Abstract
Purpose
Whether beverage quality affects changes in glycaemic traits and type 2 diabetes (T2D) risk is unknown. We examined associations of a previously developed Healthy Beverage Index (HBI) with insulin resistance, and risk of prediabetes and T2D.
Methods
We included 6769 participants (59% female, 62.0 ± 7.8 years) from the Rotterdam Study cohort free of diabetes at baseline. Diet was assessed using food-frequency questionnaires at baseline. The HBI included 10 components (energy from beverages, meeting fluid requirements, water, coffee and tea, low-fat milk, diet drinks, juices, alcohol, full-fat milk, and sugar-sweetened beverages), with a total score ranging from 0 to 100. A higher score represents a healthier beverage pattern. Data on study outcomes were available from 1993 to 2015. Multivariable linear mixed models and Cox proportional-hazards regression models were used to examine associations of the HBI (per 10 points increment) with two measurements of HOMA-IR (a proxy for insulin resistance), and risk of prediabetes and T2D.
Results
During follow-up, we documented 1139 prediabetes and 784 T2D cases. Mean ± SD of the HBI was 66.8 ± 14.4. Higher HBI score was not associated with HOMA-IR (β: 0.003; 95% CI − 0.007, 0.014), or with risk of prediabetes (HR: 1.01; 95% CI 0.97, 1.06), or T2D (HR: 1.01; 95% CI 0.96, 1.07).
Conclusion
Our findings suggest no major role for overall beverage intake quality assessed with the HBI in insulin resistance, prediabetes and T2D incidence. The HBI may not be an adequate tool to assess beverage intake quality in our population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diet constitutes an important modifiable lifestyle factor in the prevention and management of type 2 diabetes (T2D) [1]. Besides, beverages constitute an important component of the diet because they contribute to fulfill our daily fluid requirements, and they can contribute to the overall nutrient and energy intakes [2,3,4]. Commonly consumed beverages are water, coffee, tea, milk, fruit juices, sugar-sweetened beverages (SSBs), and alcoholic beverages [3].
Given the diverse nutritional composition of beverages, they have been differentially associated with cardiometabolic outcomes. SSBs are major contributors to energy and added sugars to the diet and have been consistently associated to higher risk of obesity, T2D, and other cardiometabolic diseases [5,6,7]. In line with this, replacing SSB with water has been associated with a lower risk of these diseases [8,9,10]. Fruit juices may contain as much sugar and calories as SSBs [11, 12], and have also been associated with increased risk of T2D [5] and weight gain [12]. Recent evidence has suggested that long-term consumption of artificially sweetened beverages may also be associated to increased risk of T2D [5, 13]. Both, coffee and tea have been related to lower risk of T2D and other cardiometabolic diseases [14,15,16,17,18,19]. Recent scientific evidence has shown neutral to modest inverse associations for low-fat milk consumption with T2D risk [20, 21], whereas whole-fat milk may lead to higher cardiometabolic risk because of increased risk of weight gain [22, 23]. Finally, low and moderate alcohol consumption has been associated to lower risk of T2D, but also increased risk of stroke, coronary artery disease (except MI) and hypertension [24, 25].
Although several beverage groups seem to influence cardiometabolic risk, to date few studies have investigated overall beverage intake quality and cardiometabolic outcomes, by using a priori developed scoring systems [26,27,28]. The healthy beverage index (HBI) developed by Duffey and Davy [26], is a scoring system that includes scores for recommended intakes of various beverage groups, total fluid consumption, and energy from beverages to evaluate overall beverage intake quality. A higher score in the HBI representing a healthier beverage intake pattern was cross-sectionally associated with lower cardiometabolic risk in a representative sample of the US population, as reflected by lower odds ratios of having hypertension, high fasting insulin levels, and high low-density lipoprotein cholesterol level. However, the longitudinal association between overall beverage intake quality and T2D risk has not been explored. Besides, for public health advice, it is of interest to know if a healthier beverage intake pattern may also influence T2D since early stages (insulin resistance and prediabetes). Therefore, we aimed to investigate the association of HBI score with insulin resistance, prediabetes risk and T2D risk in an adult Dutch population.
Methods
Study design and population
This study was carried-out in three sub-cohorts of the Rotterdam Study (RS). The RS is an ongoing prospective, population-based cohort study on risk factors for chronic diseases in mid and late life. Its design has been described elsewhere [29]. Briefly, the first sub-cohort (RS-I) started in 1990–1993, with the inclusion of 7983 inhabitants of the Ommoord district in the Dutch city of Rotterdam, that were 55 years or over. In 2000–2001, the second sub-cohort (RS-II) was initiated with the inclusion of 3011 new subjects who became 55 years old or moved to the study district. In 2006, the third sub-cohort (RS-III) was initiated with the inclusion of 3932 individuals aged 45 years or over. By the end of 2008, the Rotterdam Study comprised 14,926 subjects aged 45 years or over. At baseline, participants were interviewed at home and then extensively examined in a study research center. Follow-up examinations were performed every 3–5 years in each sub-cohort.
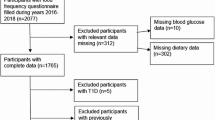
For the current study, baseline data from sub-cohorts RS-I, RS-II, and RS-III were used (n = 14,926). We excluded participants with missing data on diet (n = 5176), implausible estimated daily energy intake (cut-offs set at < 500 or > 5000 kcal/day, as previously done in the RS; n = 50), prevalent T2D at baseline (n = 963), or missing data on prevalent T2D (n = 1968), resulting in 6769 participants. For the analysis on insulin resistance, of the 6769 participants, we additionally excluded n = 2873 subjects who did not have at least two measurements of HOMA-IR, one at baseline and one at follow-up, resulting in 3896 participants. For the analysis on pre-diabetes risk, from 6769 participants, we additionally excluded subjects with prediabetes at baseline or without follow-up data on prediabetes (n = 717), resulting in 6052 participants. Finally, for the analysis of T2D, from the sub-sample of 6769, we additionally excluded subjects with missing data on follow-up T2D (n = 51), resulting in 6718 participants (Supplementary Fig. 1).
The Rotterdam Study has been approved by the Medical Ethics Committee of the Erasmus MC (registration number MEC 02.1015) and by the Dutch Ministry of Health, Welfare and Sport (Population Screening Act WBO, license number 1071272-159521-PG). The Rotterdam Study Personal Registration Data collection is filed with the Erasmus MC Data Protection Officer under registration number EMC1712001. The Rotterdam Study has been entered into the Netherlands National Trial Register (NTR; www.trialregister.nl) and into the World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP; www.who.int/ictrp/network/primary/en/) under shared catalogue number NTR6831. All subjects gave written informed consent, to participate in the study and to have their information obtained from treating physicians.
Assessment of dietary intake
At baseline, dietary intake data were collected using validated semi-quantitative food-frequency questionnaires (FFQs). A 170-item FFQ was used for sub-cohorts RS-I and RS-II, and a 389-item FFQ was used for RS-III. The preceding year served as the reference period for RS-I and RS-II, and the last month for RS-III. Details are provided elsewhere [30]. Both FFQs were previously validated for nutrient intakes against other dietary assessment methods, and demonstrated to adequately rank participants according to their intake [30,31,32]. Food and beverage intake data were converted to nutrient and energy data through linkage with the Dutch Food-Composition Table corresponding to the year the dietary evaluation was performed (1993 for RS-I, 2001 for RS-II and 2011 for RS-III).
Calculation of HBI components and scores
Based on the HBI developed by Duffey and Davy [26], 10 components were created (Table 1). Briefly, overall daily beverage intake was grouped according to the US Beverage Guidance System [2] into eight groups (components): water, coffee and tea, low-fat and skimmed milk (< 2.0% fat), diet beverages (non-calorically sweetened beverages, except for RS-I), fruit and vegetable juices, alcoholic beverages (including beer, wine and liquors), whole-fat milk (≥ 2.0% fat), and SSBs (Supplementary Table 1). Beverage intake was converted from mL/day to percentage of intake according to total fluid requirements, except for alcoholic beverages, which were assessed as glasses of alcohol consumed per day. In the Netherlands, a standard glass of alcohol contains 10 g of ethanol. Two additional components were included, total energy from beverages and meeting total fluid requirements defined as 1 mL of fluid per 1 kcal of food consumed [26].
Total score on the HBI is the sum of the individual component scores, and it ranges from 0 to 100, with a higher score indicating better adherence to beverage guidelines and, thus, a healthier beverage intake pattern. For most components, participants received the maximum score per component if they adhered to the recommended intakes, otherwise they received 0 points (dichotomous scoring system) [26]. Only the components water, energy from beverages and meeting total fluid requirements received proportional points according to reported intake (continuous scoring system). The maximum score possible was higher for those components representing aspects of beverage intake that are more critical to health, such as water and SSBs (15 points), and the additional components energy from beverages and meeting total fluid requirements (20 points).
Assessment of insulin resistance
Homeostatic model assessment of insulin resistance (HOMA-IR) was calculated using the formula: \(fasting insulin \left( \frac{mU}{L} \right)x fasting glucose \left( \frac{mmol}{L} \right)\frac{{}}{{}}22.5\). Fasting blood was drawn at the research center at two examination visits in each sub-cohort: at RS-I-3 (1997–99) and RS-I-5 (2009–11), at RS-II-1 (2000–01) and RS-II-3 (2011–12), and at RS-III-1 (2006–08) and RS-III-2 (2012–14). Because no fasting blood samples were collected in the first two visits of RS-I, we set the third visit (RS-I-3) as the baseline. Glucose levels were measured using the glucose hexokinase method. Serum insulin levels were measured by radioimmunoassay (Medgenix diagnostics, Brussels, Belgium) on a Roche Modular Analytics E170 analyzer.
Assessment of prediabetes and type 2 diabetes
Prediabetes and T2D cases at baseline and during follow-up examinations were ascertained based on general practitioners’ records, hospital discharge letters, and blood glucose measurements. Prediabetes and T2D were defined according to the WHO guidelines [33]. Diabetes was defined as fasting plasma glucose concentration ≥ 7.0 mmol/L, non-fasting plasma glucose concentration ≥ 11.1 mmol/L when fasting glucose was not available, or use of blood glucose-lowering medication (ATC code A010) [34, 35]. Information about blood glucose-lowering medication use was obtained from structured home interviews and pharmacy dispensing records. Prediabetes was defined as having fasting plasma glucose > 6.0 and < 7.0 mmol/L or non-fasting plasma glucose > 7.7 and < 11.1 mmol/L. Two study physicians independently identified all potential incident prediabetes and T2D cases. In case of disagreement, a consensus was sought by consulting diabetologists [36]. Data of incident prediabetes and T2D was collected until 01-01-2015.
Assessment of covariates
At baseline and during home interviews, trained research assistants obtained information on health status, smoking behaviour, and socioeconomic status. Self-reported smoking status was classified into three categories (never, former, or current). Educational attainment was self-reported and classified into: primary education with or without a partially completed higher education (primary); lower vocational or lower secondary education (lower); intermediate vocational education and or general secondary (intermediate); or higher vocational or university education (higher). Physical activity was assessed with an adapted version of the Zutphen Physical Activity Questionnaire at RS-I-3 and RS-II-1 [37], and with the LASA Physical Activity Questionnaire at RS-III-1 [38]. Physical activities were weighted according to the intensity with Metabolic Equivalent of Task (MET), from the Compendium of Physical Activities version 2011. To account for differences between the two questionnaires, questionnaire-specific z-scores of MET-hours per week were calculated. Overall diet quality was assessed with adherence (yes/no) to 14 items of the Dutch dietary guidelines 2015: vegetables, fruit, whole-grains, legumes, nuts, dairy, fish, tea, ratio whole-grains:total grains, ratio unsaturated fats and oils:total fats, red and processed meat, sugar-containing beverages, alcohol and salt, with a total score ranging from 0 to 14 [30]. Information on medical history and medication was obtained from interviews, medical records and pharmacy records.
Physical examinations were held at the research center. Height (meters, m) and weight (kilograms, kg) were measured while wearing indoor clothes without shoes. Body mass index (BMI) was calculated as weight divided by height squared (kg/m2), and then categorized into normal weight, overweight and obese according to the WHO standards [39].
Statistical analysis
Descriptive data were presented as mean ± SD for continuous variables with a normal distribution, as median (IQR) for continuous variables with a skewed distribution, or in absolute and relative numbers (percentages) for categorical variables. Median (IQR) scores of the 10 components of the HBI are presented across tertiles of total HBI score to identify which components differentiated healthier from less healthy HBI scores (Supplementary Table 2).
We used linear mixed-effect models (LMM) with a random-effects structure including a random intercept and slope (for participants and time of repeated measurements, respectively) to examine associations between the HBI score (per 10 points increment) and HOMA-IR over time in participants for whom at least two measurements of HOMA-IR were available. HOMA-IR values were natural log-transformed to improve distribution of the residuals (Supplementary Figs. 2 and 3), and results were presented as regression coefficients (β) with their corresponding 95% confidence intervals (CI). We used multivariable Cox proportional hazard models to analyse associations between the HBI score (per 10 points increment) and incident prediabetes and T2D. The proportional hazards assumption was confirmed visually using a log-minus-log plot of the survival time. Person-years were calculated from the time of entering the Rotterdam Study to date of prediabetes or T2D incidence, death, withdrawal from the study or end of follow-up (January 1, 2015), whichever came first. Results were presented as hazard ratios (HR) with their corresponding 95% CI. Main analyses were performed for the overall study population (pooled data of the three RS sub-cohorts). Results by sub-cohort are shown in supplementary materials. Non-linearity of the associations was explored using natural cubic splines (degrees of freedom = 3). As no indications for non-linear associations for the main models were found (p ≥ 0.05, Supplementary Figs. 4 and 5), all primary analyses were performed assuming linearity. Multivariable models included potential confounders and established risk factors for the outcomes. Model 1 was adjusted for baseline age (years), sex, total energy intake (kcal/day), Rotterdam Study sub-cohort (only for overall study population analyses), and time of repeated measurements of HOMA-IR (only for HOMA-IR analyses). Model 2 was additionally adjusted for smoking status, educational attainment, physical activity level and diet quality score. Finally, model 3 was additionally adjusted for baseline BMI (kg/m2) as a potential confounder in the associations. Effect modification was examined by including interactions of the HBI score with age, sex, and BMI (continuous and in categories) for all outcomes in model 3.
Additionally, we examined associations between individual components of the HBI and the study outcomes, by treating the component scores as dichotomous variables (adherence vs. non-adherence or partial adherence to the component criteria). We used model 3 additionally adjusting for all other components of the HBI, except for water analyses, which were not additionally adjusted for meeting total fluid requirements because of collinearity issues. Sub-cohort RS-I was excluded from the analyses of diet beverages due to lacking data on diet beverages intake in that sub-cohort.
We performed several sensitivity analyses based on model 3 to examine the robustness of our findings. First, we repeated our main analyses using different variations of the HBI. We excluded scores of the additional components ‘total energy from beverages’ and ‘meeting total fluid requirements’ from the total HBI score (alternative score 1), to see the effect of these components in our associations, because they were the most heavily weighted, additionally adjusting for the excluded components. Second, we repeated main analyses additionally excluding water component score from alternative score 1, because of limited detailed data about water consumption (alternative score 2) and additionally adjusting for the excluded components. Third, to test if the associations were driven by specific components of the HBI, we repeated main analyses by excluding each of the 10 components from the total HBI score one at a time, and adjusting for the excluded component. Fourth, we repeated main analyses assessing the HBI score in tertiles. Fifth, we excluded participants with prevalent cardiovascular diseases (CVDs) at baseline because they are closely interrelated with glucose/metabolic traits and diet. Last, to address possible reverse causation, we excluded participants that developed prediabetes and T2D within the first 3 years of follow-up.
Missing values in covariates (ranging from 0.2 to 3.9%) were imputed with tenfold multiple imputation with chained equations. The results shown correspond to the pooled results of the 10 imputed datasets. Total score in the HBI and individual components scores were calculated using SPSS statistical software, version 21.0 (IBM Corp, Armonk, NY). All other statistical analyses were performed using R version 3.3.1 (The R Foundation for Statistical Computing, Vienna, Austria). A two-sided p-value < 0.05 was considered statistically significant.
Results
Baseline characteristics
Baseline characteristics in the overall study population are shown in Table 2 and by RS sub-cohort in Supplementary Table 3. In our population of 6769 participants, the mean ± SD age was 62.0 ± 7.8 years and 59% were female. The three most consumed beverage groups were [median (IQR) in consumers]: coffee and tea [750 (550, 1000) mL/day], water [349 (175, 611) mL/day] and low-fat milk [224 (139, 352) mL/day]. Total score on the HBI (with a theoretical range from 0 to 100) ranged from 22.4 to 100, with a mean ± SD of 66.8 ± 14.4 (Table 2). The three components to which participants adhered the most to recommended intakes were diet drinks (98% adherence), SSBs (93% adherence), and fruit and vegetable juices (87% adherence) (Supplementary Table 4).
Healthy beverage index score and insulin resistance (HOMA-IR), risk of prediabetes and T2D
During 68,154 person-years of follow-up among 6052 participants (median follow-up 10 years), 1139 participants developed prediabetes (18.8%), and during 78,606 person-years of follow-up among 6718 participants (median follow-up 11 years), 784 participants developed T2D (11.7%). After multivariable adjustment (model 3), higher HBI score (per 10 points increment) was not associated with log-HOMA-IR levels (β: 0.003; 95% CI − 0.007, 0.014), nor with risk of prediabetes (HR: 1.01; 95% CI 0.97, 1.06), or T2D (HR: 1.01; 95% CI 0.96, 1.07) (Table 3). Similar results were obtained when stratifying the analyses by sub-cohort (Supplementary Tables 5 through 7). Associations did not differ by age, sex or BMI at baseline (all p-values for interaction > 0.05).
Individual component scores and insulin resistance (HOMA-IR), risk of prediabetes and T2D
When examining the individual components of the HBI, we observed that after multivariable adjustment (model 3), meeting the criteria for getting the maximum score on coffee and tea (intake ≤ 40% of total fluid requirements, Table 1) and alcohol (intake ≤ 2 glasses/day for men and ≤ 1 glass/day for women) was associated with increased log-HOMA-IR (β: 0.044; 95% CI 0.009, 0.080; and β: 0.061; 95% CI 0.028, 0.094, respectively). In contrast, meeting the HBI criteria for getting the maximum score on low-fat milk (intake ≤ 16% of total fluid requirements) and SSBs (intake ≤ 8% of total fluid requirements) was associated with lower log-HOMA-IR (β: − 0.054; 95% CI − 0.091, − 0.016; and β: − 0.064; 95% CI − 0.127, − 0.002, respectively). Meeting the criteria for coffee and tea intake was also associated with higher risk of T2D (HR: 1.27; 95% CI 1.07, 1.49). No associations were found for meeting the criteria on any of the individual components and prediabetes risk (Table 4).
Sensitivity analyses
Alternative HBI scores 1 (leaving out scores from the components total energy from beverages and meeting total fluid requirements) and 2 (alternative score 1 additionally excluding scores from water), were also not associated with the study outcomes (Supplementary Table 8). The exclusion of each one of the 10 individual components from the total HBI score one by one at a time did not substantially change the estimates for all study outcomes, except for energy from beverages and prediabetes outcome. Excluding energy from beverages from the overall HBI was associated with higher risk of prediabetes (HR: 1.08; 95% CI 1.02, 1.15) (Supplementary Table 9). Also, no associations with any of the study outcomes were found when repeating main analyses using tertiles of the HBI score (higher vs. lower tertile) (Supplementary Table 10). Likewise, no significant associations were found when excluding participants with CVDs at baseline (Supplementary table 11). Finally, similar effect estimates were found when excluding participants that developed prediabetes and T2D within the first three years of follow-up compared to those obtained for the complete study populations for prediabetes and T2D analyses (Supplementary Table 12).
Discussion
In summary, this study shows that overall beverage intake quality as reflected with total score in the Healthy Beverage Index (HBI) was not associated with insulin resistance (HOMA-IR) over time, nor with the incidence of prediabetes or T2D in our population of Dutch middle-aged and elderly people. Results from individual components showing differential associations with the study outcomes need to be interpreted with caution. Scores on individual components represent adherence to the components criteria and not actual beverage intake.
Our findings are partially in line with a previous study on the HBI and cardiometabolic health outcomes in US adults. A previous cross-sectional study that used data from the National Health and Nutrition Examination Survey (NHANES) 2005–2010 (n = 16,252 US adults), showed that higher HBI scores were associated with lower odds of high fasting insulin levels, but no association was observed with the odds of high fasting glucose levels [26]. Contrarily, in the current study we did not find any association between higher HBI scores and high fasting insulin levels in an exploratory analysis (Supplementary Table 13). Furthermore, we found no association between higher HBI scores and high glucose levels (Supplementary Table 13), nor with insulin resistance, risk of prediabetes, or T2D. In another European cohort the HBI was also computed, showing considerably higher scores compared to NHANES and our study (median: 89.7, IQR: 78.6–95). However, an association with cardiometabolic outcomes was also not found in that cohort [27]. Differences between the US and the current study might be partially explained by methodological differences such as in the study design. Our study, with a longitudinal design, had available data on repeated measurements over time of HOMA-IR as well as incidence of prediabetes and T2D, allowing us to make temporal inferences. Contrarily, the aforementioned study had a cross-sectional design and did not have data on repeated assessment of HOMA-IR over time or incident prediabetes or T2D. In addition, the use of different dietary assessment methods between the studies (average of two 24 h recalls vs. FFQs) might have led to differential misclassification of the exposure while assessing habitual beverage intake thought FFQs. However, mean ± SD HBI score was similar between the two populations (NHANES: 63 ± 13 vs. Rotterdam Study: 66.8 ± 14.4) [26].
Differences in findings between studies could also be explained by dissimilarities in beverage intake patterns, and how the HBI scoring system performs under different beverage intake patterns. Although the HBI was developed based on intake patterns and beverage recommendations for US adults, our study revealed contrasting findings in a population of Dutch middle-aged and elderly individuals. While SSBs consumption is relatively high among US adults [40], with energy from beverages accounting for up to 21% of their total energy intake [4], we observed that SSBs intake was comparatively low in our population. Instead, coffee and tea intakes were relatively high, and energy from beverages constituted approximately 10% of their total energy intake.
Concerns on whether the HBI therefore adequately reflects a healthy beverage intake pattern in our study population arise. We found a low correlation between a diet quality index score that reflects adherence to the Dutch dietary guidelines 2015 and the HBI score, which reflects adherence to healthy beverage intake recommendations (rho = 0.21, p < 0.001), while previous studies suggest that healthy beverage patterns are associated with healthy dietary patterns [41]. This may indicate that Dutch and American dietary guidelines have different healthy beverage patterns. Finally, measurement error could also have been a cause of the differences between studies. Intake of some beverages like diet beverages in RS-I and water in all sub-cohorts may not have been appropriately estimated because of the nature of the FFQs, which may have affected total scores in the HBI.
Some of our findings for scores on individual components are not directly in line with evidence from previous observational studies on actual beverages intake (e.g., in mL or glasses per day). For example, a high coffee and tea intake that surpasses the maximum limit established for this component (> 40% of total fluid requirements) results in getting the lowest score (0 points). This likely explains why we found an association for higher score on the coffee and tea component (maximum score for intake ≤ 40% of total fluid requirements) with higher HOMA-IR and T2D risk, as a higher score actually corresponds to low consumption of coffee and tea. Coffee and tea were the main contributors to beverage intake in our study (median intake: 750, IQR: 550, 1000), and similar intakes have been reported in other European populations [42, 43]. In observational cohort studies, including the RS cohort [44], higher coffee and tea consumption was consistently associated with lower T2D risk [15, 16, 18]. The associations between the individual components coffee and tea, low-fat milk, alcohol and SSBs with the study outcomes need to be interpreted with caution. We found no significant associations between the HBI score and the study outcomes when excluding individual components from the overall score (one at a time) in sensitivity analyses, in order to explore specific components as potential drivers of the associations. After exclusion of energy from beverages component, we noted a statistically significant association between the HBI score and prediabetes; however, such an association was not observed for HOMA-IR and T2D, and not in line with the main findings for prediabetes, suggesting that it is likely attributable to chance.
The HBI presents concerns for our study population. It was developed based on US intake patterns and may not reflect beverage quality accurately for populations with different intake distributions, like our Dutch participants. The scoring system's limited differentiation between healthier and less healthy beverage patterns poses challenges. Measuring total water intake is methodologically complex, impacting the accuracy of the HBI. The index places greater weight on certain components, such as low SSBs intake, while neglecting the importance of other beverages like coffee and tea. Determining the significance of beverage groups for general or cardiometabolic health is population-dependent. Previous research suggests that interventions targeting SSBs reduction and increased physical activity impact component scores beyond SSBs intake [45]. In our study, Dutch participants adhered more to recommendations for diet drinks, SSBs, and juices, while US adults adhered to recommendations for coffee and tea, milk, fruit juice, alcohol, and meeting total fluid requirements Considering these concerns, the interpretation of HBI scores should account for diverse populations and contextual factors.
Results from sensitivity analyses confirmed our main findings and showed no associations for alternative HBI scores with the study outcomes. Also, assessing HBI scores in tertiles did not change the main results.
Our study has several strengths. First, to our knowledge, this is the first study investigating longitudinal associations between overall beverage intake quality and our study outcomes for which we had available data of a large European population with repeated measurements of HOMA-IR and a long follow-up for prediabetes and T2D risk (> 10 years). Second, we added to scarce evidence on overall beverage intake quality and cardiometabolic health specifically T2D risk, by assessing associations with early risk stages of the disease, namely HOMA-IR and prediabetes risk in a longitudinal study, which in turn may help to understand how overall beverage intake quality is associated with T2D development. Third, the use of plasma glucose concentrations to assess prediabetes and T2D is another strength of our study. A high proportion of individuals with T2D is undiagnosed, and these cases would have not been detected by self-report. This is relevant for both, baseline (individuals with undiagnosed diabetes would not have been excluded from the analyses) and case ascertainment. Last, we performed a series of additional and sensitivity analyses to comprehensively examine associations between different HBI components and alternative versions of the HBI with the study outcomes, with the purpose of evaluating the HBI.
This study also had some limitations. First, measurement errors were likely to occur, as the intake of some beverages such as diet beverages in RS-I and water in all sub-cohorts may not have been appropriately estimated because of the nature of the FFQs. Although this might have affected the total HBI score, its average was similar to that of US adults in a previous study, and water intake in the Dutch population is generally low (median: 400 g/day) [46]. Second, we used two different FFQs with different numbers of items, however, the FFQs are equally capable of estimating overall beverage intake quality, since sensitivity analyses showed no major differences in beverage intake quality and in the estimates between sub-cohorts with different follow-up periods. Third, although we adjusted for many possible confounders, we cannot completely rule out residual confounding. Finally, our results cannot be generalized to other populations with different age, race and beverage intake patterns, and need further replication in other cohorts.
In conclusion, our findings within a large population-based cohort suggest that overall beverage intake quality as assessed with the healthy beverage index is not associated with insulin resistance (HOMA-IR) over time, nor with prediabetes or T2D risk in middle-age and elderly Dutch. Results of individual HBI components need to be interpreted with caution because of the complex nature of the HBI, which may not reflect true associations between actual beverage intake and disease outcomes. The HBI may not be an optimal scoring system for this population, FFQ data and/or these outcomes. Other means to evaluate overall beverage intake quality should be explored. Future studies should focus on developing country-specific scoring systems that appropriately assess overall beverage intake, and test how changes in individual components affect other components (replacement) and therefore scores and overall quality of beverage intake.
Data availability
Data described in the manuscript, code book, and analytic code will be made available upon request pending application and approval.
Abbreviations
- Β:
-
Beta coefficient
- BMI:
-
Body mass index
- CI:
-
Confidence interval
- FFQs:
-
Food-frequency questionnaires
- HBI:
-
Healthy beverage index
- HOMA-IR:
-
Homeostatic model assessment of insulin resistance
- HR:
-
Hazard ratio
- IQR:
-
Interquartile range
- LMM:
-
Linear mixed models
- MET:
-
Metabolic equivalent of task
- RS:
-
Rotterdam Study
- SD:
-
Standard deviation
- SSBs:
-
Sugar-sweteened beverages
- T2D:
-
Type 2 diabetes
- WHO:
-
World Health Organization
References
Ley SH, Hamdy O, Mohan V, Hu FB (2014) Prevention and management of type 2 diabetes: dietary components and nutritional strategies. Lancet 383:1999–2007. https://doi.org/10.1016/S0140-6736(14)60613-9
Popkin BM, Armstrong LE, Bray GM et al (2006) A new proposed guidance system for beverage consumption in the United States. Am J Clin Nutr 83:529–542. https://doi.org/10.1093/ajcn.83.3.529
Nissensohn M, Sánchez-Villegas A, Galan P et al (2017) Beverage consumption habits among the european population: association with total water and energy intakes. Nutrients 9:1–13. https://doi.org/10.3390/nu9040383
Duffey KJ, Popkin BM (2007) Shifts in patterns and consumption of beverages between 1965 and 2002. Obesity 15:2739–2747. https://doi.org/10.1038/oby.2007.326
Imamura F, O’Connor L, Ye Z et al (2015) Consumption of sugar sweetened beverages, artificially sweetened beverages, and fruit juice and incidence of type 2 diabetes: systematic review, meta-analysis, and estimation of population attributable fraction. BMJ (Online) 351:1–12. https://doi.org/10.1136/bmj.h3576
Malik VS, Hu FB (2019) Sugar-sweetened beverages and cardiometabolic health: an update of the evidence. Nutrients. https://doi.org/10.3390/nu11081840
Yin J, Zhu Y, Malik V, Li X, Peng X, Zhang FF et al (2021) Intake of sugar-sweetened and low-calorie sweetened beverages and risk of cardiovascular disease: a meta-analysis and systematic review. Adv Nutr 12:89–101. https://doi.org/10.1093/advances/nmaa084
Willett WC, Manson JE, Hu FB et al (2012) Plain-water intake and risk of type 2 diabetes in young and middle-aged women. Am J Clin Nutr 95:1454–1460. https://doi.org/10.3945/ajcn.111.032698.1
Duffey KJ, Poti J (2016) Modeling the effect of replacing sugar-sweetened beverage consumption with water on energy intake, HBI score, and obesity prevalence. Nutrients 8:395. https://doi.org/10.3390/nu8070395
Cui R, Iso H, Eshak ES et al (2018) Water intake from foods and beverages and risk of mortality from CVD: the Japan Collaborative Cohort (JACC) Study. Public Health Nutr 21:3011–3017. https://doi.org/10.1017/S1368980018001386
Gill JMR, Sattar N (2014) Fruit juice: just another sugary drink? Lancet Diabetes Endocrinol 2:444–446. https://doi.org/10.1016/S2213-8587(14)70013-0
Auerbach BJ, Dibey S, Vallila-Buchman P et al (2018) Review of 100% fruit juice and chronic health conditions: implications for sugar-sweetened beverage policy. Adv Nutr 9:78–85. https://doi.org/10.1093/advances/nmx006
Drouin-Chartier JP, Zheng Y, Li Y et al (2019) Changes in consumption of sugary beverages and artificially sweetened beverages and subsequent risk of type 2 diabetes: results from three large prospective U.S. Cohorts of women and men. Diabetes Care 42:2181–2189. https://doi.org/10.2337/dc19-0734
Shang F, Li X, Jiang X (2016) Coffee consumption and risk of the metabolic syndrome: a meta-analysis. Diabetes Metab 42:80–87. https://doi.org/10.1016/j.diabet.2015.09.001
Ding M, Bhupathiraju SN, Chen M et al (2014) Caffeinated and decaffeinated coffee consumption and risk of type 2 diabetes: a systematic review and a dose-response meta-analysis. Diabetes Care 37:569–586. https://doi.org/10.2337/dc13-1203
Poole R, Kennedy OJ, Roderick P et al (2017) Coffee consumption and health: umbrella review of meta-analyses of multiple health outcomes. BMJ 359:j5024. https://doi.org/10.1136/bmj.j5024
Geleijnse JM, Launer LJ, van der Kuip DAM et al (2002) Inverse association of tea and flavonoid intakes with incident myocardial infarction: the Rotterdam Study. Am J Clin Nutr 75:880–886. https://doi.org/10.1093/ajcn/75.5.880
Yi M, Wu X, Zhuang W et al (2019) Tea consumption and health outcomes: umbrella review of meta-analyses of observational studies in humans. Mol Nutr Food Res 63:e1900389. https://doi.org/10.1002/mnfr.201900389
Abe SK, Inoue M (2021) Green tea and cancer and cardiometabolic diseases: a review of the current epidemiological evidence. Eur J Clin Nutr 75:865–876. https://doi.org/10.1038/s41430-020-00710-7
Soedamah-Muthu SS, de Goede J (2018) Dairy consumption and cardiometabolic diseases: systematic review and updated meta-analyses of prospective cohort studies. Curr Nutr Rep 7:171–182. https://doi.org/10.1007/s13668-018-0253-y
Alvarez-Bueno C, Cavero-Redondo I, Martinez-Vizcaino V et al (2019) Effects of milk and dairy product consumption on type 2 diabetes: overview of systematic reviews and meta-analyses. Adv Nutr 10:154–163. https://doi.org/10.1093/advances/nmy107
Abargouei AS, Janghorbani M, Salehi-Marzijarani M, Esmaillzadeh A (2012) Effect of dairy consumption on weight and body composition in adults: a systematic review and meta-analysis of randomized controlled clinical trials. Int J Obes 36:1485–1493. https://doi.org/10.1038/ijo.2011.269
Geng T, Qi L, Huang T (2018) Effects of dairy products consumption on body weight and body composition among adults: an updated meta-analysis of 37 randomized control trials. Mol Nutr Food Res 62:1–10. https://doi.org/10.1002/mnfr.201700410
Gillum RF, Koenig W, Butterworth A et al (2018) Risk thresholds for alcohol consumption: combined analysis of individual-participant data for 599 912 current drinkers in 83 prospective studies. The Lancet 391:1513–1523. https://doi.org/10.1016/s0140-6736(18)30134-x
Li XH, Yu FF, Zhou YH, He J (2016) Association between alcohol consumption and the risk of incident type 2 diabetes: a systematic review and dose-response meta-analysis1. Am J Clin Nutr 103:818–829. https://doi.org/10.3945/ajcn.115.114389
Duffey KJ, Davy BM (2015) The healthy beverage index is associated with reduced cardiometabolic risk in US adults: a preliminary analysis. J Acad Nutr Diet 115:1682-1689.e2. https://doi.org/10.1016/j.jand.2015.05.005
Wagner S, Merkling T, Girerd N et al (2022) Quality of beverage intake and cardiometabolic and kidney outcomes: insights from the STANISLAS cohort. Front Nutr 8:738803. https://doi.org/10.3389/fnut.2021.738803
Hu EA, Anderson CAM, Crews DC et al (2020) A healthy beverage score and risk of chronic kidney disease progression, incident cardiovascular disease, and all-cause mortality in the chronic renal insufficiency cohort. Curr Dev Nutr 4(zaa088):1–8. https://doi.org/10.1093/cdn/nzaa088
Ikram MA, Brusselle G, Ghanbari M et al (2020) Objectives, design and main findings until 2020 from the Rotterdam Study. Eur J Epidemiol 35:483–517. https://doi.org/10.1007/s10654-020-00640-5
Voortman T, Kiefte-de Jong JC, Ikram MA et al (2017) Adherence to the 2015 Dutch dietary guidelines and risk of non-communicable diseases and mortality in the Rotterdam Study. Eur J Epidemiol 32:993–1005. https://doi.org/10.1007/s10654-017-0295-2
Klipstein-Grobusch K, den Breeijen JH, Goldbohm RA et al (1998) Dietary assessment in the elderly: validation of a semiquantitative food frequency questionnaire. Eur J Clin Nutr 52:588–596. https://doi.org/10.1038/sj.ejcn.1600611
Feunekes GI, van Staveren WA, de Vries JHM et al (1993) Relative and biomarker-based validity of a food-frequency questionnaire estimating intake of fats and cholesterol. Am J Clin Nutr 58:489–496. https://doi.org/10.1093/ajcn/58.4.489
World Health Organization and International Diabetes Federation (2006) Definition and diagnosis of diabetes mellitus and intermediate hyperglycaemia: report of a WHO/IDF consultation. World Health Organization. https://apps.who.int/iris/handle/10665/43588
Stolk RP, Pols HAP, Lamberts SWJ et al (1997) Diabetes mellitus, impaired glucose tolerance, and hyperinsulinemia in an elderly population: the Rotterdam Study. Am J Epidemiol 145:24–32. https://doi.org/10.1093/oxfordjournals.aje.a009028
World Health Organization (2009) Anatomical therapeutic chemical classification system (ATC). WHO, Oslo
Ligthart S, van Herpt TTW, Leening MJG et al (2016) Lifetime risk of developing impaired glucose metabolism and eventual progression from prediabetes to type 2 diabetes: a prospective cohort study. Lancet Diabetes Endocrinol 4:44–51. https://doi.org/10.1016/S2213-8587(15)00362-9
Caspersen CJ, Bloemberg BPM, Saris WHM et al (1991) The prevalence of selected physical activities and their relation with coronary heart disease risk factors in elderly men: the Zutphen Study, 1985. Am J Epidemiol 133:1078–1092. https://doi.org/10.1093/oxfordjournals.aje.a115821
Stel VS, Smit JH, Pluijm SMF et al (2004) Comparison of the LASA physical activity questionnaire with a 7-day diary and pedometer. J Clin Epidemiol 57:252–258. https://doi.org/10.1016/j.jclinepi.2003.07.008
World Health Organ Tech Rep Ser (2000) WHO Consultation on Obesity (1999: Geneva, Switzerland) & World Health Organization. (2000). Obesity: preventing and managing the global epidemic: report of a WHO consultation. World Health Organization
Rosinger A, Herrick K, Gahche J, Park S (2017) Sugar-sweetened Beverage Consumption Among U.S. Adults, 2011–2014. NCHS Data Brief. 1–8
Duffey KJ, Popkin BM (2006) Adults with healthier dietary patterns have healthier beverage patterns. J Nutr 136:2901–2907. https://doi.org/10.1093/jn/136.11.2901
InterAct Consortium, van Woudenbergh GJ, Kuijsten A et al (2012) Tea consumption and incidence of type 2 diabetes in Europe: the EPIC-interact case-cohort study. PLoS One 7:e36910. https://doi.org/10.1371/journal.pone.0036910
Jacobs S, Kröger J, Floegel A et al (2014) Evaluation of various biomarkers as potential mediators of the association between coffee consumption and incident type 2 diabetes in the EPIC-Potsdam Study. Am J Clin Nutr 100:891–900. https://doi.org/10.3945/ajcn.113.080317
Ochoa-Rosales C, van der Schaft N, Braun KVE et al (2023) C-reactive protein partially mediates the inverse association between coffee consumption and risk of type 2 diabetes: the UK Biobank and the Rotterdam Study cohorts. Clin Nutr 42:661–669. https://doi.org/10.1016/j.clnu.2023.02.024
Hedrick VE, Davy BM, Myers EA et al (2015) Changes in the healthy beverage index in response to an intervention targeting a reduction in sugar-sweetened beverage consumption as compared to an intervention targeting improvements in physical activity: results from the talking health trial. Nutrients 7:10168–10178. https://doi.org/10.3390/nu7125525
Van Rossum CTM, Buurma-Rethans E, Vennemann FBC et al. (2020)The diet of the Dutch. Results of the Dutch National Food Consumption Survey 2012–2016. Bilthoven: National Institute for Public Health and the Environment (RIVM)
Aknowledgements
We thank the Rotterdam Study participants, and the general practitioners and pharmacists of the Ommoord district for their contributions. The authors would like to also thank Ester de Jonge for helping in cleaning the dietary data and to Jolande Verkroost-van Heemst for helping in cleaning the disease incidence data.
Funding
The Rotterdam Study is supported by Erasmus MC and Erasmus University Rotterdam, the Netherlands Organisation for Scientific Research, the Netherlands Organisation for Health Research and Development (ZonMw), the Research Institute for Diseases in the Elderly, the Netherlands Genomics Initiative, the Ministry of Education, Culture and Science, the Ministry of Health, Welfare and Sports, the European Commission (DG XII), and the Municipality of Rotterdam. The PhD of MGJC is funded by The Mexican National Council for Science and Technology (CONACYT) through its Graduate Programme “Becas de posgrado en el Extranjero”.
Author information
Authors and Affiliations
Contributions
MGJC, TV and JMG contributed to the study conception and design. MGJC and COR performed data analyses. MGJC wrote the manuscript. TV was involved in the dietary data acquisition. MK and FA were involved in the study design and data collection of diabetes. All authors provided input for the analyses and interpretation of the data, and writing of the manuscript. All authors read, revised critically and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jacobo Cejudo, M.G., Ochoa-Rosales, C., Ahmadizar, F. et al. The healthy beverage index is not associated with insulin resistance, prediabetes and type 2 diabetes risk in the Rotterdam Study. Eur J Nutr 62, 3021–3031 (2023). https://doi.org/10.1007/s00394-023-03209-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-023-03209-6