Abstract
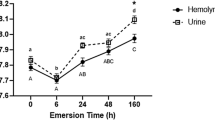
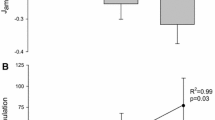
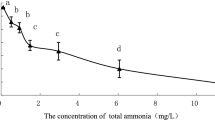
The objective of this study was to elucidate how the African lungfish, Protopterus annectens, ameliorated ammonia toxicity during 12 or 46 days of aestivation in air or in mud. Twelve days of aestivation in air led to significant increases in contents of urea, but not ammonia, in tissues of P. annectens. The estimated rate of urea synthesis increased 2.7-fold despite the lack of changes in the activities of hepatic ornithine–urea cycle enzymes, but there was only a minor change in the estimated rate of ammonia production. After 46 days of aestivation in air, the ammonia content in the liver decreased significantly and contents of urea in all tissues studied increased significantly, indicating that the fish shifted to a combination of increased urea synthesis (1.4-fold of the day 0 value) and decreased ammonia production (56% of the day 0 value) to defend against ammonia toxicity. By contrast, 12 days of aestivation in mud produced only minor increases in tissue urea contents, with ammonia contents remained unchanged. This was apparently achieved through decreases in urea synthesis and ammonia production (40 and 15%, respectively, of the corresponding day 0 value). Surprisingly, 46 days of aestivation in mud resulted in no changes in tissue urea contents, indicating that profound suppressions of urea synthesis and ammonia production (2.6 and 1.2%, respectively, of the corresponding day 0 value) had occurred. This is the first report on such a phenomenon, and the reduction in ammonia production was so profound that it could be the greatest reduction known among animals. Since fish aestivated in mud had relatively low blood pO2 and muscle ATP content, they could have been exposed to hypoxia, which induced reductions in metabolic rate and ammonia production. Consequently, fish aestivating in mud had a lower dependency on increased urea synthesis to detoxify ammonia, which is energy intensive, than fish aestivating in air.



Similar content being viewed by others
References
Anderson PM, Walsh PJ (1995) Subcellular localization and biochemical properties of the enzyme carbamoyl phosphate synthetase and urea synthesis in the batrachoidid fishes, Opsanus beta, Opsanus tau and Porichthys notatus. J Exp Biol 198:755–766
Bergmeyer HU, Beutler HO (1985) Ammonia. In: Bergmeyer HU, Bergmeyer J, Graßl M (eds) Methods of enzymatic analysis, vol VIII. Verlag Chemie, Weinheim, pp 454–461
Bradford MM (1976) A rapid and sensitive method of the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254
Brusilow SW (2002) Reviews in molecular medicine—hyperammonemic encephalopathy. Medicine 81:240–249
Campbell JW, Anderson PM (1991) Evolution of mitochondrial enzyme systems in fish: the mitochondrial synthesis of glutamine and citrulline. In: Hochahka PW, Mommsen TP (eds) Biochemistry and molecular biology of fishes. I. Phylogenetic and biochemical perspectives. Elsevier, Amsterdam, pp 43–75
Cao XY, Kemp JR, Anderson PM (1991) Subcellular localization of two glutamine-dependent carbamoyl-phosphate synthetases and related enzymes in liver of Micropterus salmoides (largemouth bass) and properties of isolated liver mitochondria: comparative relationships with elasmobranchs. J Exp Zool 258:24–33
Chew SF, Jin Y, Ip YK (2001) The loach Misgurnus anguillicaudatus reduces amino acid catabolism and accumulates alanine and glutamine during aerial exposure. Physol Biochem Zool 74:226–237
Chew SF, Ong TF, Ho L, Tam WL, Loong AM, Hiong KC, Wong WP, Ip YK (2003a) Urea synthesis in the African lungfish Protopterus dolloi: hepatic carbamoyl phosphate synthetase III and glutamine synthetase are up regulated by 6 days of aerial exposure. J Exp Biol 206:3615–3624
Chew SF, Wong MY, Tam WL, Ip YK (2003b) The snakehead Channa asiatica accumulates alanine during aerial exposure, but is incapable of sustaining locomotory activities on land through partial amino acid catabolism. J Exp Biol 206:693–704
Chew SF, Chan NKY, Loong AM, Hiong KC, Tam WL, Ip YK (2004) Nitrogen metabolism in the African lungfish (Protopterus dolloi) aestivating in a mucus cocoon on land. J Exp Biol 207:777–786
Chew SF, Gan J, Ip YK (2005) Nitrogen metabolism and excretion in the swamp eel, Monopterus albus, during 6 or 40 days of aestivation in mud. Physiol Biochem Zool 78:620–629
Chew SF, Wilson JM, Ip YK, Randall DJ (2006) Nitrogenous excretion and defense against ammonia toxicity. In: Val A, Almedia-Val V, Randall DJ (eds) Fish physiology, vol 21, The Physiology of Tropical Fishes. Academic, London, pp 307–395
Chew SF, Sim MY, Phua ZC, Wong WP, Ip YK (2007) Active ammonia excretion in the giant Mudskipper, Periophthalmodon schlosseri (Pallas), during emersion. J Exp Zool 307A:357–369
Cooper JL, Plum F (1987) Biochemistry and physiology of brain ammonia. Physiol Rev 67:440–519
DeLaney RG, Lahiri S, Fishman AP (1974) Aestivation of the African lungfish Protopterus aethiopicus: Cardiovascular and respiratory functions. J Exp Biol 61:111–128
Felipo V, Butterworth RF (2002) Neurobiology of ammonia. Progr Neurobiol 67:259–279
Felskie AK, Anderson PM, Wright PA (1998) Expression and activity of carbamoyl phosphate synthetase III and ornithine urea cycle enzymes in various tissues of four fish species. Comp Biochem Physiol 119B:355–364
Fishman AP, Pack AI, Delaney RG, Gallante RJ (1987) Estivation in Protopterus. In: Bemis WE, Burggren EE, Kemp NE (eds) The biology and evolution of lungfishes, Alan R. Liss Inc., New York, pp 237–248
Frick NT, Wright PA (2002) Nitrogen metabolism and excretion in the mangrove killifish Rivulus marmoratus I. The influence of environmental salinity and external ammonia. J Exp Biol 205:79–89
Graham JB (1997) Air-breathing Fishes: evolution, diversity and adaptation. Academic, San Diego, 299p
Greenwood PH (1987) The natural history of African lungfishes. In: Bemis WE, Burggren EE, Kemp NE (eds) The biology and evolution of lungfishes, Alan R. Liss Inc., New York, pp 163–179
Hermenegildo C, Marcaida G, Montoliu C, Grisolia S, Minana M, Felipo V (1996) NMDA receptor antagonists prevent acute ammonia toxicity in mice. Neurochem Res 21:1237–1244
Hillaby BA, Randall DJ (1979) Acute ammonia toxicity and ammonia excretion in rainbow trout (Salmo gairdneri). J Fish Res Bd Can 36:621–629
Hochachka PW (1980) Living without oxygen. Harvard University Press, Cambridge, 181p
Ip YK, Chew SF, Leong IAW, Jin Y, Wu RSS (2001a) The sleeper Bostrichthys sinensis (Teleost) stores glutamine and reduces ammonia production during aerial exposure. J Comp Physiol B 171:357–367
Ip YK, Chew SF, Randall DJ (2001b) Ammonia toxicity, tolerance, and excretion. In: Wright PA, Anderson PM (eds) Fish physiology, vol 20, nitrogen excretion. Academic, San Diego, pp 109–148
Ip YK, Lim CB, Chew SF, Wilson JM, Randall DJ (2001c) Partial amino acid catabolism leading to the formation of alanine in Periophthalmodon schlosseri (mudskipper): a strategy that facilitates the use of amino acids as an energy source during locomotory activity on land. J Exp Biol 204:1615–1624
Ip YK, Chew SF, Randall DJ (2004a) Five tropical fishes, six different strategies to defend against ammonia toxicity on land. Physiol Biochem Zool 77:768–782
Ip YK, Chew SF, Wilson JM, Randall DJ (2004b) Defences against ammonia toxicity in tropical air-breathing fishes exposed to high concentrations of environmental ammonia: a review. J Comp Physiol 174:565–575
Ip YK, Randall DJ, Kok TKT, Bazarghi C, Wright PA, Ballantyne JS, Wilson JM, Chew SF (2004c) The mudskipper Periophthalmodon schlosseri facilitates active NH4 + excretion by increasing acid excretion and decreasing NH3 permeability in the skin. J Exp Biol 207:787–801
Ip YK, Zubaidah RM, Liew PC, Loong AM, Hiong KC, Wong WP, Chew SF (2004d) The African catfish Clarias gariepinus does not detoxify ammonia to urea or amino acids during ammonia loading but is capable of excreting ammonia against an inwardly driven ammonia concentration gradient. Physiol Biochem Zool 77:242–254
Ip YK, Peh BK, Tam WL, Lee SLM, Chew SF (2005a) Changes in salinity and ionic compositions act as environmental signals to induce a reduction in ammonia production in the African lungfish Protopterus dolloi. J Exp Zool 303A:456–463
Ip YK, Peh BK, Tam WL, Wong WP, Chew SF (2005b) Effects of Intra-peritoneal injection with NH4Cl, urea or NH4Cl+urea on nitrogen excretion and metabolism in the African lungfish Protopterus dolloi. J Exp Zool 303A:272–282
Ip YK, Yeo PJ, Loong AM, Hiong KC, Wong WP, Chew SF (2005c) The interplay of increased urea synthesis and reduced ammonia production in the African lungfish Protopterus aethiopicus during 46 days of aestivation in a mucus cocoon on land. J Exp Zool 303A:1054–1065
Janssens PA (1964) The metabolism of the aestivating African lungfish. Comp Biochem Physiol 11:105–117
Janssens PA, Cohen PP (1966) Ornithine-urea cycle enzymes in the African lungfish Protopterus aethiopicus. Science 152:358–359
Janssens PA, Cohen PP (1968a) Biosynthesis of urea in the estivating African lungfish and in Xenopus laevis under conditions of water shortage. Comp Biochem Physiol 24:887–898
Janssens PA, Cohen PP (1968b) Nitrogen metabolism in the African lungfish. Comp Biochem Physiol 24:879–886
Jow LY, Chew SF, Lim CB, Anderson PM, Ip YK (1999) The marble goby Oxyeleotris marmoratus activates hepatic glutamine synthetase and detoxifies ammonia to glutamine during air exposure. J Exp Biol 202:237–245
Lim CB, Chew SF, Anderson PM, Ip YK (2001) Reduction in the rates of protein and amino acid catabolism to slow down the accumulation of endogenous ammonia: a strategy potentially adopted by mudskippers (Periophthalmodon schlosseri and Boleophthalmus boddaerti) during aerial exposure in constant darkness. J Exp Biol 204:1605–1614
Loong AM, Hiong KC, Lee SML, Wong WP, Chew SF, Ip YK (2005) Ornithine-urea cycle and urea synthesis in African lungfishes, Protopterus aethiopicus and Protopterus annectens, exposed to terrestrial conditions for 6 days. J Exp Zool 303A:354–365
McKenzie DJ, Randall DJ, Lin H, Aota S (1993) Effects of changes in plasma pH, CO2 and ammonia on ventilation in trout. Fish Physiol Biochem 10:507–515
Mommsen TP, Walsh PJ (1989) Evolution of urea synthesis in vertebrates: the piscine connection. Science 243:72–75
Perry SF, Gilmour KM, Vulesevic B, McNeil B, Chew SF, Ip YK (2005a) Circulating catecholamines in hypoxic lungfish (Protopterus dolloi): a comparison of aquatic and aerial hypoxia. Physiol Biochem Zool 78:325–334
Perry SF, Gilmour KM, Swenson ER, Vulesevic B, Chew SF, Ip YK (2005b) An investigation of the role of carbonic anhydrase in aquatic and aerial gas transfer n the African lungfish (Protopterus dolloi). J Exp Biol 208:3805–3815
Perry SF, Euverman R, Wang T, Loong AM, Chew SF, Ip YK, Gilmour KM (2007) Control of breathing in African lungfish (Protopterus dolloi): A comparison of aquatic and cocooned (terrestrialized) animals. Resp Physiol Neurobiol (in press)
Randall DJ, Wilson JM, Peng KW, Kok TWK, Kuah SSL, Chew SF, Lam TJ, Ip YK (1999) The mudskipper, Periophthalmodon schlosseri, actively transports NH4 + against a concentration gradient. Am J Physiol 277:R1562–R1567
Randall DJ, Ip YK, Chew SF, Wilson JM (2004) Air breathing and ammonia excretion in the mudskipper, Periophthalmodon schlosseri. Physiol Biochem Zool 77:783–788
Rose C (2002) Increased extracellular brain glutamate in acute liver failure: decreased uptake or increased release? Metabolic Brain Disease 17:251–261
Scheibel LW, Saz HJ, Bueding E (1968) The anaerobic incorporation of 32P into adenosine tripohosphate by Hymenolepis diminuta. J Biol Chem 243:2229–2235
Shankar RA, Anderson PM (1985) Purification and properties of glutamine synthetase from the liver of Squalus acanthias. Arch Biochem Biophys 239:248–259
Shigesada K, Tatibana M (1978) N-acetylglutamate synthetase from rat-liver mitochondria. Partial purification and catalytic properties. Eur J Biochem 84:285–291
Smith HW (1930) Metabolism of the lungfish Protopterus aethiopicus. J Biol Chem 88:97–130
Smith HW (1935) The metabolism of the lungfish II. Effect of feeding meat on metabolic rate. J Cell Comp Physiol 6:335–349
Tay SLA, Chew SF, Ip YK (2003) The swamp eel Monopterus albus reduces endogenous ammonia production and detoxifies ammonia to glutamine during 144 h of aerial exposure. J Exp Biol 206:2473–2486
Tay YL, Loong AM, Hiong KC, Lee SJ, Tng YYM, Wee NLJ, Lee SML, Wong WP, Chew SF, Wilson JM, Ip YK (2006) Active ammonia transport and excretory nitrogen metabolism in the climbing perch, Anabas testudineus, during 4 days of emersion or 10 minutes of forced exercise on land. J Exp Biol 209:4475–4489
Tsui TKN, Randall DJ, Chew SF, Jin Y, Wilson JM, Ip YK (2002) Accumulation of ammonia in the body and NH3 volatilization from alkaline regions of the body surface during ammonia loading and exposure to air in the weather loach Misgurnus anguillicaudatus. J Exp Biol 205:651–659
Walsh PJ, Wei Z, Wood CM, Loong AM, Hiong KC, Lee SML, Wong WP, Chew SF, Ip YK (2004) Nitrogen metabolism and excretion in the grunting toadfish, Allenbatrachus grunniens (L): effects of variable salinity, confinement, high pH and ammonia loading. J Fish Biol 65:1392–1411
Wilkie MP, Morgan TP, Galvez F, Smith R, Kajimura M, Ip YK, Wood CM (2007) The African Lungfish (Protopterus dolloi): Ionoregulation and osmoregulation in a fish out of water. Physiol Biochem Zool 80:90–112
Wood CM, Walsh PJ, Chew SF, Ip YK (2005) Greatly elevated urea excretion after air exposure appears to be carrier mediated in the slender lungfish (Protopterus dolloi). Physiol Biochem Zool 78:893–907
Xiong X, Anderson PM (1989) Purification and properties of ornithine carbamoyl transferase from the liver of Squalus acanthias. Arch Biochem Biophys 270:198–207
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by I.D. Hume.
Rights and permissions
About this article
Cite this article
Loong, A.M., Pang, C.Y.M., Hiong, K.C. et al. Increased urea synthesis and/or suppressed ammonia production in the African lungfish, Protopterus annectens, during aestivation in air or mud . J Comp Physiol B 178, 351–363 (2008). https://doi.org/10.1007/s00360-007-0228-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-007-0228-6