Abstract
European policy recommends that biomass production occur on marginal land, such as poorly draining Stagnosols. Compared to annual cropping, perennial crops may better mitigate N2O emissions at such sites, through more complete denitrification. To test that hypothesis, we compared N2 and N2O fluxes from the soils of a perennial crop (cup plant, Silphium perfoliatum L.) and an annual crop (silage maize, Zea mays L.). Intact soil columns (35 cm height, 14.4 cm diameter) were incubated for 37 days. The soils were fertilized with 60 or 120 kg N ha−1 and exposed to successive phases of waterlogging: free drainage, waterlogging of 1/3-, and waterlogging of 2/3- of the column. Source-specific N2O and N2 fluxes were measured using the 15 N gas flux method. Denitrification was higher in cup plant than maize soil and total N losses from denitrification were dominated by emissions from the third phase. Cup plant soil emitted 33.6 ± 78.1 mg N m−2 and 95.8 ± 64.4 mg N m−2 more N2O than maize soil in the low and high N treatments, respectively. The product ratio of denitrification (N2Oi = N2O/(N2 + N2O)) increased with waterlogging in maize soil, while remaining stable in cup plant soil. Emissions from the top 10 cm dominated the N2Oi rather than N2 fluxes from the saturated soil. This study did not show N2O mitigation in cup plant soil, instead highlighting the complexity of plant-soil effects on denitrification. We clearly showed that the application of a general N2Oi for agricultural soils across annual and perennial cropping is not recommended.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biomass cultivation for producing bioenergy gets increasingly criticized due to broad environmental concerns ranging from soil degradation to negative impact on biodiversity (Herbes et al. 2014), and due to its competition with food production for arable land (Haberl et al. 2012). To avoid increased carbon dioxide (CO2) emissions through indirect land-use change, it has been strongly recommended that biomass production be restricted to sites less suitable for food production (Directive (EU) 2018/2001, 2018).
Marginal conditions for food production can be found at sites that are difficult to cultivate due to harsh environmental conditions (Blanco-Canqui 2016; Shortall 2013). The hydromorphic, fine textured soils (Stagnosols, IUSS 2015)) in the cold, winter-wet climate of low mountain ranges are one example of marginally productive land suitable for biomass production in central Europe. However, under these conditions, the most common biomass crop in Europe, silage maize (Zea mays L.), does not reach its full yield potential due to the slow soil warming and challenging crop management at low mountain range sites. Perennial crops may overcome some of the drawbacks of annual cropping at these sites. According to Grunwald et al. (2020), cup plant (Silphium perfoliatum L.) is particularly suitable for low mountain ranges, since these sites meet the high water demand of cup plant and in turn benefit from the soil preservation of the perennial crop. Due to the perenniality of the roots and the long growing season, cup plant is reported to mitigate N leaching and erosion compared to annual crops (Grunwald et al. 2020). However, as a green harvested crop, cup plant has a relatively high nitrogen (N) demand compared with other perennial crops, e.g., miscanthus (Ruf and Emmerling 2021), leading to concerns about N fertilizer addition and related losses.
Field-derived greenhouse gas (GHG) emissions during crop production are a critical consideration when assessing the sustainability of bioenergy from biomass (Directive (EU) 2018/2001, 2018; Jin et al. 2019). While the use of marginal land may reduce carbon- (C) related emissions from indirect land-use change, these potential gains must be weighed against increases in other GHGs, particularly nitrous oxide (N2O) at the hydromorphic sites. N2O causes ozone depletion in the stratosphere, has a global warming potential 273 times greater than CO2 over a one-hundred-year time horizon (GWP100, Forster et al. (2021)), and is mainly produced by nitrification and denitrification, two microbial processes of the N cycle in soils. In agricultural soils, denitrification is usually the main source of emitted N2O (Butterbach-Bahl et al. 2013; Li et al. 2016; Ostrom et al. 2010). Denitrification is the microbial reduction of nitrate (NO3−) to N2, where N2O is an intermediate product. The key consideration, therefore, is not only whether denitrification occurs, but the product ratio (N2Oi = N2O/(N2O + N2)). While any denitrification activity represents NO3− removal from the soil–plant system, complete denitrification to N2 does not pose a risk through GHGs. Therefore, both total N2 + N2O emissions and the N2Oi are necessary to understand denitrification and its role as GHG source (Ciarlo et al. 2007; Jahangir et al. 2012; Scheer et al. 2020). On a global scale, total denitrification (N2 + N2O emissions) is currently estimated based on land-use-specific N2Oi (e.g., Scheer et al. (2020) and Schlesinger (2009)). However, cropping system-specific N2Oi under field conditions are widely unknown.
Organic C sources serve as electron donors for denitrification processes, while N oxides such as NO3−, NO2−, and N2O serve as electron acceptors. Hence, soil aeration, organic C, and NO3− availability are all key factors controlling denitrification. Land use and management has an impact on each of these: soil aeration (Soane et al. 2012), C availability (Palm et al. 2014), and N mineralization and N availability (Balesdent et al. 2000). Thus, it can be assumed that soil-derived N emissions from a system managed for annual cropping may differ substantially from those of a perennial cropping system. However, in order to predict management and cropping system effects and their potential to cause or mitigate denitrification, it is important to understand the mechanisms involved.
Soil aeration is controlled through bulk density and soil moisture. No-till systems generally have higher bulk densities than conventionally tilled systems (Palm et al. 2014). Soils that do not undergo frequent rearrangement of aggregates through tillage have fewer large pores (> 1000 µm) than tilled soils (Kay and VandenBygaart 2002). Smaller pores are saturated faster and drain slower than more transmissive pores of less compacted, tilled soil (Weninger et al. 2019). Soil under perennial crops, representing a no-till system, should then presumably have lower soil gas diffusivity than annual crops in a conventionally tilled system (Ball et al. 1999; Rochette et al. 2008), and possibly be more prone to complete denitrification, especially in fine textured soil (Chapuis-Lardy et al. 2007). Generally, denitrification emissions increase with increasing water-filled pore space (WFPS) up to an optimum of around 80% WFPS for N2O (Butterbach-Bahl et al. 2013), with N2 emissions increasing through to complete soil saturation. Thus, above the ‘tipping point’ of soil moisture for N2O emission, the reduction of N2O to N2 might exceed N2O formation thus leading to lower N2O fluxes. This tipping point can be reached through severe anoxic conditions in saturated soil or the prolonged residence time of N2O due to low soil gas diffusivity. Denitrification is known to be potentially very high in the saturated zone of hydromorphic soils (Well et al. 2005), and could be particularly relevant in the afore-mentioned, periodically waterlogged Stagnosols of low mountain ranges. With such soil conditions, the potentially lower aeration under cup plant could increase denitrification, but also increase the reduction of N2O to N2, while in maize soil, the higher pore volume may result in more aeration, limiting N2O reduction.
Th C availability for denitrifiers can be an important factor in determining the N2Oi. Since the final reduction of N2O to N2 is energy-intensive (Saggar et al. 2013), abundant available C as the energy source for denitrification can decrease the N2Oi by enabling the reduction of N2O to N2 (Firestone 1982; Firestone and Davidson 1989). However, there is no one management system (i.e., till vs. no-till) that clearly favors C availability. Tillage-induced mineralization (Balesdent et al. 2000) and incorporation of organic matter (Van Den Bossche et al. 2009) induces the release of C and NO3− and enhances O2 consumption. Perennial no-till systems are reported to facilitate soil organic C (SOC) accumulation due to increased root biomass (Don et al. 2012; Gauder et al. 2016; Monti and Zatta 2009) and canopy litter (Amougou et al. 2012; Schoo, et al. 2017a, b). The net effect of perennial no-till vs annually tilled soils on N2O and N2 fluxes is thus not clear and both maize and cup plant have the potential to enhance denitrification through C availability.
As a key substrate for denitrification, NO3− availability has a clear impact on N2O and N2 emissions and the reduction of N2O to N2 in arable soil (Firestone et al. 1979; Senbayram et al. 2012). Numerous field experiments have compared the N2O emissions from annual and perennial biomass crops (Drewer et al. 2012; Gauder et al. 2012; Walter et al. 2015), and consistently show that perennial systems have lower N2O emissions. However, in these field studies, the perennial crops received less N. Therefore, the effect of the different cropping systems on other soil properties affecting denitrification (i.e., bulk density, C availability) is unclear. In studies where perennial biomass crops received comparable amounts of N fertilizer to annual crops, perennial crops exhibited equal or higher N2O emissions than the annual crops (Ferchaud et al. 2020; Jørgensen et al. 1997). Given the higher N demand of cup plant as compared to other perennial crops (Ruf and Emmerling 2021), cup plant relative to maize may provide a considerable risk for high denitrification losses. To assess this risk, more information is needed on the interactions between N availability and other soil properties related to the cropping system.
In the last decades, numerous denitrification studies have been conducted under fully saturated or constantly submerged soil conditions, as these conditions are assumed to cause substantial gaseous N losses to the environment (Friedl et al. 2016; Reddy et al. 1978; Well et al. 2005). In the field, however, soil water conditions are more dynamic. Varying stages of soil saturation are likely to be more common than full waterlogging, particularly on slow draining Stagnosols. Information on denitrification with changing soil saturation at different soil depths, which would be typical for Stagnosols and more representative of field conditions than static waterlogging, is lacking. To assess the effects of cup plant and maize cultivation on N2 and N2O emissions, an incubation experiment using the 15 N gas flux method was conducted, focusing on the potential for complete denitrification during periods of waterlogging. Field conditions occurring between fall and spring were mimicked as closely as possible using a dynamic soil water regime ranging from moist soil up to waterlogging 10 cm below the soil surface. The main question of the present study was to test if the cultivation of cup plant on Stagnosols results in soil conditions that mitigate N2O emissions due to more complete reduction of N2O to N2 compared to maize cultivation. To answer this question, we had three specific objectives: (1) to describe the impact of waterlogging in different soil depths on denitrification and the emission of N2O and N2; (2) to test, using intact cores, the effect of N availability on the formation and reduction of N2O, given the different soil properties that develop under cup plant and maize cultivation; and finally (3) to compare denitrification, particularly the reduction of N2O to N2, from maize and cup plant soil.
Materials and methods
Soil selection, sampling of soil cores, and their preparation
Intact soil cores were taken from a maize (Zea mays) and an adjacent cup plant (Silphium perfoliatum) field in Gronig (49.520° N, 7.073° E, 365 m.a.s.l., mean annual temperature 9 °C, mean annual precipitation 1031 mm), western Germany. The extraction sites were in close vicinity of each other (< 80 m). The cup plant stand was established in 2017, while the maize field followed maize in rotation with a rye winter cover crop (Table S.1). The soil monoliths extracted either from the maize or the cup plant field are referred as maize or cup plant soil. The soil at both sites is a Hypereutric Stagnic Cambisol (Loamic, Aric, Humic) (IUSS 2015). The fine textured soil (silt loam) is characterized by temporal water logging due to a reduced total pore volume and a higher bulk density below ploughing depth of 25 cm (Table S.2). Plant row spacing was 75 cm for both crops. To minimize the number of factors affecting N cycling (e.g., plant uptake, rhizosphere effects), living plants were excluded from the experiment by taking the columns from the inter-row area representative for > 50% of both fields.
In September 2021, an area of 7.5 m2 was marked out in the adjacent fields, and was irrigated by drip irrigation (approximately 20–25 mm) to minimize structural damage while pushing cylinders in the soil. Following irrigation, intact, 35-cm-high soil columns were collected in Plexiglas cylinders/inner diameter = 14.4 cm, height = 40 cm height). Cylinders were used as liners in a steel auger that was pushed into the soil with an electric motor hammered auger. Columns were carefully extracted by digging. Parallel to the column extraction, bulk density measurements were conducted with 100 cm3 steel cylinders at the following depths: 5–10 cm, 15–20 cm, and 30–35 cm (each depth n = 4).
After transport to the incubation facility, each column was irrigated with 4 L solution of 0.01 M CaCl2, equivalent to 245.6 mm, for 13 days to remove mineral NO3− from the soil, create comparable conditions in cup plant and maize soil, and allow homogenous 15 N labeling. The amount of irrigated water is comparable to the precipitation over the winter at the extraction sites. To exclude the effects from growing plants during the subsequent incubation, emerging weeds were carefully removed and few individual, sprouting cup plants were killed selectively by brushing the emerging leaves with glyphosate (24 g glyphosate/l, Roundup Powerflex, Bayer AG, Leverkusen, Germany) to avoid soil disturbance by removing the plants due to their relatively deep rooting. In total, six cup plant soil cores were treated with the herbicide; three were used to determine initial N content, leaving only three for flux measurements (highest share of treated cores in one treatment was 22% in the 60 kg N treatment in the first phase).
Incubation set up and experimental design
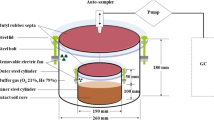
The Plexiglas cylinders (volume = 6515 cm3) served as incubation vessel, as previously described in Kemmann et al. (2021). Briefly, a needle plate as irrigation lid on the top and a base plate at the bottom of the Plexiglas cylinder were sealed by flat rubber sealings airtight. The porous base plate contained a polyamide membrane with a 0.45 µm pore size and bubble point of 100 kPa (ecoTech, Bonn, Germany) and was connected to leachate collection bottles held at defined negative pressure to allow controlled drainage. In addition to the connections for the irrigation and fertilizer solution, the lid contained an inlet and outlet for the synthetic gas flow. The artificial atmosphere containing 20% O2, 2.7% N2, 77% He, 350 ppm CO2, and 250 ppb N2O flowed through the 815 cm3 headspace and had a mean flow rate of 11 ml min−1. The reduced N2 concentration was used to improve the sensitivity of the isotope ratio mass spectrometry (IRMS) measurements (Lewicka-Szczebak et al. 2017), whereas CO2 and N2O were added to establish atmospheric conditions of these gases. Gas chromatography (GC) measurements, flow measurements, valve control, irrigation, and temperature were controlled by the fully automated system (Hantschel et al. 1994; Kemmann et al. 2021) (see supplement). The experiment was conducted constantly at 10 °C. Temperature and soil moisture regimes were set to mimic field conditions occurring from mid-October to mid-March.
Treatments were two N levels, 60 kg NO3-N ha−1 (low N) and 120 kg NO3-N ha−1 (high N), and soils from the two cropping systems: cup plant soil and maize soil; in total four treatment combinations with a minimum of five replicates (Fig. 1).
The dynamic experiment (Fig. 1) consisted of three phases with different water levels: drainage with pressure head of – 80 kPa for 239 h (phase WL0), waterlogging in the lower 10 cm (25–35 cm) for 336.5 h (phase WL10), and waterlogging in the lower 25 cm (10–35 cm) for 321.5 h (phase WL25). The waterlogging was established by slowly pumping water (approximately 50 to 100 ml for 10 cm of waterlogging) through the base plate into the soil in order to avoid entrapped air bubbles in the waterlogged soil. Detailed information about the experimental setup can be found in the supplement (Fig. S.7). The number of days in phase WL10 and WL25 include the time for adjusting the water level (around 48–72 h). As soil columns designated to be used for soil sampling were included in the incubation, the start of the dynamic experiment consisted of a total of 36 soil columns (Fig. 1). Additional eight soil columns were destructively sampled after label application but prior the start of the experiment (time 0 sample).
Soil analysis
Two days after finishing 15 N labeling (time 0, t0) and after each phase, two mesocosms per treatment combination were sampled destructively. At t0, during phase transition, or at the end of the experiment, columns were sampled destructively in three soil depths, where each depth represented 1/3 of the soil column (0–11.7, 11.7–23.3, 23.3–35 cm). For simplicity, throughout the rest of the paper, these depths are referred to as depth 1 (0–12 cm), depth 2 (12–23 cm), and depth 3 (23–35 cm). All analyses were conducted in each depth of the sampled column. Soil moisture was determined immediately after dividing columns into the three soil layers. Gravimetric water content was determined by drying the soil for 48 h at 105 °C. Free water in the column at the time of destructive sampling could not be quantified, due to loss during the sampling process. Therefore, water content in the waterlogged soil layers could not be precisely determined. One subsample of fresh soil was stored at – 20 °C and thawed over night at 4 °C for determination of soil mineral N content (Nmin = NO3−-N + NH4+-N). For the Nmin extraction, 400 g of homogenized fresh soil was filled into a 1 L PE bottle. The extractions were carried out by shaking the sample with 2 M KCl at a ratio 1:1.5 (w/v) for 1 h in an overhead shaker. The extraction solution was filtered (MN 614¼ filters, Macherey & Nagel, Düren, Germany) and stored afterwards at – 20 °C until analysis. NO3− and NH4+-N concentrations were analyzed colorimetrically with a continuous flow analyzer (SA 5000, Skalar Analytical B.V., Netherlands). The large soil sample size was used to account for soil heterogeneity, since soil cores were not disturbed and mixed prior to incubation. 15 N enrichment of extractable NO3− (15aNO3) and NH4+ (15aNH4) was quantified by analyzing the same extract by chemical conversion of NO3− to N2O and NH4+ to N2, respectively, and online analysis by mass spectrometry (Dyckmans et al. 2021).
Another fresh soil subsample for determination of the water extractable organic C (WEOC) was stored at 4 °C overnight after destructive sampling. WEOC was extracted by gently shaking 5 g (dry mass basis) of fresh soil for one minute in 20 ml in pure water. Afterwards, solution was centrifuged for 10 min at 12,000 g and the supernatant was filtered through a 45 µm syringe filter. For analysis of the WEOC concentration in the extractant, WEOC was oxidized and C emissions quantified by NDIR detection (Dimatoc 2000, Dimatec, Essen, Germany). WEOC analysis was only conducted on the remaining 20 columns after phase WL25. Soil acidity was measured with pH-meter (FE20, Mettler Toledo, Urdorf, Switzerland) after shaking 5 g (dry mass basis) of fresh soil in 20 ml in 0.01 M CaCl2 for 1 h. Soil for the determination of total C and N was air dried and ground before analyzing in an elemental analyzer (TruMac CN analyzer, Leco, St. Joseph, USA). Soil organic C (SOC) was determined indirectly by dry combustion as the difference of total C and total inorganic C. Soil pH, SOC, and total N were only analyzed on three columns per treatment combination (n = 3).
Gas analysis
Gas samples from all mesocosms connected to the automated incubation system were analyzed online with a gas chromatograph (GC, Shimadzu GC-2014, Shimadzu, Duisburg, Germany) equipped with a flame ionization detector (FID), electron capture detector (ECD), and a thermal conductivity detector (TCD).
Each column was measured in intervals of 4.5, 3.5, and 2.5 h in phase WL0, WL10, and WL25, respectively. In each measuring sequence, three measurements of empty vessels with a representative headspace exchange rate as mesocosms were integrated for monitoring background N2O and CO2 concentrations in the gas mixture. Furthermore, for calibration six standards (0.33, 0.55, 2.01, 6.94, 40.4, 130 ppm N2O) were measured regularly. Repeated measurements of these standards determined an analytical precision of the GC of consistently < 2% CV.
15 N labeling and isotope analysis of N 2 and N 2 O
For 15 N labeling the soil columns were amended with a K15NO3 solution containing 0.01 M CaCl2 to achieve a target enrichment of 60 atom% 15 N. Since the 15 N gas flux method (15NGF) assumes homogeneous distribution of 15 N, Br− percolation pre-tests were conducted to identify the best practicable irrigation scheme to reach homogeneously labeling. Based on these pre-tests, the 15 N tracer solution was applied by replacing soil water 1.33 times with an alternating regime of two irrigation and flooding cycles. The irrigation was conducted by applying 1 L of fertilizer solution through the overhead irrigation plate. The flooding was conducted from the bottom by slowly pumping fertilizer solution through the filter membrane in the base plate into the mesocosm (Fig. S.7).
For 15 N gas analysis, 12 ml Exetainers (Labco Ltd., Lampeter, UK) were connected to the gas flow at the outlet of each mesocosm, guaranteeing a flushing of at least 1200 times the Exetainer’s volume in 24 h. Samples from each mesocosm plus two blanks (background concentration of the gas mixture) were analyzed at t0 and then in a three day intervals. Therefore, each phase had four sampling dates. Sampling dates were selected to cover each phase equally during the time period when soil moisture conditions were stable. Therefore, the time for establishing the water level of each phase was excluded, because individual columns behaved differently in establishing the water level and thus did not provide comparable conditions.
Dissolved N2O and N2 in the soil water were analyzed at the end of incubation to estimate accumulation of denitrification products in the water-saturated layers. Soil water samples were drawn from the baseplate with a syringe and immediately transferred into a 100 ml serum bottle containing a gas mixture of 3% atmospheric air in helium with slight overpressure of 40 hPa. Serum bottles with the samples were shaken for 1 h intensively on a horizontal lab shaker at constant 10 °C to equilibrate dissolved gases with the headspace. After shaking, two aliquots of 12.5 ml of headspace air were transferred into evacuated Exetainers for gas analysis by GC and IRMS.
IRMS analysis of gas samples was conducted as described in Lewicka-Szczebak et al. (2013) with a modified GasBench II (Thermo Scientific, Bremen, Germany) with an automated sampling and online sample preparation (PAL Systems, Zwingen, Switzerland). Before samples were analyzed in the triple collector IRMS (MAT 253, Thermo Scientific, Bremen, Germany), N2O is reduced in Cu oven to N2. Therefore, the isotopocule mass ratios 29R (29N2/28N2) and 30R (30N2/28N2) from N2, N2 + N2O, and N2O were measured. Through these measurements, the fraction originating of the labeled pool (fp) of N2 (fp_N2), N2 + N2O (fp_N2 + N2O), and N2O (fp_N2O) were quantified. The analytical precision of the IRMS measurements were < 7% and < 0.01% CV for 30R and 29R, respectively, which corresponds to a standard deviation of < 10−6 for both ratios.
Calculations and statistics
The bulk densities (BD) measured in the field were systematically higher than the mean column average BDs, presumably due to slight loosening during sampling of the soil cores or due to more pronounced soil swelling following (drip) irrigation. Therefore, column average BD was used for calculations of the 15 N labeling. However, the BD trends observed in the field were also observed in the extracted soil columns.
For flux calculations, the mass concentrations (C) were calculated according the ideal gas law from the mole fraction (n) of CO2, N2O, and N2 provided from the GC and IRMS measurement:
where M is the molar mass and T the temperature (°C). Mass flow (f, mg C or µg N m−2 h−1), therefore, was calculated by C and the flow rate (Q) provided from the flow meter:
where A denotes the soil surface of the column in m2 and Q is the flow rate in ml min−1.
Cumulated fluxes were obtained by integrating the emissions according the trapezoidal law from each phase separately or from the whole observation period after linear interpolation. Treatments were evaluated based on their cumulated emissions.
The 15 N enrichment of N2 and N2O and of the active NO3−-pool undergoing denitrification (ap: apN2, apN2 + N2O, and apN2O) were calculated based on the assumption of the non-random distribution of isotopocules in the gas samples (Hauck and Bouldin 1961). Calculations of N2, N2 + N2O, and N2O fluxes were conducted according to Spott et al. (2006) and Russow et al. (1996). N2O flux from other sources than the labeled pool (fn_N2O) were calculated as (1-Fp_N2O)*N2Ot (where Fp_N2O is the ratio of pool derived N2O at total N2O in the sample (background corrected), lower case t = total, see Kemmann et al. (2021)). Detailed information about the calculation of the IRMS data can be found in the supplement.
Cumulated fluxes of pool derived N2, N2 + N2O, and N2O as well as N2O from other sources than the labeled pool were calculated with the formulas 1 and 2 using fp_N2, fp_N2 + N2O, fp_N2O, and fn_N2O. Where concentrations in the samples were below detection limit of the IRMS, half of the detection limit was imputed for calculation of cumulated fluxes. The product ratio of denitrification (N2Oi: fp_N2O/fp_N2 + N2O) and Fp_N2O were calculated for each sample and corresponding cumulated fluxes. When the imputation of half detection limit had a significant effect on the cumulated emissions, these fluxes were omitted for further calculations of the two ratios. This was the case when the cumulated flux was not 5 times greater than the theoretical cumulated flux of half the detection limit. The IRMS data set with four sampling dates per phases overestimates cumulated N2O emissions from the GC measurements with a higher temporal resolution by 3.2 ± 10.6%. Therefore, calculation and comparisons were all based on the IRMS dataset, unless otherwise stated.
Dissolved gas concentrations were calculated from headspace concentrations according the Henry’s law using the fp_N2 and the N2O concentrations from the GC (further information in the supplement). The molar solubilities were obtained according to Wilhelm et al. (1977).
The observation period of phase WL0 started two days after the t0 sampling and 4 days after finishing fertilizer application because soil moisture conditions did not reach stable conditions before that date.
Calculations and statistical analysis were conducted using the version 4.1.1 of the statistic software R (R core team, 2020). Normal distributed explanatory variables were analyzed using an analysis of variance (ANOVA). Cumulated fluxes were analyzed using a linear mixed effect models with soil core as random effect. When assumption of normality were violated (e.g., N2Oi, Fp_N2O), generalized linear mixed models were fitted with quasibinomial distribution family using a logit link function with the R package nlme (Pinheiro et al. 2020) or the glmmTMB package (Magnusson et al. 2017) with a beta distribution and logit link function when random effects were implemented. To handle the high variability of N emissions, flux data was log10 transformed. Data transformation for CO2 data was in most cases not necessary. Statistic tests in each phase were conducted with all replicates in each phase,; however, the fluxes were only plotted with those columns that lasted for the whole incubation time. All values, if not mentioned differently, are the arithmetic means ± 1 standard deviation.
Results
Physical soil properties and water content
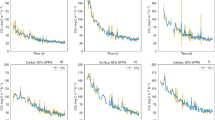
Soil properties varied substantially between the soils and within soils. The bulk density (BD) was 1.44 ± 0.03 g cm−3 and 1.42 ± 0.02 g cm−3 in cup plant and maize soil (Fig. S.1), respectively, and thus significantly (p < 0.01) higher in cup plant soil. Depth-specific bulk densities (BD) in the mesocosm could not be determined accurately. Density measurements in the field indicated following trends in BD and total pore volume (PV): maize 5–10 cm (BD = 1.43 g cm−3, PV = 46.0%) < 15–20 cm (BD = 1.44 g cm−3, PV = 41.0%) < 30–35 cm (BD = 1.69 g cm−3, PV = 36.2%); and cup plant 5–10 cm (BD = 1.49 g cm−3, PV = 43.9%) > 15–20 cm (BD = 1.43 g cm−3, PV = 46.2%) < 30–35 cm (BD = 1.64 g cm−3, PV = 38.0%). While the analysis of variance did not show significant differences in WC between soils in the WL0 and WL10 phases, the WC in WL25 supported the BD field measurements, showing significantly higher WC in depth 1 in maize than cup plant soil but not in depth 2 and 3 (Fig. 2). In phase WL25, the WC in depth 1 increased from phase WL10 by 2.8% (w/w, p = 0.001) in maize soil and by 1.6% (w/w, p = 0.21) in cup plant soil. Soil WC for each phase is shown in Fig. 2. From t0 to the end of the drainage phase (WL0), the WC in maize soil decreased significantly (p = 0.01), while no change (p = 0.70) in WC could be observed in cup plant soil. There were no significant differences in phase WL0 and WL10, although depth 2 in phase WL10 appeared to be higher than in the first phase. In phase WL10 and WL25, water saturation was visually present in the waterlogged depths.
Chemical soil properties and C availability
Soil pH was significantly lower (p < 0.001) in cup plant (4.9 ± 0.1) compared to maize soil (5.2 ± 0.1, Table 1). Total N decreased with soil depth. This decrease was more pronounced in cup plant soil than maize soil, while the decrease in SOC was comparable between the soils (Table 1). Due to a higher (p = 0.002) total N content in maize compared to cup plant soil, the C:N ratio also differed (p < 0.001) between the soils, especially in depth 3 where the C:N ratio was lower in maize soil than cup plant soil. Total C and N contents in depth 3 were about 50% lower in both soils than in the soil above (Table 1). Water extractable organic C (WEOC) did not differ between the soils (p = 0.77). However, as with SOC, WEOC decreased significantly (p < 0.03) with depth (Table 1).
Soil mineral N
The target mineral N (Nmin = NO3-N + NH4-N) contents for the high and low N treatments were 24 and 12 mg N kg−1, respectively. With 26.2 mg N kg−1 and 14.5 mg N kg−1 (n = 2), the maize Nmin contents in phase WL0 were slightly above target. In contrast, Nmin contents in phase WL0 in cup plant soil were with 26.0 mg N kg−1 and 8.9 mg N kg−1 (n = 2), both above and below the initial targets. Coinciding with the decreasing gravimetric water content, NO3− decreased with soil depth, resulting in lower NO3− content in the denser depth 3. However, the 15 N enrichment of the NO3− (15aNO3) in the lower depths (2 and 3) did not differ from depth 1, indicating that fertilizer solution replaced soil water sufficiently in the lower soil. In depth 2 and 3 of cup plant soil, NO3− content, especially in the high N treatment, started to decrease during WL10 and WL25 (Table 2). In maize soil, the NO3− content increased in depth 1 and 2 in the absence of waterlogging, while NO3− content in depth 3 remained relatively stable (Table 2). Ammonium (NH4+) content did not differ (p = 0.96) between the N treatments. NH4+ contents were affected by the soil (p < 0.001) and were non-significantly lower in maize soil treatments (1.2 ± 1.4 mg NH4-N kg−1) than in cup plant soil (2.4 ± 1.3 mg NH4-N kg−1). In cup plant soil, NH4+ remained at a constant level (2.1 and 2.7 mg NH4-N kg−1 in phase WL0 and WL25, respectively), whereas NH4+ content in maize soil decreased (2.1 and 1.0 mg NH4-N kg−1 in phase WL0 and WL25, respectively) throughout the observation period (Table S.3).
In cup plant, soil NO3− content was negatively correlated (r = − 0.53, p < 0.001) with the NH4+ content. The 15aNH4+ in cup plant soil was with 17.6 ± 6.5 atom% significantly higher (p < 0.001) than in maize soil with 0.6 ± 0.4 atom% (Fig. S.2).
CO 2 fluxes
Cumulated CO2 emissions were significantly (p < 0.001) higher from cup plant soil (41.8 ± 11.2 mg C m−2 h−1) than from maize soil (17.7 ± 3.6 mg C m−2 h−1) (Table S.5). Furthermore, there was a significant interaction of soil x phase (p < 0.001) on the CO2 emissions. The N level had no effect on the CO2 emissions (p = 0.13) (Fig. 3). CO2 fluxes were the highest at the beginning of phase WL0, especially from cup plant soil. Flux rates showed a decreasing trend with each phase. In both soils, CO2 dropped markedly with the establishment of phase WL25 but increased again afterwards. Cup plant soil exhibited higher variability in CO2 fluxes than maize soil (Fig. 3).
N 2 O fluxes dynamics from the GC dataset
Similar to the CO2 emissions, there was a significant effect of the soil x phase (p < 0.001) interaction, and additionally, an interaction of soil x N level (p = 0.03), whereas the main effect of N level was not significant (p = 0.31). However, corresponding to the significant main effect of soil (p < 0.001), N2Ot emissions from cup plant soil were always equal or higher than from maize soil (Fig. 3). In cup plant soil, the N level had a significant effect (p = 0.04) on the N2Ot emissions while N2Ot emissions maize soil were not affected by the N level (p = 0.44).
At the beginning of phase WL0, N2Ot were 109.6 ± 97.7 µg N m−2 h−1 and 15.1 ± 15.0 µg N m−2 h−1 from cup plant and maize soil, respectively, and declined slowly toward the end of phase WL0. Pairwise comparison of the cumulated N2Ot emissions in phase WL0 showed that emissions from cup plant soil at both N levels were significantly (p < 0.001) higher than those from maize soil at both N levels (Fig. 3). During the establishment of WL10, fluxes from cup plant soil dropped and increased again after 2 days reaching a level comparable to the end of phase WL0 (Fig. 3). Emissions from maize soil did not show a similar dynamic during establishment of phase WL10. In phase WL10, the only significant difference observed was that emissions from high-N maize soil were lower than the high-N cup plant soil (p = 0.01). In contrast to WL10, only N2Ot from maize dropped due to the increase of the water level in WL25, whereas fluxes from cup plant soil were apparently not affected (Fig. 4). After raising the water level in WL25, fluxes from both soils increased substantially. The N2Ot from cup plant soil peaked between day 31–33 with a maximum of 1302.4 µg N m−2 h−1 and 1194.1 µg N m−2 h−1 for the high and low N treatment, respectively. Fluxes from maize soil were still increasing at the end of incubation and thus ended with maximum flux rates of 809.4 µg N m−2 h−1 and 686.2 µg N m−2 h−1 in the high and low N treatment, respectively.
N2O flux over the time course (37 days) of an incubation of intact soil cores from a maize and cup plant cropping system. Soils were fertilized with 60 kg or 120 kg N ha−1 and underwent three phases of increasing waterlogging (WL; 0, 10, and 25 cm of soil saturation). Error bars show ± 1 standard deviation (n = 5)
N 2 and N 2 O fluxes from the 15 N labeled pool
The pool-derived N2O fluxes (fp_N2O) closely followed the total N2O fluxes (N2Ot) (Fig. 4; Fig. S.6) as indicated by the fraction of pool-derived fluxes in the total fluxes (fp_N2O/N2Ot = Fp_N2O). Fp_N2O was relatively high (> 0.84) throughout the observation period and did not differ (p = 0.38) between cup plant soil (0.93 ± 0.08) and maize soil (0.90 ± 0.12). The N level had a significant effect on the Fp_N2O (p = 0.02), but Fp_N2O did not differ between phases (p = 0.66, Fig. S.5).
The soil × phase interaction (p < 0.001) and the soil × N level interaction (p = 0.02) had a significant effect on the cumulated fp_N2O emissions. While fp_N2O emissions from cup plant soil was always equal or higher than from maize soil (soil effect p < 0.001, Fig. 5). As shown with the N2Ot, the N level only had an effect on the fp_N2O in cup plant soil (p = 0.04), not in maize (p = 0.3). Aside from the high N cup plant treatment, the cumulated fp_N2O emissions in phase WL0 and WL10 were relatively low (3 times the detection limit, Table S.6).
Cumulated emissions of pool-derived gaseous N fluxes (fp_N2O, fp_N2, and fp_N2 + N2O) from intact soil cores incubated for 37 days. Soils were fertilized with 60 kg or 120 kg N ha−1 and underwent three phases of increasing waterlogging (WL; 0, 10 and 25 cm of soil saturation). Black dots and error bars show estimated marginal means (n ≥ 5) and 95% confidence interval
The fp_N2 emissions were only affected by soil (p < 0.001) and phase (p < 0.001). There were no significant interactions. The coefficients of variation of fp_N2 (94.9 ± 50.7%) and fp_N2O (75.5 ± 36.8%) were comparable (p = 0.12).
In cup plant soil, fp_N2 decreased during phase WL0 from 357.8 ± 515.6 µg N m−2 h−1 to 91.51 ± 148.1 µg N m−2 h−1. During phase WL0 and WL10, fp_N2 from cup plant soil ranged between 100 and 270 µg N m−2 h−1 (Table S.6) and increased significantly (p < 0.02) by 100–200% in phase WL25 compared to phase WL10. In phase WL10, the greatest increase of fp_N2 was observed in the cup plant soil, at the high N level. In maize soil, fp_N2 started phase WL0 at 17.2 ± 17.5 µg m−2 h−1 and remained constant during phase WL0 and WL10. In phase WL25, fp_N2 in maize increased significantly (p < 0.001) by 600% and 1700% in the low and high N treatments, respectively (Table S.6). Pairwise comparison within the phases revealed that fp_N2 in phase WL0 and WL10 from maize soil were, with < 20 µg N m−2 h−1, significantly (p < 0.003) lower than those from cup plant soil (Fig. 5). In phase WL25, cup plant and maize soil only differed in fp_N2 emissions at the high N level (p = 0.001), although cumulated fluxes from maize soil were nearly one magnitude lower (Table S.6). Compared to the fp_N2O dynamic, fp_N2 in cup plant soil apparently reacted more to the establishment of phase WL10 than fp_N2O (Fig. S.8). Furthermore, in phase WL25, fp_N2 was constantly increasing toward the end of incubation, unlike fp_N2O. However, in maize soil, the increase of fp_N2 by establishing WL25 was not as pronounced as it was for fp_N2O (Fig. S.8).
The soil × phase interaction (p = 0.001) had a significant effect on the cumulated fp_N2 + N2O. Testing fp_N2 + N2O showed that cup plant soil exhibited a higher denitrification rate in phase WL0 and WL10 (both p < 0.001) and marginally higher in phase WL25 (p = 0.055) than maize soil (Fig. 5).
The N 2 Oi in the head space
In phase WL0, the product ratio of denitrification in the head space gas (N2Oi: fp_N2O/(fp_N2 + N2O)) did not differ between cup plant and maize soil (Fig. 6). In phase WL10, the N2Oi in maize (p > 0.14) and cup plant (p > 0.11) was not significantly different than in phase WL0 (Fig. 6). In phase WL25, the N2Oi in maize soil was 102.7 ± 68.1% higher (p < 0.002) than in phase WL0 (Fig. 6), corresponding to the substantial increase of fp_N2O emissions in relation to fp_N2 in phase WL25 (Table S.6). In contrast, increasing waterlogging in the cup plant soil caused simultaneous increases in both fp_N2O and fp_N2 (Fig. S.6), resulting in no change in N2Oi (p > 0.41). The N level had no effect (p = 0.40) on the N2Oi in either soil.
The mean product ratio (N2Oi = fp_N2O/(fp_N2 + N2O) and 95% confidence interval (n ≥ 5) of denitrification in intact soil cores incubated for 37 days. Soils were fertilized with 60 kg or 120 kg N ha−1 and underwent three phases of increasing waterlogging (WL; 0, 10, and 25 cm of soil saturation). Letters show significant differences between the treatments within one phase
Dissolved N 2 and N 2 O from the 15 N labeled pool
Measured concentration of dissolved fp_N2 ranged from 3.7 to 7649.3 µg N L−1 and the concentration of N2Ot ranged from 15.51 to 376.0 µg N L−1. The N2Ot and fp_N2 dissolved gas concentrations showed the same treatment effects as the N2Ot and fp_N2 emissions (Table S.9). As with the cumulated emissions in phase WL25, dissolved N2Ot did not differ between the soils (p = 0.26, Fig. 7) but they were slightly affected by N level (p = 0.04). N level had no effect on dissolved fp_N2 (p = 0.68), but dissolved N2 concentrations were higher (p < 0.001, Fig. 7) in cup plant soil than in maize soil.
Dissolved gas concentrations of pool derived N2 (fp_N2) and total N2O (N2Ot) in the soil solution of cup plant and maize soil at the end of a 37-day incubation of intact soil cores from a cup plant and maize cropping system (n = 5). For simplification, box plots were averaged over the 120 and 60 kg N ha−1 treatment
Total N emissions to the head space
Total emitted N (fp_N2 + N2Ot) was clearly dominated by the emissions in phase WL25, which masked the differences of the previous phases since only 34.5% (154.3 ± 215.6 mg N m−2 24 days−1) in cup plant and 10.4% (11.8 ± 9.4 mg N m−2 24 days−1) of total emissions in maize soil were emitted in the first two phases (Table S.7). Furthermore, total emitted N was not affected by the N level (p = 0.13). Cup plant soil emitted significantly more N (equivalent to 4.2 ± 3.0 kg N ha−1 37.4 days−1, p < 0.001) than maize soil (equivalent to 1.1 ± 0.3 kg N ha−1 37.4 days−1). In contrast to the individual N2O emissions in the single phases and N levels where soil had a significant effect (p < 0.001) on fp_N2O and N2Ot, total cumulated N2O emissions over the entire observation period were only slightly higher in cup plant than in maize soil (fp_N2O p = 0.04; N2Ot p = 0.03, see Table 3).
Discussion
Denitrification in partially waterlogged soils
The aim of our experimental setup was to mimic field conditions at marginally productive, wet to waterlogged sites, only excluding the effect of active plants. Under the tested conditions, high N2 and N2O emissions could be expected (Bronson and Fillery 1998; Friedl et al. 2016; Well et al. 2003), due to extremely reduced aeration in saturated soil favoring denitrification. However, although theory states that high denitrification is likely, there are few studies reporting direct measurements of total denitrification (N2 and N2O), which can be compared with the presented results. Anderson et al. (2014) addressed annual denitrification losses from landscapes that are prone to temporal waterlogging, comparable to the conditions mimicked in this study, by a mass balance approach. They reported landscape averaged annual denitrification losses around 45 kg N ha−1 a−1. Wendland et al. (2009) reported in a study that focused on N removal from groundwater that > 50 kg N ha−1 a−1 denitrification losses can be expected from periodically waterlogged Stagnosols of temperate climates. Interestingly, directly upscaling the results of this study results in 49 kg N ha−1 a−1, matching very closely with the values reported by Anderson et al. (2014) and Wendland et al. (2009). However, upscaling our results (or other short-term incubations) to a per-year basis almost certainly results in an overestimation, since we had waterlogged soils for over 2/3 of the incubation period, whereas field sites would only be expected to be partially waterlogged for perhaps 3–4 months of the year and the soil mineral N contents during these 3–4 months are usually lower than in our study. A more accurate upscaling of our results (assuming that our conditions are representative for 2 months in a year: early fall where high soil mineral N contents coincides with high precipitation; and late spring where fertilizer is applied on wet soils) would contribute less than 10 kg N to total annual denitrification losses. Additionally, the relatively high product ratio of denitrification in this study (N2Oi > 0.3, Fig. 6) contradicts reports from other denitrification incubations (albeit with re-packed soils, not comparable conditions or measured in micro lysimeters), under high soil moisture, where N2 was by far the predominant product of denitrification (Bronson and Fillery 1998; Friedl et al. 2016; Weier et al. 1993). Possible explanations for these differences are discussed in the next section.
The N2O flux rates from cup plant (0.6–1302.4 µg N m−2 h−1) and maize soil (0.0–809.4 µg N m−2 h−1) in the present study were in the range of the N2O fluxes from the same cup plant (− 19.7 to 371.1 µg N m−2 h−1) and maize (− 25.2 to 1955.6 µg N m−2 h−1) field, observed in a 2-year field experiment at the extraction sites (Kemmann 2022). The fact that the observed emissions were not significantly different from reported field emissions suggests that we were able to achieve our goal of field-like conditions with incubation of intact soil cores. Furthermore, emissions in the current study were much lower (measured N2O fluxes were 5 × and × lower in cup plant and maize soil, respectively) than a previous incubation study that also used intact cores from the same extraction sites (Kemmann et al. 2021). The observed low emissions in the current study could be explained by the absence of rapid changes in soil moisture or N supply throughout the observation period, which are known to boost mineralization and microbial activity (Appel 1998; Kuzyakov et al. 2000). Rapid changes, also known as hot moments, can disproportionally contribute to cumulative emissions (Groffman et al. 2009). Such rapid changes did occur in the previous study, through a dry pre-incubation and higher NO3− addition (Kemmann et al. 2021).
N 2 O and N 2 emissions with consecutively increasing water level
Denitrification products and their ratio measured in headspace gas concentrations are generally interpreted as being representative of denitrification from the entire soil. However, incubating intact soil cores under different waterlogging levels does provide very different conditions for denitrification in the individual soil depths. Therefore, our first objective was to analyze and discuss N2O and N2 emissions for each phase individually.
Drainage phase (WL0)
Prolonged phases of high N2 and N2O emissions can been observed if soil structure (i.e., due to compacted or no-till managed soil) results in persistent anaerobic conditions in the soil due to a high water content through reduced drainage (Harrison-Kirk et al. 2015; Rochette 2008). In this study, the WL0 phase was designed to specifically observe whether differences in physical soil properties between the maize and cup plant soil affected drainage and related emissions. Although the bulk densities and pore structure of the individual soil depths could not be determined, particularly the drainage phase but also the two waterlogging phases gave an approximation of differences in soil moisture (Fig. 2, t0 moisture not shown) that would be controlled by soil structure. Using those differences as proxies, we were able to show, based on changes in gravimetric water content, that the annually tilled maize soil drained faster than the denser cup plant soil. The faster drainage was presumably due to the 3.8 vol.% more total pore space in 0–20 cm previously observed by Kemmann et al. (2021) in the same soil, and may explain the consistently low fp_N2 + N2O emissions from maize soil (< 35 µg N m−2 h−1), in contrast to cup plant soil, where slower drainage may have caused the gradual decrease in fp_N2 + N2O emissions from 475 µg N m−2 hv1 at the start of WL0, to 130 µg N m−2 h−1 at the end of WL0. Although the difference in BD between the two soils was seemingly marginal (cup plant was 0.02 g cm−3 higher than maize; p = 0.01), a strong negative effect of compaction > 1.3 g cm−3 on soil aeration has been reported for fine textured soils with a moisture tension < 100 kPa (Stępniewski 1981). Therefore, the effect of reduced drainage on gaseous N emission might be particularly relevant for the tested soils (BD > 1.4 g cm−3), explaining the higher fp_N2O and fp_N2 emissions in cup plant compared with maize soil in phase WL0. Additionally, the soil gas diffusivity (Dp/D0) as calculated by Moldrup et al. (2013) could be estimated from a previous study (Kemmann et al. 2021). In 0–20 cm, under comparable conditions (moisture and texture) as in phase WL0, Dp/D0 was 0.009 and 0.019 for cup plant and maize, respectively. Therefore, at any later point than phase WL0, increased soil moisture would have caused a lower Dp/D0 that was far below the critical anaerobicity value of < 0.02 (Stępniewski 1981). Given the lower soil aeration of the cup plant compared to the maize soil, it can be concluded that the tested soil, from a relatively young cup plant stand (no tillage in the last 5 years), is already more prone to denitrification than the annually tilled soil. However, the role of bioturbation, bio-pores, and other biotic aspects affecting soil aeration might become increasingly important with stand age (Blanco-Canqui 2010; Bonin et al. 2012).
Phase of 10 cm waterlogging (WL10)
The WL10 phase tested the potential for significant denitrification losses in the lower soil depths of cup plant and maize soil. The saturated zone of hydromorphic soils provides conditions for intense denitrification (Well et al. 2005). However, while the potential for microbial denitrification is present in subsoils (Barrett et al. 2016), denitrification rates often decrease exponentially with soil depth (Jahangir et al. 2012; Luo et al. 1998). In the present study, although there was potential for denitrification in the subsoil, as seen by the slightly increased fp_N2 emissions in cup plant soil in WL10 compared to WL0 (Fig. 5), water saturation in the lower 10 cm of soil had only an insignificant effect on cumulated fp_N2 and fp_N2O emissions to the headspace (Fig. 5; Fig. S.6). This may be reflecting changes in C availability. Given anoxic conditions and the presence of NO3−, denitrifier abundance and activity, particularly in subsoils, is often constrained by C availability (Barrett et al. 2016; Dhondt et al. 2004; Jahangir et al. 2012). In the present study, N was added to all 3 depths of both tested soils, but there were significantly lower C concentrations (ca. 50%) measured in depth 3 compared to the soil above. Furthermore, root biomass under perennial crops is often higher than annual crops (Don et al. 2012; Gauder et al. 2016; Monti and Zatta 2009). Along with increased root biomass under perennial crops, Jesus et al. (2016) and Liang et al. (2012) reported a higher microbial biomass in soil of perennial biomass systems compared to an annual maize soil. The higher CO2 emissions from cup plant soil in this study (Fig. 3) suggest that a higher microbial biomass and/or root biomass was present. Not only the presence of roots, but also the quality of the C source (Barrett et al. 2016; Morley and Baggs 2010) and likely the distribution, will have a significant effect on denitrification (Loecke and Robertson 2009). Schoo et al. (2017a, b) observed a higher fine roots fraction at total root biomass in maize than cup plant, while cup plant forms thick roots resulting in a patchy distribution of root litter. Patches of organic matter provide conditions of high microbial activity where O2 consumption exceeds O2 supply by diffusion and thus favors denitrification (Kravchenko et al. 2017; Loecke and Robertson 2009). Hence, C availability and distribution is a plausible explanation for the difference between the two soils and the lack of any changes in fp_N2 or fp_N2O emissions in maize soil (Fig. 5; Fig. S.6) during phase WL10, despite the presence of strong reducing conditions in 25–35 cm. We note that three methodological points need to be considered with respect to C availability. First, we did measure soil C, and saw no significant difference between the soils (Table 1). The standard procedures for soil C measurement that we used (SOC, WEOC), however, did not account for large root fragments (visible roots are excluded when subsampling soil for analysis). Second, the simulated rain prior to the experiment, with which NO3− was washed out, could have aggravated existing C limitation due to the removal of soluble organic C (Kindler et al. 2011). Third, since the different phases were established sequentially and not in parallel, substrates for denitrification (labile C and NO3−) might have been depleted to some extent before the start of phase WL10, especially in denitrifying microsites. Increasing substrate limitations might thus have balanced increasing oxygen limitation.
Phase of 25 cm waterlogging (WL25)
The WL25 phase highlighted the potential for significant denitrification losses in the upper soil depths of cup plant and maize soil. The uppermost layer of soil is known to have the highest microbial activity and denitrifying potential because of high substrate availabilities (Luo et al. 1998; Parkin 1987; Staley et al. 1990). Jahangir et al. (2012) observed significantly higher denitrification rates from 0 to 10 cm of soil than from soil layers below. Dobbie and Smith (2006) also observed exponentially increased N2O emissions caused by increased WFPS in the uppermost soil due to a shallow ground water table in the field. In this study, near-surface waterlogging resulted in more pronounced increases in the fluxes of fp_N2O than fp_N2 from both soils and both N levels; these were, on average, between 5 × and 23 × higher compared to phase WL10 (Fig. 5).
The stronger increase of fp_N2O compared to fp_N2 with the establishment of phase WL25 was not in line with observations from previous denitrification studies, where the predominant product of denitrification was N2 at high soil moistures (Bronson and Fillery 1998; Friedl et al. 2016; Rudaz et al. 1999; Weier et al. 1993). This difference may be reflecting the specific soil depth where denitrification occurred. In contrast to the experimental design of Bronson and Fillery (1998), while the gravimetric water content in the top 10 cm increased during WL25 (2.8% (p = 0.001) in maize and by 1.6% (p = 0.06) in cup plant soil; Fig. 2), the top 10 cm of soil was not itself waterlogged. Therefore, due to capillary rise in the top 10 cm of soil, soil moisture conditions may have been more favorable for N2O emissions (i.e., < 80% WFPS; Butterbach-Bahl et al. (2013)), than for N2 emissions. The capillary rise, which is closely associated with soil structure, was apparently more pronounced in maize than cup plant soil. Since no similar observation of N emissions from the boundary layer between depths 2 and 3 were made in phase WL10, it is likely that the fp_N2O emissions from depth 1 dominated the N2Ot emissions in phase WL25.
The increase in fp_N2 due to the establishment of phase WL25 was comparable or less pronounced than the increase in fp_N2O (5 × in cup plant soil and 11 × in maize soil higher than in phase WL10). In saturated soil, a time lag between the actual denitrification (NO3− consumption) and the emission of N2 at the soil surface can be expected, due to impeded gas diffusion (Friedl et al. 2016). However, in this study, fp_N2 and fp_N2O emissions in both soils started to increase simultaneously around day 27, which does not indicate a significant time lag between the emission of fp_N2 and fp_N2O (Fig. S.6). This suggests that both species were produced in close temporal and spatial proximity to each other. Given the assumption that the N2O was produced under the less reducing conditions in depth 1, it is likely that, especially at the beginning of phase WL25 (day 27–33), the top 10 cm was also the origin of the majority of fp_N2. Further indications for this assumption are given by the fact that the active pool producing N2 (apN2) was more comparable to the labeled NO3− (15aNO3) of depth 1 than that of the soil below. For example, in the high N treatment for cup plant, apN2 was 47 atom% and 15aNO3 of depth 1 was 48 atom%, whereas 15aNO3 was 49 and 55 atom% in depths 2 and 3, respectively (Table S.4). Therefore, the contribution from N2 produced and/or N2O reduced in the saturated zone to the head space emissions was unexpectedly low.
Nitrate consumption in saturated soil
This experiment was conducted using two N levels (60 kg NO3-N ha−1 and 120 kg NO3-N ha−1), in order to explore the effect of a differing NO3− concentration on denitrification. Both the stimulation of N2O formation through increased substrate availability and the inhibition of N2O reduction are possible with higher N (Senbayram et al. 2012; Weier et al. 1993). Interestingly, there was only a slight increase in N2O emissions (both N2Ot and fp_N2O) at the higher N level in cup plant soil (p < 0.05), while no effect was observed in maize soil (p > 0.3). The N rate had no significant effect on the N2Oi (p = 0.40, Fig. 6) in either soil, suggesting that no substantial inhibition of N2O reduction occurred. Overall, these observations support other studies that have shown that mineral N content is not always the most important explanatory variable for N2O emissions (Rochette et al. 2008) or the N2Oi (Kemmann et al. 2021).
While NO3− availability did not clearly affect measured gas emissions in the headspace, there was evidence that it affected NO3− consumption in the saturated soil. Mineral N was highly variable between columns in each of the four treatments (Table 2). However, based on the average change in NO3− content in high N cup plant soil, there was more NO3− consumption in the saturated soil depths (loss of 24.7 ± 20.9 kg N ha−1 from the beginning of WL10 to the end of WL25), than in the other three treatments (in maize a net NO3− production of 0.6 ± 22.0 kg N ha−1 occurred). As increased NO3− consumption was not reflected in the N2 + N2O surface emissions, we explore here other potential explanations of that NO3− loss, focusing on the high N cup plant treatment.
Separated by depth, NO3− losses were observed both in depth 2 (loss of 9.0 ± 12.9 kg N ha−1 during phase WL25) and in depth 3 (loss of 15.4 ± 16.2 kg N ha−1 across phase WL10 and WL25, Table 2). Our measured surface fluxes only explained a NO3-N loss of 5.4 ± 3.9 kg N ha−1 (Table 3). However, with an expected lag time between denitrification and emission (Friedl et al. 2016), it is possible that our incubation period may have been too short to see activity from the saturated layers, due to the extremely low gas diffusivity in water and resulting accumulation of produced N2 (Well et al. 2001). This is indicated both by the increase in fp_N2 fluxes that we observed near the end of our incubation (Fig. S.6), and by the dissolved gas concentrations at the end of WL25 (Fig. 7), which showed that some of the denitrification products had built up in the saturated layers (Well and Myrold 1999). Our estimated upscaling of the dissolved gases (Table S.9) accounted for another 1.2 ± 1.8 kg N ha−1 of NO3− loss. This increases the directly measured denitrification losses to 6.6 ± 5.8 kg N ha−1, which represents just under 27% of the observed 24.7 kg N ha−1 loss. However, measurement of dissolved gases shows only those gaseous denitrification products that are trapped in the liquid phase, and excludes the gases accumulated in encapsulated gas bubbles. Bubbles typically occur during the rise of the water table in the soil (Fayer and Hillel 1986). Therefore, a possible source of ‘missed’ denitrification activity may have been entrapped N2O and N2. It is possible for a significant portion of produced N2 to accumulate in gas bubbles that are either entrapped during saturation (Well and Myrold 1999) or newly formed when N2 production by denitrification is in excess of gas solubility (Blicher-Mathiesen et al. 1998) due to the low solubility of N2 (Wilhelm et al. 1977). Assuming a WFPS between 90 and 100% in the waterlogged soil layers, dissolved N2 may only reflect part of the total accumulated N2 in the saturated layers (between 15 and 100%; see calculations in Table S.10). Thus, N2 trapped in bubbles could theoretically account for up to 7.1 kg N ha−1 of additional denitrification that occurred, but was not captured through headspace or dissolved gas measurements. Measured (dissolved gas + headspace flux, 6.6 kg N ha−1) and estimated (trapped N2 in bubbles, 7.1 kg N ha) denitrification losses account for up to 55% (13.7 of 24.7 kg N ha−1) of total NO3− loss observed in the saturated soil in the high N cup plant soil, which was in the range of N losses explained by denitrification reported by Matheson et al. (2002) in a mass balance study from submerged riparian soil. Future studies should target methods to directly measure entrapped N2 and N2O, as it has the potential to cause significant underestimations of denitrification activity in saturated soil.
Aside from denitrification, other processes that consume NO3− include dissimilatory nitrate reduction to NH4+ (DNRA) and immobilization. At the end of the incubation, the much higher recovery of 15 N in the cup plant soil extractable NH4+ (Fig. S.2) shows that much more of the 15 N-labeled NO3− was transferred to the NH4+-pool in cup plant soil than in maize soil. In the saturated soil layers, the high 15aNH4 could be due to DNRA (Rütting et al. 2011). DNRA transforms NO3− into the more biologically available NH4+ (Burgin and Hamilton 2007), and has been shown to account for 50% of NO3 loss in unplanted riparian soil (Matheson et al. 2002). The retention of N in the form of less mobile NH4+ in the soil via DNRA can substantially differ between soils under different plant species (Shi et al. 2020). Since NH4+ formed through DNRA can be preferably immobilized in organic forms (Burgin and Hamilton 2007), DNRA occurring in the cup plant soils of this study could explain why the total extractable NH4+ content did not increase significantly, but the pool was enriched by 18 atom% 15 N. Microbial immobilization and re-mineralization (Azam et al. 1988) might additionally explain up to 25% of NO3− loss under waterlogged conditions (Matheson et al. 2002). Therefore, in the saturated layers of cup plant soil, NO3− loss was likely caused by a combination of denitrification, DNRA, and immobilization. In comparison, these processes appeared to be less active in maize soil, where net NO3− production was observed (Table 2).
N 2 O emissions and its reduction to N 2
In view of the higher N2O emissions from cup plant soil than maize soil, the main hypothesis of the presented study was not confirmed. Cup plant soil did not emit less N2O than the reference maize soil due to an increased reduction to N2 with increasing waterlogging. Cup plant soil had, in general, higher fp_N2 + N2O fluxes than maize soil (p < 0.02, Table 3), and at no point during the experiment did this enhanced N2O reduction balance the higher gross N2O formation (Fig. 6). Denitrification from soil depth 1 was disproportionally contributing to emissions of N2O and to a smaller extent also N2, so that other aspects, such as prolonged residence time and strong reducing conditions in the saturated soil below the 10 cm of soil depth, were not decisive for total N emissions at the soil surface.
Although not associated with less N2O emissions, the conditions in the soil cultivated with cup plant were more favorable for the reduction of N2O to N2, which was evident from the surface emissions (Fig. 6), but also from dissolved gas analysis (Fig. 7). As previously discussed, this was likely due to C availability and soil structure, pointing out another substantial difference between the soils. The lower N2O reduction in maize soil cannot simply be explained by an inhibitory effect of O2 on N2O reductase (Morley et al. 2008) in the better aerated maize soil, since the N2Oi measured in the dissolved gas in the saturated soil was also significantly higher than in cup plant soil (Table S.9). An inhibition of the expression of N2O reductase due to a low soil pH (Raut et al. 2012) in maize can also be excluded, because the pH was significantly higher than in cup plant soil. However, the slightly lower pH might have had an effect on the Cu mobility in cup plant soil. Shen et al. (2020) reported an increased N2O reduction with increased Cu availability. Along with the different N cycling (e.g., occurrence of DNRA) in cup plant soil, limited N2O reduction in maize soil could be due to land use and management related effects on the composition of the microbial community (Cavigelli and Robertson 2000; Domeignoz-Horta et al. 2015; Hargreaves and Hofmockel 2014; Jangid et al. 2008; Maul et al. 2019). In comparison with annual systems, perennial cropping systems have a higher abundance of N2O reducers of the clade II, but not of the more common clade I (Domeignoz-Horta et al. 2015). The recent development of new primers for the clades I (Zhang et al. 2021) and II (Chee-Sanford et al. 2020) of nosZ gene might further elucidate the diversity of the denitrifier community under different cropping systems.
Conclusion
In the absence of living plants, the soil from the perennial cup plant field generally exhibited higher denitrification rates than the soil from maize cropping, thus providing an increased risk of N loss via denitrification. With increasing waterlogging, the reduction of N2O to N2, under the tested conditions, did not increase as much as total denitrification in either soil. The cultivation of the perennial biomass crop did not result in soil properties that caused less N2O emissions than maize soil. Therefore, the potential to mitigate N2O emissions through more complete reduction by changing from an annual silage maize cropping to perennial cup plant cropping remains hypothetical.
In the direct comparison of maize and cup plant soil, maize soil exhibited a significantly lower reduction of N2O to N2 under waterlogged conditions than cup plant soil. Given that the parent material was the same in the two cropping systems, this suggests that each cropping systems had a strong influence on the mechanism controlling denitrification in that soil. This mechanism is likely related to differences in C availability, soil structure, and the composition of the microbial community, but we are unable to identify a clear explanation for the inhibited N2O reduction in the maize soil. Nonetheless, this study clearly demonstrated that even under the same soil and climatic conditions, annual and perennial land use systems can strongly influence the product ratio of denitrification. Conclusively, our results therefore stressed that denitrification losses and its implication for the cropping system’s N use efficiency in biomass cultivation should not simply be estimated based on constant product ratios of denitrification across annual and perennial cropping systems.
Data availability
The datasets generated during and/or analyzed during the current study are available in the OpenAgrar repository, https://doi.org/10.3220/DATA20220922104041 or is available from the corresponding author.
References
Amougou N, Bertrand I, Cadoux S, Recous S (2012) Miscanthus× giganteus leaf senescence, decomposition and C and N inputs to soil. GCB Bioen 4:698–707
Anderson TR, Goodale CL, Groffman PM, Walter MT (2014) Assessing denitrification from seasonally saturated soils in an agricultural landscape: a farm-scale mass-balance approach. Agricult Ecosyst Environ 189:60–69
Appel T (1998) Non-biomass soil organic N—the substrate for N mineralization flushes following soil drying–rewetting and for organic N rendered CaCl2-extractable upon soil drying. Soil Biol Biochem 30:1445–1456
Azam F, Mahmood T, Malik K (1988) Inmobilization-remineralization of NO3-N and total N balance during the decomposition of glucose, sucrose and cellulose in soil incubated at different moisture regimes. Plant Soil 107:159–163
Balesdent J, Chenu C, Balabane M (2000) Relationship of soil organic matter dynamics to physical protection and tillage. Soil till Res 53:215–230
Ball BC, Scott A, Parker JP (1999) Field N2O, CO2 and CH4 fluxes in relation to tillage, compaction and soil quality in Scotland. Soil till Res 53:29–39
Barrett M, Khalil M, Jahangir MMR, Lee C, Cárdenas LM, Collins G, Richards KG, O’Flaherty V (2016) Carbon amendment and soil depth affect the distribution and abundance of denitrifiers in agricultural soils. Environ Sci Pollut Res 23:7899–7910
Blanco-Canqui H (2010) Energy crops and their implications on soil and environment. Agron J 102:403–419
Blanco-Canqui H (2016) Growing dedicated energy crops on marginal lands and ecosystem services. Soil Sci Soc Am J 80:845–858
Blicher-Mathiesen G, McCarty G, Nielsen L (1998) Denitrification and degassing in groundwater estimated from dissolved dinitrogen and argon. J Hydrol 208:16–24
Bonin C, Lal R, Schmitz M, Wullschleger S (2012) Soil physical and hydrological properties under three biofuel crops in Ohio. Acta Agriculturae Scandinavica, Section B - Soil Plant Sci 62:595–603
Bronson K, Fillery I (1998) Fate of nitrogen-15-labelled urea applied to wheat on a waterlogged texture-contrast soil. Nutr Cycl Agroecosyst 51:175–183
Burgin AJ, Hamilton SK (2007) Have we overemphasized the role of denitrification in aquatic ecosystems? A review of nitrate removal pathways. Front Ecol Environ 5:89–96
Butterbach-Bahl K, Baggs EM, Dannenmann M, Kiese R, Zechmeister-Boltenstern S (2013) Nitrous oxide emissions from soils: how well do we understand the processes and their controls? Philos Trans R Soc B 368:20130122
Cavigelli MA, Robertson GP (2000) The functional significance of denitrifier community composition in a terrestrial ecosystem. Ecology 81:1402–1414
Chapuis-Lardy L, Wrage N, Metay A, Chotte JL, Bernoux M (2007) Soils, a sink for N2O? A review. Glob Chang Biol 13:1–17
Chee-Sanford JC, Connor L, Krichels A, Yang WH, Sanford RA (2020) Hierarchical detection of diverse Clade II (atypical) nosZ genes using new primer sets for classical-and multiplex PCR array applications. J Microbiol Meth 172:105908
Ciarlo E, Conti M, Bartoloni N, Rubio G (2007) The effect of moisture on nitrous oxide emissions from soil and the N2O/(N2O+N2) ratio under laboratory conditions. Biol Fertil Soils 43:675–681
Dhondt K, Boeckx P, Hofman G, Van Cleemput O (2004) Temporal and spatial patterns of denitrification enzyme activity and nitrous oxide fluxes in three adjacent vegetated riparian buffer zones. Biol Fertil Soils 40:243–251
Directive (EU) 2018/2001 of the European Parliament and of the council of 11 December 2018 on the promotion of the use of energy from renewable sources (2018)
Dobbie K, Smith K (2006) The effect of water table depth on emissions of N2O from a grassland soil. Soil Use Manag 22:22–28
Domeignoz-Horta L, Spor A, Bru D, Bizouard F, Leonard J, Philippot L (2015) The diversity of the N2O reducers matters for the N2O: N2 denitrification end-product ratio across an annual and a perennial cropping system. Front Microbiol 6:971
Don A, Osborne B, Hastings A, Skiba U, Carter MS, Drewer J, Flessa H, Freibauer A, Hyvönen N, Jones MB (2012) Land-use change to bioenergy production in Europe: implications for the greenhouse gas balance and soil carbon. GCB Bioen 4:372–391
Drewer J, Finch JW, Lloyd CR, Baggs EM, Skiba U (2012) How do soil emissions of N2O, CH4 and CO2 from perennial bioenergy crops differ from arable annual crops? GCB Bioen 4:408–419
Dyckmans J, Eschenbach W, Langel R, Szwec L, Well R (2021) Nitrogen isotope analysis of aqueous ammonium and nitrate by membrane inlet isotope ratio mass spectrometry (MIRMS) at natural abundance levels. Rapid Commun Mass Spectrom 35:e9077
Fayer M, Hillel D (1986) Air encapsulation: II. Profile water storage and shallow water table fluctuations. Soil Sci Soc Am J 50:572–577
Ferchaud F, Peyrard C, Léonard J, Gréhan E, Mary B (2020) Large variations in N2O fluxes from bioenergy crops according to management practices and crop type. Atmosphere 11:675–699
Firestone M (1982) Biological denitrification. In: Stevenson FJ (ed) Nitrogen in agricultural soils. American Society of Agronomy, Madison, WI, pp 289–326
Firestone M, Smith M, Firestone R, Tiedje J (1979) The influence of nitrate, nitrite, and oxygen on the composition of the gaseous products of denitrification in soil. Soil Sci Soc Am J 43:1140–1144
Firestone M, Davidson E (1989) Microbiological basis of NO and N2O production and consumption in soil. In: Andreae MO, Schimel DS (eds) Exchange of the trace gases between terrestrial ecosystems and the atmosphere. Report for the Dahlem Workshop on Exchange of Gases Between Terrestrial Ecosystems and the Atmosphere. Wiley, New York, pp 7–22
Forster P, Storelvmo T, Armour K, Collins W., Dufresne JL, Frame D, Lunt DJ, Mauritsen T, Palmer MD, Watanabe M, Wild M, Zhang H (2021) The Earth’s energy budget, climate feedbacks, and climate sensitivity. In Masson-Delmotte V, Zhai P, Pirani A, Connors S.L, Péan C, Berger S, Caud N, Chen Y, Goldfarb L, Gomis M.I, Huang M, Leitzell K, Lonnoy E, Matthews JBR, Maycock TK, Waterfield T, Yelekçi O, Yu R, Zhou B (eds), Climate change 2021: the physical science basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, UK, pp. 923–1054
Friedl J, Scheer C, Rowlings DW, McIntosh HV, Strazzabosco A, Warner DI, Grace PR (2016) Denitrification losses from an intensively managed sub-tropical pasture–Impact of soil moisture on the partitioning of N2 and N2O emissions. Soil Biol Biochem 92:58–66
Gauder M, Butterbach-Bahl K, Graeff-Hönninger S, Claupein W, Wiegel R (2012) Soil-derived trace gas fluxes from different energy crops—results from a field experiment in southwest Germany. GCB Bioen 4:289–301
Gauder M, Billen N, Zikeli S, Laub M, Graeff-Hönninger S, Claupein W (2016) Soil carbon stocks in different bioenergy cropping systems including subsoil. Soil till Res 155:308–317
Groffman PM, Butterbach-Bahl K, Fulweiler RW, Gold AJ, Morse JL, Stander EK, Tague C, Tonitto C, Vidon P (2009) Challenges to incorporating spatially and temporally explicit phenomena (hotspots and hot moments) in denitrification models. Biogeochemistry 93:49–77
Grunwald D, Panten K, Schwarz A, Bischoff WA, Schittenhelm S (2020) Comparison of maize, permanent cup plant and a perennial grass mixture with regard to soil and water protection. GCB Bioen 12:694–705
Haberl H, Sprinz D, Bonazountas M, Cocco P, Desaubies Y, Henze M, Hertel O, Johnson RK, Kastrup U, Laconte P (2012) Correcting a fundamental error in greenhouse gas accounting related to bioenergy. Energy Policy 45:18–23
Hantschel RE, Flessa H, Beese F (1994) An automated microcosm system for studying soil ecological processes. Soil Sci Soc Am J 58:401–404
Hargreaves SK, Hofmockel KS (2014) Physiological shifts in the microbial community drive changes in enzyme activity in a perennial agroecosystem. Biogeochemistry 117:67–79
Harrison-Kirk T, Thomas SM, Clough TJ, Beare MH, van der Weerden TJ, Meenken ED (2015) Compaction influences N2O and N2 emissions from 15N-labeled synthetic urine in wet soils during successive saturation/drainage cycles. Soil Biol Biochem 88:178–188
Hauck R, Bouldin D (1961) Distribution of isotopic nitrogen in nitrogen gas during denitrification. Nature 191:871–872
Herbes C, Jirka E, Braun JP, Pukall K (2014) Der gesellschaftliche Diskurs um den, Maisdeckel “vor und nach der Novelle des Erneuerbare-Energien-Gesetzes (EEG) 2012, The social discourse on the “Maize Cap “ before and after the 2012 amendment of the German Renewable Energies Act (EEG). GAIA-Ecol Perspect Sci Soc 23:100–108
IUSS WG (2015) World reference base for soil resources 2014 (update 2015). International soil classification system for naming soils and creating legends for soil maps, World Soil Resource Reports 106
Jahangir MM, Khalil MI, Johnston P, Cardenas L, Hatch D, Butler M, Barrett M, O’flaherty V, Richards KG (2012) Denitrification potential in subsoils: a mechanism to reduce nitrate leaching to groundwater. Agricult Ecosyst Environ 147:13–23
Jangid K, Williams MA, Franzluebbers AJ, Sanderlin JS, Reeves JH, Jenkins MB, Endale DM, Coleman DC, Whitman WB (2008) Relative impacts of land-use, management intensity and fertilization upon soil microbial community structure in agricultural systems. Soil Biol Biochem 40:2843–2853
Jesus EDC, Liang C, Quensen JF, Susilawati E, Jackson RD, Balser TC, Tiedje JM (2016) Influence of corn, switchgrass, and prairie cropping systems on soil microbial communities in the upper Midwest of the United States. GCB Bioen 8:481–494
Jin VL, Schmer MR, Stewart CE, Mitchell RB, Williams CO, Wienhold BJ, Varvel GE, Follett RF, Kimble J, Vogel KP (2019) Management controls the net greenhouse gas outcomes of growing bioenergy feedstocks on marginally productive croplands. Sci Advances 5:eaav9318
Jørgensen RN, Jørgensen BJ, Nielsen NE, Maag M, Lind A-M (1997) N2O emission from energy crop fields of Miscanthus “Giganteus” and winter rye. Atmos Environ 31:2899–2904
Kay B, VandenBygaart A (2002) Conservation tillage and depth stratification of porosity and soil organic matter. Soil till Res 66:107–118
Kemmann B, Wöhl L, Fuß R, Schrader S, Well R, Ruf T (2021) N2 and N2O mitigation potential of replacing maize with the perennial biomass crop Silphium perfoliatum—an incubation study. GCB Bioen 13:1649–1665
Kemmann B (2022) Assessment of field-derived greenhouse gas mitigation potential in biomass production by replacing maize with cup plant (Silphium perfoliatum) in low mountain ranges. Dissertation, University of Kiel
Kindler R, Siemens J, Kaiser K, Walmsley DC, Bernhofer C, Buchmann N, Cellier P, Eugster W, Gleixner G, Grunwald T (2011) Dissolved carbon leaching from soil is a crucial component of the net ecosystem carbon balance. Glob Change Biol 17:1167–1185
Kravchenko A, Toosi E, Guber A, Ostrom N, Yu J, Azeem K, Rivers M, Robertson G (2017) Hotspots of soil N2O emission enhanced through water absorption by plant residue. Nature Geosci 10:496–500
Kuzyakov Y, Friedel J, Stahr K (2000) Review of mechanisms and quantification of priming effects. Soil Biol Biochem 32:1485–1498
Lewicka-Szczebak D, Well R, Giesemann A, Rohe L, Wolf U (2013) An enhanced technique for automated determination of 15N signatures of N2,(N2+N2O) and N2O in gas samples. Rapid Commun Mass Spectrom 27:1548–1558
Lewicka-Szczebak D, Augustin J, Giesemann A, Well R (2017) Quantifying N2O reduction to N2 based on N2O isotopocules—validation with independent methods (helium incubation and 15N gas flux method). Biogeosciences 14:711–732
Li X, Sørensen P, Olesen JE, Petersen SO (2016) Evidence for denitrification as main source of N2O emission from residue-amended soil. Soil Biol Biochem 92:153–160
Liang C, Jesus EDC, Duncan DS, Jackson RD, Tiedje JM, Balser TC (2012) Soil microbial communities under model biofuel cropping systems in southern Wisconsin, USA: impact of crop species and soil properties. Appl Soil Ecol 54:24–31
Loecke TD, Robertson GP (2009) Soil resource heterogeneity in terms of litter aggregation promotes nitrous oxide fluxes and slows decomposition. Soil Biol Biochem 41:228–235
Luo J, Tillman R, White R, Ball P (1998) Variation in denitrification activity with soil depth under pasture. Soil Biol Biochem 30:897–903
Magnusson A, Skaug H, Nielsen A, Berg C, Kristensen K, Maechler M, van Bentham K, Bolker B, Brooks M, Brooks MM. (2017) Package ‘glmmTMB’. R Package Version 0.2. 0
Matheson F, Nguyen M, Cooper A, Burt T, Bull D (2002) Fate of 15N-nitrate in unplanted, planted and harvested riparian wetland soil microcosms. Ecol Eng 19:249–264
Maul JE, Cavigelli MA, Vinyard B, Buyer JS (2019) Cropping system history and crop rotation phase drive the abundance of soil denitrification genes nirK, nirS and nosZ in conventional and organic grain agroecosystems. Agricult Ecosyst Environ 273:95–106
Moldrup P, Chamindu Deepagoda TKK, Hamamoto S, Komatsu T, Kawamoto K, Rolston DE, de Jonge LW (2013) Structure-dependent water-induced linear reduction model for predicting gas diffusivity and tortuosity in repacked and intact soil. Vadose Zone J 12:0026
Monti A, Zatta A (2009) Root distribution and soil moisture retrieval in perennial and annual energy crops in Northern Italy. Agricult Ecosyst Environ 132:252–259
Morley N, Baggs EM (2010) Carbon and oxygen controls on N2O and N2 production during nitrate reduction. Soil Biol Biochem 42:1864–1871
Morley N, Baggs E, Dörsch P, Bakken L (2008) Production of NO, N2O and N2 by extracted soil bacteria, regulation by NO2− and O2 concentrations. FEMS Microbiol Ecol 65:102–112
Ostrom NE, Sutka R, Ostrom PH, Grandy AS, Huizinga KM, Gandhi H, von Fischer JC, Robertson GP (2010) Isotopologue data reveal bacterial denitrification as the primary source of N2O during a high flux event following cultivation of a native temperate grassland. Soil Biol Biochem 42:499–506
Palm C, Blanco-Canqui H, DeClerck F, Gatere L, Grace P (2014) Conservation agriculture and ecosystem services: an overview. Agricult Ecosyst Environ 187:87–105
Parkin TB (1987) Soil microsites as a source of denitrification variability. Soil Sci Soc Am J 51:1194–1199
Pinheiro J, Bates D, DebRoy S, Sarkar D (2020) R Core Team. 2020. nlme: linear and nonlinear mixed effects models. R package version 3.1–147. URL: https://CRAN.R-project.org/package=nlme.
Raut N, Dörsch P, Sitaula BK, Bakken LR (2012) Soil acidification by intensified crop production in South Asia results in higher N2O/(N2+N2O) product ratios of denitrification. Soil Biol Biochem 55:104–112
Reddy K, Patrick W Jr, Phillips R (1978) The role of nitrate diffusion in determining the order and rate of denitrification in flooded soil: I. Experimental results. Soil Sci Soc Am J 42:268–272
Rochette P (2008) No-till only increases N2O emissions in poorly-aerated soils. Soil till Res 101:97–100
Rochette P, Angers DA, Chantigny MH, Bertrand N (2008) Nitrous oxide emissions respond differently to no-till in a loam and a heavy clay soil. Soil Sci Soc Am J 72:1363–1369
Rudaz A, Wälti E, Kyburz G, Lehmann P, Fuhrer J (1999) Temporal variation in N2O and N2 fluxes from a permanent pasture in Switzerland in relation to management, soil water content and soil temperature. Agric Ecosyst Environ 73:83–91
Ruf T, Emmerling C (2021) Different life-form strategies of perennial energy crops and related nutrient exports require a differentiating view specifically concerning a sustainable cultivation on marginal land. GCB Bioen 13:893–904
Russow R, Stevens R, Laughlin R (1996) Accuracy and precision for measurements of the mass ratio 30/28 in dinitrogen from air samples and its application to the investigation of N losses from soil by denitrification. Isotopes Environ Health Stud 32:289–297
Rütting T, Boeckx P, Müller C, Klemedtsson L (2011) Assessment of the importance of dissimilatory nitrate reduction to ammonium for the terrestrial nitrogen cycle. Biogeosciences 8:1779–1791
Saggar S, Jha N, Deslippe J, Bolan N, Luo J, Giltrap D, Kim D-G, Zaman M, Tillman R (2013) Denitrification and N2O: N2 production in temperate grasslands: processes, measurements, modelling and mitigating negative impacts. Sci Tot Environ 465:173–195
Scheer C, Fuchs K, Pelster DE, Butterbach-Bahl K (2020) Estimating global terrestrial denitrification from measured N2O:(N2O+N2) product ratios. Curr Opin in Environ Sustain 47:72–80
Schlesinger WH (2009) On the fate of anthropogenic nitrogen. Proc Natl Academy Sci 106:203–208
Schoo B, Kage H, Schittenhelm S (2017) Radiation use efficiency, chemical composition, and methane yield of biogas crops under rainfed and irrigated conditions. Eur J Agron 87:8–18
Schoo B, Schroetter S, Kage H, Schittenhelm S (2017) Root traits of cup plant, maize and lucerne grass grown under different soil and soil moisture conditions. J Agron Crop Sci 203:345–359
Senbayram M, Chen R, Budai A, Bakken L, Dittert K (2012) N2O emission and the N2O/(N2O+N2) product ratio of denitrification as controlled by available carbon substrates and nitrate concentrations. Agric Ecosyst Environ 147:4–12
Shen W, Xue H, Gao N, Shiratori Y, Kamiya T, Fujiwara T, Isobe K, Senoo K (2020) Effects of copper on nitrous oxide (N2O) reduction in denitrifiers and N2O emissions from agricultural soils. Biol Fertil Soils 56:39–51
Shi X, Wang J, Müller C, Hu H-W, He J-Z, Wang J, Huang Z (2020) Dissimilatory nitrate reduction to ammonium dominates soil nitrate retention capacity in subtropical forests. Biol Fertil Soils 56:785–797
Shortall O (2013) “Marginal land” for energy crops: exploring definitions and embedded assumptions. Energy Policy 62:19–27
Soane BD, Ball BC, Arvidsson J, Basch G, Moreno F, Roger-Estrade J (2012) No-till in northern, western and south-western Europe: a review of problems and opportunities for crop production and the environment. Soil till Res 118:66–87
Spott O, Russow R, Apelt B, Stange CF (2006) A 15N-aided artificial atmosphere gas flow technique for online determination of soil N2 release using the zeolite Köstrolith SX6®. Rapid Commun Mass Spectrom 20:3267–3274
Staley T, Boyer D, Caskey W (1990) Soil denitrification and nitrification potentials during the growing season relative to tillage. Soil Sci Soc Am J 54:1602–1608
Stępniewski W (1981) Oxygen diffusion and strength as related to soil compaction. II. Oxygen diffusion coefficient. Polish J Soil Sci 14:3–13
Van Den Bossche A, De Bolle S, De Neve S, Hofman G (2009) Effect of tillage intensity on N mineralization of different crop residues in a temperate climate. Soil till Res 103:316–324
Walter K, Don A, Flessa H (2015) Net N2O and CH4 soil fluxes of annual and perennial bioenergy crops in two central German regions. Biomass Bioen 81:556–567
Weier K, Doran J, Power J, Walters D (1993) Denitrification and the dinitrogen/nitrous oxide ratio as affected by soil water, available carbon, and nitrate. Soil Sci Soc Am J 57:66–72
Well R, Myrold DD (1999) Laboratory evaluation of a new method for in situ measurement of denitrification in water-saturated soils. Soil Biol Biochem 31:1109–1119
Well R, Augustin J, Davis J, Griffith S, Meyer K, Myrold D (2001) Production and transport of denitrification gases in shallow ground water. Nutr Cycl Agroecosyst 60:65–75
Well R, Augustin J, Meyer K, Myrold D (2003) Comparison of field and laboratory measurement of denitrification and N2O production in the saturated zone of hydromorphic soils. Soil Biol Biochem 35:783–799
Well R, Höper H, Mehranfar O, Meyer K (2005) Denitrification in the saturated zone of hydromorphic soils—laboratory measurement, regulating factors and stochastic modeling. Soil Biol Biochem 37:1822–1836
Wendland F, Behrendt H, Gömann H, Hirt U, Kreins P, Kuhn U, Kunkel R, Tetzlaff B (2009) Determination of nitrogen reduction levels necessary to reach groundwater quality targets in large river basins: the Weser basin case study, Germany. Nutr Cycl Agroecosyst 85:63–78
Weninger T, Kreiselmeier J, Chandrasekhar P, Julich S, Feger K-H, Schwärzel K, Bodner G, Schwen A (2019) Effects of tillage intensity on pore system and physical quality of silt-textured soils detected by multiple methods. Soil Res 57:703–711
Wilhelm E, Battino R, Wilcock RJ (1977) Low-pressure solubility of gases in liquid water. Chem Reviews 77:219–262
Zhang B, Penton CR, Yu Z, Xue C, Chen Q, Chen Z, Yan C, Zhang Q, Zhao M, Quensen JF (2021) A new primer set for Clade I nosZ that recovers genes from a broader range of taxa. Biol Fertil Soils 57:523–531
Acknowledgements
We would like to thank Andreas Kirch for the support in the field and Stefan Burkart for the assistance with the mesocosm facility. Furthermore, we would like to thank the technicians from the isotope and the soil laboratory at the Thünen-Institute for Climate-Smart Agriculture for analyzing all the samples: Martina Heuer, Jennifer Giere and Ute Rieß, Ute Tambor, Andrea Niemeyer, and Claudia Wiese.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was funded by the Fachagentur Nachwachsende Rohstoffe e.V. (FNR, Agency for Renewable Resources, Germany) through the project BESTLAND (FKZ: 22403618 and 22404217): Greenhouse gas mitigation potential, soil and biodiversity conservation of perennial biomass cropping on temporal waterlogged (pseudogleyic) sites.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kemmann, B., Ruf, T., Matson, A. et al. Waterlogging effects on N2O and N2 emissions from a Stagnosol cultivated with Silphium perfoliatum and silage maize. Biol Fertil Soils 59, 53–71 (2023). https://doi.org/10.1007/s00374-022-01673-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-022-01673-6