Abstract
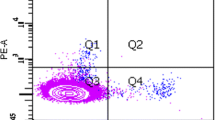
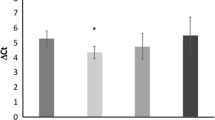
The association between levels of circulating endothelial progenitor cells (EPCs) and heart transplant recipients (HTX) with cardiac allograft vasculopathy (CAV) is under debate. The chemokine receptor CXCR4 plays an important role in the mobilization of progenitor cells and is implicated in pathological conditions, including cardiovascular disease. This study aims to evaluate the association between EPCs and CXCR4-positive cells in HTX patients. Peripheral blood mononuclear cells (PBMCs) from 34 HTX patients and 25 control participants were analyzed by flow cytometry for CXCR4-positive cells and EPCs. Endothelial progenitor cells were defined by the expression of a range of hematopoietic and endothelial lineage markers in different combinations. The ability to form endothelial cell colonies in vitro was also assessed by colony-forming unit (CFU) assay. Phenotypic analysis of EPCs by flow cytometry revealed similar levels in HTX patients compared to controls. In addition, no difference was observed between levels of EPCs or CFU number in patients with and without CAV. By contrast, CFU assay revealed a reduced number of CFUs in HTX patients compared to controls (3.3% ± 0.95 and 13.3% ± 4.5%, respectively, P = 0.014). Likewise, levels of CXCR4-positive cells were significantly reduced (15.9 ± 1.4 in patients vs 24.8 ± 3.3% in controls, P < 0.01), negatively correlated with Framingham risk score (rho = −0.4, P = 0.02) and the number of risk factors (rho = −0.3, P = 0.049). Levels of CXCR4-positive cells were also correlated with CFU number (r = 0.65, P = 0.0005). These findings further develop our understanding of the role of EPCs and endothelial CFUs in cardiovascular disease, in addition to highlighting the potential importance of CXCR4 in heart transplantation.



Similar content being viewed by others
References
Keck BM, Bennett LE, Rosendale J, Daily OP, Novick RJ, Hosenpud JD (1999) Worldwide thoracic organ transplantation: a report from the UNOS/ISHLT International Registry for Thoracic Organ Transplantation. Clin Transpl 35–49
Julius BK, Attenhofer Jost CH, Sütsch G, Brunner HP, Kuenzli A, Vogt PR, Turina M, Hess OM, Kiowski W (2000) Incidence, progression and functional significance of cardiac allograft vasculopathy after heart transplantation. Transplantation 69:847–853
Libby P, Zhao DX (2003) Allograft arteriosclerosis and immune-driven angiogenesis. Circulation 107:1237–1239
Asahara T, Masuda H, Takahashi T, Kalka C, Pastore C, Silver M, Kearne M, Magner M, Isner JM (1999) Bone marrow origin of endothelial progenitor cells responsible for postnatal vasculogenesis in physiological and pathological neovascularization. Circ Res 85:221–228
Hu Y, Davison F, Zhang Z, Xu Q (2003) Endothelial replacement and angiogenesis in arteriosclerotic lesions of allografts are contributed by circulating progenitor cells. Circulation 25:3122–3127
Urbich C, Dimmeler S (2004) Endothelial progenitor cells: characterization and role in vascular biology. Circ Res 95:343–353
Egan CG, Lavery R, Caporali F, Fondelli C, Laghi-Pasini F, Dotta F, Sorrentino V (2008) Generalised reduction of putative endothelial progenitors and CXCR4-positive peripheral blood cells in type 2 diabetes. Diabetologia 51:1296–1305
Vasa M, Fichtlscherer S, Aicher A, Adler K, Urbich C, Martin H, Zeiher AM, Dimmeler S (2001) Number and migratory activity of circulating endothelial progenitor cells inversely correlate with risk factors for coronary artery disease. Circ Res 89:E1–E7
Fadini GP, Baesso I, Albiero M, Sartore S, Agostini C, Avogaro A (2008) Technical notes on endothelial progenitor cells: ways to escape from the knowledge plateau. Atherosclerosis 197:496–503
Charo IF, Taubman MB (2004) Chemokines in the pathogenesis of vascular disease. Circ Res 95:858–866
Kucia M, Ratajczak J, Ratajczak MZ (2005) Bone marrow as a source of circulating CXCR4+ tissue-committed stem cells. Biol Cell 97:133–146
Yamaguchi J, Kusano KF, Masuo O, Kawamoto A, Silver M, Murasawa S, Bosch-Marce M, Masuda H, Losordo DW, Isner JM, Asahara T (2003) Stromal cell-derived factor-1 effects on ex vivo expanded endothelial progenitor cell recruitment for ischemic neovascularization. Circulation 107:1322–1328
Walter DH, Haendeler J, Reinhold J, Rochwalsky U, Seeger F, Honold J, Hoffmann J, Urbich C, Lehmann R, Arenzana-Seisdesdos F, Aicher A, Heeschen C, Fichtlscherer S, Zeiher AM, Dimmeler S (2005) Impaired CXCR4 signaling contributes to the reduced neovascularisation capacity of endothelial progenitor cells from patients with coronary artery disease. Circ Res 97:1142–1151
Wang Y, Haider HKh, Ahmad N, Zhang D, Ashraf M (2006) Evidence for ischemia induced host-derived bone marrow cell mobilization into cardiac allografts. J Mol Cell Cardiol 41:478–487
Sakihama H, Masunaga T, Yamashita K, Hashimoto T, Inobe M, Todo S, Uede T (2004) Stromal cell-derived factor-1 and CXCR4 interaction is critical for development of transplant arteriosclerosis. Circulation 110:2924–2930
Yamani MH, Ratliff NB, Cook DJ, Tuzcu EM, Yu Y, Hobbs R, Rincon G, Bott-Silverman C, Young JB, Smedira N, Starling RC (2005) Peritransplant ischemic injury is associated with up-regulation of stromal cell-derived factor-1. J Am Coll Cardiol 46:1029–1035
Chen MC, Chen CJ, Yang CH, Liu WH, Fang CY, Hsieh YK, Chang HW (2008) Relationship of the percentage of circulating endothelial progenitor cell to the severity of coronary artery disease. Heart Vessels 23:47–52
Simper D, Wang S, Deb A, Holmes D, McGregor C, Frantz R, Kushwaha SS, Caplice NM (2003) Endothelial progenitor cells are decreased in blood of cardiac allograft patients with vasculopathy and endothelial cells of noncardiac origin are enriched in transplant atherosclerosis. Circulation 108:143–149
Thomas HE, Parry G, Dark JH, Arthur HM, Keavney BD (2009) Circulating endothelial progenitor cell numbers are not associated with donor organ age or allograft vasculopathy in cardiac transplant recipients. Atherosclerosis 202:612–616
Ventura HO, Mehra MR, Smart FW, Stapleton DD (1995) Cardiac allograft vasculopathy: current concepts. Am Heart J 129:791–799
Grundy SM, Pasternak R, Greenland P, Smith S Jr, Fuster V (1999) Assessment of cardiovascular risk by use of multiple-risk-factor assessment equations: a statement for healthcare professionals from the American Heart Association and the American College of Cardiology. Circulation 100:1481–1492
Grundy SM, Cleeman JI, Merz CN, Brewer HB Jr, Clark LT, Hunninghake DB, Pasternak RC, Smith SC Jr, Stone NJ (2004) Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III guidelines. Circulation 110:227–239
Schillaci G, Reboldi G, Verdecchia P (2001) High-normal serum creatinine concentration is a predictor of cardiovascular risk in essential hypertension. Arch Intern Med 161:886–891
Egan CG, Caporali F, Garcia-Gonzalez E, Galeazzi M, Sorrentino V (2008) Endothelial progenitor cells and colony-forming units in rheumatoid arthritis: association with clinical characteristics. Rheumatology 47:1484–1488
Hristov M, Weber CJ (2004) Endothelial progenitor cells: characterization, pathophysiology, and possible clinical relevance. Cell Mol Med 8:498–508
Dimmeler S, Aicher A, Vasa M, Mildner-Rihm C, Adler K, Tiemann M, Rütten H, Fichtlscherer S, Martin H, Zeiher AM (2001) HMG-CoA reductase inhibitors (statins) increase endothelial progenitor cells via the PI 3-kinase/Akt pathway. J Clin Invest 108:391–397
Tura O, Barclay GR, Roddie H, Davies J, Turner ML (2007) Absence of a relationship between immunophenotypic and colony enumeration analysis of endothelial progenitor cells in clinical haematopoietic cell sources. J Transl Med 5:37
Damås JK, Waehre T, Yndestad A, Ueland T, Müller F, Eiken HG, Holm AM, Halvorsen B, Frøland SS, Gullestad L, Aukrust P (2002) Stromal cell-derived factor-1alpha in unstable angina: potential antiinflammatory and matrix-stabilizing effects. Circulation 106:36–42
Egan CG, Caporali F, Huqi A, Zito MC, Focardi M, Mondillo S, Pierli C, Marzilli M, Sorrentino V (2009) Reduced levels of putative endothelial progenitor and CXCR4+ cells in coronary artery disease: kinetics following percutaneous coronary intervention and association with clinical characteristics. Thromb Haemost 101:1138–1146
Miriuka SG, Rao V, Peterson M, Tumiati L, Delgado DH, Mohan R, Ramzy D, Stewart D, Ross HJ, Waddell TK (2006) mTOR inhibition induces endothelial progenitor cell death. Am J Transpl 6:2069–2079
Hegner B, Lange M, Kusch A, Essin K, Sezer O, Schulze-Lohoff E, Luft FC, Gollasch M, Dragun D (2009) mTOR regulates vascular smooth muscle cell differentiation from human bone marrow-derived mesenchymal progenitors. Arterioscler Thromb Vasc Biol 29:232–238
Pinney SP, Mancini D (2004) Cardiac allograft vasculopathy: advances in understanding its pathophysiology, prevention, and treatment. Curr Opin Cardiol 19:170–176
António N, Fernandes R, Rodriguez-Losada N, Jiménez-Navarro MF, Paiva A, de Teresa Galván E, Gonçalves L, Ribeiro CF, Providência LA (2010) Stimulation of endothelial progenitor cells: a new putative effect of several cardiovascular drugs. Eur J Clin Pharmacol 66:219–230
Acknowledgments
This work was supported by a grant from Regione Toscana for the Center for Stem Cell Research, University of Siena.
Author information
Authors and Affiliations
Corresponding author
Additional information
C. G. Egan and F. Caporali contributed equally to this study.
Rights and permissions
About this article
Cite this article
Egan, C.G., Caporali, F., Capecchi, P.L. et al. Levels of circulating CXCR4-positive cells are decreased and negatively correlated with risk factors in cardiac transplant recipients. Heart Vessels 26, 258–266 (2011). https://doi.org/10.1007/s00380-010-0053-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-010-0053-9