volkers M, Herzog N, Mahler M, Tsoporis JN, Parker TG, Linke B, Giannitsis E, Gao E, Peppel K, Katus HA, Most P (2014) S100A1 is released from ischemic cardiomyocytes and signals myocardial damage via Toll-like receptor 4. EMBO Mol Med 6:778–794. doi:10.15252/emmm.201303498
Serebruany VL, Solomon SR, Herzog WR, Gurbel PA (1998) Plasma fibronectin during myocardial ischemia–reperfusion: effects of magnesium, diltiazem, and a novel Mac-1 inhibitor. Am J Hematol 57:309–314
Shang L, Ananthakrishnan R, Li Q, Quadri N, Abdillahi M, Zhu Z, Qu W, Rosario R, Toure F, Yan SF, Schmidt AM, Ramasamy R (2010) RAGE modulates hypoxia/reoxygenation injury in adult murine cardiomyocytes via JNK and GSK-3beta signaling pathways. PLoS One 5:e10092. doi:10.1371/journal.pone.0010092
Sharma AK, LaPar DJ, Stone ML, Zhao Y, Kron IL, Laubach VE (2013) Receptor for advanced glycation end products (RAGE) on iNKT cells mediates lung ischemia–reperfusion injury. Am J Transplant 13:2255–2267. doi:10.1111/ajt.12368
Swirski FK, Nahrendorf M, Etzrodt M, Wildgruber M, Cortez-Retamozo V, Panizzi P, Figueiredo JL, Kohler RH, Chudnovskiy A, Waterman P, Aikawa E, Mempel TR, Libby P, Weissleder R, Pittet MJ (2009) Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science 325:612–616. doi:10.1126/science.1175202
Tian Y, French BA, Kron IL, Yang Z (2015) Splenic leukocytes mediate the hyperglycemic exacerbation of myocardial infarct size in mice. Basic Res Cardiol 110:39. doi:10.1007/s00395-015-0496-3
Tian Y, Gigliotti JC, Wu D, Klibanov AL, Kron IL, Yang Z (2015) Abstract 12972: ultrasound of the spleen reduces myocardial ischemia–reperfusion injury by stimulating a splenic anti-inflammatory pathway. Circulation 132:A12972
Tian Y, Zhang W, Xia D, Modi P, Liang D, Wei M (2011) Postconditioning inhibits myocardial apoptosis during prolonged reperfusion via a JAK2-STAT3-Bcl-2 pathway. J Biomed Sci 18:53. doi:10.1186/1423-0127-18-53
van der Laan AM, Ter Horst EN, Delewi R, Begieneman MP, Krijnen PA, Hirsch A, Lavaei M, Nahrendorf M, Horrevoets AJ, Niessen HW, Piek JJ (2014) Monocyte subset accumulation in the human heart following acute myocardial infarction and the role of the spleen as monocyte reservoir. Eur Heart J 35:376–385. doi:10.1093/eurheartj/eht331
Wang H, Bloom O, Zhang M, Vishnubhakat JM, Ombrellino M, Che J, Frazier A, Yang H, Ivanova S, Borovikova L, Manogue KR, Faist E, Abraham E, Andersson J, Andersson U, Molina PE, Abumrad NN, Sama A, Tracey KJ (1999) HMG-1 as a late mediator of endotoxin lethality in mice. Science 285:248–251
Wu H, Ma J, Wang P, Corpuz TM, Panchapakesan U, Wyburn KR, Chadban SJ (2010) HMGB1 contributes to kidney ischemia reperfusion injury. J Am Soc Nephrol 21:1878–1890. doi:10.1681/ASN.2009101048
Wystrychowski W, Filipczyk L, Cierpka L, Obuchowicz E, Wiecek A, Wystrychowski A (2014) Splenectomy attenuates the course of kidney ischemia–reperfusion injury in rats. Transplant Proc 46:2558–2561. doi:10.1016/j.transproceed.2014.09.056
Xiao L, Zhang Y, Berr SS, Chordia MD, Pramoonjago P, Pu L, Pan D (2012) A novel near-infrared fluorescence imaging probe for in vivo neutrophil tracking. Mol Imaging 11:372–382
Xu H, Yao Y, Su Z, Yang Y, Kao R, Martin CM, Rui T (2011) Endogenous HMGB1 contributes to ischemia–reperfusion-induced myocardial apoptosis by potentiating the effect of TNF-α/JNK. Am J Physiol Heart Circ Physiol 300:H913–H921. doi:10.1152/ajpheart.00703.2010
Xu Y, Huo Y, Toufektsian MC, Ramos SI, Ma Y, Tejani AD, French BA, Yang Z (2006) Activated platelets contribute importantly to myocardial reperfusion injury. Am J Physiol Heart Circ Physiol 290:H692–H699. doi:10.1152/ajpheart.00634.2005
Yang H, Antoine DJ, Andersson U, Tracey KJ (2013) The many faces of HMGB1: molecular structure-functional activity in inflammation, apoptosis, and chemotaxis. J Leukoc Biol 93:865–873. doi:10.1189/jlb.1212662
Yang XM, Cui L, White J, Kuck J, Ruchko MV, Wilson GL, Alexeyev M, Gillespie MN, Downey JM, Cohen MV (2015) Mitochondrially targeted Endonuclease III has a powerful anti-infarct effect in an in vivo rat model of myocardial ischemia/reperfusion. Basic Res Cardiol 110:3. doi:10.1007/s00395-014-0459-0
Yang Z, Day YJ, Toufektsian MC, Ramos SI, Marshall M, Wang XQ, French BA, Linden J (2005) Infarct-sparing effect of A2A-adenosine receptor activation is due primarily to its action on lymphocytes. Circulation 111:2190–2197. doi:10.1161/01.CIR.0000163586.62253.A5
Yang Z, Day YJ, Toufektsian MC, Xu Y, Ramos SI, Marshall MA, French BA, Linden J (2006) Myocardial infarct-sparing effect of adenosine A2A receptor activation is due to its action on CD4+ T lymphocytes. Circulation 114:2056–2064. doi:10.1161/CIRCULATIONAHA.106.649244
Yang Z, Linden J, Berr SS, Kron IL, Beller GA, French BA (2008) Timing of adenosine 2A receptor stimulation relative to reperfusion has differential effects on infarct size and cardiac function as assessed in mice by MRI. Am J Physiol Heart Circ Physiol 295:H2328–H2335. doi:10.1152/ajpheart.00091.2008
Yang Z, Tian Y, Liu Y, Hennessy S, Kron IL, French BA (2013) Acute hyperglycemia abolishes ischemic preconditioning by inhibiting Akt phosphorylation: normalizing blood glucose before ischemia restores ischemic preconditioning. Oxid Med Cell Longev 2013:329183. doi:10.1155/2013/329183
Zhang BJ, Men XJ, Lu ZQ, Li HY, Qiu W, Hu XQ (2013) Splenectomy protects experimental rats from cerebral damage after stroke due to anti-inflammatory effects. Chin Med J (Engl) 126:2354–2360
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding sources
This study was funded in part by a University of Virginia School of Medicine Collaborative Science Pilot Grant and an NIH R01 HL130082 to ZY and a National Natural Science Foundation of China Grant (81400213) to YT.
Conflict of interest
None.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tian, Y., Pan, D., Chordia, M.D. et al. The spleen contributes importantly to myocardial infarct exacerbation during post-ischemic reperfusion in mice via signaling between cardiac HMGB1 and splenic RAGE. Basic Res Cardiol 111, 62 (2016). https://doi.org/10.1007/s00395-016-0583-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-016-0583-0