Abstract
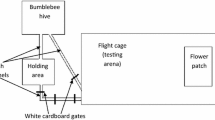
We previously studied a conditioning paradigm to associate the proboscis extension reflex (PER) with monochromatic light (conditioned stimulus; CS) in harnessed honeybees. Here, we established a novel conditioning paradigm to associate the PER with a motion cue generated using graphics interchange format (GIF) animations with a speed of 12 mm/s speed and a frame rate of 25 Hz as the CS, which were projected onto a screen consisting of a translucent circular cone that largely covered the visual field of the harnessed bee using two liquid crystal projectors. The acquisition rate reached a plateau at approximately 40% after seven trials, indicating that the bees were successfully conditioned with the motion cue. We demonstrated four properties of the conditioning paradigm. First, the acquisition rate was enhanced by antennae deprivation, suggesting that sensory input from the antennae interferes with the visual associative learning. Second, bees conditioned with a backward-direction motion cue did not respond to the forward-direction, suggesting that bees can discriminate the two directions in this paradigm. Third, the bees can retain memory for motion cue direction for 48 h. Finally, the acquisition rate did not differ significantly between foragers and nurse bees.







Similar content being viewed by others
Abbreviations
- CS:
-
Conditioned stimulus
- GIF:
-
Graphics interchange format
- ITI:
-
Intertrial interval
- PER:
-
Proboscis extension reflex
- US:
-
Unconditioned stimulus
References
Abel JR, Menzel R (2001) Structure and response patterns of olfactory interneurons in the honeybee, Apis mellifera. J Comp Neurol 437:363–383
Ben-Shahar Y (2005) The foraging gene, behavioral plasticity, and honeybee division of labor. J Comp Physiol A 191:987–994
Ben-Shahar Y, Robichon A, Sokolowski MB, Robinson GE (2002) Influence of gene action across different time scales on behavior. Science 296:741–744
Ben-Shahar Y, Leung HT, Pak WL, Sokolowski MB, Robinson GE (2003) cGMP-dependent changes in phototaxis: a possible role for the foraging gene in honey bee division of labor. J Exp Biol 206:2507–2515
Esch HE, Zhang S, Srinivasan MV, Tautz J (2001) Honeybee dances communicate distances measured by visual motion. Nature 411:581–583
Faber T, Menzel R (2001) Visualizing mushroom body response to a conditioned odor in honeybees. Naturewissenschaften 88:472–476
Faber T, Joerges J, Menzel R (1999) Associative learning modifies neural representations of odors in the insect brain. Nat Neurosci 2:74–78
Farooqui T, Robinson K, Vaessin H, Smith BH (2003) Modulation of early olfactory processing by an octopaminergic reinforcement pathway in the honeybee. J Neurosci 23:5370–5380
Farooqui T, Vaessin H, Smith BH (2004) Octopamine receptors in the honeybee (Apis mellifera) brain and their disruption by RNA-mediated interference. J Insect Physiol 50:701–713
Galizia CG, Kimmerle B (2004) Physiological and morphological characterization of honeybee olfactory neurons combining electrophysiology, calcium imaging and confocal microscopy. J Comp Physiol A 190:21–38
Galizia CG, Sachse S, Rappert A, Menzel R (1999) The glomerular code for odor representation is species specific in the honeybee Apis mellifera. Nat Neurosci 2:473–478
Giurfa M, Malun D (2004) Associative mechanosensory conditioning of the proboscis extension reflex in honeybees. Learn Mem 11:294–302
Grünbaum L, Müller U (1998) induction of a specific olfactory memory leads to a long-lasting activation of protein kinase C in the antennal lobe of the honeybee. J Neurosci 18:4384–4392
Guerrieri F, Schubert M, Sandoz JC, Giurfa M (2005) Perceptual and neural olfactory similarity in honeybees. PLoS Biol 3:60
Hammer M (1993) An identified neuron mediates the unconditioned stimulus in associative olfactory learning in honeybees. Nature 366:59–63
Hammer M, Menzel R (1998) Multiple sites of associative odor learning as revealed by local brain microinjections of octopamine in honeybees. Learn Mem 5:146–156
Hellstern F, Malaka R, Hammer M (1998) Backward inhibitory learning in honeybees: a behavioral analysis of reinforcement processing. Learn Mem 4:429–444
Hori S, Takeuchi H, Arikawa K, Kinoshita M, Ichikawa N, Sasaki M, Kubo T (2006) Associative visual learning, color discrimination, and chromatic adaptation in the harnessed honeybee Apis mellifera L. J Comp Physiol A 192:691–700
Horridge A (2000) Pattern vision of the honeybee (Apis mellifera). What is an oriented edge? J Comp Physiol A 186:521–534
Lehrer M (1994) Spatial vision in the honeybee: the use of different cues in different tasks. Vision Res 34:2363–2385
Lozono VC, Armengaug C, Gauthier M (2001) Memory impairment induced by cholinergic antagonists injected into the mushroom bodies of the honeybee. J Comp Physiol A 187:249–254
Lunney GH (1970) Using analysis of dependent variable: An empirical study. J Educat Meas 7:263–269
Mauelshagen J (1993) Neural correlates of olfactory learning paradigms in an identified neuron in the honeybee brain. J Neurophysiol 69:609–625
Müller U (1996) Inhibition of nitric oxide synthase impairs a distinct form of long-term memory in the honeybee, Apis mellifera. Neuron 16:541–549
Müller U (2000) Prolonged activation of cAMP-dependent protein kinase during conditioning induces long-term memory in honeybees. Neuron 27:159–168
Müller R, Abel R, Brandt M, Zöckler M, Menzel R (2002) Differential parallel processing of olfactory information in the honeybee, Apis mellifera L. J Neurobiol 188:359–370
Ray S, Ferneyhough B (1997) The effects of age on olfactory learning in the honeybee Apis mellifera. Neuroreport 8:789–793
Ray S, Ferneyhough B (1999) Behavioral development and olfactory learning in the honeybee (Apis mellifera). Dev Psychobiol 34:21–27
Ronacher B (1998) How do bees learn and recognize visual patterns? Biol Cybern 79:477–485
Sachse S, Galizia CG (2003) The coding of odour-intensity in the honeybee antennal lobe: local computation optimizes odour representation. Eur J Neurosci 18:2119–2132
Sandoz JC, Galizia CG, Menzel R (2003) Side-specific olfactory conditioning leads to more specific odor representation between sides but not within sides in the honeybee antennal lobes. Neuroscience 120:1137–1148
Seeley TD, Visscher PK (1998) Assessing the benefits of cooperation in honeybee foraging: search costs, forage quality, and competitive ability. Behav Ecol Sociobiol 22:229–237
Srinivasan MV, Lehrer M (1984) Temporal acuity of honeybee vision: behavioural studies using moving stimuli. J Comp Physiol A 155:297–312
Srinivasan MV, Zhang S, Altwein M, Tautz J (2000) Honeybee navigation: nature and calibration of the “Odometer”. Science 287:851–853
Tautz J, Zhang S, Spaethe J, Brockmann A, Si A, Srinivasan M (2004) Honeybee odometry: performance in varying natural terrain. PLoS 2:E211
von Frisch K (1967a) The dance language and orientation of bees. Harvard University Press, Cambridge
von Frisch K (1967b) Honeybee: Do they use direction and distance information provided by their dancers? Science 158:1072–1077
Winston ML (1987) The biology of the honeybee. Harvard University Press, Cambridge
Acknowledgement
This work was supported by Grants-in Aid from Bio-oriented Technology Research Advancement Institution (BRAIN) and Grants-in Aid for Scientific Research from the Japanese Ministry of Education, Culture, Sports, Science and Technology, Scientific Research on Priority Areas (Area No.454, Mobiligence Project).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
359_2007_234_MOESM1_ESM.tif
Electronic Supplementary Material Figure S1 Components of the projector light. (a) Wavelength and intensity of the white stripes measured inside the cone. (b) Wavelength and intensity of the black stripes measured inside the cone. (TIFF 1139 kb)
359_2007_234_MOESM2_ESM.tif
Electronic Supplementary Material Figure S2. Comparison of 2-min and 5-min ITIs. (a) Accumulated data (N=53 each) of several experiments performed in December of 2006 (N=24, maintained outside, and N=4 each maintained in the phytotron), and January of 2007 (N=25, maintained in the phytotron). The acquisition rates were plotted for both the 2-min ITI (N=53) and 5-min ITI (N=53) groups. Significant increases were observed in the 2-min ITI (one-way repeated measures ANOVA: F = 5.109, p < 0.01) and 5-min ITI groups (one-way repeated measures ANOVA: F = 4.606, p < 0.01). There was no significant difference between the two groups (p > 0.05). (b) The acquisition rates of selected experiments performed in January of 2007 (N=25, maintained in the phytotron) of (a) were plotted. Acquisition rates were low and there were no significant increases in the 2-min ITI (one-way repeated measures ANOVA: F = 1.931, p > 0.05) and 5-min ITI groups (one-way repeated measures ANOVA: F = 2.010, p > 0.05). (TIFF 1139 kb)
359_2007_234_MOESM3_ESM.tif
Electronic Supplementary Material Table S1 Response of individual bees over trials with 2-min ITIs (paired and unpaired). (TIFF 143 KB)
359_2007_234_MOESM4_ESM.tif
Electronic Supplementary Material Table S2 Response of individual bees to visual motion presented by animation and to manual motion with 2-min ITIs. (TIFF 133 KB)
Electronic Supplementary Material Movie S1 Movie showing a typical PER response of a bee to the CS in the 3rd trial (not yet conditioned) and in the 7th trial (conditioned). In the 7th trial, the bee exhibited a PER upon presentation of the motion cue. (MOV 2754 kb)
Electronic Supplementary Material Movie S2 Movie showing a typical PER response of a bee in the motion cue direction discrimination trials. The bee conditioned with the backward-direction motion cue did not exhibit a PER to the backward-direction motion cue. (MOV 8591 kb)
Rights and permissions
About this article
Cite this article
Hori, S., Takeuchi, H. & Kubo, T. Associative learning and discrimination of motion cues in the harnessed honeybee Apis mellifera L.. J Comp Physiol A 193, 825–833 (2007). https://doi.org/10.1007/s00359-007-0234-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-007-0234-x