Abstract
Polarization vision is used by a wide range of animals for navigating, orienting, and detecting objects or areas of interest. Shallow marine and semi-terrestrial crustaceans are particularly well known for their abilities to detect predator-like or conspecific-like objects based on their polarization properties. On land, some terrestrial invertebrates use polarization vision for detecting suitable habitats, oviposition sites or conspecifics, but examples of threat detection in the polarization domain are less well known. To test whether this also applies to crustaceans that have evolved to occupy terrestrial habitats, we determined the sensitivity of two species of land and one species of marine hermit crab to predator-like visual stimuli varying in the degree of polarization. All three species showed an ability to detect these cues based on polarization contrasts alone. One terrestrial species, Coenobita rugosus, showed an increased sensitivity to objects with a higher degree of polarization than the background. This is the inverse of most animals studied to date, suggesting that the ecological drivers for polarization vision may be different in the terrestrial environment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The use of the polarization of light for object detection has been demonstrated in a range of animal groups, including cephalopods, crustaceans and insects (How et al. 2012; Temple et al. 2012; Drerup and How 2021; Shashar and Cronin 1996; Cronin et al. 2002; Kelber et al. 2001). These ‘small-field’ tasks require the detection or identification of objects such as predators, food, or conspecifics in small parts of the visual field, roughly analogous to the colour vision systems that many animals use to detect objects based on chromatic contrasts. Small-field tasks differ from wide-field tasks, such as navigation and orientation using the pattern of polarization in the sky (Wehner 1976).
Object-based polarization vision is particularly widespread in aquatic and semi-aquatic species, suggesting that these habitats are well-suited for the detection of objects using contrasts in polarization (Marshall et al. 2019). One explanation for this is that these environments offer predictably polarized backgrounds. For example, in clear water, the background illumination produced by the scattering of down-welling light from the sun and sky is generally polarized (up to 50–70%) and is predominantly oriented along the horizontal axis (Cronin and Shashar 2001). Similarly, on intertidal mudflats the damp substrate reflects light from the sun and sky also with a predominantly horizontal angle of polarization (How et al. 2015). In terrestrial and semi-terrestrial habitats, the blue sky also provides a stable and predictable backdrop of polarization, with angle and degree relating to the position of the sun (Wehner 1976; Homberg 2004). These predictably polarized backgrounds provide an ideal scene against-which to spot unpolarized contrasts associated with predators, food, or conspecifics (How et al. 2015). A second explanation for why polarization vision may be so prevalent underwater is that, particularly in shallow aquatic environments, this sensory modality may help to mitigate challenges associated with dynamic lighting (Vincent Venebles et al. 2022), a theory first proposed by Maximov (2000) to explain the evolution of colour (rather than polarization) vision underwater.
In many terrestrial environments, polarization may be a less reliable source of information for threat detection than colour, due to high levels of polarization ‘noise’ generated by background objects such as vegetation. On land, the large difference in refractive index between air and natural objects is often responsible for complex polarization cues across visual scenes. This differs from underwater, where the difference in refractive index between objects and water is much lower, leading to much reduced levels of polarized reflections (Marshall et al. 2019). Despite this, there are terrestrial species that have evolved small-field polarization vision that mediate specific behavioural tasks. For example, some butterflies combine polarization and spectral information to help locate appropriate vegetation for oviposition (Kelber et al. 2001; Blake et al. 2020; Nagaya et al. 2021) and many insect species use small-field polarization cues for locating aquatic habitats or bloodmeal hosts (Schwind 1984; Kriska et al. 1998; Horváth et al. 2008; Mathejczyk and Wernet 2017; Heinloth et al. 2018; Meglič et al. 2019; Yadav and Shein-Idelson 2021).
The apparent rarity of polarization vision systems for threat detection in terrestrial environments opens the question of whether marine species that have evolved to occupy terrestrial habitats would lose any survival advantage conferred by their polarization vision, and therefore lose the ability to detect predators using the polarization of light through the process of genetic drift. A candidate group of animals for investigating this question is the superfamily of hermit crabs, the Paguroidea (Latreille 1802).
There are some 800 species of hermit crab, most of which occupy marine or intertidal habitats (Hazlett 1981). One family, the Coenobitidae (Dana 1851), have evolved to exploit terrestrial ecosystems (Hartnoll 1988) and display a range of adaptations for life away from water (Burggren and McMahon 1988). These species spend their entire lives in terrestrial habitats, returning to the ocean only to release larvae (Nio et al. 2019). Two species of terrestrial hermit crab are abundant at our study site on the island of Mahé, Seychelles. Coenobita rugosus (Milne Edwards 1837) tend to occupy forest understory from the top of the shoreline to approximately 100 m inshore, and Coenobita brevimanus (Dana 1852) occupy more inland forest habitats > 50 m inshore (Hsu et al. 2018). Whether or not these species make use of polarization vision is unknown, but it is likely that—much like their marine counterparts (Kerz 1950)—ancestral marine hermit crabs would have had this sensory capability.
To determine whether C. rugosus and C. brevimanus make use of the polarization of light, we determined their sensitivity to predator-like visual cues presented only in contrasts of polarization using an established playback assay (How et al. 2012; Shragai et al. 2017; Wilby et al. 2018) and compared this to the polarization responses of a fully marine species, Pagurus bernhardus (Linnaeus 1758). To interpret our findings, we analysed the distribution of polarization cues in the visual habitats of the two terrestrial species by collecting footage with a polarization camera on the island of Mahé, Seychelles.
Materials and methods
Animal collection
Terrestrial hermit crabs (C. rugosus and C. brevimanus) were collected at Anse Petit Police (4°48′12″S; 55°31′05″E) and Anse Marie-Louise (4°47′24″S; 55°31′50″E), Mahé Island, Seychelles and housed for < 48 h in a terrarium with damp plant detritus. Animals were maintained in natural light cycles and fed ad libitum on fruit scraps (coconut, papaya and mango). Marine hermit crabs (P. bernhardus) were collected from intertidal rockpools at Looe, Cornwall, UK (50°20′57″N; 4°27′01″W), housed for < 2 weeks in an aquarium with artificial seawater at salt concentrations of 35ppt, and fed twice weekly with defrosted shrimp. All animals were returned to the site of collection after experimentation.
Experimental procedure
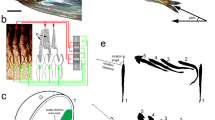
The response of hermit crabs to visual stimuli was tested using an approach similar to previous studies on other crab species (How et al. 2012, 2015; Shragai et al. 2017; Wilby et al. 2018; Smithers et al. 2019). Briefly, animals were tethered above a treadmill (for terrestrial species) or underwater over a slippery surface (for the marine species) so that they faced a display screen on which moving stimuli could be presented (Fig. 1). For terrestrial crabs, the stimulus screen consisted of a field-portable patterned-vertical-alignment type liquid crystal display (LCD) screen (1905fp, Dell Technologies, Round Rock, USA) with the front-most polarizing filter replaced with a removable sheet of polaroid filter (see Foster et al. 2018 for details). For the marine species, a lab-based version of the screen was constructed, consisting of 1) an LCD panel (1905fp, same as above) with the outer most polaroid filter removed and dissembled from the panel housing to allow illumination from 2) a digital projector (CP-WX3030WN, Hitachi Ltd., Tokyo, Japan) (see Smithers et al. 2019 for further details).
A visual stimulus simulating the looming movement of an approaching predator was created by animating an expanding disc on the display. The disc expanded around a central point with a geometric expansion profile over a period of 3 s for terrestrial hermits and 10 s for P. bernhardus, from a visual angle of 0° to approximately 19° (12 cm disc diameter viewed from ~ 35 cm), followed by a 2 s period at maximum size before disappearing. By modifying the RGB (R = G = B) values of the pixels of the background and foreground of the stimulus, we could systematically adjust either the intensity contrast (presented with the removable polarizer in place or on the digital projector screen) or the polarization contrast (presented with the removable polarizer removed or on the polarization screen) of the expanding disc. Polarization and intensity properties of the screen were measured using a spectrophotometer (Flame, Ocean Optics, Orlando, USA) coupled to a Glan Thompson linear polarizer (GTH10M-B, Thorlabs, Newton, USA).
After acclimatising to the experimental arena for 3 min, animals were presented with a randomised series of expanding disc stimuli varying in contrast with the background, with time intervals of 1–3 min. Animals only saw one series of stimuli in either intensity or polarization and so each series of contrasts provides an independent set of results. For intensity-based (I) stimuli, the background was set to near the mid-point of the radiance levels measured across the greyscale range, which corresponded to 8 bit RGB values of [158, 158, 158]. Expanding disc stimuli varied from this in both the negative (darker) and positive (lighter) weber contrast scale, defined as
For polarization-based stimuli, the background was set to 50% polarized in the horizontal plane, with stimuli differing in degree of polarization (DoP) in the negative (less polarized) and positive (more polarized) scale (for details on all pixel values, weber contrast and DoP of stimuli, see Supplementary Table 1). Crab responses were recorded with a digital video camera filming from above, and stimulus timing was passed to the video stream both by audio signals conveyed through the microphone port (inaudible to the outside world) and via a red flashing light-emitting-diode (LED) placed outside the field of view of the hermit crab. The LED emitted no wavelengths of light below 600 nm and so falls outside the main spectral sensitivity range of the animals.
Behavioural data analysis
Responses of hermit crabs to the visual stimuli were determined using a blind-scoring technique, and for terrestrial crabs, the timing of onset of three different behaviours was determined relative to the stimulus. These behaviours were: stop, the animal stopped its previous behaviour and became stationary; retreat, the animal pulled itself partially or fully into its shell refuge; and walk, the animal began walking on the treadmill. A crab was said to have responded to a stimulus if it exhibited any of these behaviours. A small number of trials were excluded if the animal performed a full retreat before the onset of the visual stimulus, a behaviour presumed to be associated with hydration.
Contrast response curves were modelled (where possible) with sigmoid functions using the sigm_fit script (Pavao 2017) in Matlab (r2021, Mathworks, Natick, USA) and habituation was modelled by fitting a line to response probability as a function of stimulus order using Matlab’s ‘polyfit’ function. Data for intensity responses of P. bernhardus were not collected over a suitable range of contrasts for comparison with terrestrial species, so the graph is not included for this species.
For statistical analysis, we used mixed effects binary logistic regression, fitted using the lme4 package (Bates et al. 2015) in R (R Core Team 2022). We used the DHARMa package (Hartig 2022) to run residual diagnostics that are often overlooked in logistic regression. The binary response variable was crab response. Stimulus contrast (i.e. intensity or polarization contrast) and presentation order were included as fixed effects, the latter was included to test for habituation. Crab ID was included as a random effect to control for repeated measures. When fitting the full model for the intensity experiment with C. rugosus a quadratic effect was included to control for an irregular pattern in the residuals. To test for a significant effect of stimulus contrast and presentation order we used a likelihood ratio test (LRT) to compare the full model with the same model but with the effect of interest removed.
Polarization camera recording
To investigate the polarization environment of C. rugosus and C. brevimanus, a monochromatic polarization camera (Triton TRI050S-PC, LucidVisionLabs, Richmond, Canada) was used to take snapshots from the location at which animals were collected for study. The polarization camera collects information via subpixels etched with polarization filters oriented at 0°, 45°, 90° and 135° and the local degree and angle of polarization values for each super-pixel were then calculated using stokes equations and displayed in false-colour images (see Foster et al. 2018 and supplementary information for details).
Results
Behavioural experiments
Both the marine P. bernhardus and the terrestrial C. rugosus showed evidence of good polarization vision responding reliably to predator-like stimuli varying only in polarization contrast (Fig. 2a, left; mixed effects binary logistic regression- effect of polarization contrast for P. bernhardus: χ2(1) = 12.31, p < 0.001; effect of polarization contrast for C. rugosus: χ2(1) = 32.16, p < 0.001). Both species exhibited an asymmetric response curve indicating that the effect of polarization contrast is driven by both the size and polarity of the contrast. The terrestrial crab C. brevimanus also exhibited evidence of polarization vision (Fig. 2a, left), although polarization contrast was not found to be statistically significant (χ2(1) = 2.41, p = 0.12). However, as is evident from Fig. 2a, we argue that this result is not due to a lack of response to polarization contrasts. Unlike for the other two species, the response curve for C. brevimanus is relatively symmetrical on either side of the control suggesting that contrast polarity is less important for this species. Given the reduced effect of contrast polarity for C. brevimanus, it is possible that the smaller sample size meant that the model did not have enough power to detect the overall effect of polarization contrast. This suggestion is supported by the fact that the effect of polarization contrast is highly significant when we fit separate models for positive (χ2(1) = 28.36, p < 0.001) and negative (χ2(1) = 25.4, p < 0.001) contrasts. Half-maximum values extracted from fitted sigmoidal curves indicate threshold response values of DoP between 0.17 and 0.22 for all species. Interestingly, the terrestrial C. rugosus showed a stronger response to positive polarization contrasts (stimuli with a higher DoP than the background), while the marine species showed a stronger response to stimuli with a lower polarization contrast than the background.
Responses of hermit crab species to visual stimuli presented in a polarization contrast only (DoP contrast is calculated as stimulus-background DoP values), and b intensity contrast only (negative and positive values equate to darker and lighter stimuli than the background respectively). Left panels: response as a function of stimulus contrast. Solid red lines show (where possible) fitted sigmoid curves. Vertical dotted red lines show steepest point of sigmoid slope. Middle panels: response as a function of stimulus order. Blue lines show lines of best fit. Note the differences in x-axis scale. ‘h’ = habituation rate, defined as the change in response probability per trial. Right panels: timing and frequency of ‘stop’ (red), ‘retreat’ (yellow), and ‘walk’ (blue) behaviours relative to loom expansion (black line). Note that ‘retreat’ behaviour is scored as positive values below the x-axis. Sample sizes encompass data arranged by contrast and by order of presentation. Inset photos by M How and H Hillewaert
Both terrestrial species responded more typically to intensity-only stimuli (effect of intensity contrast for C. rugosus: χ2(1) = 81.53, p < 0.001; effect of intensity contrast for C. brevimanus: χ2(1) = 30.4, p < 0.001), showing the strongest response to darker expanding discs than the background (Fig. 2b, left). The types of behaviour and their timing relative to stimulus onset was similar between species and across both polarized and intensity-based stimuli (Fig. 2a,b, right). In the polarization experiment both terrestrial species displayed a significant amount of habituation (Fig. 2a, middle; effect of order for C. rugosus: χ2(1) = 25.46, p < 0.001; effect of order for C. brevimanus: χ2(1) = 4.93, p = 0.026), while P. bernhardus did not (effect of order for P. bernhardus: χ2(1) = 0.79, p = 0.375) (Fig. 2a, middle). Coenobita rugosus also showed signs of habituation in the intensity experiment (effect of order for C. rugosus: χ2(1) = 5.43, p = 0.02), but C. brevimanus did not (effect of order for C. brevimanus: χ2(1) = 1.6, p = 0.207).
Terrestrial polarization
Polarization images taken from a crab’s perspective at the site of animal collection indicates relatively low levels of DoP (Fig. 3, middle). These locations consisted of leaf litter, soil and sand under a canopy of palms and deciduous trees. The locations were also characteristically dry at the time of measurement.
Polarimetry from the terrestrial habitats of C. rugosus (top) and C. brevimanus (bottom). Left: Intensity image. Middle: Degree of polarization (DoP) false-colour image (scale along right of panel). 0 = unpolarized, 1 = fully polarized. Right: Angle of polarization (AoP) false-colour image; 0° is horizontal. Over- and under-exposed pixels are assigned white and black respectively in the DoP and AoP images
Discussion
Our results demonstrate that terrestrial hermit crabs can use polarization contrasts in the context of threat detection, despite evolving to occupy terrestrial habitats with very different polarization content to marine and intertidal locations. Whether this is because polarization vision conveys a fitness benefit to these species, or whether this sensory capacity is an evolutionary hang-over from their previous marine adaptations remains to be demonstrated.
One result of note is that the terrestrial species exhibited standard responses to intensity stimuli, showing stronger responses to dark objects viewed against light backgrounds than vice versa. This behavioural filter is likely due to silhouetted predators appearing darker than the background and can similarly be found in many other species (Layne 1998; Santer et al. 2005; Yilmaz and Meister 2013; Temizer et al. 2015; Smithers et al. 2019). However, the terrestrial crab C. rugosus differed from the marine species, and indeed other crustaceans (Layne 1998; Smithers et al. 2019), by responding more strongly to stimuli with higher DoP than the background (Fig. 2a, left). In other contexts, such as underwater or in semi-terrestrial mudflats, animals benefit from polarization vision by being able to detect objects with low levels of polarization (be they conspecifics or predators) viewed against a uniformly polarized background of reflected or scattered light (How et al. 2015). Perhaps the visual ecology of terrestrial hermit crabs is different and salient objects in the environment tend to be more polarized than the unpolarized background, causing behavioural filters to favour responding to cues that are more polarized than the background. This possibility is supported by the polarimetry (Fig. 3) which shows very low levels of polarization reflected from the dry understory of inland forest habitats occupied by these species. How these polarization and intensity channels (and indeed any chromatic input if present) of information are integrated in the optic lobes of the hermit crab is unknown, but could be tested using a similar approach to Smithers et al (2019).
Another possibility is that polarization vision serves alternative ‘small field’ visual tasks such as the location of damp understory refuges or fresh water sources needed during dry conditions (Mathejczyk and Wernet 2017). Or alternatively, polarization vision may play a role in navigation when moving towards or away from shore during reproductive behaviour (Vannini and Chelazzi 1981). Future research should target the ecological function of polarization vision in these species.
Data availability
All datasets and analysis code are available at the github repository https://github.com/EcologyOfVisionBristol/HermitCrabPolarizationVision2023.git.
References
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67(1):1–48. https://doi.org/10.18637/jss.v067.i01
Blake AJ, Hahn GS, Grey H, Kwok SA, McIntosh D, Gries G (2020) Polarized light sensitivity in Pieris rapae is dependent on both color and intensity. J Exp Biol 223(13):220350. https://doi.org/10.1242/jeb.220350
Burggren WW, McMahon BR (eds) (1988). Cambridge University Press, Cambridge
Cronin TW, Shashar N (2001) The linearly polarized light field in clear, tropical marine waters: Spatial and temporal variation of light intensity, degree of polarization and e-vector angle. J Exp Biol 204(14):2461–2467. https://doi.org/10.1242/jeb.204.14.2461
Cronin TW, Shashar N, Caldwell RL, Cheroske AG, Chiou TH (2002) Polarization vision and its role in underwater signaling. Integr Comp Biol 42(6):1215–1215
Dana JD (1851) Paguridea: Conspectus Crustaceorum quae in Orbis Terrarum circumnavigatione, Carolo Wilkes e classe Reipublicae Foederatae Duce, lexit et descripsit J.D. Dana Proc Acad Nat Sci Phila 5:267–272
Dana JD (1852) On the classification of the Crustacea Choristopoda or Tetrapoda. Am J Sci 2(14):197–306
Drerup C, How MJ (2021) Polarization contrasts and their effect on the gaze stabilization of crustaceans. J Exp Biol. https://doi.org/10.1242/jeb.229898
Foster JJ, Temple SE, How MJ, Daly IM, Sharkey CR, Wilby D, Roberts NW (2018) Polarisation vision: overcoming challenges of working with a property of light we barely see. Naturwissenschaften 105(3–4):27–27. https://doi.org/10.1007/s00114-018-1551-3
Hartig F (2022) DHARMa: Residual diagnostics for hierarchical (multi-level/mixed) regression models. R package version 0.4.6
Hartnoll RG (1988) Evolution, systematics and geographical distribution. In: Burggren WW, McMahon BR (eds) Biology of the Land Crabs, Cambridge University Press
Hazlett BA (1981) The behavioral ecology of hermit crabs. Annu Rev Ecol Syst 12:1–22
Heinloth T, Uhlhorn J, Wernet MF (2018) Insect responses to linearly polarized reflections: orphan behaviors without neural circuits. Front Cell Neurosci. https://doi.org/10.3389/fncel.2018.00050
Homberg U (2004) In search of the sky compass in the insect brain. Naturwissenschaften 91(5):199–208. https://doi.org/10.1007/s00114-004-0525-9
Horváth G, Majer J, Horváth L, Szivák I, Kriska G (2008) Ventral polarization vision in tabanids: horseflies and deerflies (Diptera: Tabanidae) are attracted to horizontally polarized light. Naturwissenschaften 95(11):1093–1100. https://doi.org/10.1007/s00114-008-0425-5
How MJ, Pignatelli V, Temple SE, Marshall NJ, Hemmi JM (2012) High e-vector acuity in the polarisation vision system of the fiddler crab Uca vomeris. J Exp Biol 215(12):2128–2134. https://doi.org/10.1242/jeb.068544
How MJ, Christy JH, Temple SE, Hemmi JM, Marshall NJ, Roberts NW (2015) Target detection is enhanced by polarization vision in a fiddler crab. Curr Biol 25:3069–3073. https://doi.org/10.1016/j.cub.2015.09.073
Hsu CH, Otte ML, Liu CC, Chou JY, Fang WT (2018) What are the sympatric mechanisms for three species of terrestrial hermit crab (Coenobita rugosus, C. brevimanus, and C. cavipes) in coastal forests? PLOS ONE 13(12):e0207640. https://doi.org/10.1371/journal.pone.0207640
Kelber A, Thunell C, Arikawa K (2001) Polarisation-dependent colour vision in Papilio butterflies. J Exp Biol 204(14):2469–2480. https://doi.org/10.1242/jeb.204.14.2469
Kerz M (1950) Wahrnehmung polarisierten Lichtes durch Eupagurus. Experientia 6(11):427–427. https://doi.org/10.1007/BF02150125
Kriska G, Horváth G, Andrikovics S (1998) Why do mayflies lay their eggs en masse on dry asphalt roads? Water-imitating polarized light reflected from asphalt attracts Ephemeroptera. J Exp Biol 201(15):2273–2286. https://doi.org/10.1242/jeb.201.15.2273
Layne JE (1998) Retinal location is the key to identifying predators in fiddler crabs (Uca pugilator). J Exp Biol 201:2253–2261. https://doi.org/10.1242/jeb.201.15.2253
Linnaeus C (1758) Systema naturae, vol 1. 10th edn. Laurentius Salvius, Stockholm
Marshall NJ, Powell SB, Cronin TW, Caldwell RL, Johnsen S, Gruev V, Chiou T-HS, Roberts NW, How MJ (2019) Polarisation signals: a new currency for communication. J Experim Biol 222(3):134213. https://doi.org/10.1242/jeb.134213
Mathejczyk TF, Wernet MF (2017) Sensing polarized light in insects. Oxford University Press. https://doi.org/10.1093/acrefore/9780190264086.013.109
Maximov VV (2000) Environmental factors which may have led to the appearance of colour vision. Philos Trans R Soc Lond, Ser b: Biol Sci 355:1239–1242. https://doi.org/10.1098/rstb.2000.0675
Meglič A, Ilić M, Pirih P, Škorjanc A, Wehling MF, Kreft M, Belušič G (2019) Horsefly object-directed polarotaxis is mediated by a stochastically distributed ommatidial subtype in the ventral retina. Proc Natl Acad Sci 116(43):21843. https://doi.org/10.1073/pnas.1910807116
Milne Edwards H (1837) Historie naturelle des crustaces comprenant l-anatomie, la physiologie et las classification des ces animaux, vol 2. Librarie Encyclopedique de Roret, Paris
Nagaya H, Stewart FJ, Kinoshita M (2021) Swallowtail butterflies use multiple visual cues to select oviposition sites. Insects 12(11):1047. https://doi.org/10.3390/insects12111047
Nio T, Doi W, Mizutani A, Kohno H (2019) Seaward migration and larval release of the land hermit crab Coenobita brevimanus Dana, 1852 (Anomura: Coenobitidae) on Iriomote Island, Japan. Crustac Res 48:67–80. https://doi.org/10.18353/crustacea.48.0_67
Pavao R (2017) sigm_fit. MATLAB Central File Exchange
R Core Team (2022) R: A language and environment for statistical computing. Foundation for Statistical Computing, Vienna
Santer RD, Simmons PJ, Rind FC (2005) Gliding behaviour elicited by lateral looming stimuli in flying locusts. J Comp Physiol A 191(1):61–73. https://doi.org/10.1007/s00359-004-0572-x
Schwind R (1984) Evidence for true polarization vision based on a two-channel analyzer system in the eye of the water bug, Notonecta glauca. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 154(1):53–57. https://doi.org/10.1007/bf00605390
Shashar N, Cronin TW (1996) Polarization contrast vision in Octopus. J Exp Biol 199(4):999–1004. https://doi.org/10.1242/jeb.199.4.999
Shragai T, Ping X, Arakaki C, Garlick D, Blumstein DT, Blaisdell AP (2017) Hermit crab response to a visual threat is sensitive to looming cues. PeerJ 5:e4058. https://doi.org/10.7717/peerj.4058
Smithers SP, Roberts NW, How MJ (2019) Parallel processing of polarization and intensity information in fiddler crab vision. Sci Adv 5(8):eaax3572. https://doi.org/10.1126/sciadv.aax3572
Temizer I, Donovan Joseph C, Baier H, Semmelhack Julia L (2015) A visual pathway for looming-evoked escape in larval zebrafish. Curr Biol 25(14):1823–1834. https://doi.org/10.1016/j.cub.2015.06.002
Temple SE, Pignatelli V, Cook T, How MJ, Chiou T-H, Roberts NW, Marshall NJ (2012) High-resolution polarisation vision in a cuttlefish. Curr Biol 22(4):R121–R122. https://doi.org/10.1016/j.cub.2012.01.010
Vannini M, Chelazzi G (1981) Orientation of Coenobita rugosus (Crustacea: Anomura): A field study on Aldabra. Mar Biol 64(2):135–140. https://doi.org/10.1007/BF00397102
Vincent Venebles S, Drerup C, Powell SB, Marshall NJ, Herbert-Read JE, How MJ (2022) Polarization vision mitigates visual noise from flickering light underwater. Sci Adv 8 (36):eabq2770. https://doi.org/10.1126/sciadv.abq277
Wehner R (1976) Polarized-light navigation by insects. Sci Am 235:106–115. https://doi.org/10.1038/scientificamerican0776-106
Wilby D, Riches S, Daly IM, Bird A, Wheelwright M, Foster JJ (2018) Hermit crabs (Pagurus bernhardus) use visual contrast in self-assessment of camouflage. J Exp Biol 221(13):173831. https://doi.org/10.1242/jeb.173831
Yadav P, Shein-Idelson M (2021) Polarization vision in invertebrates: beyond the boundaries of navigation. Curr Opin Insect Sci 48:50–56. https://doi.org/10.1016/j.cois.2021.09.005
Yilmaz M, Meister M (2013) Rapid innate defensive responses of mice to looming visual stimuli. Curr Biol 23(20):2011–2015. https://doi.org/10.1016/j.cub.2013.08.015
Acknowledgements
Our thanks go to the Seychelles Marine Conservation Society for help with finding hermit crabs on Mahé. Thanks to Jack Vaughan for helping to catch UK hermit crabs. MJH was supported by a Royal Society University Research Fellowship (grant UF140558). SPS was supported by an Engineering and Physical Sciences Research Council iCase studentship with the Defence Science and Technology Laboratory (Dstl), Ministry of Defence (grant DSTLX-1000096651).
Author information
Authors and Affiliations
Contributions
MJH designed and performed the experiments on terrestrial hermit crab species. SPS, AR and DW designed and perfomed the expeirments on the marine species of hermit crab. All authors contributed to data analysis and manuscript preparation.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict interests.
Additional information
Handling Editor: Mathias F. Wernet.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
How, M.J., Robertson, A., Smithers, S.P. et al. Polarization vision in terrestrial hermit crabs. J Comp Physiol A 209, 899–905 (2023). https://doi.org/10.1007/s00359-023-01631-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-023-01631-z