Abstract
Adaptive mechanisms for unfavorable environments have evolved in plants for thousands of generations, primarily in the form of endogenous chemical signals and the coordination of physiological processes. Signaling peptides (SPs) are diverse molecular messengers in various stress responses which have been identified in different plant families. SPs are recognized by the membrane-localized receptors and co-receptors, leading to downstream signaling for various plant responses. Progress in in silico analysis, along with other factors, has increased our understanding of the signaling peptide-mediated regulatory mechanisms underlying the entire plant life cycle. SPs mediate both long-distance (root-to-shoot-to-root) and local cell–cell communication via vascular system to communicate and coordinate with plant organs at distant locations. During abiotic stress, SPs inside plant cells perceive stress signals and transfer information at short and long physiological ranges through the signal transduction pathway, causing stress-responsive gene expression. SPs interact with pathogens and mediate cell-to-cell communication via signaling pathways. There are intriguing relationships between phytohormones and the secondary signaling cascades which are mediated by SPs. During biotic or abiotic stress, different peptides trigger jasmonic acid, ethylene, and ABA signaling, involving several secondary messengers. These messengers mediate the stress response via shared signaling components of ROS, Ca2+, and MAPKs, and they modify the gene expression for different phytohormones. In this review, we highlight current knowledge on the role of signaling peptides in plant adaptation, growth, and development. We aim to analyze the SP-receptor interactions and the significance of crosstalk between a few sample SPs and phytohormones. Potential directions on how scientists can use this information for crop improvement are also suggested.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Signaling peptides (SPs), also known as ‘peptide hormones,’ are emerging growth hormones. These are small biological molecules of proteomes present in plants (usually < 20 amino acids when mature and rarely > 120 amino acids as a full-length precursor). SPs occur in very low concentrations at the physiological level (Olsen et al. 2002; Albert 2013; Matsubayashi 2014). Biosynthesis of these peptides involves proteolytic cleavage and post-translational modification, including tyrosine sulfation PHYTOSULPHOKINE (PSK), PLANT PEPTIDE CONTAINING SULFATED TYROSINE (PSY), INFLORESCENCE DEFICIENT IN ABSCISSION (IDA), ROOT GROWTH FACTOR (RGF) or CLEL/GLV and CASPARIAN STRIP INTEGRITY FACTOR (CIF) sulfated) by TPST, proline hydroxylation (Hyp) (proline-hydroxylated-systemin, CLAVATA 3 (CLV3)/EMBRYO SURROUNDING REGION RELATED (CLE), C-TERMINALLY ENCODED PEPTIDE (CEP), ROOT GROWTH FACTOR (RGFs), and PLANT PEPTIDE CONTAINING SULFATED TYROSINE 1 (PSY1), and hydroxyproline glycosylation (O-arabinosylation) (CLV3 and CLE2), in the primary structure of peptides for the generation of mature and active peptides for the bioactivation and perception of ligands (peptides) by receptors (Fig. 1) (Pearce et al. 2001a; Pearce and Ryan 2003; Ito et al. 2006; Kondo et al. 2006; Amano et al. 2007; Rojo and Denecke 2008; Ohyama et al. 2008, 2009; Komori et al. 2009; Matsuzaki et al. 2010; Tavormina et al. 2015; Patel et al. 2018). The plant genome contains several secreted peptide genes that encode SPs. In the model plant Arabidopsis thaliana, > 1000 genes encoding SPs have been reported, but only a limited number of signaling peptides have been characterized at the structural and functional levels (Lease and Walker 2006; Ghorbani et al. 2015). The secreted peptides orchestrate peptide-mediated cellular communication or signaling and bind to the corresponding plasma membrane-bound receptors (Czyzewicz et al. 2013). SPs act as ligands in this process, binding to specific membrane-localized receptors to make a ligand–receptor duo, which changes the configuration of receptors and activates their catalytic domains, whereas the spatial expression patterns of SPs lead to some specific interactions and functions in plants (Czyzewicz et al. 2013).
Receptors are the main switches initiating downstream short-range and long-distance signaling in the cell and modulating cellular activities (Czyzewicz et al. 2013). However, temporal receptor expression and limited peptides restrict the activity of peptide signaling (Breiden and Simon 2016). Various small signaling peptides are yet to be functionally characterized (Czyzewicz et al. 2013). Several biochemical, genetic, and bioinformatics-based methodologies can help identify and characterize SP receptors in plants.
SPs play an essential role in plant reproduction, growth, and development, including homeostasis maintenance, stomatal aperture control (Takahashi et al. 2018a; Yu et al. 2018; Brito et al. 2018; Zeng et al. 2022; Fedoreyeva 2023), development of seeds (Doll et al. 2020), shoots (Brand et al. 2000), roots (Fletcher 2020; Shao et al. 2020), and pollen (Ge et al. 2017; Li et al. 2020) and, thus, yield (Yu et al. 2019), through specific mobile cell-to-cell communication (Matsubayashi et al. 2001). SPs and receptor kinase-mediated cell-to-cell communication play key roles in the initial stages of plant growth (Escobar-Restrepo et al. 2007; Okuda et al. 2009; Kessler et al. 2010). Plant intercellular communication is based on the signaling of non-peptide plant hormones (auxin, gibberellin, ethylene, abscisic acid, cytokinin, and brassinolides), small peptides, and specific receptors (Matsubayashi 2011). Small SPs interact with mobile transcription factors and noncoding RNAs, playing a crucial role in plant cell-to-cell communication (Vanneste and Friml 2009; Busch and Benfey 2010; Van Norman et al. 2011).
SPs act as silent heroes in biotic and abiotic stress defense and tolerance under diverse climatic conditions (Lindsey et al. 2002; Murphy et al. 2012; Albert 2013; Matsubayashi 2014; Ghorbani et al. 2014). During abiotic stress, SPs inside plant cells perceive stress signals and transfer signal information at short and long physiological ranges through the signal transduction pathway, causing stress-responsive gene expression (Akpinar et al. 2012). For example, SPs can interact with pathogens (Goyal and Mattoo 2014) and mediate cell-to-cell communication via signaling pathways (Murphy et al. 2012; Araya et al. 2014a, b; Tavormina et al. 2015). Cys-rich peptides are antimicrobial peptides owing to their antibacterial, antiviral, and antifungal (e.g., PR12 or defensins) properties in plant–microbe interactions for disease resistance (Hu et al. 2018). Rapid alkalinization factors (RALFs) in Arabidopsis act as negative regulators of the plant immune response to bacterial infection (Mang et al. 2017). Plant defensins (PDFs)—antimicrobial and host defense peptides—enhance fungal (i.e., Fusarium spp., Botrytis cinerea, and Verticillium dahlia) and bacterial (i.e., Pectobacterium carotovorum) resistance in model plants (Carvalho Ade and Gomes 2011; Ahmed et al. 2012; Gaspar et al. 2014). While numerous studies on SPs have been conducted, only few reviews have integrated the available information. Here, we have highlighted the current knowledge on how plants use these molecular networks and cues to respond to major biotic and abiotic stresses.
Signaling Peptides: Classification, Structure, and Function
Most SPs have been characterized in angiospermic plants, while symbolic SPs have been identified in green algae and mosses. Several peptide classes show high interspecific and intraspecific sequence diversity, while others contain single peptides or are only present in distinct plant families (Wheeler and Irving 2012). The first signal peptide molecule was discovered by isolating the 18-amino-acid systemin peptide (TomSys) from Solanum lycopersicum (tomato); this SP is restricted to Solanaceae family of plants (Ryan and Pearce 1998). Systemin regulates signaling events in systemic response, induces proteinase inhibitor biosynthesis in tomato leaves (Pearce et al. 1991) and production of jasmonic acid (Farmer et al. 1992a, b), defends plants against both the abiotic (salinity and UltraViolet (UV)) (Holley et al. 2003; Dombrowski 2003), and biotic (herbivory) stresses and also modifies root growth in plants (Ryan and Pearce 2003; Narváez-Vásquez and Orozco-Cárdenas 2008). In general, SPs are categorized on their structural characteristics as cysteine-rich peptides (CRPs) and small post-translationally modified (PTM) peptides (Fig. 1) (Matsubayashi 2012; Olsson et al. 2019). CRPs have even numbers (2–16) of Cysteine/Cys residues, with each CRP class having a distinct number and linear organization of amino acids. Mature CRPs are generally 40-amino-acid long but sometimes have > 100 but < 160 amino-acid residues and larger than small PTM peptides possessing intermolecular disulfide bonds, which determine the three-dimensional structure of mature proteins (Pearce et al. 2001a, b). CRPs are cationic with a conserved N-terminal. The C-terminal domain is Cys-rich, generally requiring 4–16 Cys residues to establish disulfide bridges to keep mature peptides in an active conformation (Fig. 1) (Pearce et al. 2001a). Disulfide bridges selectively degrade variable regions of pre-pro-peptides. The active peptide then becomes available for receptor binding. These peptides have several uniting structures regardless of higher amino-acid sequence divergence between individual peptides (Pearce et al. 2001a). CRPs act as antimicrobial compounds during plant–microbe interactions (Van Der Weerden et al. 2013; Tavormina et al. 2015), leaf stomata patterns and density, symbiosis, germination, guidance and burst of pollen tubes, plant gamete activation and seed development. CRPs are predominantly copious during plant reproduction. Due to their role in various reproductive processes, CRPs occur in female and male gametophytes, unlike PTM peptides that are mainly present in vegetative tissues (Hara et al. 2007; Sugano et al. 2010; Maróti et al. 2015; Bircheneder and Dresselhaus 2016). CRPs do not undergo post-translational modifications. They are further characterized into two subgroups based on proteolytic processing. The first subgroup includes CRPs that undergo proteolytic cleavage. For example, STOMAGEN/EPIDERMAL PATTERNING FACTORS (EPF)-LIKE9 (EPFL9) of the epidermal patterning factor (EPF) peptide family. STOMAGEN is a positive regulator of leaf stomata density (Hara et al. 2007; Kondo et al. 2010; Sugano et al. 2010). Another example is RAPID ALKALINIZATION FACTOR 1 (RALF1), recognized by the FERONIA (FER) receptor (Haruta et al. 2014; Pearce et al. 2001b), which also shows proteolytic cleavage and plays a role in cell expansion. The second subgroup contains CRPs that lack proteolytic cleavage such as S-locus cysteine-rich protein/S- locus protein 11 (SCR/SP11) and LUREs (Schopfer et al. 1999; Takayama et al. 2001; Okuda et al. 2009).
Small PTM peptides are small mature peptides (~ 10–20 amino-acid residues) produced by proteolytic processing and encoded by multiple paralogous genes. The longer translated precursor/propeptides correspond to mature peptides encompassing ~ 70–120 amino acids with few or no Cysteine/Cys residues, share conjoint tripartite structure, and undergo one or more PTMs in the C-terminal region; for instance, tyrosine sulfation, hydroxyproline glycosylation, and proline hydroxylation (Fig. 1). Non-CRPs have a tripartite structure of signal peptide at the N-terminal region, conserved C-terminal motif yielding the mature peptide, and frequently have proline residues in different genera within a plant family and a variable segment connecting the two (N and C) terminal domains (Matsubayashi 2014). PTMs increase the binding capacity of the signaling peptide to the receptor through structural conformation of peptides or direct interaction with receptors (Matsubayashi 2011). Non-CRP peptides form a major group of signaling peptides involved in plant growth and development through the regulation of many intercellular communication processes (Matsubayashi 2011). Examples include TRACHEARY ELEMENT DIFFERENTIATION INHIBITORY FACTOR (TDIF) (Matsubayashi and Sakagami 1999), ADVENTITIOUS ROOT FORMATION FACTOR (Amano et al. 2007), PLANT PEPTIDE CONTAINING SULFATED TYROSINE 1 (PSY1) (Amano et al. 2007), CLAVATA3CLV3/ EMBRYO SURROUNDING REGION RELATED (CLE) (Katsir et al. 2011), C-TERMINALLY ENCODED PEPTIDE (CEP) (Akker et al. 2016), and ROOT GROWTH FACTOR (RGF)/GOLVEN (GLV)/CLE-like (CLEL) (Fernandez et al. 2015), PHYTOSULFOKINE (PSK), and INFLORESCENCE DEFICIENT IN ABSCISSION (IDA). Table 1 lists the functions of various plant-signaling peptides.
Identification of SP Receptors and Analysis of SP-receptor Interactions for Plant Signaling
The identification and functional annotation of SP receptors are key to understanding the mechanisms of signal transduction systems in plants. In the last two decades, various biochemical, genetics, and bioinformatics-based methods have been used to identify and characterize SP receptors in plant genomes, accelerating the discovery of SP receptors corresponding to secreted peptide hormones. Chow and McCourt (2006) defined hormone receptors based on their potential to recognize the subtle structural diversity between small molecules in a specific manner. As per this standard definition, molecular interactions between receptors and hormones should be non-covalent and reversible, producing an activated complex that induces the primary hormone response. From the receptor side, functional annotation of peptide signaling pathways can overcome the genetic redundancy of ligand peptides that are found exclusively as multi-gene families in plant genomes. Receptors are classified into three major categories based on their biochemical properties: two-component receptor system (TCS), leucine-rich repeat (LRR)-based receptors, and ubiquitination-based hormone receptors. Systematic classification of receptors allows researchers to compare their structure and function within plant hormone systems and other kingdoms (Chow and McCourt 2006).
TCS are well-known receptors in prokaryotes, fungi, slime molds, and plants (Stock et al. 2000; Ahmad et al. 2020). Members of this system play a principal role in regulating and modulating a diverse range of growth and developmental processes, including biotic and abiotic stress (particularly drought, high salinity, and high or low temperature) responses. TCS regulators were initially reported in prokaryotes and represent up to 1% of the genome size (Mizuno 1997). TCS systems of fungi and plants are more complicated than the canonical prokaryotic system (Chow and McCourt 2006). In this system, histidine kinases (HKs), an ancient and ancestrally conserved signaling mechanism, sense a signal input and response regulator that play an important role in output. In plants, TCS regulators typically comprise three types of proteins, including HKs, phosphotransfers (HPs), and response regulator proteins (RRs) (Ahmad et al. 2020). The functional members of TCS have been identified and characterized in the model plant Arabidopsis using different genomics methods, with three transmembrane HKs (AHK2, AHK3, and AHK4) characterized as cytokinin receptors (Schaller et al. 2008; He et al. 2020). These HKs respond negatively to biotic and abiotic stresses in Arabidopsis cytokinin signaling (Urao et al. 2000). In the transmitter functional domain, these three HKs perceive stimuli and are autophosphorylated at a conserved histidine residue, followed by the transfer of phosphoryl groups to HPs at a conserved residue of aspartate (He et al. 2016). Histidine–aspartate signaling mediated by TCS regulators controls a wide range of biological processes, including cell division, vascular differentiation, leaf senescence, male and female gametogenesis, seed development, and responses to environmental stimuli (He et al. 2016; Liu et al. 2020; Huo et al. 2020). TCS regulators are reported from several plants, including Arabidopsis, rice, maize, soybean, wheat, sesame, Chinese cabbage, tomato, cucumber, and Brassica rapa, among others (Pareek et al. 2006; Mochida et al. 2010; Chu et al. 2011; Liu et al. 2014; Gahlaut et al. 2014; He et al. 2016). However, studies are needed to determine the exact role and signal transduction mechanisms of each TCS element in plants. TCS signaling machinery in Arabidopsis is a complex system comprising 11 AtHKs, five AtHPTs, and 23 AtRRs (Hwang et al. 2002). Most TCS members in Arabidopsis interact with abscisic acid (ABA) in response to different abiotic stresses i.e., drought, salt, and low temperature (Nishiyama et al. 2013; Nguyen et al. 2016; He et al. 2016). The expression levels of three TCS elements (AHP1, AHP2, and AHP3) decrease significantly under heat stress (Miyata et al. 1998; He et al. 2020). Functional characterization of AHK1, a functional member of the AHK family, demonstrated its crucial role in stress responses, including drought, high salinity, and osmotic stress (Huo et al. 2020). Zhao et al. (2020a, b) reviewed the crosstalk between ethylene and other plant hormones focusing on rice and Arabidopsis mechanisms. Sharan et al. (2017) assembled an interactome map of TCS proteins in rice using a highly stringent Y2H platform and extensive in planta BiFC assays. The results revealed different sensory OsHK-mediated crosstalk in signaling. The same study reported OsHPTs as interaction hubs with OsRRs and highlighted interactions between type-A and type-B OsRRs, which led to a comprehensive molecular investigation of signal(s) transmitted by different TCS elements in a wide range of biological processes. Zhao et al. (2020a, b) analyzed a rice ethylene-response mutant mhz1 and reported that MHZ1 positively modulates root ethylene responses; MHZ1/OsHK1 works with the OsEIN2-mediated pathway to regulate root growth on ethylene perception. A more recent study demonstrated that HISTIDINE KINASE1 (HK1) coordinates the transfer cell specification in young barley endosperm (Hertig et al. 2020). Using RNA interference-mediated downregulation of Barley HISTIDINE KINASE1 (HvHK1), function of HvHK1 was investigated in ETC fate acquisition.
The leucine-rich repeat receptor kinase (LRR-RK) is one of the largest membrane integral receptor families evolutionary conserved in plants and animals. In plants, these receptors are essential for the intercellular signaling network as they perceive secreted hormone peptides (Chakraborty et al. 2019) and are involved in diverse external and internal signal responses to regulate plant growth and development (Ganie et al. 2021). Plant RKs are classified into 14 subfamilies based on ectodomain structure (Shiu and Bleecker 2001; Gou et al. 2010; Chakraborty et al. 2019). Previous genomic and biochemical studies report the XI class of the LRR-RK family as receptors of different SP signals, including CLE/TDIF (Ogawa et al. 2008; Hirakawa and Bowman 2015), IDA (Santiago et al. 2016), CEP (Tabata et al. 2014), Pep1 (Yamaguchi et al. 2006), RGF (Shinohara et al. 2016), PIP1 (Hou et al. 2014), and CASPARIAN STRIP INTEGRITY FACTORS (CIF) (Nakayama et al. 2017). While subclass X of the LRR-RK family is known for the PSK receptor PSKR (Matsubayashi et al. 2002). Through specific binding with ligands and hormones, LRR-RK controls diverse biological processes, such as cell division, proliferation, differentiation, and stem cell balance (Chakraborty et al. 2019). Recent efforts have identified novel LRR genes at the genome scale using a forward genetic approach. Three LRR-RKs (RGFR1, RGFR2, and RGFR3) were overexpressed in different root tissues, including the proximal meristem, elongation zone, and differentiation zone. In Arabidopsis, these three LRR-RKs directly interacted with the ROOT MERISTEM GROWTH FACTOR (RGF) peptide and regulated root meristem development (Shinohara et al. 2016). In Arabidopsis, cell wall LRR extensions (LRX) 3/4/5 are important for plant salt tolerance (Zhao et al. 2018a, b). Using immunoprecipitation–mass spectrometry (IP–MS), this study reported that LRX3/4/5 proteins interact with RALF peptides and defined an important signaling pathway for regulating plant growth and salt tolerance. Trihemasava et al. (2020) analyzed N-glycans on MIK1 and reported clusters of glycosylation that may explicate the regions participating in MIK1 ectodomain binding. Several studies have identified the crystal structure of different peptide-LRR-RK receptor pairs using X-ray crystallography and NMR (Song et al. 2016; Chakraborty et al. 2019). Peptide ligands act as molecular glue to stabilize the interaction between each corresponding receptor and its co-receptor (Fig. 2) (Morita et al. 2016). Song et al. (2016) reported the structure-guided discovery of RGF receptors for sustainable plant development. At a resolution of 2.6 Å, the same authors identified the crystal structure of RGF1 in a complex with LRR domain of RGFR1. Interaction between RGF1 and LRR domain suggests that the Arg-x-Gly-Gly (RxGG) motif is important for recognizing the sulfate group of RGF1 by RGFR1.
Crystal structure of the probable LRR receptor-like serine/threonine-protein kinase At4g26540 (Chain B) with peptide PTR-SER-ASN-PRO-GLY-HIS-HIS-PRO-HYP-ARG-HIS-ASN (Chain A); a Hydrophobicity surface representation of the overall structure of hormone receptor RGFR1 in complex with RGF1 (PDB: 5HYX); b A close view of pocket with peptide structure in ball-and-stick model colored by atom types, yellow dashed lines indicate the hydrogen bond networks; c Peptide molecule as ball-and-stick model with interacting receptor residues labeled. This structure was rendered in different CPK using UCSF-Chimera, PyMol, and Ligplot + programs
Some plant hormones have unique signaling mechanisms, evidenced after discovering a cluster of genes responsive to exogenous auxin (Chow and McCourt 2006). In general, these types of genes encode two classes of molecules, Aux/IAAs and SCF (Skp1/Cullin/F-box) ubiquitin proteins (Rouse et al. 1998; Tian and Reed 1999; Nagpal et al. 2000; Ruegger et al. 1998; Gray et al. 2001; Rogg et al. 2001). Aux/IAAs belong to primary/early auxin-response genes, such as GH3 (Gretchen Hagen 3) and SAUR (small auxin up RNA) (Abel and Theologis 1996), which play a crucial role in the early stages of auxin signal transduction and respond quickly to auxin treatment. Functional members of Aux/IAAs mediate phytohormone signaling pathways in plants, including the jasmonic acid, salicylic acid, ethylene, and brassinosteroid pathways (Kazan and Manners 2009; Waseem et al. 2018).
The molecular weight of Aux/IAA proteins ranges from 18 to 36KD and is localized in nuclear compartment (Guifoyle and Hagen 2007). The number of Aux/IAA domains leads to classification of the whole family into four groups (I to IV). A typical domain of these proteins comprises 7–40 amino-acid residues (Gray et al. 2001), whereas on the basis of presence of four functional domains, Aux/IAAs are classified into canonical Aux/IAAs (with domains) and non-canonical Aux/IAAs (without domains). In general, Domain I of Aux/IAAs comprises LRRs (LxLxLx) that function as transcriptional repressors (Song et al. 2009). Domain II interacts with F-box proteins and plays an important role in Aux/IAA stability (Tiwari et al. 2004). Among Aux/IAAs and ARFs, Domains III and IV are responsible for homodimerization and heterodimerization (Rinaldi et al. 2012). The availability of whole-genome sequencing data in public repositories has accelerated genome-wide identification and functional annotation of Aux/IAAs. The auxin-insensitive mutant iaa1/axr5 revealed auxin-mediated growth effects in Arabidopsis (Yang et al. 2004). OsIAA9 or OsIAA13, a functional member of the Aux/IAA family, affects starch accumulation and participates in lateral root intonation in rice (Luo et al. 2015). Recently, at the TIR1 N- and C-termini, essential residues were identified with attractive interfaces for no-native interaction with IDRs followed by folded PB1 domain found in Aux/IAA (Niemeyer et al. 2020). Sun et al. (2020) produced a comprehensive protein–protein interaction map that mediates environmental and hormonal responses and cloned 113 phytohormone-related genes and 29 light signaling components in Arabidopsis. After individual testing, 141 interactions were identified using an Y2H approach. The results of these studies demonstrate that peptide–receptor compatibility is crucial for understanding peptide-mediated signaling in plants.
Post-translational Modifications of Signaling Peptides for the Orchestration of Peptide-Signaling and other Signaling Pathways
Signaling peptides are emerging growth hormones that mediate cell-to-cell signal transmission and regulate plant growth and development. The SPs act as ligands and make a ligand–receptor duo with the membrane-localized receptors, which are main switches that initiate the downstream signaling in the cell. Peptide biosynthesis often involves two major events: proteolytic cleavage and PTM. Analysis of peptide primary structure indicates that these two modifications are important to produce a mature and active peptide (Rojo and Denecke 2008).
Proteolytic Cleavage
The proteolytic cleavage is an important event in the life of precursor proteins to release an active and mature peptide. It is carried out with the involvement of different protease enzymes. The precursor proteins consist of a signal located at the N-terminus, which is cleaved by the peptidases once the precursor or pre-pro protein enters the endoplasmic reticulum (Tavormina et al. 2015). Proteolytic cleavage of the signal from N-terminus is necessary for further PTM. A peptide may be cleaved at single point or multi-points depending upon the location of peptide on pre-pro-protein. For example, in some of the RGF and CLE family peptides, situated at the C-terminus, proteolytic cleavage at only one point is sufficient to release the mature peptide (Fig. 3). In most cases, the peptide is located near the C-terminus and not at the exact end of the pre-pro-protein, and in such cases, proteolytic lysis at two points is needed to get a mature peptide (Ghorbani et al. 2015).
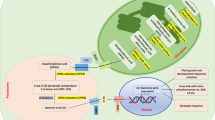
A signaling peptide (SP). Nucleus-localized genes encode pro-preproteins peptides, undergoing cleavage processing for cleaving signal peptide in endoplasmic reticulum (ER). Maturation of peptides includes cleavage of the signal peptide (SP) in the ER during preproprotein synthesis, tyrosine sulfation by TPST in the Golgi apparatus (Komori et al. 2009), proline hydroxylation by prolyl-4-hydroxylase (P4H) and tri-arabinosylation by hydroxyproline O-arabinosyltransferase (HPAT) in the case of PSYs in the Golgi apparatus, and cleavage of the N- and C-terminals by subtilases (SBT) in the apoplast to release the mature peptide. Peptides are perceived at the plasma membrane by a receptor/co-receptor pair that mutually transphosphorylate each other and activate the receptor. LRR-RLKs-HEA and proton-pumping H+-ATPase (AHA) have been identified as direct targets of the peptide’s receptors PSKR1 and PSY1R. Modified peptides are predicted to regulate Pep-responsive genes (SPRGs) via unknown signaling intermediates and transcription factors (MYB15/MY36-CIFs), and via MAPK, Transphoprylation signaling pathway produces a biological response
Post-Translational Modifications (PTMs)
Till now most of the peptides reported are derived from non-functional longer precusor; and these peptides are grouped into three categories viz., (1) peptides subjected to specific PTMs, (2) Cys-rich peptides without PMT, and (3) non-Cys-rich peptides without PMTs (Tavormina et al. 2015). However, the post-translational modified peptides are derived from 70 to 120 amino-acid long pre-proproteins. Three types of PTM have been identified: tyrosine sulfation, proline hydroxylation (Hyp), and hydroxyproline glycosylation (O-arabinosylation) (Tavormina et al. 2015) (Fig. 1). While the enzymes responsible for PTMs have been identified, how they affect peptide bioactivity and signaling is unknown.
The first type of PTM is tyrosine sulfation, which occurs via peptide sulfation of tyrosine residues in most eukaryotes, including higher plant peptides. Peptides undergo sulfation in secretory pathways by a transmembrane enzyme, tyrosylprotein sulfotransferase (TPST), which transfers the sulfate group from 3′-phosphoadenosine 5′-phosphosulfate (PAPS) to the tyrosine residue (Moore 2003). Four peptide classes—PSKs (PHYTOSULFOKINES), PSYs (PLANT PEPTIDES CONTAINING SULFATED TYROSINE), RGF (ROOT MERISTEM GROWTH FACTORS), and CLEL (CLAVATA3/EMBRYO SURROUNDING REGION-LIKE) /GLV (GOLVEN), and CASPARIAN STRIP INTEGRITY FACTOR (CIF) are reportedly sulfated by TPST in tyrosine residues. The PSK and PSY precursor proteins are sulfated using TPST as the catalyzing enzyme and PAPS as the sulfur donor (Komori et al. 2009). Sulfation of tyrosine increases the hydrophilic binding of peptides, enhancing their affinity to bind more strongly to their receptors (Wang et al. 2015). For example, the binding of PSK to PSKR1 is mediated by H-bond formation and interaction of the sulfate group with tyrosine residue at the sub-domain of PSKR1 (Stührwohldt et al. 2011; Igarashi et al. 2012). Similarly, the binding affinity of unsulfated RGF peptides to RGFR1 decreased by approximately 200 times (Shinohara et al. 2016). These studies suggest the importance of sulfation for activating signaling peptides and peptide–receptor interactions. Because TPST plays a crucial role in tyrosine sulfation of signaling peptides, analysis of loss-of-function tpst mutants is useful for elucidating the bioactivity and impact of sulfated peptides on plant growth. Arabidopsis Attpst knockout mutants showed pleotropic phenotypes with dwarf roots, small hypocotyls, smaller veins, pale green leaves, premature senescence, and fewer flowers and fruits (Stührwohldt et al. 2011; 2015). However, all defective phenotypes could be recovered partially by applying sulfated peptides to tpst knockout plants (Komori et al. 2009; Doblas et al. 2017). Besides, plants with unsulfated and non-hydroxylated CLE8 peptides have altered lateral root growth and development; this phenotype was rescued by applying sulfated CLE8 (Matsuzaki et al. 2010). These results indicate the crucial role of tyrosine sulfation in signaling peptides for plant growth and development and stress resistance.
The second type of PTM is proline hydroxylation (Hyp), the most common and stable type in plant peptides, growth factors, and hormones (Myllyharju 2003). Various Hyp peptides, including HypSys (proline-hydroxylated-systemin), CLV3, CLE, CEPs, RGFs, and PSY1 have been isolated from plant tissues (Pearce et al. 2001a; Pearce and Ryan 2003; Ito et al. 2006; Kondo et al. 2006; Amano et al. 2007; Ohyama et al. 2008, 2009; Matsuzaki et al. 2010; Whitford et al. 2012; Patel et al. 2018). Accumulating evidence reveals that Hyp is crucial for peptide bioactivation and ligand perception (Mohd-Radzman et al. 2015; Patel et al. 2018). Peptides have been isolated in PTM (hydroxylated or glyoxylated) or unmodified (Patel et al. 2018) forms. This variation in peptide structure marks the significance of post-translational modification in the biogenesis of structurally different peptides or the inefficiency of PTM to make the peptides functionally active. Although Hyp is the most commonly occurring PTM in plants, the enzyme hydroxylating proline residues is unknown. Putative prolyl 4-hydroxylases (P4Hs) are involved in the biosynthesis of hydroxyproline-rich glycoproteins, including extensins, arabinogalactan proteins, and proline-rich proteins (Velasquez et al. 2015). Arabidopsis has 13 P4H-encoding genes, including those encoding enzymes for modifying precursor proteins (Myllyharju 2003; Matsubayashi 2012). P4Hs are Golgi-localized proteins (Yuasa et al. 2005), hydroxylating proline at different positions in precursor peptides, an initial step for further O-linked glycosylation (Pearce et al. 2001a; Amano et al. 2007; Ohyama et al. 2009). Among the Golgi-localized P4Hs, those which require P4H for peptide biogenesis have not been discovered. The INFLORESCENCE DEFICIENT IN ABSCISSION (IDA) is a 14-mer peptide post-translationally hydroxylated at the ninth proline residue and functions in the abscission of senesced or damaged floral parts (Butenko et al. 2003). IDA acts as a ligand, inducing the expression of genes encoding LRR-RLKs-HAE and HAESA-like 2 (HSL2) receptors in the abscission zone (Santiago et al. 2016). Post-translational proline hydroxylation is important for the functional activation of IDA, its perception by receptors, and activation of the downstream-signaling cascade required for cell separation in the abscission zone (Schardon et al. 2016). Besides HAE and HSL2 receptors, IDA appoints SOMATIC EMBRYOGENESIS RECEPTOR KINASE (SERKs) which act as act co-receptors downstream of IDA. The ligand–receptor complex produced by IDA-HAESA-SERK generates a signal initiating a downstream-signaling pathway involving MITOGEN-ACTIVATED PROTEIN KINASE (MAPK). In turn, MAPK and MKK4/MKK5 act in the signaling pathway to regulate cell separation that leads to the abscission of floral parts (Stenvik et al. 2006; Cho et al. 2008; Meng et al. 2016). Analysis of the structural activity of the IDA and receptors show that proline hydroxylation at position 9 enhances IDA activity in Nicotiana bethemiana (Santiago et al. 2016). On the other hand, the abscission activity in floral parts remained unaffected in hydroxylated or non-hydroxylated IDA (Stührwohldt et al. 2018). Post-translational hydroxylation of proline residues at positions 4 and 11 is highly effective in the bioactivation of CEPs to increase legume nodulation (Imin et al. 2013; Mohd-Radzman et al. 2015). In contrast, modifying proline at position 11 with the tri-arabinosyl group decreases CEP activity (Patel et al. 2018), suggesting that not all PTMs are involved in peptide activation.
The last major type of PTM is hydroxyproline arabinosylation, common in several classes of abundant cell wall proteins and frequently observed in plant peptide hormones and growth factors (Kieliszewski et al. 2011). The enzyme involved in Hyp-arabinosylation is hydroxyproline O-arabinosyltransferase (HPAT), a golgi-targeted type II transmembrane protein which triggers the arabinosylation signaling cascade resulting in tri-arabinosylation at the hydroxyl group of proline. In Arabidopsis, three genes encode HPAT and hpat knockout mutants with loss-of-function of signaling peptides, and other cell proteins as well showed various defective phenotypes with cell wall thickness, altered flowering time, early senescence, hypocotyl elongation, and impaired pollen tube development (Ogawa-Ohnishi et al. 2013). CLV3 is 13-amino-acid long peptide, hydroxylated at seventh proline residue and triarabinosylated and is the first identified hydroxyproline-arabinosylated peptide growth factor purified from Arabidopsis plants overexpressing CLV3 precursor peptide. Similarly, another proline hydroxylated and triarabinosylated glycopeptide, CLE2, was purified from apoplastic extracellular fluids. Constitutive expression of CLV3 and CLV2 in knockout clv3 mutants revealed the importance of O-arabinosylation in the activation and maturation of CLV3/CLV2 peptides (Ohyama et al. 2009). CLAVATA3/Endosperm Surrounding Region (CLE) peptides are 13-amino-acid long peptides modified with triarabinose at Hyp at position 7. They are involved in the orchestration of signaling between roots and shoots to induce nodulation (Imin et al. 2018). The importance of glycosylation in CLE40 activity (Okamoto et al. 2013) and perception by ligands in soyabean was reported (Corcilius et al. 2017). Functional analysis of clv3 knockout mutants showed that the resumption of normal phenotypes occurs by complementation with glycosylated CLV3/CLV2 only, indicating that CLV peptides involve hydroxylation of proline residues and arabinosylation of hydroxyproline (Ohayama et al. 2009). Molecular studies suggest that triarabinosylation induces conformational changes in CLV3, influencing CLV3 binding specificity for receptors which was evaluated in an in vitro study of chemically synthesized CLV3, which interacts more strongly with CLV1 (CLAVATA1, receptor kinase) than non-arabinosylated CLV3 (Ohayama et al. 2009), restoring large meristems in the loss-of-function clv3 mutants upon exogenous application (Prusinkiewicz et al. 2007). These findings suggest that a considerable proportion of structurally related CLV peptides may be arabinosylated. Post-translational glycosylation affects peptide conformation through steric interactions with the peptide backbone, modulating the binding ability and specificity of peptides to target proteins (Seitz 2000). Tyrosine-sulfated peptides, including PSY1, CEPs, and HypSys, need additional modification of arabinosylation at Hyp residues to enhance their activity (Amano et al. 2007; Pearce and Ryan 2003). It is noteworthy that the first identified HypSys in tomato was found without Hyp or Hyp-glycosylation (Pearce et al. 1991, 2001a). These reports highlight that glycosylation plays a reversible (arabinosylation/dearabinosylation) role in regulating peptide activity and ligand-receptor binding.
Mobility of the Small Peptides: Short Range (intercellular) or Long-Distance (from source tissue to target tissue)
Signaling peptides mediate both long-distance (root-to-shoot-to-root) and local signal; cell–cell communication systems and peptide-based signaling communication are dominant in plants (Lease and Walker 2006; Oh et al. 2018). Signaling peptides can be mobile or bound to the membranes. Peptides are recognized by the receptors localized on membranes and co-receptors of shape-complementary nature. This ligand-receptor/co-receptor connotation starts signaling at intracellular levels for various plant responses (Kim et al. 2021). Signaling peptides regulate vascular development and abiotic stress responses by localized cell-to-cell communication pathways (Fukuda et al. 2007; Fukuda and Hardtke 2020; Kim et al. 2021). Tracheary Element Differentiation Inhibitory Factor (TDIF) produced in phloem translocate to cambium and attaches to the PHLOEM INTERCALATED WITH XYLEM/TDIF RECEPTOR (PXY/TDR) on the cell membrane (Hirakawa et al. 2008; Etchells et al. 2016; Fletcher 2020).
Long-distance signals transport rapidly through mass flow in xylem or phloem pathways. Thus, movement of peptides within vascular system allows one part to communicate and coordinate with plant organs at distant locations (Ham and Lucas 2017; Winter and Kragler 2018). Organ-to-organ communication mediated by long-distance signals in xylem and phloem is crucial to maintain homeostasis in plants (Caetano-Anollés and Gresshoff 1990; Oka-Kira and Kawaguchi 2006; Ruffel et al. 2011). There is a functional connection between xylem and phloem signaling pathways. Small mobile signaling peptides specifically are usually recognized and bind to an array of extracellular receptor domains of transmembrane proteins of receptor-like kinase family which are important components of their perception machineries. The peptide–receptor interaction triggers various processes at biochemical and physiological levels. SERK family receptor-like kinases act as co-receptors for the activation of peptide–receptor-like kinase pair through heterodimerization and transphosphorylation (Oh et al. 2018; Chen et al. 2020b).
Systemin signal peptides are also supposed to show long-distance signaling by transport through phloem (Chen and Kim 2006; Lough and Lucas 2006). Signal peptides applied exogenously to stems transport through xylem to the leaves (Pearce et al. 2001a, b; Scheer et al. 2005; Huffaker et al. 2006). The SHORTROOT protein is synthesized in the xylem, procambium, and pericycle of roots and transported locally to phloem poles and quiescent center to regulate development of phloem (Kim et al. 2020). Arabidopsis CLE genes encode peptides which are recognized by transmembrane receptors at the cell surface and in turn trigger signal transduction at intracellular level. This regulates growth and development in plants (Yamaguchi et al. 2016; Fletcher 2020; Fukuda and Hardtke 2020). The CLAVATA3/ESR-related 25 peptides (CLE-RS) and C-terminally Encoded Peptide (CEP) arrays are translocated from the root vascular system (xylem) as long-distance (root to shoot) mobile signals of drought or Nitrogen starvation to the shoot wherein ascending signals are recognized and directly bound to membrane-associated receptor kinases (HAR1, CLV1-homologous receptor) and CEP receptors, respectively (Okamoto et al. 2013; Tabata et al. 2014; Takahashi et al. 2018a, b, c). A CLE-RS receptor (HAR1) and CEP1 receptor (XIP1/CEPR1) are specifically expressed in the phloem (Nontachaiyapoom et al. 2007; Bryan et al. 2012). The peptides derived from roots are converted to secondary signal messengers in phloem and transported via phloem sap from “shoot-to-root” (Sasaki et al. 2014). The ascending signals trigger shoot–to-root secondary signals and induce systemic response of plant roots to upregulate nitrate transport. In nitrogen status signaling network, plant hormone cytokinin operates alongside CEP DOWNSTREAM peptides which send long-range communication signals from shoot to root through phloem (Ruffel et al. 2016; Ohkubo et al. 2017; Poitout et al. 2018). CLE-RS/HAR1 cascade plays vital role in autoregulation of nodulation in legumes (Sasaki et al. 2014).
The CEP genes upregulate in response to N-starvation and express in lateral root stele. Then CEP family peptides translocate from xylem vessels to shoots wherein these are recognized by the CEP Receptor 1 (CEPR1) kinase of leaf vascular tissue (Roberts et al. 2013; Delay et al. 2013; Tabata et al. 2014). The putative shoot-derived secondary signals upregulate nitrate transporter gene (NRT2.1) in plant roots to compensate for local N-starvation (Roberts et al. 2013; Delay et al. 2013). CEP-CEPR-signaling module works in all seed plants. According to Takahashi et al. (2018a, b, c), CLE25 is drought activated to mobile signal which moves from plant roots to the leaves wherein these are perceived by BAM1 and BAM3 receptors and CLE25 interactions activate carotenoid-cleaving enzyme NCED3 expression. NCED3 generates an ABA precursor molecule, active ABA signal, which enables plant to cope with a water shortage (Nambara & Marion-Poll 2005).
Signaling Peptide-Mediated Regulation of Plant Growth and Development
Small SPs help regulate plant growth and development through specific mobile cell-to-cell communication (Brand et al. 2000; Matsubayashi et al. 2001; Brito et al. 2018; Zeng et al. 2022; Fedoreyeva 2023). Knowledge of signaling peptide-mediated regulation pathways underlying the whole-plant life cycle is expanding, mostly due to progress in in silico analysis, in vitro design, and in planta verification (Murphy et al. 2012; De Coninck and De Smet 2016; Boschiero et al. 2020; Fletcher 2020). Here, we focus on biological function, interactions, and crosstalk between some representative SPs and phytohormones, target receptors, and downstream changes to illustrate their role in plant growth and development (Table 2).
Seed Development
Seed development requires deep crosstalk between the embryo and endosperm (Moussu et al. 2017). During seed development, the embryonic cuticle serves as a hydrophobic barrier for de novo deposition of the embryo, likely controlled by a signaling pathway involving ABNORMAL LEAF SHAPE1 subtilase (ALS1) and two GASSHO receptor-like kinases (GSO1 and GSO2) (Creff et al. 2019; Doll et al. 2020). A recent report showed that a sulfated peptide TWISTED SEED1 (TWS1) acts as a GASSHO ligand in the embryo, but ALS1 modulates its precursor in the neighboring endosperm to release active peptides (Doll et al. 2020). Thus, the signaling peptide TWS1 mediates a bidirectional molecular dialog between the embryo and endosperm before seed germination.
Shoot Development
The CLAVATA3 (CLV3) peptide is a well-known and major regulator in shoot apical meristem (SAM), determining stem cell fate in the division of aboveground leaves, stems, and floral parts (Brand et al. 2000). The secreted peptide CLV3 from the outermost meristem cell layers binds with the CLV1 receptor (containing tandem LRR domain) in deeper cell layers to repress the expression of homeodomain transcription factor WUSCHEL (WUS), restricting stem cell expansion (Yadav et al. 2011). In contrast, WUS promotes the expression of CLV3 and, thus, leads to a negative feedback signaling pathway in shoot growth and development by modulating CLV1 and WUS in flowering plants (Murphy et al. 2012). However, a recent paper reported that CLV3/CLE is a haploid stem cell-promoting signal in the liverwort Marchantia polymorpha, suggesting a critical role in the evolution of land plants (Hirakawa et al. 2020).
Root Development
In the root apical meristem (RAM), CLV3/EMBRYO SURROUNDING REGIONRELATED (CLE) peptides act synergistically with a set of specific receptors, hormones, or other catalytic substances (Fletcher 2020). Similar to the CLV3 signaling pathway, CLE40 interacts with the RLK ARABIDOPSIS CRINKLY4 (ACR4) to activate the expression of WUSCHEL-related homeobox 5 (WOX5) in controlling cell meristematic activity (Murphy et al. 2012). A recent study suggested that ginsenosides modulate a novel PgCLE45–PgWOX11 regulatory loop for adventitious root branching (Liu et al. 2020). Furthermore, RAM involves crosstalk between CLE members and cytokinin. CLE10 inhibits root protoxylem differentiation by repressing the expression of ARABIDOPSIS RESPONSE REGULATOR (ARR5 and ARR6) to negatively regulate cytokinin signaling (Cammarata et al. 2019). In addition, the peptide ROOT GROWTH FACTOR 1 (RGF1) binds with LRR-RLK INSENSITIVE (RGI) to modulate the expression of PLT1 and PLT2, thereby guiding the YDA-MKK4/MKK5-MPK3/MPK6 cascade for primary root growth (Song et al. 2016; Shao et al. 2020).
Pollen Development
During the meeting of sperm and ovule by pollen tube delivery, 5-kDa cysteine-rich peptide RALFs and their RLK1-like receptor kinases (RLK1Ls) modulate pollen growth via the autocrine and paracrine signaling pathway (Ge et al. 2017). Two secreted peptides (RALF4 and RALF19) are autocrine ligands of BUPS1/2–ANX1/2 receptor complexes that maintain pollen tube integrity; when sperm enters the pollen tube for fertilization, another peptide (RALF34) acts as a paracrine signal competing with RALF4/19 to induce timely tube rupture for the release of sperm (Ge et al. 2019). In addition, the grass-specific EXINE PATTERN DESIGNER 1 (EPAD1), containing N-terminal signaling peptide and C-terminal glycosylphosphatidylinositol (GPI)-anchor sites, can bind with plasma membrane lipids to form primexine for pollen exine (Li et al. 2020).
Yield
Yield formation mediated by signaling peptides requires a full-screen signaling crosstalk at whole-plant growth and development stages. Similarly, two secreted signaling peptides (EPFL2 and EPFL9) coordinate ovule patterning and seed number with gynoecium and fruit growth by binding with ERL1 and ERL2 (LRR receptor-like) kinases (Kawamoto et al. 2020). Correspondingly, heterologous expression of soybean phytosulfokine (GmPSK1), a sulfated pentapeptide hormone with the sequence YIYTQ, markedly increased seed growth (seed size and weight) and yield in transgenic Arabidopsis and tobacco (Yu et al. 2019). Understanding the signaling peptide-mediated regulation pathway in growth and development might help improve yield production.
Abscission
The phytosulfokine (PSK) regulates fruit abscission on the activation by subtilisin like proteinase (SBTs) known as phytaspase 2, and provides insight into the induced expression of hydrolases for the degradation of cell walls in the abscission zones of the flowers and fruits of tomato plants (Reichardt et al. 2020). Inflorescence Deficient in Abscission (IDA)-like peptides and HAESA (HAE) and HAESA-LIKE2 (HSL2) receptor-like kinases are reported to be associated with abscission of tomato flowers (Lu et al. 2023) floral organs and cauline leaves in Arabidopsis thaliana (Patharkar and Walker 2018; Shi et al. 2019), corolla abscission in flowers of Nicotiana benthamiana (Ventimilla et al. 2021) and IDA-LIKE genes, RbIDL1 and RbIDL4 regulate petal abscission in Rosa bourboniana (Singh et al. 2023). Both PSK and IDL6 induce tomato pedicel abscission (Li et al. 2021).
Role of Peptide Signaling in Biotic Stress
Diverse pathogenic microorganisms threaten plants (e.g., bacteria, fungi, viruses, and oomycetes), endangering their existence and efficiency (Cramer et al. 2011). These pathogens decrease annual crop production and pose a serious threat to food security. Plants use diverse defense mechanisms to defend against enemies to survive or retain their efficiency (Roux et al. 2014; Ganie and Reddy 2021). Pathogen-associated molecular pattern-triggered immunity (PTI) and effector-triggered immunity (ETI) are two forms of plant immunity. In general, pathogen-associated molecular patterns (PAMPs) comprise microbial or pathogenic assemblies such as flagellins, lipopolysaccharides, and fungal cell wall components (chitins and glucans), recognized by unique plant receptors called pattern recognition receptors (PRRs), which auxiliary activate PTII (Zipfel and Felix 2005).
Furthermore, microbial pathogens secrete effector proteins, recognized by resistance (R) proteins that accelerate the activation of induced resistance responses (ETI) (Dangl and Jones 2001). The effector proteins are essential components of the fungal pathogen’s virulence alongside plants and are significant during the biotrophic process of infection (Sonah et al. 2016). The importance of pathogenesis-related (PR) proteins during plant–fungal pathogen interactions has been documented, with an increasing list of known pathogen effector proteins that interact precisely with PR proteins through infection (Breen et al. 2017). Within plant species, the complexity and effectiveness of the plant protection mechanism for combating pathogen attacks vary (Jones and Dangl 2006; Segonzac and Monaghan 2019). Several PR proteins are classified as antimicrobial peptides (AMPs). They typically have a wide range of antimicrobial activity as cysteine-rich molecules, including the families of PR6 proteins (proteinase inhibitors), PR12 proteins (plant defensins), PR13 proteins (plant thionins), and PR 14 proteins (lipid transfer proteins). AMPs are ubiquitous and form a significant part of the host defense against many microbial pathogens and pests in various living forms, from microbes to plants (Egorov et al. 2005).
Antimicrobial Peptides and Disease Resistance
Antimicrobial peptides (AMPs) are abundant in eukaryotic organisms as various types of PR peptides (Bulet et al. 2004). Usually, their mode of action involves disrupting the pathogen membrane in particular and non-specific electrostatic and hydrophobic connections with cell surface groups (Thevissen et al. 2003). AMPs are receiving more attention for enhancing disease resistance due to their all-rounder efficiency against several biotic stresses, such as bacterial, viral, fungal, and their function in abiotic stress tolerance. For example, the transcript levels of AMPs in tomato plants increase after bacterial and fungal infections, suggesting their role in disease resistance (Chan et al. 2005). PR6 peptides have demonstrated efficient antimicrobial activity against various fungal pathogens in in vitro studies (Terras et al. 1993). The most important antifungal peptides in plants are the PR12 or plant defensins. In vitro experiments have shown that plant defenses demonstrate antifungal activity against many fungal pathogens (Terras et al. 1995; Jha and Chattoo 2009). Furthermore, overexpression of plant defense peptides has had improved and long-lasting disease tolerance in model and crop plants (Anuradha et al. 2008; Ghag et al. 2012; Kaur et al. 2016). PAMP is commonly used to refer to molecules that induce natural immune responses. PAMPs are evolutionarily conserved pathogen-derived molecules that differentiate hosts from pathogens, as classically defined. They include lipopolysaccharides, bacterial flagellins, peptidoglycans, and yeast mannans. However, since these molecules are often synthesized by nonpathogens, the term ‘pathogen-associated’ is a contradiction, and a more precise term would be ‘microbe-associated molecular pattern.’ Therefore, it makes sense that hosts will have identified receptors for truly pathogen-specific molecules, but only in plants if their conclusive evidence of immune receptors that recognize virulence-related pathogen-encoded molecules such as type III effectors. The expression ‘microbe-associated molecule(s)’ is used here instead of ‘PAMP.’
Arabidopsis reacts to flagellin and a highly conserved flagellin protein fragment of 22 amino acids called Flg222 (Felix et al. 1999). Flg22 activates a signal transduction cascade, containing a MAP kinase cascade, transmembrane LRR receptor kinase (FLS2), so-called WRKY transcription factors, and downstream effector proteins (Asai et al. 2002; Gomez-Gomez and Boller 2000, 2002) (Fig. 4). Studies have shown that fungal pathogenicity in plants imposed a conserved MAPK signaling cascade homologous to the filamentation/pheromone response pathways in Saccharomyces cerevisiae (Turra et al. 2014; Xu and Hamer 1996). The processes regulating MAPK signaling through the fungus–plant interaction are mostly unidentified (Turra et al. 2014). However, Masachis et al. (2016) found that F. oxysporum increases extracellular pH during plant infection, activating the pathogenicity-related MAPK signaling cascade, thus, furthering invasive hyphal growth and virulence.
A general schematic of plant cell responses to biotic stress. Signaling pathways downstream of PRRs in plants. Plants have receptor family like kinases, such as the FLS2 flagellin receptor. Although the overall structure of the FLS2 signaling pathway appears similar to the animal PRR signaling pathway, no individual components are preserved, and the similarity most likely reflects the ubiquity of eukaryotic MAPK stress-response cassettes that respond to environmental signals. The graph has been modified from Rejeb et al. (2014)
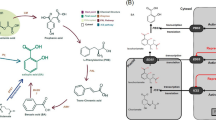
RALF peptides include four conserved Cys residues that can verify two disulfide bridges; they were first identified in tobacco (Nicotiana tabacum) for their ability to trigger rapid extracellular alkalinization in suspension-cultured cells (Pearce et al. 2001b). RALF peptides were subsequently found ubiquitous in the plant kingdom, with 37 members recognized in the Arabidopsis thaliana genome alone (Sharma et al. 2016; Campbell and Turner 2017). Plant RALFs are secreted as pre-propeptides, discharging the mature peptide by proteolytic processing (Srivastava et al. 2009). RALF peptides are involved in the signal sequence for extracellular extrusion and also contain special amino-acid motifs, such as the RRILA motif for S1P protease recognition (Srivastava et al. 2009) and YISY motif, important for signaling cascade activation (Pearce et al. 2001b) (Fig. 5). Furthermore, four conserved cysteines form two disulfide bonds that stabilize mature RALF proteins. Therefore, RALF peptides are classified into four major groups (Campbell and Turner 2017): Groups I, II, and III encompass typical RALF peptides, while Group IV contains the most deviating RALF peptides, missing RRILA and YISY motifs, and, in some cases, only three cysteines. RALF peptides engaged in plant growth and development regulation, including root cell elongation, pollen tube growth, and stress responses (Ge et al. 2019; Haruta et al. 2014). In recent years, studies have shown that plant–microbe interaction control involves RALF-mediated signaling. Stegmann et al. (2017) stated that RALF peptides in Arabidopsis act as negative regulators of the plant immune response to bacterial infection, as the binding to the FER receptor of processed RALF23 prevents the formation of the complex between kinases of the immune receptor FLAGELLIN-SENSING2 (FLS2) and EF-TU RECEPTOR (EFR) with their co-receptor BRASSINOSTEROID INSENSITIVE 1–ASSOCIATED KINASE 1 (BAK1), essential for initiating immune signaling. Furthermore, ANXUR1 (ANX1) RALF receptors, the nearest FER homolog, are associated with PRRs and nucleotide-binding domain leucine-rich repeat (NLR) type R proteins to inhibit PTI and ETI (Mang et al. 2017).
Schematic representation of a comparative account of the signaling mechanisms mediated by signaling peptides and phytohormones in plant cells. Peptide and hormone-mediated signaling (A-B) resemble their receptors, which are mostly leucine-rich repeats (LRRs) with receptor-like kinases (RLKs; co-receptors) that transduce secondary signals in the cytoplasm. The secondary messengers and other signaling components commonly include ROS, Ca2+ influx, and MAPK activity associated with both pathways (A–B)
PLANT DEFENSINS (PDFs), with about 5 kDa small peptides, appear to be the best-studied cysteine-rich peptides and AMPs (Tavormina et al. 2015). PDFs are distributed extensively and one of the largest and most complex protein families associated with pathogenesis in monocots and dicots (van Loon et al. 2006). PDFs are primarily antifungal, while some have reported antibacterial activity. The heterologous overexpression of different PDFs improved resistance to various fungi and bacteria in both the model plants and crops (Carvalho Ade and Gomes 2011; Gaspar et al. 2014). PDFs are internalized or connected to intracellular targets by the fungal cell or remain outside the cell and stimulate cell death by triggering a signaling cascade (Vriens et al. 2014). As such, they represent a typical example of host defense peptides acting more specifically than the classically suggested specific lipid bilayer disturbance for AMPs (Wilmes et al. 2011).
The Arabidopsis genome has ~ 285 PDF and defensin-like genes (Mondragon-Palominoet al. 2017). PDFs have been involved in various model plants and crops, displaying their participation in inherent immune responses to fungal pathogens such as Fusarium spp., Botrytis cinerea, and Verticillium dahlia, and bacterial pathogens, such as Pectobacterium carotovorum (Gaspar et al. 2014; Ahmed et al. 2012). PDFs have shown antimicrobial activity because they bind host intracellular targets that activate defense signals, such as inducing cell death, and interact with different fungal sphingolipids and phospholipids (Tavormina et al. 2015). Table 3 and Fig. 6 summarize peptides associated with biotic stress responses.
Role of Peptide Signaling in Abiotic Stress
Peptide hormones are involved in plant adaptation to abiotic stress (Fig. 7). They are encoded by small coding genes, secreted from cells, and translocated to other targeted plant cells, where they bind to a receptor protein (Matsubayashi and Sakagami 2006; Matsubayashi 2014; Tavormina et al. 2015) and induce physiological responses (Matsubayashi 2014; Tör et al. 2009). Several peptide hormones function as long-distance signal molecules in organ-to-organ communication (Okamoto et al. 2013, 2015). Characterization of hormone-like signaling peptides in abiotic stress responses (e.g., drought, heat, salinity etc.) is in its infancy.
Salinity Stress
CAP-DERIVED PEPTIDE 1 (CAPE1) belonging to CRP family is involved in the salinity stress response in Arabidopsis thaliana (Chen et al. 2014; Chien et al. 2015). AtCAPE1 (11-amino-acid) induced germination, produced yellowish cotyledons, and decreased growth under high salinity in A. thaliana, and PROAtCAPE1 is downregulated under salinity conditions (Chien et al. 2015). AtCAPE1 peptide and CEP negatively regulate plant salt tolerance response under high salinity by suppressing salt tolerance genes namely, DELTA1-PYRROLINE-5-CARBOXYLATE SYNTHASE 1 (P5CS1) and GALACTINOLSYNTHASE2 (GolS2)) (involved in production of osmolytes), ALDEHYDE DEHYDROGENASE 7B4 (ALDH7B4) (for detoxification), ABSCISIC ACID–RESPONSIVE ELEMENT BINDING PROTEIN 1 (AREB1) and ABA INSENSITIVE 5 (ABI5)) (regulates stomatal closure), and RESPONSIVE TO DESICCATION 20/CALEOSIN 3 (RD20/CLO3) (protects the plasma membrane) (Tavormina et al. 2015; Chien et al. 2015). CAPE1 (proatcape1) knockout mutants in Solanum lycopersicum tolerated high salt stress whereas PROAtCAPE1 overexpression or exogenous application of AtCAPE1 peptide restores the salinity response in it (Chien et al. 2015).
The CEP peptide family shows differential regulation under salt stress (Delay et al. 2013; Aggarwal et al. 2020). CEP knockdown improves salinity stress tolerance partially in cep3 mutant of Arabidopsis thaliana which had longer primary roots (Ohyama et al. 2008), while CEP3 overexpression decreased primary root length and increased shoot length and salinity tolerance (Delay et al. 2013). Systemin, CEP, the C-terminus of a cysteine-rich secretory protein antigen 5, and genes within the pathogenesis-related 1 protein (CAP) superfamily are associated with the salinity stress response (Orsini et al. 2010; Delay et al. 2013; Chien et al. 2015). When overexpressed in plants, systemin positively regulates salinity stress tolerance, whereas C-TERMINALLY ENCODED peptide (CEP) and CAP GENE FAMILY DERIVED Peptide (CAPE) negatively regulate salinity stress tolerance GRIM REAPER PEPTIDE (GRIp) induced oxidative stress and reactive oxygen species (ROS)-dependent cell death in Arabidopsis thaliana (Wrzaczek et al. 2015). Several defense-responsive peptides modulate salinity tolerance. Systemin peptide overexpression confers better salinity tolerance in Solanum lycopersicum (Pearce et al. 1991).
Overexpression of OSIP108 (oxidative stress-induced peptide) in Arabidopsis thaliana enhances oxidative stress tolerance (Spincemaille et al. 2014a, b). Plant Elicitor Peptides (PEPs) are involved in starvation stress and biotic stress. PEPs crosstalk with salicylic acid (SA), jasmonic acid (JA), and ethylene during abiotic stress (Huffaker et al. 2006; Flury et al. 2013; Liu et al. 2013; Tintor et al. 2013). AtPROPEP3 recognized by the PEP RECEPTOR 1 (PEPR1) receptor, induced salinity stress tolerance and biotic stress tolerance in plants in response to exogenous application with 13-synthetic AtPROPEP3/AT13 peptide fragments (KPTPSSGKGGKHN) in a culture-based salinity stress assay by regulating salinity-induced genes and activating Na+ influx (Nakaminami et al. 2018). Multiple small coding genes are involved in salinity stress-related signal cascades. AtPROPEP3 knock down makes Arabidopsis plants hypersensitive to salinity whereas AtPROPEP3 overexpression or exogenous application of AtPEP3 peptide induces tolerance to salinity (Nakaminami et al. 2018).
Salt stress leads to downregulation of Rapid Alkalinization Factor (RALF)1 in plant root system. On exogenous use of active RALF1 peptide which is perceived by FERONIA (FER) receptor, salinity stress toxicity increases due to Na + accumulation by inhibition of activities of ARABIDOPSIS H + -ATPASE 2 (AHA2) and Na + /K + transporters (Yu and Assmann 2018). The fer mutants are RALF1 insensitive and hypersensitive to salinity due to loss of cell integrity (Feng et al. 2018; Yu and Assmann 2018).
Salt conditions activate RALF22/23 peptide accumulation and which in turn on interaction with LEUCINE-RICH REPEAT EXTENSINS (LRX) regulates FER-mediated integrity of cell wall, ABA signaling, and Reactive Oxygen Species (ROS), and leads to salinity tolerance. Ca2 + signaling cascade is also maintained (Zhao et al. 2018a, b, 2020a; Feng et al. 2018).
Drought Stress
Drought stress causes serious reductions in crop productivity and plants employ various ways in response to this stress (Ganie and Ahammed 2021). SPs have been demonstrated over years to play important roles in minimizing the drought-induced damages in plants. The CLAVATA3/EMBRYO SURROUNDING REGION RELATED 25 (CLE25) peptides move from roots to leaves as long-distance mobile signal and controls stomatal closure. In leaves, CLE25 (12-amino-acid peptide) is recognized and binds to plasma membrane-localized BARELY ANY MERISTEM (BAM) 1 and BAM3 receptor-like protein kinases under dehydration stress. Induction of CLE25 gene expression modulates ABA accumulation in vascular tissues and NINE‐CIS‐EPOXY-CAROTENOID DIOXYGENASE 3 (NCED3) in Arabidopsis leaves (Christmann and Grill 2018; McLachlan et al. 2018; Takahashi et al. 2018b; Yoshida and Fernie 2018). The root-derived CLE25 peptide functions as a long-distance signal and transmits water-deficiency signals under dehydration through vascular tissues to induce stomatal closure due to more ABA production for decreasing water loss in leaves by transpiration and enhances dehydration stress resistance. The CLE25–BAM module functions as a signaling molecule for long-distance signaling under dehydration (Christmann and Grill 2018; McLachlan et al. 2018; Takahashi et al. 2018b; Yoshida and Fernie 2018). CLE9 peptide expression in guard cells of plant leaves has also an important role in ABA signaling-dependent drought response and salinity (Zhang et al. 2019). The overexpression of CLE9 in transgenic plants leads to stomatal closure and stronger resistance to water deficiency (Zhang et al. 2019).
CEP5 has antagonistic crosstalk with auxin signaling. CEP5-dependent signaling stabilizes AUX/IAAs transcriptional repressors to control the auxin response under drought and osmotic stress (Smith et al. 2020). Novel peptide-dependent control mechanisms contribute to the fine tuning of auxin signaling with a role in osmotic and drought stress tolerance. CEP5 phenotypes include auxin-mediated control of root architecture (Roberts et al. 2016). Exogenous CEP5 peptide application in seedlings and CEP5-overexpressed plants shows drought and osmotic stress tolerance in CEPR receptor dependent or independent mode (Smith et al. 2020).
Drought stress-induced phytosulfokine (PSK) regulates fruit abscission on the activation by subtilisin like proteinase (SBTs) known as phytaspase 2, and provides insight into the induced expression of hydrolases to degrade cell walls in the flower and fruit abscission zones of tomato plants (Reichardt et al. 2020). The overexpression proPSK1 or SBT3.8 (SUBTILISIN) transgenic plant positively regulates drought stress resistance (Stührwohldt et al. 2021).
Overexpression of the EPIDERMAL PATTERNING FACTOR (EPF) signaling pathway modified leaf stomata density and size, decreased transpiration, increased growth and biomass, and improved tolerance to drought and high CO2 in Arabidopsis thaliana (Doheny-Adams et al. 2012). Excess Fe induces CASPARIAN STRIP INTEGRITY FACTOR 1 (CIF1) and CIF2, expressed in the root stele, binding to GASSHO1 (GSO1)/SCHENGEN3 receptor kinases, which control water and Fe permeability in the vascular stele of roots (Nakayama et al. 2017).
Nitrogen (N), Phosphate (Pi), Iron (Fe), and Sulfur (S) Stress
C-TERMINALLY ENCODED PEPTIDE (CEP) family acts as root-derived nitrogen (N)-demand signal in N-deficient soil conditions and ascends to shoots. The perception of CEP signals by leaf expressed XYLEM INTERMIXED WITH PHLOEM 1 (XIP1)/CEP RECEPTOR (CEPR1 and CEPR 2) produces putative shoot-derived phloem-specific polypeptides CEPD proteins. Putative shoot-derived phloem-specific polypeptides upregulate nitrate transporter genes in the roots (nitrogen sensing). If part of the plant root system is N-starved while the rest grows in N-rich soil, then the N-starved roots under nitrate deficiency use CEP hormones produced by root-derived ascending N-demand signals to signal distant CEPR receptors in shoots development (Mohd-Radzman et al. 2015; Ohyama et al. 2008; Tabata et al. 2014; Delay et al. 2013; Ohkubo et al. 2017). CEP1 functions in root-to-shoot signaling and regulates root development in plants under nitrate starvation (Tabata et al. 2014). CEPR1 and 2 perceive CEP1 to increase nitrate uptake in Arabidopsis (Ohkubo et al. 2017). In turn, CEP1-CEPR signaling induces CEP DOWNSTREAM-LIKE 1 (CEPDL1) and LIKE 2 (CEPDL2) proteins in leaves. CEPDL1 and CEPDL2 act as a leaf-derived phloem-mobile descending signals to N-rich roots, and induce nitrate transporter NITRATE TRANSPORTER 2.1 (NRT2.1) gene expression in the roots. The NRT2.1 in Arabidopsis thaliana mediates root N uptake and transport. CEP1 plays a role in all three N-stress tolerance processes. CEP1 overexpression in legumes inhibits lateral root emergence and enhances nodule development (Mohd-Radzman et al. 2015; Ohyama et al. 2008; Tabata et al. 2014; Delay et al. 2013; Ohkubo et al. 2017). When roots cannot uptake sufficient N, CEPDL2 and NRT1.5 and NRT2.1 are upregulated to absorb and transport N (Ota et al. 2020).
In Arabidopsis thaliana and Medicago spp., signaling by CEP-CEPR module regulates root system architecture, lateral root gravitropic set-point, shoot auxin content, and auxin transport to roots (Chapman et al. 2020). CLE1/3/4/7 expression increases in plants roots during N-starvation. There is increase in transcription levels of CLE3 which is recognized by CLAVATA (CLV1) receptor located in phloem. CLE-CLV1 signaling optimizes nitrate-dependent lateral root development, elongation, and emergence during N availability. CLV1-mediated N-demanding signal represses CLE-CLV1 cascade during low nitrate condition forming a feedback loop (Araya et al. 2014a, b; Chapman et al. 2020). The clv1 receptor mutant produces lateral roots under severe N deficiency due to the overaccumulation of CLE3 and CLE2 peptides. CEP peptide overexpression induced by low N reduces lateral root number in Medicago truncatula (Chapman et al. 2020). In Arabidopsis, ROOT MERISTEM GROWTH FACTOR 1 (RGF1, a 13-amino-acid peptide) is perceived by RGF1 INSENSITIVE 1–5 (RGI 1–5) receptors, and this cascade controls root development under phosphate (Pi) deficiency (Matsuzaki et al. 2010; Cederholm and Benfey 2015; Ou et al. 2016). CLE14 controls differentiation of the Root Apical Meristem (RAM) in P signaling (Gutiérrez-Alanís et al. 2017). In P. starvation environments, CLE14-CLV2/PEPR2 signaling attenuate POLTERGEIST (POLL) and POLTERGEIST-LIKE 1 (PLL1) which leads to downstream exhaustion of root meristems (Gutiérrez-Alanís et al. 2017).
In S starvation scenarios, expression levels of CLE2/3 peptide are controlled and repressed. CLE2/3 is perceived by CLV1 receptors (CLE-CLV1 module) to reduce lateral root density (Czyzewicz et al. 2015; Dong et al. 2019) but diminishes in clv1 mutants.
In Arabidopsis, CASPARIAN STRIP INTEGRITY FACTORS (CIF1 and CIF2 are 21 amino-acid peptides) function as a signal increase iron (Fe) tolerance by controlling Casparian strip formation and lignification in roots under excess iron (Doblas et al. 2017; Nakayama et al. 2017). GASSHO1 (GSO1)/SCHENGEN3 (SGN3) and GSO2 act as the receptor for synthetic CIF1. CIF1 and 2 restore iron homeostasis in cif1cif2 mutants by lignification of Casparian strip after treatment with these peptides (Doblas et al. 2017; Nakayama et al. 2017).
Heat Stress
The expression of CLE45 in floral stigma is activated by heat stress and mediates pollen tube growth wherein CLE45 is recognized by two receptors namely STERILITY-REGULATING KINASE MEMBER1 (SKM1) and SKM2 under heat conditions (Endo et al. 2013). Exogenous application of synthetic CLE45 peptide increases growth of pollen grain tubes under heat scenarios whereas skm mutants are insensitive to this treatment (Endo et al. 2013).
Do SPs and Phytohormones exhibit similar Signaling Mechanisms Accompanying Defense and Stress Tolerance in Plants?
The number of signaling peptides exceeds the number of conventional phytohormones in plants (Olsson et al. 2018), indicative of their myriad functions. In addition to phytohormones, SPs take part in short and long-distance signaling associated with developmental changes and stress sensing in plants (Olsson et al. 2018). Furthermore, various tissue-specific proteases are involved in precursor (prepeptides) processing, regulating the functional redundancy of SPs in plant tissues precisely with different ontogenic phases (Chen et al. 2019). During evolution, increasing genome complexity and diverse functions are accompanied by gene duplication for SP gene families in various plant species (Olsson et al. 2018). SPs also exhibit various forms produced by gene duplication, but some exhibit functional redundancy (Olsson et al. 2018). For instance, the CLE gene cluster produces different peptides with similar amino-acid sequences. CLE42 and CLE41/CLE44 inhibit tracheary element differentiation but do not inhibit root growth (Breiden and Simon 2016).
Despite the current knowledge on the role of SPs and their mechanisms of action in different plant biological processes, their differences with phytohormones in accomplishing these processes need deciphering. Emerging evidence suggests similarities in the mechanisms of perception, communication, and expression of SPs and phytohormones in plants subjected to normal or challenging environments (Chen et al. 2019). Nevertheless, future investigations may reveal some differences. SPs and other phytohormones have co-evolved in various plant groups, where increased structural and functional complexity has led to diverse functions and signaling routes (Olsson et al. 2018). Like phytohormones, SPs can move between the apoplast and symplast of cells (Stahl and Simon 2013).
Interesting connections exist between the secondary signaling cascades mediated by SPs and phytohormones. Various peptides elicit JA, ET, and ABA signaling during biotic or abiotic stress, involving several secondary messengers (Chen et al. 2019), which mediate the stress response via shared signaling components of ROS, Ca2+, and MAPKs, and modulate the gene expression for various phytohormones (Kandoth et al. 2007; Chen et al. 2019). IAA, JA, SA, ET, and ABA are the major phytohormones involved in crosstalk with various stress-induced peptides in plants (Chen et al. 2019). One or more PTM events are likely to regulate peptide activity; thus, peptides have a unique regulation system compared with other phytohormones. Unlike SPs, hormone signaling is regulated primarily by PTMs in their receptors or transcription factors (Gou and Li 2020; Semeradova et al. 2020). Similar to stress-induced hormone signaling, SPs bind specific cellular receptors (LRRs) to transduce the downstream cascade of secondary signals via co-receptors (RLKs), transcription factors, and various secondary messengers (Olsson et al. 2018). Thus, no remarkable differences exist for the downstream-signaling cascades mediated by peptides and hormones. Certain stress-induced SPs are involved in phytohormone crosstalk, either upregulating or downregulating their expression patterns (Chen et al. 2019). Systemins bind with SYSTEMOIN RECEPTOR 1 (SYR 1), triggering ROS accumulation, ethylene biosynthesis, and protease inhibitor expression. Furthermore, systemins trigger Ca2+ influx and MAPK phosphorylation to activate JA biosynthesis. PEP1 peptides trigger downstream defense responses to wound stimuli, involving JA, ET, and SA pathways accompanied by a burst of H2O2 (Bartels et al. 2013). Similarly, phytosulfokines (PSKs) induce Ca2+ signaling and mediate auxin biosynthesis to initiate necrotrophic responses (Zhang et al. 2018). Moreover, SPs also exert precise effects on root growth and architecture (Oh et al. 2018). CLE25 and CAPE1 exert associative effects on drought and salinity tolerance by regulating ABA homeostasis (Chen et al. 2019). Thus, it is evident that SPs and phytohormones operate at the crossroads of defense and stress tolerance signaling pathways in plants. While SP- and phytohormone-mediated signaling share some components, further investigations are needed to decipher the interaction of SPs with various other molecular components involved in hormone signaling pathways.
Noncoding RNA-encoded SPs
Previously, regulatory SPs were considered to be derived from precursor proteins as their processed products, ORFs via direct translation, and sometimes untranslated regions of mRNAs (Ren et al. 2021). However, many studies have documented that SPs are also encoded by noncoding RNAs (ncRNAs), including primary microRNAs (miRNAs), long ncRNAs, and circular RNAs (Legnini et al. 2017). Among the various ncRNA-derived SPs, only the role of pri-miRNA-derived peptides in regulating plant biological processes has been documented (Sharma et al. 2020). The pri-miRNAs possess a short ORF in their 5’ upstream region, which encodes regulatory peptides known as miPEPs (Ren et al. 2021), found in plant species such as grapes, Arabidopsis, soybean, and Medicago (reviewed by Ren et al. 2021). Endogenous miPEPs have been detected using the Western blotting technique and found to accumulate in plant tissues (Sharma et al. 2020). miPEPs may act as internal signals in a feedback mechanism to regulate miRNA accumulation in plants (Ormancey et al. 2020). These miPEPs are believed to control/activate pri-miRNA transcription, inducing mature miRNA upregulation and miPEP accumulation. The role of miPEPs in regulating the transcription of genes encoding pri-miRNAs has been reported in some plant species. For example, cordycepin (RNA synthesis inhibitor) supplementation inhibits the miPEP165a-mediated accumulation of pri-miR165a in Arabidopsis, indicating a positive effect of miPEPs on miRNA expression (Lauressergues et al. 2015). Similarly, GUS gene expression is activated by the miR858a promotor in two reporter lines of Arabidopsis, which is fused with only the start code or entire ORF encoded miPEP858a (Sharma et al. 2020). Sharma et al. (2020) reported that synthetic miPEP858a supplementation enhanced GUS activity, suggesting activation of the promotor and, thus, enhanced transcription of miR858a by miPEP858a. miPEPs may directly or indirectly function as trans-acting factors, such as transcription factors (TFs), to enhance miRNA gene transcription (Sharma et al. 2020). The actual regulatory mechanisms of miPEP activity remain unclear.
An investigation of pri-miRNAs in plants such as Arabidopsis (Sharma et al. 2020), grapevine (Chen et al. 2020a, b), Dimocarpus longan (Zhang et al. 2020) has revealed that several putative pri-miRNAs possess functional sORFs that code for regulatory micropeptides. The role of some miPEPs in plant biological processes has been elucidated. For example, exogenous application and overexpression of miPEP165a (in Arabidopsis) and miPEP171b (in Medicago truncatula) significantly induce the expression of their corresponding miRNAs, impairing main root growth and lateral root formation (Lauressergues et al. 2015). Similarly, application of miPEP171d1 in grapes upregulates MIR171d expression, and enhances adventitious root development (Chen et al. 2020a). Supplementation of synthetic miPEP172c increases nodule number in soybean roots (Couzigou et al. 2016). Sharma et al. (2020) demonstrated that miPEP858a regulates the phenylpropanoid pathway and auxin signaling genes, thus, controlling flavonoid biosynthesis and plant development.
Despite this progress in determining the biological function of miPEPs, many important questions remain unanswered. For example, do miPEPs specifically encode pri-miRNAs, and if so, how does it occur? There are few reports on the specific activation of miRNAs (Lauressergues et al. 2015). Application of synthetic miPEP171d1 to tissue-cultured grape plantlets reduces the expression of some miRNAs (MIR171i, MIR171a, and MIR160c), but it is not clear whether the incubation period causes this reduction (Chen et al. 2020a). Hence, studies are needed to elucidate whether miPEPs negatively affect their corresponding miRNAs or other miRNAs.
Does Peptide-Signaling Involve Multiple Crosstalk Events with Plant Growth Regulators? Future Prospects
Secreted peptides and membrane-bound receptors orchestrate cellular communication. Peptides bind to receptors and change their conformation, initiating downstream communication (for short-range and long-distance signaling) and modulating cellular activities and processes. Connections exist between the secondary signaling cascades mediated by SPs and phytohormones.
Higher plants contain several SP genes. Most SP research has been conducted on Arabidopsis, with > 1000 genes encoding SPs reported to date. However, few SPs have been structurally and functionally characterized. Peptide biosynthesis involves two major events—proteolytic cleavage and PTM—crucial for producing mature and active peptides. Proteolytic cleavage (single or multiple) is essential for releasing mature peptides as in RGFs, CLE, CEPs, and PSK families. However, the identified number of proteases in plants is less; further research is needed to identify more proteases. PTMs, including tyrosine sulfation, proline hydroxylation, and hydroxyproline glycosylation, are crucial for cell peptide biogenesis, activation, and subsequent downstream signaling. Several studies have demonstrated that PTMs contribute to plant adaptation to various biotic and abiotic stresses. In addition, PTMs of SPs have been associated with phenotypic plasticity related to roots, leaves, fruit ripening, flowering time, and early senescence.
Enzymes regulate the biosynthesis of various conventional phytohormones. However, questions remain unanswered about tissue-specific production and secretion mechanisms of SPs in plant tissues. In general, all plant cells can produce and deliver peptides, as with other phytohormones. SPs also exhibit dose-dependent reactions in cells. However, little information is available on the conjugation mechanisms or stability of SPs in plants.
Several questions remain unanswered on the mechanism of peptides which can trigger the functioning of secondary messengers and gene actions pertaining to hormonal actions. Thus, it is imperative to decipher the spatio-temporal regulation of the production of several peptides being originated from the precursor proteins under normal and challenging environment. The extent or intensity of response mediated by peptides and their regulation by a positive feedback loop should undergo detailed investigations. Future investigations are required to unravel the molecular signaling routes of various peptides under stress conditions in plants. It would be interesting to perform detailed investigations on the fate of mature peptides associated with cell secretion, transport, and long-distance signaling under abiotic stress. In this context, works for some insights into peptide-receptor interactions regulating specific developmental aspects will be worth to be undertaken in future. Challenges exist in deciphering the role of peptides involved in canonical signaling pathways to that of phytohormones. Furthermore, it would be interesting to investigate the associative role of peptides and novel plant growth regulators, including gasotransmitters (NO, H2S, and CO), melatonin, and strigolactone. Thus, a holistic approach needs to be undertaken to extend our current understand of mechanisms of peptide formation, receptor recognition, long-distance signaling, and crosstalk with other plant-signaling molecules. Integrating various ‘omics’ technologies with SPs research would enhance existing knowledge, which could be translated into applied research for plant breeding and genetics.
References
Abel S, Theologis A (1996) Early genes and auxin action. Plant Physiol 111:9–17. https://doi.org/10.1104/pp.111.1.9
Adie BAT, Perez-Perez J, Perez-Perez MM, Godoy M, Sanchez-Serrano JJ, Schmelz EA, Solano R (2007) ABA is an essential signal for plant resistance to pathogens affecting JA biosynthesis and the activation of defenses in Arabidopsis. Plant Cell 19:1665–1681
Aggarwal S, Kumar A, Jain M, Sudan J, Singh K, Kumari S, Mustafz A (2020) C-terminally encoded peptides (CEPs) are potential mediators of abiotic stress response in plants. Physiol Mol Biol Plants 26:2019–2033
Ahmad B, Azeem F, Ali MA, Nawaz MA, Nadeem H, Abbas A et al (2020) Genome-wide identification and expression analysis of two component system genes in cicer arietinum. Genomics 112:1371–1383. https://doi.org/10.1016/j.ygeno.2019.08.006
Ahmed NU, Park JI, Jung HJ, Seo MS, Kumar TS, Lee IH, Nou IS (2012) Identification and characterization of stress resistance related genes of Brassica rapa. Biotechnol Lett 34:979–987
Akker SEV, Lilley C, Yusup HB, Jones JT, Urwin PE (2016) Functional C-TERMINALLY ENCODED PEPTIDE (CEP) plant hormone domains evolved de novo in the plant parasite Rotylenchulus reniformis. Mol Plant Pathol 17(8):1265–1275
Akpinar B, Bihter A, Lucas SJ, Hikmet B (2012) Plant abiotic stress signaling. Plant Signal Behav 7:1450–1455
Albert M (2013) Peptides as triggers of plant defence. J Exp Bot 64:5269–5279
Amano Y, Tsubouchi H, Shinohara H, Ogawa M, Matsubayashi Y (2007) Tyrosine-sulfated glycopeptide involved in cellular proliferation and expansion in Arabidopsis. Proc Natl Acad Sci USA 104:18333–18338
Anuradha TS, Divya K, Jami SK, Kirti PB (2008) Transgenic tobacco and peanut plants expressing a mustard defensin show resistance to fungal pathogens. Plant Cell Rep 27:1777–1786
Araya T, Miyamoto M, Wibowo J, Suzuki A, Kojima S et al (2014a) CLE-CLAVATA1 peptide- receptor signaling module regulates the expansion of plant root systems in a nitrogen dependent manner. Proc Natl Acad Sci U S A 111:2029–2034
Araya T, Miyamoto M, Wibowo J, Suzuki A, Kojima S, Tsuchiya YN, Sawa S, Fukuda H, von Wirén N, Takahashi H (2014b) CLE-CLAVATA1 peptide-receptor signaling module regulates the expansion of plant root systems in a nitrogen-dependent manner. Proc Natl Acad Sci USA 111:2029–2034
Asai T, Tena G, Plotnikova J, Willmann MR, Chiu WL, Gomez-Gomez L, Boller T, Ausubel FM, Sheen J (2002) MAP kinase signaling cascade in Arabidopsis innate immunity. Nature 415(6875):977–983
Atkinson NJ, Lilley CJ, Urwin PE (2013) Identification of genes involved in the response of Arabidopsis to simultaneous biotic and abiotic stresses. Plant Physiol 162:2028–2041
Bartels S, Lori M, Mbengue M, van Verk M, Klauser D, Hander T, Boni R, Robatzek S, Boller T (2013) The family of Peps and their precursors in Arabidopsis: Differential expression and localization but similar induction of pattern-triggered immune responses. J Exp Bot 64:5309–5321
Bhattacharya R, Koramutla MK, Negi M, Pearce G, Ryan CA (2013) Hydroxyproline-rich glycopeptide signals in potato elicit signaling associated with defense against insects and pathogens. Plant Sci 207:88–97
Bircheneder S, Dresselhaus T (2016) Why cellular communication during plant reproduction is particularly mediated by CRP signaling. J Exp Bot 67:4849–4861
Blanvillain R, Young B, Cai YM, Hecht V, Varoquaux F, Delorme V, Lancelin JM, Delseny M, Gallois P (2011) The Arabidopsis peptide kiss of death is an inducer of programmed cell death. EMBO J 30:1173–1183
Boschiero C, Dai XB, Lundquist PK, Roy S, de Bang TC, Zhang S, Zhuang ZH, Torres-Jerez I, Udvardi MK, Scheible WR, Zhao PX (2020) The Medicago truncatula small secreted peptide database. Plant Physiol 183(1):399–413
Brand U, Fletcher JC, Hobe M, Meyerowitz EM, Simon R (2000) Dependence of stem cell fate in Arabidopsis on a feedback loop regulated by CLV3 activity. Science 289:617–619
Breen S, Williams SJ, Outram M, Kobe B, Solomon PS (2017) Emerging insights into the functions of pathogenesis-related protein 1. Trends Plant Sci 22:871–879
Breiden M, Simon R (2016) Q&A: How does peptide signaling direct plant development? BMC Biol 14:58. https://doi.org/10.1186/s12915-016-0280-3
Brito MS, DePaoli HC, Cossalter V, Avanci NC, Ferreira PB, Azevedo MS, Strini EJ, Quiapim AC, Goldman GH, Peres LEP et al (2018) A novel cysteine-rich peptide regulates cell expansion in the tobacco pistil and influences its final size. Plant Sci 277:55–67
Bryan AC, Obaidi A, Wierzba M, Tax FE (2012) XYLEM INTERMIXED WITH PHLOEM1, a leucine-rich repeat receptor-like kinase required for stem growth and vascular development in Arabidopsis thaliana. Planta 235:111–122. https://doi.org/10.1007/s00425-011-1489-6
Bulet P, Stocklin R, Menin L (2004) Anti-microbial peptides: from invertebrates to vertebrates. Immunol Rev 198:169–184
Busch W, Benfey PN (2010) Information processing without brains—The power of intercellular regulators in plants. Development 137:1215–1226
Butenko MA, Patterson SE, Grini PE, Stenvik GE, Amundsen SS et al (2003) Inflorescence deficient in abscission controls floral organ abscission in Arabidopsis and identifies a novel family of putative ligands in plants. Plant Cell 15:2296–2307
Caetano-Anollés G, Gresshoff PM (1990) Early induction of feedback regulatory responses governing nodulation in soybean. Plant Sci 71:69–81
Cammarata J, Roeder AHK, Scanlon MJ (2019) Cytokinin and CLE signaling are highly intertwined developmental regulators across tissues and species. Curr Opin Plant Biol 51:96–104
Campbell L, Turner SR (2017) A comprehensive analysis of RALF proteins in green plants suggests there are two distinct functional groups. Front Plant Sci 8:1–14
Carvalho Ade O, Gomes VM (2011) Plant defensins and defensin-like peptides - biological activities and biotechnological applications. Curr Pharm Des 17:4270–4293
Casson SA, Chilley PM, Topping JF, Evans IM, Souter MA et al (2002) The POLARIS gene of Arabidopsis encodes a predicted peptide required for correct root growth and leaf vascular patterning. Plant Cell 14:1705–1721
Cederholm HM, Benfey PN (2015) Distinct sensitivities to phosphate deprivation suggest that RGF peptides play disparate roles in Arabidopsis thaliana root development. New Phytol 207:683–691
Chakraborty S, Nguyen B, Wasti SD, Xu G (2019) Plant Leucine-Rich Repeat Receptor Kinase (LRR-RK): Structure, ligand perception, and activation mechanism. Molecules 24:3081. https://doi.org/10.3390/MOLECULES24173081
Chan YL, Prasad V, Chen KH, Liu PC, Chan MT, Cheng CP (2005) Transgenic tomato plants expressing an Arabidopsis thionin (Thi2. 1) driven by fruit-inactive promoter battle against phytopathogenic attack. Planta 221:386–393
Chapman K, Ivanovici A, Taleski M, Sturrock CJ, Ng JLP, Mohd-Radzman NA, Frugier F, Bennett MJ, Mathesius U, Djordjevic MA (2020) CEP receptor signaling controls root system architecture in Arabidopsis and Medicago. New Phytol 226:1809–1821
Chen XY, Kim JY (2006) Transport of macromolecules through plasmodesmata and the phloem. Physiol Plant 126:560–571
Chen YC, Siems WF, Pearce G, Ryan CA (2008) Six peptide wound signals derived from a single precursor protein in Ipomoea batatas leaves activate the expression of the defense gene sporamin. J Biol Chem 283:11469–11476
Chen YL, Lee CY, Cheng KT, Chang WH, Huang RN, Nam HG et al (2014) Quantitative peptidomics study reveals that a wound induced peptide from PR-1 regulates immune signaling in tomato. Plant Cell 26:4135–4148. https://doi.org/10.1105/tpc.114.131185
Chen Y, Fan K, Hung S, Chen Y (2019) The role of peptides cleaved from protein precursors in eliciting plant stress reactions. New Phytol. https://doi.org/10.1111/nph.16241
Chen QJ, Deng BH, Gao J, Zhao ZY, Chen ZL, Song SR, Wang L, Zhao LP, Xu WP, Zhang CX, Ma C (2020a) A miRNA-encoded small peptide, vvi-miPEP171d1, regulates adventitious root formation. Plant Physiol 183(2):656–670
Chen YL, Fan KT, Hung SC, Chen YR (2020b) The role of peptides cleaved from protein precursors in eliciting plant stress reactions. New Phytol 225:2267–2282
Chien PS, Nam HG, Chen YR (2015) A salt-regulated peptide derived from the CAP superfamily protein negatively regulates salt-stress tolerance in Arabidopsis. J Exp Bot 66(17):5301–5313. https://doi.org/10.1093/jxb/erv263
Chilley PM, Casson SA, Tarkowski P, Hawkins N, Wang KL et al (2006) The POLARIS peptide of Arabidopsis regulates auxin transport and root growth via effects on ethylene signaling. Plant Cell 18:3058–3072
Cho SJ, Juillerat MA, Lee CH (2008) Identification of LDL-receptor transcription stimulating peptides from soybean hydrolysate in human hepatocytes. J Agric Food Chem 56(12):4372–4376
Chow B, McCourt P (2006) Plant hormone receptors: perception is everything. Genes Dev 20(15):1998–2008. https://doi.org/10.1101/gad.1432806
Christmann A, Grill E (2018) Peptide signal alerts plants to drought. Nature 556(7700):178–179. https://doi.org/10.1038/d41586-018-03872-4
Chu ZX, Ma Q, Lin YX, Tang XL, Zhou YQ, Zhu SW, Fan J, Cheng BJ (2011) Genome-wide identification, classification, and analysis of two-component signal system genes in maize. Genet Mol Res 10(4):3316–3330. https://doi.org/10.4238/2011
Colling J, Tohge T, De Clercq R, Brunoud G, Vernoux T, Fernie AR, Makunga NP, Goossens A, Pauwels L (2015) Overexpression of the Arabidopsis thaliana signaling peptide TAXIMIN1 affects lateral organ development. J Exp Bot 66(17):5337–5349. https://doi.org/10.1093/jxb/erv291
Corcilius L, Hastwell AH, Zhang M, Williams J, Mackay JP, Gresshoff PM, Ferguson BJ, Payne RJ (2017) Arabinosylation modulates the growth regulating activity of the peptide hormone CLE40a from Soybean. Cell Chem Biol 24:1347–1355
Costa LM, Marshall E, Tesfaye M, Silverstein KA, Mori M (2014) Central cell-derived peptides regulate early embryo patterning in flowering plants. Science 344:168–172
Couzigou JM, André O, Guillotin B, Alexandre M, Combier JP (2016) Use of microRNA-encoded peptide miPEP172c to stimulate nodulation in soybean. New Phytol 211(2):379–381
Craik DJ, Daly NL, Bond T, Waine C (1999) Plant cyclotides: a unique family of cyclic and knotted proteins that defines the cyclic cystine knot structural motif. J Mol Biol 294:1327–1336
Cramer GR., Urano K, Delrot S, Pezzotti M, Shinozaki K (2011) Effects of abiotic stress onplants: a systems biology perspective. BMC Plant Biol 11–163
Creff A, Brocard L, Joubes J, Taconnat L, Doll NM, Marsollier AC, Pascal S, Galletti R, Boeuf S, Moussu S, Widiez T, Domergue F, Ingram G (2019) A stress-response-related inter-compartmental signaling pathway regulates embryonic cuticle integrity in Arabidopsis. PLoS Genet 15(4):e1007847
Cui Y, Li M, Yin X, Song S, Xu G, Wang M, Li C, Peng C, Xia X (2018) OsDSSR1, a novel small peptide, enhances drought tolerance in transgenic rice. Plant Sci 270:85–96. https://doi.org/10.1016/j.plantsci.2018.02.015
Czyzewicz N, Yue K, Beeckman T, De Smet I (2013) Message in a bottle: small signaling peptide outputs during growth and development. J Exp Bot 64:5281–5296. https://doi.org/10.1093/jxb/ert283
Czyzewicz N, Shi CL, Vu LD, Van De Cotte B, Hodgman C, Butenko MA, De Smet I (2015) Modulation of Arabidopsis and monocot root architecture by CLAVATA3/EMBRYO SURROUNDING REGION 26 peptide. J Exp Bot 66:5229–5243
Dangl JL, Jones JDG (2001) Plant pathogens and integrated defence responses to infection. Nature 411:826–833
De Coninck B, De Smet I (2016) Plant peptides—taking them to the next level. J Exp Bot 67(16):4791–4795
De Vleesschauwer D, Seifi HS, Filipe O, Haeck A, Huu SN, Demeestere K, Höfte M (2016) The DELLA protein SLR1 integrates and amplifies salicylic acid- and jasmonic acid-dependent innate immunity in rice. Plant Physiol 170(3):1831–1847. https://doi.org/10.1104/pp.15.01515
Delay C, Imin N, Djordjevic MA (2013) CEP genes regulate root and shoot development in response to environmental cues and are specific to seed plants. J Exp Bot 64(17):5383–5394
DeYoung BJ, Clark SE (2008) BAM receptors regulate stem cell specification and organ development through complex interactions with CLAVATA signaling. Genetics 180:895–904
Doblas VG, Smakowska-Luzan E, Fujita S, Alassimone J, Barberon M, Madalinski M, Belkhadir Y, Geldner N (2017) Root diffusion barrier control by a vasculature-derived peptide binding to the SGN3 receptor. Science 355(6322):280–284
Doheny-Adams T, Hunt L, Franks PJ, Beerling DJ, Gray JE (2012) Genetic manipulation of stomatal density influences stomatal size, plant growth and tolerance to restricted water supply across a growth carbon dioxide gradient. Philos Trans Royal Soc Lond b: Biol Sci 367(1588):547–555
Doll NM, Royek S, Fujita S, Okuda S, Chamot S, Stintzi A, Widiez T, Hothorn M, Schaller A, Geldner N, Ingram G (2020) A two-way molecular dialogue between embryo and endosperm is required for seed development. Science 367:431–435
Dombrowski JE (2003) Salt stress activation of wound-related genes in tomato plants. Plant Physiol 132(4):2098–2107
Dong W, Wang Y, Takahashi H (2019) CLE-CLAVATA1 signaling pathway modulates lateral root development under sulfur defciency. Plants (basel) 8:10
Egorov TA, Odintsova TI, Pukhalsky VA, Grishin EV (2005) Diversity of wheat anti-microbial peptides. Peptides 26:2064–2073
Endo S, Shinohara H, Matsubayashi Y, Fukuda H (2013) A novel pollen pistil interaction conferring high-temperature tolerance during reproduction via CLE45 signaling. Curr Biol 23:16
Escobar-Restrepo JM, Huck N, Kessler S, Gagliardini V, Gheyselinck J, Yang WC, Grossniklaus U (2007) The FERONIA receptor-like kinase mediates male-female interactions during pollen tube reception. Science 317:656–660
Etchells JP, Smit ME, Gaudinier A, Williams CJ, Brady SM (2016) A brief history of the TDIF-PXY signalling module: balancing meristem identity and differentiation during vascular development. New Phytol 209:474–484. https://doi.org/10.1111/nph.13642
Farkas A, Maróti G, Durgő H, Györgypál Z, Lima RM, Medzihradszky KF, Kereszt A, Mergaert P, Kondorosi É (2014) Medicago truncatula symbiotic peptide NCR247 contributes to bacteroid differentiation through multiple mechanisms. Proc Natl Acad Sci U S A 111:5183–5188
Farmer EE, Ryan CA (1992) Octadecanoid precursors of jasmonic acid activate the synthesis of wound-inducible proteinase inhibitors. Plant Cell 4:129–134
Farmer EE, Johnson RR, Ryan CA (1992) Regulation of expression of proteinase inhibitor genes by methyl jasmonates and jasmonic acid. Plant Physiol 98:995–1002
Fedoreyeva LI (2023) Molecular mechanisms of regulation of root development by plant peptides. Plants 12(6):1320. https://doi.org/10.3390/plants12061320
Felix G, Duran JD, Volko S, Boller T (1999) Plants have a sensitive perception system for the most conserved domain of bacterial flagellin. Plant J 18:265–276
Feng W, Kita D, Peaucelle A, Cartwright HN, Doan V, Duan Q, Liu MC, Maman J, Steinhorst L, Schmitz-Thom I, Yvon R, Kudla J, Wu HM, Cheung AY, Dinneny JR (2018) The FERONIA receptor kinase maintains cell-wall integrity during salt stress through Ca2+ signaling. Curr Biol 28:666–675
Fernandez A, Drozdzecki A, Hoogewijs K et al (2013) Transcriptional and functional classification of the GOLVEN/ROOT GROWTH FACTOR/CLE-like signaling peptides reveals their role in lateral root and hair formation. Plant Physiol 161(2):954–970. https://doi.org/10.1104/pp.112.206029
Fernandez A, Drozdzecki A, Hoogewijs K, Vassileva V, Madder A, Beeckman T, Hilson P (2015) The GLV6/RGF8/CLEL2 peptide regulates early pericycle divisions during lateral root initiation. J Exp Bot 66(17):5245–5256
Fletcher JC (2020) Recent advances in Arabidopsis CLE peptide signaling. Trends Plant Sci 25(10):1005–1016. https://doi.org/10.1016/j.tplants.2020.04.014
Flury P, Klauser D, Schulze B, Boller T, Bartels S (2013) The anticipation of danger: microbe associated molecular pattern perception enhances AtPep-triggered oxidative burst. Plant Physiol 161(4):2023–2035
Fukuda H, Hardtke CS (2020) Peptide signaling pathways in vascular differentiation. Plant Physiol 182:1636–1644. https://doi.org/10.1104/pp.19.01259
Fukuda H, Hirakawa Y, Sawa S (2007) Peptide signaling in vascular development. Curr Opin Plant Biol 10:477–482. https://doi.org/10.1016/j.pbi.2007.08.013
Furukawa T, Sakaguchi N, Shimada H (2006) Two OsGASR genes, rice GAST homologue genes that are abundant in proliferating tissues, show different expression patterns in developing panicles. Genes Genet Syst 81:171–180
Furumizu C, Sawa S (2021) The RGF/GLV/CLEL family of short peptides evolved through lineage-specific losses and diversification and yet conserves its signaling role between vascular plants and bryophytes. Front Plant Sci 12:1491
Furumizu C, Krabberød AK, Hammerstad M, Alling RM, Wildhagen M, Sawa S et al (2021) The sequenced genomes of non-flowering land plants reveal the innovative evolutionary history of peptide signaling. Plant Cell. https://doi.org/10.1093/plcell/koab173
Gahlaut V, Mathur S, Dhariwal R, Khurana JP, Tyagi AK, Balyan HS, Gupta PK (2014) A multi-step phosphorelay two-component system impacts on tolerance against dehydration stress in common wheat. Funct Integr Genomics 14(4):707–716. https://doi.org/10.1007/s10142-014-0398-8
Ganie SA, Ahammed GJ (2021) Dynamics of cell wall structure and related genomic resources for drought tolerance in rice. Plant Cell Rep 40:437–459
Ganie SA, Reddy AS (2021) Stress-induced changes in alternative splicing landscape in rice: Functional significance of splice isoforms in stress tolerance. Biology 10:309
Ganie SA, Bhat JA, Devoto A (2021) The influence of endophytes on rice fitness under environmental stresses. Plant Mol Biol. https://doi.org/10.1007/s11103-021-01219-8
García AN, Ayub ND, Fox AR, Gómez MC, Diéguez MJ, Pagano EM, Berini CA, Muschietti JP, Soto G (2014) Alfalfa snakin-1 prevents fungal colonization and probably coevolved with rhizobia. BMC Plant Biol 14:248
Gaspar YM, McKenna JA, McGinness BS, Hinch J, Poon S, Connelly AA, Anderson MA, Heath RL (2014) Field resistance to Fusarium oxysporum and Verticillium dahliae in transgenic cotton expressing the plant defensin NaD1. J Exp Bot 65:1541–1550
Ge ZX, Bergonci T, Zhao YL, Zou YJ, Du S, Liu MC, Luo XJ, Ruan H, Garcia-Valencia LE, Zhong S et al (2017) Arabidopsis pollen tube integrity and sperm release are regulated by RALF-mediated signaling. Science 358:1596–1599
Ge Z, Cheung AY, Qu LJ (2019) Pollen tube integrity regulation in flowering plants: insights from molecular assemblies on the pollen tube surface. New Phytol 222(2):687–693
Ghag SB, Shekhawat UKS, Ganapathi TR (2012) Petunia floral defensins with unique Prodomains as novel candidates for development of Fusarium wilt resistance in transgenic banana plants. PLoS ONE 7:e39557
Ghorbani S, Fernandez A, Hilson P, Beeckman T (2014) Signaling peptides in plants. Cell Dev Biol 3:2
Ghorbani S, Lin YC, Parizot B, Fernandez A, Njo MF, van de Peer Y, Beeckman T, Hilson P (2015) Expanding the repertoire of secretory peptides controlling root development with comparative genome analysis and functional assays. J Exp Bot 66:5257–5269
Godiard L, Sauviac L, Torii KU, Grenon O, Mangin B, Grimsley NH, Marco Y (2003) ERECTA, an LRR receptor-like kinase protein controlling development pleiotropically affects resistance to bacterial wilt. Plant J 36:353–365
Gomez-Gomez L, Boller T (2000) FLS2: an LRR receptor-like kinase involved in the perception of the bacterial elicitor flagellin in Arabidopsis. Mol Cell 5:1003–1011
Gomez-Gomez L, Boller T (2002) Flagellin perception: a paradigm for innate immunity. Trends Plant Sci 7:251–256
Gottig N, Garavaglia BS, Daurelio LD, Valentine A, Gehring C et al (2008) Xanthomonas axonopodis pv. citri uses a plant natriuretic peptide-like protein to modify host homeostasis. Proc Natl Acad Sci USA 105:18631–18636
Gou X, Li J (2020) Paired receptor and coreceptor kinases perceive extracellular signals to control plant development. Plant Physiol 01343:2019. https://doi.org/10.1104/pp.19.01343
Gou X, He K, Yang H, Yuan T, Lin H, Clouse SD, Li J (2010) Genome-wide cloning and sequence analysis of leucine-rich repeat receptor-like protein kinase genes in Arabidopsis thaliana. BMC Genomics. https://doi.org/10.1186/1471-2164-11-19
Goyal RK, Mattoo AK (2014) Multitasking antimicrobial peptides in plant development and host defense against biotic/abiotic stress. Plant Sci 228:135–149
Gray WM, Kepinski S, Rouse D, Leyser O (2001) Estelle M (2001) Auxin regulates SCFTIR1-dependent degradation of AUX/IAA proteins. Nature 414:6861
Gray-Mitsumune M, Matton DP (2006) The egg apparatus 1 gene from maize is a member of a large gene family found in both monocots and dicots. Planta 223:618–625
Guilfoyle TJ, Hagen G (2007) Auxin response factors. Curr Opin Plant Biol 10(5):453–460. https://doi.org/10.1016/j.pbi.2007.08.014
Gully K, Hander T, Boller T, Bartels S (2015) Perception of Arabidopsis AtPep peptides, but not bacterial elicitors, accelerates starvation-induced senescence. Front Plant Sci 6:14
Gultyaev AP, Roussis A (2007) Identification of conserved secondary structures and expansion segments in enod40 RNAs reveals new enod40 homologues in plants. Nucleic Acids Res 35:3144–3152
Gutiérrez-Alanís D, Yong-Villalobos L, Jiménez-Sandoval P, Alatorre-Cobos F, Oropeza-Aburto A, Mora-Macías J, Sánchez-Rodríguez F, Cruz-Ramírez A, Herrera-Estrella L (2017) Phosphate starvation-dependent iron mobilization I-induces CLE14 expression to trigger root meristem differentiation through CLV2/PEPR2 signaling. Dev Cell 41:555–570
Haeffner E, Karlovsky P, Splivallo R, Traczewska A, Diederichsen E (2014) ERECTA, salicylic acid, abscisic acid, and jasmonic acid modulate quantitative disease resistance of Arabidopsis thaliana to Verticillium longisporum. BMC Plant Biol 14:85
Ham B-K, Lucas WJ (2017) Phloem-mobile RNAs as systemic signaling agents. Annu Rev Plant Biol 68:173
Hara K, Kajita R, Torii KU, Bergmann DC, Kakimoto T (2007) The secretory peptide gene EPF1 enforces the stomatal one-cell-spacing rule. Genes Dev 21:1720–1725
Hara K, Yokoo T, Kajita R, Onishi T, Yahata S et al (2009) Epidermal cell density is autoregulated via a secretory peptide, EPIDERMAL PATTERNING FACTOR 2 in Arabidopsis leaves. Plant Cell Physiol 50:1019–1031
Haruta M, Constabel CP (2003) Rapid alkalinization factors in poplar cell cultures. Peptide isolation, cDNA cloning, and differential expression in leaves and methyl jasmonate treated cells. Plant Physiol 131:814–823
Haruta M, Sabat G, Stecker K, Minkoff BB, Sussman M (2014) A peptide hormone and its receptor protein kinase regulate plant cell expansion. Science 343:408–411
Hashidume T, Sakano T, Mochizuki A et al (2018) Identification of soybean peptide leginsulin variants in different cultivars and their insulin-like activities. Sci Rep 8:16847. https://doi.org/10.1038/s41598-018-35331-5
He Y, Liu X, Ye L, Pan C, Chen L, Zou T, Lu G (2016) Genome-wide identification and expression analysis of two-component system genes in tomato. Int J Mol Sci. https://doi.org/10.3390/IJMS17081204
He L, Zhang F, Wu X, Hu Y, Dong L, Dewitte W, Wen B (2020) Genome-wide characterization and expression of two-component system genes in cytokinin-regulated gall formation in zizania latifolia. Plants 9:1–26. https://doi.org/10.3390/PLANTS9111409
Hertig C, Melzer M, Rutten T, Erbe S, Hensel G, Kumlehn J, Weschke W, Weber H, Thiel J (2020) Barley HISTIDINE KINASE 1 (HvHK1) coordinates transfer cell specification in the young endosperm. Plant J 103:1869–1884. https://doi.org/10.1111/TPJ.14875
Higashiyama T (2010) Peptide signaling in pollen-pistil interactions. Plant Cell Physiol 51:177–189
Hirakawa Y, Bowman JL (2015) A role of TDIF peptide signaling in vascular cell differentiation is conserved among euphyllophytes. Front Plant Sci 6:1048. https://doi.org/10.3389/fpls.2015.01048
Hirakawa Y, Shinohara H, Kondo Y, Inoue A, Nakanomyo I, Ogawa M et al (2008) Non-cell-autonomous control of vascular stem cell fate by a CLE peptide/receptor system. Proc Natl Acad Sci U S A 105:15208–15213. https://doi.org/10.1073/pnas.0808444105
Hirakawa Y, Kondo Y, Fukuda H (2011) Establishment and maintenance of vascular cell communities through local signaling. Curr Opin Plant Biol 14(1):17-23. doi: https://doi.org/10.1016/j.pbi.2010.09.011.Hirakawa Y, Fujimoto T, Ishida S, Uchida N, Sawa S, Kiyosue T, Ishizaki K, Nishihama R, Kohchi T, Bowman JL (2020) Induction of multichotomous branching by CLAVATA peptide in Marchantia polymorpha. Current Biology 33(19): 3383
Holley SR, Yalamanchili RD, Moura DS, Ryan CA, Stratmann JW (2003) Convergence of signaling pathways induced by systemin, oligosaccharide elicitors, and ultraviolet-B radiation at the level of mitogen-activated protein kinases in Lycopersicon peruvianum suspension-cultured cells. Plant Physiol 132:1728–1738
Hou S, Wang X, Chen D, Yang X, Wang M, Turrà D, di Pietro A, Zhang W (2014) The secreted peptide PIP1 amplifies immunity through receptor-like kinase. PLoS Pathog. https://doi.org/10.1371/JOURNAL.PPAT.1004331
Hu Z, Zhang H, Shi K (2018) Plant peptides in plant defense responses. Plant Signal Behav 13:e1475175
Huffaker A, Pearce G, Ryan CA (2006) An endogenous peptide signal in Arabidopsis activates components of the innate immune response. Proc Natl Acad Sci USA 103:10098–10103
Hunt L, Gray JE (2009) The signaling peptide EPF2 controls asymmetric cell divisions during stomatal development. Curr Biol 19:864–869
Huo R, Liu Z, Yu X, Li Z (2020) The interaction network and signaling specificity of two-component system in arabidopsis. Int J Mol Sci 21:4898
Hwang I, Chen HC, Sheen J (2002) Two-component signal transduction pathways in Arabidopsis. Plant Physiol 129(2):500–515. https://doi.org/10.1104/pp.005504
Igarashi D, Tsuda K, Katagiri F (2012) The peptide growth factor, phytosulfokine, attenuates pattern-triggered immunity. Plant J 71:194–204
Ikeuchi M, Yamaguchi Y, Kazama T, Ito T, Horiguchi G, Tsukaya H (2011) ROTUNDIFOLIA4 regulates cell proliferation along the body axis in Arabidopsis shoot. Plant Cell Physiol 52:59–69
Imin N, Mohd-Radzman NA, Ogilvie HA, Djordjevic MA (2013) The peptide-encoding CEP1 gene modulates lateral root and nodule numbers in Medicago truncatula. J Exp Bot 64:5395–5409
Imin N, Patel N, Corcilius L, Payne RJ, Djordjevic MA (2018) CLE peptide tri-arabinosylation and peptide domain sequence composition are essential for SUNN-dependent autoregulation of nodulation in Medicago truncatula. New Phytol 218:73–80
Ito Y, Nakanomyo I, Motose H, Iwamoto K, Sawa S, Dohmae N, Fukuda H (2006) Dodeca-CLE peptides as suppressors of plant stem cell differentiation. Science 313:842–845
Jha S, Chattoo BB (2009) Expression of a plant defensin in rice confers resistance to fungal phytopathogens. Transgenic Res 19:373–384
Jia G, Liu X, Owen HA, Zhao D (2008) Signaling of cell fate determination by the TPD1 small protein and EMS1 receptor kinase. Proc Natl Acad Sci USA 105:2220–2225
Jones JD, Dangl JL (2006) The plant immune system. Nature 444(7117):323–329
Kandoth PK, Ranf S, Pancholi SS, Jayanty S, Walla MD, Miller W, Howe GA, Lincoln DE, Stratmann JW (2007) Tomato MAPKs LeMPK1, LeMPK2, and LeMPK3function in the systemin-mediated defense response against herbivorous insects. Proc Natl Acad Sci U S A 104:12205–12210
Katsir L, Davies KA, Bergmann DC, Laux T (2011) Peptide signaling in plant development. Curr Biol 21(9):356–364
Kaur J, Fellers J, Adholeya A, Velivelli SL, El-Mounadi K, Nersesian N, Clemente T, Shah D (2016) Expression of apoplast- targeted plant defensin MtDef4. 2 confers resistance to leaf rust pathogen Puccinia triticina but does not affect mycorrhizal symbiosis in transgenic wheat. Trans Res 26:37–49
Kawamoto N, Del Carpio DP, Hofmann A, Mizuta Y, Kurihara D, Higashiyama T, Uchida Y, Torii KU, Colombo L, Groth G, Simon R (2020) A peptide pair coordinates regular ovule initiation patterns with seed number and fruit size. Curr Biol 30(22):4352–4361
Kazan K, Manners JM (2009) Linking development to defense: auxin in plant-pathogen interactions. Trends Plant Sci 14:373–382. https://doi.org/10.1016/J.TPLANTS.2009.04.005
Kessler SA, Shimosato-Asano H, Keinath NF, Wuest SE, Ingram G, Panstruga R, Grossniklaus U (2010) Conserved molecular components for pollen tube reception and fungal invasion. Science 330:968–971
Kieliszewski MJ, Lamport DTA, Tan L, Cannon MC (2011) Hydroxyproline-rich glycoproteins: form and function. In: Ulvskov P., Plant Polysaccharides. Annual Plant Reviews: Plant Polysaccharides, Biosynthesis and Bioengineering 41, Wiley, Chichester, UK, pp 321–342
Kim H, Zhou J, Kumar D, Jang G, Ryu KH, Sebastian J, Miyashima S, Helariutta Y, Lee JY (2020) SHORTROOT-mediated intercellular signals coordinate phloem development in Arabidopsis roots. Plant Cell 32:1519–1535
Kim JS, Jeon BW, Kim J (2021) Signaling peptides regulating abiotic stress responses in plants. Front Plant Sci. https://doi.org/10.3389/fpls.2021.704490
Komori R, Amano Y, Ogawa-Ohnishi M, Matsubayashi Y (2009) Identification of tyrosylprotein sulfotransferase in Arabidopsis. Proc Natl Acad Sci U S A 106:15067–15072
Kondo T, Sawa S, Kinoshita A, Mizuno S, Kakimoto T, Fukuda H, Sakagami Y (2006) A plant peptide encoded by CLV3 identified by in situ MALDI-TOF MS analysis. Science 313:845–848
Kondo T, Kajita R, Miyazaki A, Hokoyama M, Nakamura-Miura T et al (2010) Stomatal density is controlled by a mesophyll-derived signaling molecule. Plant Cell Physiol 51:1–8
Kutschmar A, Rzewuski G, Stuhrwohldt N, GTS B, Inze D, Sauter M, (2009) PSK-a promotes root growth in Arabidopsis. New Phytol 181:820–831
Lauressergues D, Couzigou JM, Clemente HS, Martinez Y, Dunand C, Becard G, Combier JP (2015) Primary transcripts of microRNAs encode regulatory peptides. Nature 520(7545):90–93
Lease KA, Walker JC (2006) The Arabidopsis unannotated secreted peptide database, a resource for plant peptidomics. Plant Physiol 142:831–838
Lee JS, Hnilova M, Maes M, Lin YC, Putarjunan A, Han SK, Avila J, Torii KU (2015) Competitive binding of antagonistic peptides fine-tunes stomatal patterning. Nature 522(7557):439–443. https://doi.org/10.1038/nature14561
Lee KP, Liu K, Kim EY, Medina-Puche L, Dong H, Duan J, Li M, Dogra V, Li Y, Lv R, Li Z, Lozano-Duran R, Kim C (2020) PLANT NATRIURETIC PEPTIDE A and its putative receptor PNP-R2 antagonize salicylic acid-mediated signaling and cell death. Plant Cell 32(7):2237–2250. https://doi.org/10.1105/tpc.20.00018
Legnini I, Di Timoteo G, Rossi F, Morlando M, Briganti F, Sthandier O, Fatica A, Santini T, Andronache A, Wade M, Laneve P (2017) Circ-ZNF609 is a circular RNA that can be translated and functions in myogenesis. Mol Cell 66(1):22–37
Li KL, Bai X, Li Y, Cai H, Ji W, Tang LL, Wen YD, Zhu YM (2011) GsGASA1 mediated root growth inhibition in response to chronic cold stress is marked by the accumulation of DELLAs. J Plant Physiol 168:2153–2160. https://doi.org/10.1016/j.jplph.2011.07.006
Li Z, Chakraborty S, Xu G (2017) Differential CLE peptide perception by plant receptors implicated from structural and functional analyses of TDIF-TDR interactions. PLoS ONE. https://doi.org/10.1371/journal.pone.0175317
Li H, Kim YJ, Yang L, Liu Z, Zhang J, Shi H, Huang G, Persson S, Zhang D, Liang W (2020) Grass-specific EPAD1 is essential for pollen exine patterning in rice. Plant Cell 32(12):3961–3977
Li R, Shi CL, Wang X, Meng Y, Cheng L, Jiang CZ et al (2021) Inflorescence abscission protein SlIDL6 promotes low light intensity-induced tomato flower abscission. Plant Physiol 186(2):1288–1301
Liao Z, Yu H, Duan J et al (2019) SLR1 inhibits MOC1 degradation to coordinate tiller number and plant height in rice. Nat Commun 10:2738. https://doi.org/10.1038/s41467-019-10667-2
Lindsey K, Casson S, Chilley P (2002) Peptides: new signaling molecules in plants. Trends Plant Sci 7:78–83
Liu Z, Wu Y, Yang F, Zhang Y, Chen S, Xie Q, Tian X, Zhou JM (2013) BIK1 interacts with PEPRs to mediate ethylene-induced immunity. PNAS USA 110:6205–6210
Liu Z, Zhang M, Kong L, Lv Y, Zou M, Lu G et al (2014) Genome-wide identification, phylogeny, duplication, and expression analyses of two-component system genes in Chinese cabbage (Brassica rapa ssp. pekinensis). DNA Res 21:379–396. https://doi.org/10.1093/dnares/dsu004
Liu J, Chen T, Zhang J, Li C, Xu Y, Zheng H, Zhou J, Zha L, Jiang C, Jin Y, Nan T, Yi J, Sun P, Yuan Y, Huang L (2020) Ginsenosides regulate adventitious root formation in Panax ginseng via a CLE45-WOX11 regulatory module. J Exp Bot 71(20):6396–6407
Llorente F, Alonso-Blanco C, Sanchez-Rodriguez C, Jorda L, Molina A (2005) ERECTA receptor-like kinase and heterotrimeric G protein from Arabidopsis are required for resistance to the necrotrophic fungus Plectosphaerella cucumerina. Plant J 43:165–180
Lough TJ, Lucas WJ (2006) INTEGRATIVE PLANT BIOLOGY: Role of phloem long-distance macromolecular trafficking. Annu Rev Plant Biol 57(1):203–232
Lu X, Shi H, Ou Y, Cui Y, Chang J, Peng L et al (2020) RGF1-RGI1, a peptide-receptor complex, regulates Arabidopsis root meristem development via a MAPK signaling cascade. Mol Plant 13:1594–1607. https://doi.org/10.1016/j.molp.2020.09.005
Lu L, Arif S, Yu JM, Lee JW, Park YH, Tucker ML, Kim J (2023) Involvement of IDA-HAE module in natural development of tomato flower abscission. Plants (basel) 12(1):185. https://doi.org/10.3390/plants12010185
Luo S, Li Q, Liu S, Pinas NM, Tian H, Wang S (2015) Constitutive expression of OsIAA9 affects starch granules accumulation and root gravitropic response in arabidopsis. Front Plant Sci. https://doi.org/10.3389/FPLS.2015.01156
Ma T, Ma H, Zhao H et al (2014) Identification, characterization, and transcription analysis of xylogen-like arabinogalactan proteins in rice (Oryza sativa L.). BMC Plant Biol 14:299. https://doi.org/10.1186/s12870-014-0299-y
Mang HG, Feng BM, Hu ZJ, Boisson-Dernier A, Franck CM, Meng XZ, Huang YY, Zhou JG, Xu GY, Wang TT et al (2017) Differential regulation of two-tiered plant immunity and sexual reproduction by ANXUR receptor-like kinases. Plant Cell 29:3140–3156
Maróti G, Downie JA, Kondorosi É (2015) Plant cysteine-rich peptides that inhibit pathogen growth and control rhizobial differentiation in legume nodules. Curr Opin Plant Biol 26:57–63
Márton ML, Fastner A, Uebler S, Dresselhaus T (2012) Overcoming hybridization barriers by the secretion of the maize pollen tube attractant ZmEA1 from Arabidopsis ovules. Curr Biol 22:1194–1198
Masachis S, Segorbe D, Turrà D, Leon-Ruiz M, Fürst U, El Ghalid M, Leonard G, López-Berges MS, Richards TA, Felix G, Di Pietro A (2016) A fungal pathogen secretes plant alkalinizing peptides to increase infection. Nat Microbiol 1(6):16043
Matos JL, Fiori CS, Silva-Filho MC, Moura DS (2008) A conserved dibasic site is essential for correct processing of the peptide hormone AtRALF1 in Arabidopsis thaliana. FEBS 582:3343–3347
Matsubayashi Y (2010) MBSJ MCC Young Scientist Award 2012. Recent progress in research on small post-translationally modified peptide signals in plants. Genes Cells 17(1):1–10
Matsubayashi Y (2011) Post-translational modifications in secreted peptide hormones in plants. Plant Cell Physiol 52:5–13
Matsubayashi Y (2014) Posttranslationally modified small-peptide signals in plants. Ann Rev Plant Biol 65:385–413
Matsubayashi Y, Sakagami Y (1996) Phytosulfokine, sulfated peptides that induce the proliferation of single mesophyll cells of Asparagus officinalis L. Proc Natl Acad Sci USA 93:7623–7627
Matsubayashi Y, Sakagami Y (1999) Characterization of specific binding sites for a mitogenic sulfated peptide, phytosulfokine-alpha, in the plasma-membrane fraction derived from Oryza sativa L. Eur J Biochem 262:666–671
Matsubayashi Y, Sakagami Y (2006) Peptide hormones in plants. Annu Rev Plant Biol 57:649–674
Matsubayashi Y, Takagi L, Omura N, Morita A, Sakagami Y (1999) The endogenous sulfated pentapeptide phytosulfokine-α stimulates tracheary element differentiation of isolated mesophyll cells of Zinnia. Plant Physiol 120(4):1043–1048
Matsubayashi E, Yang P, Sakagami Y (2001) Peptide signals and their receptors in higher plants. Trends Plant Sci 6(12):573–577
Matsubayashi Y, Ogawa M, Morita A, Sakagami Y (2002) An LRR receptor kinase involved in perception of a peptide plant hormone, phytosulfokine. Science 296:1470–1472
Matsuzaki Y, Ogawa-Ohnishi M, Mori A, Matsubayashi Y (2010) Secreted peptide signals required for maintenance of root stem cell niche in Arabidopsis. Science 329:1065–1067
McLachlan DH, Pridgeon AJ, Hetherington AM (2018) How arabidopsis talks to itself about its water supply. Mol Cell 70(6):991–992. https://doi.org/10.1016/j.molcel.2018.06.011
Meng X, Zhou J, Tang J, Li B, de Oliveira MVV, Chai J, He P, Shan L (2016) Ligand-induced receptor-like kinase complex regulates floral organ abscission in Arabidopsis. Cell Rep 14:1330–1338
Miyata SI, Urao T, Yamaguchi-Shinozaki K, Shinozaki K (1998) Characterization of genes for two-component phosphorelay mediators with a single HPt domain in Arabidopsis thaliana. FEBS Lett 437:11–14. https://doi.org/10.1016/S0014-5793(98)01188-0
Mizuno T (1997) Compilation of all genes encoding two-component phosphotransfer signal transducers in the genome of Escherichia coli. DNA Res 4:161–168
Mochida K, Yoshida T, Sakurai T, Yamaguchi-Shinozaki K, Shinozaki K, Tran LS (2010) Genome-wide analysis of two-component systems and prediction of stress-responsive two-component system members in soybean. DNA Res 17:303–324
Mohd-Radzman NA, Binos S, Truong TT, Imin N, Mariani M, Djordjevic MA (2015) Novel MtCEP1 peptides produced in vivo differentially regulate root development in Medicago truncatula. J Exp Bot 66(17):5289–5300. https://doi.org/10.1093/jxb/erv008
Mondragon-Palomino M, Stam R, John-Arputharaj A, Dresselhaus T (2017) Diversification of defensins and NLRs in Arabidopsis species by different evolutionary mechanisms. BMC Evol Biol 17:255
Moore KL (2003) The biology and enzymology of protein tyrosine O -sulfation. J Biol Chem 278:24243–24246
Morita J, Kato K, Nakane T, Kondo Y, Fukuda H, Nishimasu H, Ishitani R, Nureki O (2016) Crystal structure of the plant receptor-like kinase TDR in complex with the TDIF peptide. Nat Commun 17:1–9. https://doi.org/10.1038/ncomms12383
Motose H, Fukuda H, Sugiyama M (2001) Involvement of local intercellular communication in the differentiation of zinnia mesophyll cells into tracheary elements. Planta 213:121–131. https://doi.org/10.1007/s004250000482
Moussu S, Doll NM, Chamot S, Brocard L, Creff A, Fourquin C, Widiez T, Nimchuk ZL, Ingram G (2017) ZHOUPI and KERBEROS mediate embryo/endosperm separation by promoting the formation of an extracuticular sheath at the embryo surface. Plant Cell 29(7):1642–1656
Murphy E, Smith S, De Smet I (2012) Small signaling peptides in Arabidopsis development: how cells communicate over a short distance. Plant Cell 24(8):3198–3217
Muschietti J, Dircks L, Vancanneyt G, McCormick S (1994) LAT52 protein is essential for tomato pollen development: Pollen expressing antisense LAT52 RNA hydrates and germinates abnormally and cannot achieve fertilization. Plant J 6:321–338
Myllyharju J (2003) Prolyl 4-hydroxylases, the key enzymes of collagen biosynthesis. Matrix Biol 22:15–24
Nagpal P, Walker LM, Young JC, Sonawala A, Timpte C, Estelle M, Reed JW (2000) AXR2 encodes a member of the Aux/IAA protein family. Plant Physiol 123:563–574. https://doi.org/10.1104/PP.123.2.563
Nahirnak V, Almasia NI, Fernandez PV, Hopp HE, Estevez JM, Carrari F, Vazquez-Rovere C (2012a) Potato snakin-1 gene silencing affects cell division, primary metabolism, and cell wall composition. Plant Physiol 158:252–263
Nahirnak V, Almasia NI, Hopp HE, Vazquezrovere C (2012b) Snakin/GASA proteins involvement in hormone crosstalk and redox homeostasis. Plant Signal Behav 7:1004–1008
Nakaminami K, Okamoto M, Higuchi-Takeuchi M, Yoshizumi T, Yamaguchi Y, Fukao Y, Shimizu M, Ohashi C, Tanaka M, Matsui M, Shinozaki K, Seki M, Hanada K (2018) AtPep3 is a hormone-like peptide that plays a role in the salinity stress tolerance of plants. Proc Natl Acad Sci USA 22:5810–5815. https://doi.org/10.1073/pnas.1719491115
Nakayama T, Shinohara H, Tanaka M, Baba K, Ogawa-Ohnishi M, Matsubayashi Y (2017) A peptide hormone required for Casparian strip diffusion barrier formation in Arabidopsis roots. Science 355:284–286
Nambara E, Marion-Poll A (2005) Abscisic acid biosynthesis and catabolism. Annu Rev Plant Biol 56:165–185. https://doi.org/10.1146/annurev.arplant.56.032604.144046
Narita NN, Moore S, Horiguchi G, Kubo M, Demura T, Fukuda H, Goodrish J, Tsukaya H (2004) Overexpression of a novel small peptide ROTUNDIFOLIA4 decreases cell proliferation and alters leaf shape in Arabidopsis thaliana. Plant J 38:699–713
Narváez-Vásquez J., Orozco-Cárdenas ML (2008) Systemins and AtPeps: Defense-Related Peptide Signals. In: Schaller A. (eds) Induced Plant Resistance to Herbivory. Springer, Dordrecht. https://doi.org/10.1007/978-1-4020-8182-8_15
Nguyen KH, van Ha C, Nishiyama R, Watanabe Y, Leyva-González MA, Fujita Y, Tran UT, Li W, Tanaka M, Seki M, Schaller GE, Herrera-Estrella L, Tran LSP (2016) Arabidopsis type B cytokinin response regulators ARR1, ARR10, and ARR12 negatively regulate plant responses to drought. Proc Natl Acad Sci USA 113:3090–3095. https://doi.org/10.1073/PNAS.1600399113
Niemeyer M, Moreno Castillo E, Ihling CH, Iacobucci C, Wilde V, Hellmuth A, Hoehenwarter W, Samodelov SL, Zurbriggen MD, Kastritis PL, Sinz A (2020) Calderón Villalobos LIA (2020) Flexibility of intrinsically disordered degrons in AUX/IAA proteins reinforces auxin co-receptor assemblies. Nat Commun 11:1–18. https://doi.org/10.1038/s41467-020-16147-2
Nishiyama R, Watanabe Y, Leyva-Gonzalez MA, van Ha C, Fujita Y, Tanaka M, Seki M, Yamaguchi-Shinozaki K, Shinozaki K, Herrera-Estrella L, Tran LSP (2013) Arabidopsis AHP2, AHP3, and AHP5 histidine phosphotransfer proteins function as redundant negative regulators of drought stress response. Proc Natl Acad Sci U S A 110:4840–4845. https://doi.org/10.1073/PNAS.1302265110
Nontachaiyapoom S, Scott PT, Men AE, Kinkema M, Schenk PM, Gresshoff PM (2007) Promoters of orthologous Glycine max and Lotus japonicus nodulation autoregulation genes interchangeably drive phloem-specific expression in transgenic plants. Mol Plant Microbe Interact 20:769–780. https://doi.org/10.1094/mpmi-20-7-0769
Ogawa M, Shinohara H, Sakagami Y, Matsubayashi Y (2008) Arabidopsis CLV3 peptide directly binds CLV1 ectodomain. Science 319:294. https://doi.org/10.1126/science.1150083
Ogawa-Ohnishi M, Matsushita W, Matsubayashi Y (2013) Identification of three hydroxyproline O-arabinosyltransferases in Arabidopsis thaliana. Nat Chem Biol 9:726–730
Oh E, Seo PJ, Kim J (2018) Signaling peptides and receptors coordinating plant root development. Trends Plant Sci 23(4):337–351. https://doi.org/10.1016/j.tplants.2017.12.007
Ohkubo Y, Tanaka M, Tabata R, Ogawa-Ohnishi M, Matsubayashi Y (2017) Shoot-to-root mobile polypeptides involved in systemic regulation of nitrogen acquisition. Nature Plants 3:17029
Ohyama K, Ogawa M, Matasubayashi Y (2008) Identification of a biologically active, small, secreted peptide in Arabidopsis by in silico gene screening, followed by LC-MS-based structure analysis. Plant J 55:152–160
Ohyama K, Shinohara H, Ogawa-Ohnishi M, Matsubayashi Y (2009) A glycopeptide regulating stem cell fate in Arabidopsis thaliana. Nat Chem Biol 5:578–580
Oka-Kira E, Kawaguchi M (2006) Long-distance signaling to control root nodule number. Curr Opin Plant Biol 9:496–502
Okamoto S, Shinohara H, Mori T, Matsubayashi Y, Kawaguchi M (2013) Root-derived CLE glycopeptides control nodulation by direct binding to HAR1 receptor kinase. Nat Commun 4:2191
Okamoto S, Suzuki T, Kawaguchi M, Higashiyama T, Matsubayashi Y (2015) A comprehensive strategy for identifying long-distance mobile peptides in xylem sap. Plant J 84:611–620
Okuda S, Higashiyama T (2010) Pollen tube guidance by attractant molecules: LUREs. Cell Struct Funct 35(1):45–52. https://doi.org/10.1247/csf.10003
Okuda S, Tsutsui H, Shiina K, Sprunck S, Takeuchi H, Yui R, Higashiyama T et al (2009) Defensin-like polypeptide LUREs are pollen tube attractants secreted from synergid cells. Nature 458(7236):357–361
Olsen AN, Mundy J, Skriver K (2002) Peptomics, identification of novel cationic Arabidopsis peptides with conserved sequence motifs. In Silico Biol (gedrukt) 2:441–451
Olsson V, Joos L, Zhu S, Gevaert K, Butenko MA, DeMA DS, I, (2018) Look closely, the beautiful may be small: precursor-derived peptides in plants. Annu Rev Plant Biol. https://doi.org/10.1146/annurev-arplant-042817-040413
Olsson V, Joos L, Zhu S, Gevaert K, Butenko MA, De Smet I (2019) Look closely, the beautiful may be small: precursor-derived peptides in plants. Annu Rev Plant Biol 70:153–218
Onrubia M, Pollier J, Van den Bossche R, Goethals M, Gevaert K, Moyano E, Vidal-Limon H, Cusidó RM, Palazón J, Goossens A (2014) Taximin, a conserved plant-specific peptide is involved in the modulation of plant-specialized metabolism. Plant Biotechnol J 12:971–983
Ormancey M, Le Ru A, Duboé C, Jin H, Thuleau P, Plaza S, Combier JP (2020) Internalization of miPEP165a into Arabidopsis roots depends on both passive diffusion and endocytosis-associated processes. Int J Mol Sci 21(7):2266
Orsini F, Cascone P, De Pascale S, Barbieri G, Corrado G, Rao R, Maggio A (2010) Systemin-dependent salinity tolerance in tomato: Evidence of specific convergence of abiotic and biotic stress responses. Physiol Plant 138:10–21
Ota R, Ohkubo Y, Yamashita Y, Ogawa-Ohnishi M, Matsubayashi Y (2020) Shoot-to-root mobile CEPD-like 2 integrates shoot nitrogen status to systemically regulate nitrate uptake in Arabidopsis. Nat Commun 11:641
Ou Y, Lu X, Zi Q, Xun Q, Zhang J, Wu Y, Shi H, Wei Z, Zhao B, Zhang X et al (2016) RGF1 INSENSITIVE 1 to 5, a group of LRR receptor-like kinases, are essential for the perception of root meristem growth factor 1 in Arabidopsis thaliana. Cell Res 26:686–698
Pan H, Wang D (2017) Nodule cysteine-rich peptides maintain a working balance during nitrogen-fixing symbiosis. Nature Plants 3:17048. https://doi.org/10.1038/nplants.2017.48
Pareek A, Singh A, Kumar M, Kushwaha HR, Lynn AM, Singla-Pareek SL (2006) Whole-genome analysis of Oryza sativa reveals similar architecture of two-component signaling machinery with Arabidopsis. Plant Physiol 142(2):380–397. https://doi.org/10.1104/pp.106.086371
Patel N, Mohd-Radzman NA, Corcilius L, Crossett B, Connolly A, Cordwell SJ, Ivanovici A, Taylor K, Williams J, Binos S, Mariani M, Payne RJ, Djordjevic MA (2018) Diverse peptide hormones affecting root growth identified in the Medicago truncatula secreted peptidome. Mol Cell Proteomics 17:160–174
Patharkar OR, Walker JC (2018) Advances in abscission signaling. J Exp Bot 69(4):733–740. https://doi.org/10.1093/jxb/erx256. (PMID: 28992277)
Pearce G, Ryan CA (2003) Systemic signaling in tomato plants for defense against herbivores. Isolation and characterization of three novel defense-signaling glycopeptide hormones coded in a single precursor gene. J Biol Chem 278:30044–30050
Pearce G, Strydom D, Johnson S, Ryan CA (1991) A polypeptide from tomato leaves induces wound-inducible proteinase inhibitor proteins. Science 253:895–898
Pearce G, Moura DS, Stratmann J, Ryan CA (2001a) RALF, a 5-kDa ubiquitous polypeptide in plants, arrests root growth and development. Proc Natl Acad Sci U S A 98(22):12843–12847. https://doi.org/10.1073/pnas.201416998
Pearce G, Moura DS, Stratmann J, Ryan CA (2001b) Production of multiple plant hormones from a single polyprotein precursor. Nature 411:817–820
Pearce G, Yamaguchi Y, Munske G, Ryan CA (2008) Structure-activity studies of AtPep1, a plant peptide signal involved in the innate immune response. Peptides 29:2083–2089
Pearce G, Yamaguchi Y, Barona G, Ryan CA (2010) A subtilisin-like protein from soybean contains an embedded, cryptic signal that activates defense-related genes. Proc Natl Acad Sci U S A 107(33):14921–14925
Poitout A, Crabos A, Petřík I, Novák O, Krouk G, Lacombe B, Ruffel S (2018) Responses to systemic nitrogen signaling in arabidopsis roots involve trans-Zeatin in Shoots. Plant Cell 30(6):1243–1257. https://doi.org/10.1105/tpc.18.00011
Prel A, Dozier C, Combier JP, Plaza S, Besson A (2021) Evidence that regulation of Pri-miRNA/miRNA expression is not a general rule of miPEPs function in humans. Int J Mol Sci 22:3432
Prusinkiewicz P, Erasmus Y, Lane B, Harder LD, Coen E (2007) Evolution and development of inflorescence architectures. Science 316:1452–1456
Qi Z, Verma R, Gehring C, Yamaquchi Y, Zhao Y, Ryan CA, Berkowitz GA (2010) Ca2+ signaling by plant Arabidopsis thaliana pep peptides depends on AtPepR1, a receptor with guanylyl cyclase activity, and cGMP-activated Ca2+ channels. Proc Natl Acad Sci U S A 107:21193–21198
Qu X, Cao B, Kang J, Wang X, Han X, Jiang W, Shi X, Zhang L, Cui L, Hu Z, Zhang Y, Wang G (2019) Fine-tuning stomatal movement through small signaling peptides. Front Plant Sci. https://doi.org/10.3389/fpls.2019.00069
Rafudeen S, Gxaba G, Makgoke G, Bradley G, Pironcheva G et al (2003) A role for plant natriuretic peptide immuno-analogues in NaCl- and drought-stress responses. Physiol Plant 119:554–562
Reichardt S, Piepho HP, Stintzi A, Schaller A (2020) Peptide signaling for drought-induced tomato flower drop. Science 367(6485):1482–1485. https://doi.org/10.1126/science.aazscience.aaz5641
Rejeb I, Pastor V, Mauch-Mani B (2014) Plant responses to simultaneous biotic and abiotic stress: molecular mechanisms. Plants 3(4):458–475
Ren Y, Song Y, Zhang L, Guo D, He J, Wang L, Song S, Xu W, Zhang C, Lers A, MaA MC (2021) Coding of non-coding RNA: insights into the regulatory functions of Pri-MicroRNA-encoded peptides in plants. Front Plant Sci 12:186
Rinaldi MA, Liu J, Enders TA, Bartel B, Strader LC (2012) A gain-of-function mutation in IAA16 confers reduced responses to auxin and abscisic acid and impedes plant growth and fertility. Plant Mol Biol 79:359–373. https://doi.org/10.1007/S11103-012-9917-Y
Roberts I, Smith S, De Rybel B, Van Den Broeke J, Smet W et al (2013) The CEP family in land plants: evolutionary analyses, expression studies, and role in Arabidopsis shoot development. J Exp Bot 64:5371–5381
Roberts I, Smith S, Stes E, De Rybel B, Staes A, van de Cotte B, Njo MF, Dedeyne L, Demol H, Lavenus J et al (2016) CEP5 and XIP1/CEPR1 regulate lateral root initiation in Arabidopsis. J Exp Bot 67:4889–4899
Rogg LE, Lasswell J, Bartel B (2001) A gain-of-function mutation in IAA28 suppresses lateral root development. Plant Cell 13:465–480. https://doi.org/10.1105/TPC.13.3.465
Rohrig H, Schmidt J, Miklashevichs E, Schell J, John M (2002) Soybean ENOD40 encodes two peptides that bind to sucrose synthase. Proc Natl Acad Sci U S A 99:1915–1920
Rojo E, Denecke J (2008) What is moving in the secretory pathway of plants? Plant Physiol 147:1493–1503
Ross A, Yamada K, Hiruma K, Yamashita-Yamada M, Lu X, Takano Y, Tsuda K, Saijo Y (2014) The Arabidopsis PEPR pathway couples local and systemic plant immunity. EMBO J 33:62–75
Rouse D, Mackay P, Stirnberg P, Estelle M, Leyser O (1998) Changes in auxin response from mutations in an AUX/IAA gene. Science 279:1371–1373. https://doi.org/10.1126/SCIENCE.279.5355.1371
Roux F, Voisin D, Badet TC, Balagué X, Barlet C, Huard-Chauveau, et al (2014) Resistance to phytopathogens e tutti quanti: placing plant quantitative disease resistance on the map. Mol Plant Pathol 15:427–432
Ruegger M, Dewey E, Gray WM, Hobbie L, Turner J, Estelle M (1998) The TIR1 protein of Arabidopsis functions in auxin response and is related to human SKP2 and yeast grr1p. Genes Dev 12:198–207. https://doi.org/10.1101/GAD.12.2.198
Ruffel S, Krouk G, Ristova D, Shasha D, Birnbaum KD, Coruzzi GM (2011) Nitrogen economics of root foraging: transitive closure of the nitrate-cytokinin relay and distinct systemic signaling for N supply vs. demand. Proc Natl Acad Sci U S A 108:18524–18529
Ruffel S, Poitout A, Krouk G, Coruzzi GM, Lacombe B (2016) Long-distance nitrate signaling displays cytokinin dependent and independent branches. J Integr Plant Biol 58:226–229
Ryan CA, Pearce G (1998) Systemin: a polypeptide signal for plant defensive genes. Ann Rev Cell Dev Biol 14:1–17
Ryan CA, Pearce G (2003) Systemins: a functionally defined family of peptide signals that regulate defensive genes in Solanaceae species. Proc Natl Acad Sci U S A 2:14577–14580
Sanchez-Rodriguez C, Estevez JM, Llorente F, Hernandez-Blanco C, Jorda L, Pagan I, Berrocal M, Marco Y, Somerville S, Molina A (2009) The ERECTA receptor-like kinase regulates cell wall-mediated resistance to pathogens in Arabidopsis thaliana. Mol Plant Microbe Interact 22:953–963
Santiago J, Brandt B, Wildhagen M, Hohmann U, Hothorn LA, Butenko MA, Hothorn M (2016) Mechanistic insight into a peptide hormone signaling complex mediating floral organ abscission. eLife, 5: e15075.
Sasaki T, Suzaki T, Soyano T, Kojima M, Sakakibara H, Kawaguchi M (2014) Shoot-derived cytokinins systemically regulate root nodulation. Nat Commun 5:4983. https://doi.org/10.1038/ncomms5983
Schaller GE, Kieber JJ, Shiu SH (2008) Two-component signaling elements and histidyl-aspartyl phosphorelays. Arabidopsis Book 6: e0112.Schardon K., Hohl M., Graff L., Pfannstiel J., Schulze W., Stintzi A., Schaller A (2016) Precursor processing for plant peptide hormone maturation by subtilisin- like serine proteinases. Science 354:1594–1597
Scheer JM, Pearce G, Ryan CA (2005) LeRALF, a plant peptide that regulates root growth and development, specifically binds to 25 and 120 kDa cell surface membrane proteins of Lycopersicon peruvianum. Planta 221:667–674. https://doi.org/10.1007/s00425-004-1442-z
Schmelz EA, Carroll MJ, LeClere S, Phipps SM, Meredith J, Chourey PS, Alborn HT, Teal PEA (2006) Fragments of ATP synthase mediate plant perception of insect attack. Proc Natl Acad Sci U S A 103:8894–8899
Schmelz EA, Huffaker A, Carroll MJ, Alborn HT, Ali JG, Teal PEA (2012) An amino acid substitution inhibits specialist herbivore production of an antagonist effector and recovers insect- induced plant defenses. Plant Physiol 160:1468–1478
Schopfer CR, Nasrallah ME, Nasrallah JB (1999) The male determinant of self-incompatibility in Brassica. Science 286:1697–1700
Segonzac C, Monaghan J (2019) Modulation of plant innate immune signaling by small peptides. Curr Opin Plant Biol 51:22–28
Seitz O (2000) Glycopeptide synthesis and the effects of glycosylation on protein structure and activity. ChemBioChem 1(4):214–246
Semeradova H, Montesinos JC, Benkova E (2020) All roads lead to auxin: post-translational regulation of auxin transport by multiple hormonal pathways. Plant Commun. https://doi.org/10.1016/j.xplc.2020.100048
Shao Y, Yu X, Xu X, Li Y, Yuan W, Xu Y, Mao C, Zhang S, Xu J (2020) The YDA-MKK4/MKK5-MPK3/MPK6 cascade functions downstream of the RGF1-RGI ligand-receptor pair in regulatingc mitotic activity in root apical meristem. Mol Plant 13(11):1608–1623
Sharan A, Soni P, Nongpiur RC, Singla-Pareek SL, Pareek A (2017) Mapping the ‘Two-component system’ network in rice. Sci Rep 7:1–13. https://doi.org/10.1038/s41598-017-08076-w
Sharma A, Hussain A, Mun BG, Imran QM, Falak N, Lee SU et al (2016) Comprehensive analysis of plant rapid alkalization factor (RALF) genes. Plant Physiol Biochem 106:82–90
Sharma A, Badola PK, Bhatia C, Sharma D, Trivedi PK (2020) Primary transcript of miR858 encodes regulatory peptide and controls flavonoid biosynthesis and development in Arabidopsis. Nature Plants 6(10):1262–1274
Shi L, Gast RT, Gopalraj M, Olszewski NE (1992) Characterization of a shoot-specific, GA3- and ABA-regulated gene from tomato. Plant J 2:153–159
Shi CL, Alling RM, Hammerstad M, Aalen RB (2019) Control of organ abscission and other cell separation processes by evolutionary conserved peptide signaling. Plants (basel) 8(7):225. https://doi.org/10.3390/plants8070225
Shinohara H, Mori A, Yasue N, Sumida K, Matsubayashi Y (2016) Identification of three LRR-RKs involved in perception of root meristem growth factor in Arabidopsis. Proc Natl Acad Sci USA 113:3897–3902
Shiu SH, Bleecker AB (2001) Receptor-like kinases from Arabidopsis form a monophyletic gene family related to animal receptor kinases. Proc Natl Acad Sci USA 98:10763–10768
Silverstein KAT, Moskal WJ Jr, Wu HC, Underwood BA, Graham MA, Town CD, Vanden Bosch KA (2007) Small cysteine-rich peptides resembling antimicrobial peptides have been underpredicted in plants. Plant J 51:262–280
Singh P, Maurya SK, Singh D, Sane AP (2023) The rose INFLORESCENCE DEFICIENT IN ABSCISSION-LIKE genes, RbIDL1 and RbIDL4, regulate abscission in an ethylene-responsive manner. Plant Cell Rep. https://doi.org/10.1007/s00299-023-03017-6
Smith S, Zhu S, Joos L, Roberts I, Nikonorova N, Vu LD, Stes E, Cho H, Larrieu A, Xuan W, Goodall B, van de Cotte B, Waite JM, Rigal A, Ramans Harborough S, Persiau G, Vanneste S, Kirschner GK, Vandermarliere E, Martens L, Stahl Y, Audenaert D, Friml J, Felix G, Simon R et al (2020) The CEP5 peptide promotes abiotic stress tolerance, as revealed by quantitative proteomics, and attenuates the AUX/IAA equilibrium in Arabidopsis. Mol Cell Proteom 19:1248–1262. https://doi.org/10.1074/mcp.ra119.001826
Sonah H, Zhang X, Deshmukh RK, Borhan MH, Fernando WG, Bélanger RR (2016) Comparative transcriptomic analysis of virulence factors in Leptosphaeria maculans during compatible and incompatible interactions with canola. Front Plant Sci 7:1784
Song Y, You J, Xiong L (2009) Characterization of OsIAA1 gene, a member of rice Aux/IAA family involved in auxin and brassinosteroid hormone responses and plant morphogenesis. Plant Mol Biol 70:297–309. https://doi.org/10.1007/S11103-009-9474-1
Song W, Liu L, Wang J, Wu Z, Zhang H, Tang J, Lin G, Wang Y, Wen X, Li W, Han Z, Guo H, Chai J (2016) Signature motif-guided identification of receptors for peptide hormones essential for root meristem growth. Cell Res 26:674–685
Spincemaille P, Chandhok G, Newcomb B, Verbeek J, Vriens K, Zibert A, Schmidt H, Hannun YA, van Pelt J, Cassiman D, Cammue BP, Thevissen K (2014a) The plant decapeptide OSIP108 prevents copper-induced apoptosis in yeast and human cells. Biochim Biophys Acta 1843:1207–1215
Spincemaille P, Pham DH, Chandhok G, Verbeek J, Zibert A, Libbrecht L, Schmidt H, Esguerra CV, de Witte PAM, Cammue BPA, Cassiman D, Thevissen K (2014b) The plant decapeptide OSIP108 prevents copper-induced toxicity in various models for Wilson disease. Toxicol Appl Pharmacol 280:345–351
Sprunck S, Hackenberg T, Englhart M, Vogler F (2014) Same same but different: sperm-activating EC1 and ECA1 gametogenesis-related family proteins. Biochem Soc Trans 42(2):401–407. https://doi.org/10.1042/BST20140039
Srivastava R, Liu JX, Guo H, Yin Y, Howell SH (2009) Regulation and processing of a plant peptide hormone, AtRALF23, in Arabidopsis. Plant J 59:930–939
Stahl Y, Simon R (2013) Gated communities: apoplastic and symplastic signals converge at plasmodesmata to control cell fates. Jour Exp Botany 64(17):5237–5241
Stegmann M, Monaghan J, Smakowska-Luzan E, Rovenich H, Lehner A, Holton N, Belkhadir Y, Zipfel C (2017) The receptor kinase FER is a RALF-regulated scaffold controlling plant immune signaling. Science 355:287–289
Stenvik GE, Butenko MA, Urbanowicz BR, Rose JK, Aalen RB (2006) Overexpression of INFLORESCENCE DEFICIENT IN ABSCISSION activates cell separation in vestigial abscission zones in Arabidopsis. Plant Cell 18:1467–1476
Stock AM, Robinson VL, Goudreau PN (2000) Two-component signal transduction. Annu Rev Biochem 69:183–215. https://doi.org/10.1146/annurev.biochem.69.1.183
Stuhrwohldt N, Dahlke RI, Steffens B, Johnson A, Sauter M (2011) Phytosulfokine-alpha controls hypocotyl length and cell expansion in Arabidopsis thaliana through phytosulfokine receptor 1. PLoS ONE 6:e21054
Stuhrwohldt N, Dahlke RI, Kutschmar A, Peng X, Sun MX, Sauter M (2015) Phytosulfokine peptide signaling controls pollen tube growth and funicular pollen tube guidance in Arabidopsis thaliana. Physiol Plant 153:643–653
Stuhrwohldt N, Hohl M, Schardon K, Stintzi A, € Schaller A, (2018) Post-translational maturation of IDA, a peptide signal controlling floral organ abscission in Arabidopsis. Communicative & Integrative Biology 11:e1395119
Stührwohldt N, Bühler E, Sauter M, Schaller A (2021) Phytosulfokine (PSK) precursor processing by subtilase SBT3.8 and PSK signaling improve drought stress tolerance in Arabidopsis. J Exp Bot 72:3427–3440
Sugano SS, Shimada T, Imai Y, Okawa K, Tamai A, Mori M, Hara-Nishimura I (2010) Stomagen positively regulates stomatal density in Arabidopsis. Nature 463:241–244
Sui Z, Wang T, Li H, Zhang M et al (2016) Overexpression of peptide encoding OsCEP6.1 results in pleiotropic effects on growth in rice (O. sativa). Front Plant Sci 7:228
Sun L, Feraru E, Feraru MI, Waidmann S, Wang W, Passaia G, Wang ZY, Wabnik K, Kleine-Vehn J (2020) PIN-LIKES coordinate brassinosteroid signaling with nuclear auxin input in arabidopsis thaliana. Curr Biol 30:1579-1588.e6. https://doi.org/10.1016/J.CUB.2020.02.002
Tabata R, Sumida K, Yoshii T, Ohyama K, Shinohara H, Matsubayashi Y (2014) Perception of root-derived peptides by shoot LRRRKs mediates systemic N-demand signaling. Science 346:343–346
Takahashi T, Shibuya H, Ishikawa A (2016) ERECTA contributes to non-host resistance to Magnaporthe oryzae in Arabidopsis. Biosci Biotechno Biochem 80:1390–1392
Takahashi F, Kuromori T, Sato H, Shinozaki K (2018a) Regulatory gene networks in drought stress responses and resistance in plants. Adv Exp Med Biol 1081:189–214
Takahashi F, Suzuki T, Osakabe Y, Betsuyaku S, Kondo Y, Dohmae N, Fukuda H, Yamaguchi-Shinozaki K, Shinozaki K (2018b) A small peptide modulates stomatal control via abscisic acid in long-distance signaling. Nature 556(7700):235–238
Takahashi F, Suzuki T, Osakabe Y, Betsuyaku S, Kondo Y, Dohmae N, Fukuda H, Yamaguchi-Shinozaki K, Shinozaki K (2018c) A small peptide modulates stomatal control via abscisic acid in long-distance signaling. Nature 556:235–238
Takayama S, Shiba H, Iwano M, Shimosato H, Che FS, Kai N, Watanabe M, Suzuki G, Hinata K, Isogai A (2000) The pollen determinant of self-incompatibility in Brassica campestris. Proc Natl Acad Sci USA 97:1920–1925
Takayama S, Shimosato H, Shiba H, Funato M, Che FS, Watanabe M, Isogai A (2001) Direct ligand- receptor complex interaction controls Brassica self-incompatibility. Nature 413(6855):534–538
Tavormina P, Coninck B, de Nikonorova N, Smet I, de Cammue BPA (2015) The plant peptidome: an expanding repertoire of structural features and biological functions. Plant Cell 27:2095–2118
Terras F, Schoofs H, Thevissen K, Osborn RW, Vanderleyden J, Cammue B, Broekaert WF (1993) Synergistic enhancement of the antifungal activity of wheat and barley thionins by radish and oilseed rape 2S albumins and by barley trypsin inhibitors. Plant Physiol 103:1311–1319
Terras FR, Eggermont K, Kovaleva V, Raikhel NV, Osborn RW, Kester A, Rees SB, Vanderleyden J, Cammue BP, Broekaert WF (1995) Small cysteine-rich antifungal proteins from radish: their role in host defense. Plant Cell 7:573–588
Thevissen K, Ferket KK, Francois IE, Cammue B (2003) Interactions of antifungal plant defensins with fungal membrane components. Peptides 24:1705–1712
Tian Q, Reed JW (1999) Control of auxin-regulated root development by the Arabidopsis thaliana SHY2/IAA3 gene. Development 126:711–721. https://doi.org/10.1242/DEV.126.4.711
Tintor N, Ross A, Kanehara K, Yamada K, Fan L, Kemmerling B, Nurnberger T, Tsuda K, Saijo Y (2013) Layered pattern receptor signaling via ethylene and endogenous elicitor peptides during Arabidopsis immunity to bacterial infection. PNAS USA 110(15):6211–6216
Tiwari SB, Hagen G, Guilfoyle TJ (2004) Aux/IAA proteins contain a potent transcriptional repression domain. Plant Cell 16:533. https://doi.org/10.1105/TPC.017384
Tör M, Lotze MT, Holton N (2009) Receptor-mediated signaling in plants: Molecular patterns and programmes. J Exp Bot 60(13):3645–3654
Trihemasava K, Chakraborty S, Blackburn K, Xu G (2020) Expression, purification, and phylogenetic analysis of MDIS1-INTERACTING RECEPTOR-LIKE KINASE1 (MIK1). Protein J 39:461. https://doi.org/10.1007/S10930-020-09926-9
Turra D, Segorbe D, Di Pietro A (2014) Protein kinases in plant pathogenic fungi: conserved regulators of infection. Annu Rev Phytopathol 52:267–288
Urao T, Yamaguchi-Shinozaki K, Shinozaki K (2000) Two-component systems in plant signal transduction. Trends Plant Sci 5(2):67–74. https://doi.org/10.1016/s1360-1385(99)01542-3
Van Der Weerden NL, Bleackley MR, Anderson MA (2013) Properties and mechanisms of action of naturally occurring antifungal peptides. Cell Mol Life Sci 70:3545–3570
van Loon LC, Rep M, Pieterse CMJ (2006) Significance of inducible defense-related proteins in infected plants. Annu Rev Phytopathol 44:135–162
Van Norman JM, Breakfield NW, Benfey PN (2011) Intercellular communication during plant development. Plant Cell 23:855–864
Vanneste S, Friml J (2009) Auxin: A trigger for change in plant development. Cell 136:1005–1016
Vanoosthuyse V, Miege C, Dumas C, Cock JM (2001) Two large Arabidopsis thaliana gene families are homologous to the Brassica gene superfamily that encodes pollen coat proteins and the male component of the self-incompatibility response. Plant Mol Biol 46:17–34
Velasquez SM, Ricardi MM, Poulsen CP, Oikawa A, Dilokpimol A, Halim A, Mangano S, Denita Juarez SP, Marzol E, Salgado Salter JD, Dorosz JG, Borassi C, Moller SR, Buono R, Ohsawa Y, Matsuoka K, Otegui MS, Scheller HV, Geshi N, Petersen BL, Iusem ND, Estevez JM (2015) Complex regulation of prolyl-4-hydroxylases impacts root hair expansion. Mol Plant 8:734–746
Ventimilla D, Velázquez K, Ruiz-Ruiz S et al (2021) IDA (INFLORESCENCE DEFICIENT IN ABSCISSION)-like peptides and HAE (HAESA)-like receptors regulate corolla abscission in Nicotiana benthamiana flowers. BMC Plant Biol 21:226. https://doi.org/10.1186/s12870-021-02994-8
Vie AK, Najafi J (2015) The IDA/IDA-LIKE and PIP/PIP-LIKE gene families in Arabidopsis: phylogenetic relationship, expression patterns, and transcriptional effect of the PIPL3 peptide. J Exp Bot 66(17):5351–5365
Vriens K, Cammue BP, Thevissen K (2014) Antifungal plant defensins: mechanisms of action and production. Molecules 19:12280–12303
Wang YH, Irving HR (2011) Developing a model of plant hormone interactions. Plant Signal Behav 6:494–500
Wang L, Wang Z, Xu YY, Joo SH, Kim SK, Xue Z, Xu Z, Wang Z, Chong K (2009) OsGSR1 is involved in crosstalk between gibberellins and brassinosteroids in rice. Plant J 57:498–510
Wang YH, Gehring C, Irving HR (2011) Plant natriuretic peptides are apoplastic and paracrine stress response molecules. Plant Cell Physiol 52:837–850
Wang J, Li H, Han Z, Zhang H, Wang T, Lin G, Chang J, Yang W, Chai J (2015) Allosteric receptor activation by the plant peptide hormone phytosulfokine. Nature 525:265–268
Waseem M, Ahmad F, Habib S (2018) Li Z (2018) Genome-wide identification of the auxin/indole-3-acetic acid (Aux/IAA) gene family in pepper, its characterization, and comprehensive expression profiling under environmental and phytohormones stress. Sci Rep 8:1–12. https://doi.org/10.1038/s41598-018-30468-9
Weidmann J, Craik DJ (2016) Discovery, structure, function, and applications of cyclotides: circular proteins from plants. J Exp Bot 124:215–227
Wen J, Lease KA, Walker JC (2004) DVL, a novel class of small polypeptides: overexpression alters Arabidopsis development. Plant J 37:668–677
Wheeler JI, Irving HR (2012) Plant peptide signaling: An evolutionary adaptation. In: Irving HR, Gehring C (eds) Plant signaling peptides. Springer-Verlag, Heidelberg, Berlin, pp 1–19
Whitford R, Fernandez A, De Groodt R, Ortega E, Hilson P (2008) Plant CLE peptides from two distinct functional classes synergistically induce division of vascular cells. Proc Natl Acad Sci USA 105:18625–18630
Whitford R, Fernandez A, Tejos R, Perez AC, Kleine-Vehn J, Vanneste S, Drozdzecki A, Leitner J, Abas L, Aerts M, Hoogewijs K, Baster P, de Groodt R, Lin YC, Storme V et al (2012) GOLVEN secretory peptides regulate auxin carrier turnover during plant gravitropic responses. Dev Cell 22:678–685
Wilmes M, Cammue BPA, Sahl HG, Thevissen K (2011) Antibiotic activities of host defense peptides: more to it than lipid bilayer perturbation. Nat Prod Rep 28:1350–1358
Winter N, Kragler F (2018) Conceptual and methodological considerations on mRNA and proteins as intercellular and long-distance signals. Plant Cell Physiol 59:1700–1713
Woriedh M, Merkl R, Dresselhaus T (2015) Maize EMBRYO SAC family peptides interact differentially with pollen tubes and fungal cells. J Exp Bot 66(17):5205–5216. https://doi.org/10.1093/jxb/erv268
Wrzaczek M, Brosché M, Kollist H, Kangasjärvi J (2009) Arabidopsis GRI is involved in the regulation of cell death induced by extracellular ROS. Proc Natl Acad Sci USA 106(13):5412–5417. https://doi.org/10.1073/pnas.0808980106
Wrzaczek M, Vainonen JP, Stael S, Tsiatsiani L, Help-Rinta-Rahko H, Gauthier A, Kaufholdt D, Bollhöner B et al (2015) GRIM REAPER peptide binds to receptor kinase PRK5 to trigger cell death in Arabidopsis. EMBO J 34(1):55–66. https://doi.org/10.15252/embj.20148858
Wu J, Kurten EL, Monshausen G, Hummel GM, Gilroy S, Baldwin IT (2007) NaRALF, a peptide signal essential for the regulation of root hair tip apoplastic pH in Nicotiana attenuata, is required for root hair development and plant growth in native soils. Plant J 52:877–890
Xu JR, Hamer JE (1996) MAP kinase and cAMP signaling regulate infection structure formation and pathogenic growth in the rice blast fungus Magnaporthe grisea. Genes Dev 10:2696–2706
Yadav RK, Perales M, Gruel J, Girke T, Jönsson H, Reddy GV (2011) WUSCHEL protein movement mediates stem cell homeostasis in the Arabidopsis shoot apex. Genes Development 25:2025–2030
Yamaguchi Y, Huffaker A (2011) Endogenous peptide elicitors in higher plants. Curr Opin Plant Biol 14:351–357
Yamaguchi Y, Pearce G, Ryan CA (2006) The cell surface leucine-rich repeat receptor for AtPep1, an endogenous peptide elicitor in Arabidopsis, is functional in transgenic tobacco cells. Proc Natl Acad Sci USA 103:10104–10109
Yamaguchi YL, Ishida T, Sawa S (2016) CLE peptides and their signaling pathways in plant development. J Exp Bot 67:4813–4826. https://doi.org/10.1093/jxb/erw208
Yang SL, Xie LF, Mao HZ, Puah CS, Yang WC, Jiang L, Sundaresan V, Ye D (2003) Tapetum determinant1 is required for cell specialization in the Arabidopsis anther. Plant Cell 15(12):2792–2804. https://doi.org/10.1105/tpc.016618
Yang X, Lee S, So J, Dharmasiri S, Dharmasiri N, Ge L, Jensen C, Hangarter R, Hobbie L, Estelle M (2004) The IAA1 protein is encoded by AXR5 and is a substrate of SCFTIR1. Plant J 40:772–782
Ying P, Li C, Liu X et al (2016) Identification and molecular characterization of an IDA-like gene from litchi, LcIDL1, whose ectopic expression promotes floral organ abscission in Arabidopsis. Sci Rep 6:37135
Yoshida T, Fernie AR (2018) Remote control of transpiration via ABA. Trends Plant Sci 23(9):755–758
Yu Y, Assmann SM (2018) Inter-relationships between the heterotrimeric Gbeta subunit AGB1, the receptor-like kinase FERONIA, and RALF1 in salinity response. Plant Cell Environ 41:2475–2489
Yu Y, Chakravorty D, Assmann SM (2018) The G protein β subunit, AGB1, interacts with FERONIA in RALF1-regulated stomatal movement. Plant Physiol 176:2426–2440. https://doi.org/10.1104/pp.17.01277
Yu LL, Liu YM, Zeng S, Yan JH, Wang ET, Luo L (2019) Expression of a novel PSK-encoding gene from soybean improves seed growth and yield in transgenic plants. Planta 249(4):1239–1250
Yuasa K, Toyooka K, Fukuda H, Matsuoka K (2005) Membrane-anchored prolyl hydroxylase with an export signal from the endoplasmic reticulum. Plant J Cell Mol Biol 41:81–94
Zeng Y, Tang Y, Shen S, Zhang M, Chen L, Ye D, Zhang X (2022) Plant-specific small peptide AtZSP1 interacts with ROCK1 to regulate organ size in Arabidopsis. New Phytol 234:1696–1713
Zhang SC, Wang XJ (2008) Expression pattern of GASA, downstream genes of DELLA, in Arabidopsis. Chinese Sci Bull 53:3839–3846
Zhang H, Hu Z, Lei C, Zheng C, Wang J, Shao S, Li X, Xia X, Cai X, Zhou J, Zhou Y, Yu J, Foyer CH, Shi K (2018) A plant phytosulfokine peptide initiates Auxin-dependent immunity through cytosolic Ca2 + signaling in tomato. Plant Cell 30:652–667
Zhang L, Shi X, Zhang Y, Wang J, Yang J, Ishida T, Jiang W, Han X, Kang J, Wang X, Pan L, Lv S, Cao B, Zhang Y, Wu J, Han H, Hu Z, Cui L, Sawa S, He J, Wang G (2019) CLE9 peptide induced stomatal closure is mediated by abscisic acid, hydrogen peroxide, and nitric oxide in Arabidopsis thaliana. Plant Cell Environ 42:1033–1044
Zhao C, Zayed O, Yu Z, Jiang W, Zhu P, Hsu CC, Zhang L, Tao WA, Lozano-Durán R, Zhu JK (2018a) Leucine-rich repeat extensin proteins regulate plant salt tolerance in Arabidopsis. Proc Natl Acad Sci USA 115(51):13123–13128. https://doi.org/10.1073/pnas.1816991115
Zhao C, Zayed O, Yu Z, Jiang W, Zhu P, Hsu CC, Zhang L, Tao WA, Lozano-Durán R, Zhu JK (2018b) Leucine-rich repeat extensin proteins regulate plant salt tolerance in Arabidopsis. Proc Natl Acad Sci USA 115:13123–13128
Zhao H, Duan KX, Ma B, Yin CC, Hu Y, Tao JJ, Huang YH, Cao WQ, Chen H, Yang C, Zhang ZG (2020b) Histidine kinase MHZ1/OsHK1 interacts with ethylene receptors to regulate root growth in rice. Nat Commun 11(1):1–13
Zhao C, Jiang W, Zayed O, Liu X, Tang K, Nie W, Li Y, Xie S, Li Y, Long T, Liu L, Zhu Y, Zhao Y, Zhu JK (2020a) The LRXs-RALFs-FER module controls plant growth and salt stress responses by modulating multiple plant hormones. Natl Sci Rev 8: nwaa149
Ziemann S, van der Linde K, Lahrmann U, Acar B, Kaschani F, Colby T, Kaiser M, Ding Y, Schmelz E, Huffaker A, Holton N, Zipfel C, Doehlemann G (2018) An apoplastic peptide activates salicylic acid signaling in maize. Nat Plants 4(3):172–180
Zimmermann R, Sakai H, Hochholdinger F (2010) The Gibberellic Acid Stimulated-Like gene family in maize and its role in lateral root development. Plant Physiol 152(1):356–365. https://doi.org/10.1104/pp.109.149054
Zipfel C, Felix G (2005) Plants and animals: a different taste for microbes? Curr Opin Plant Biol 8:353–360
Acknowledgement
Funding to pay the Open Access publication charges for this article was provided by the Royal Holloway University of London, U.K
Author information
Authors and Affiliations
Contributions
All authors have equally contributed to this article.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests relevant to the content in this article.Acknowledgement: Funding to pay the Open Access publication charges for this article was provided by the Royal Holloway University of London, U.K
Additional information
Handling Editor: Sudhir K. Sopory
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pandita, D., Bhat, J.A., Wani, S.H. et al. Mobile Signaling Peptides: Secret Molecular Messengers with a Mighty Role in Plant Life. J Plant Growth Regul 42, 6801–6834 (2023). https://doi.org/10.1007/s00344-023-11069-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-023-11069-x