Abstract
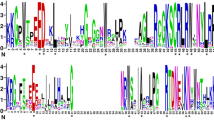
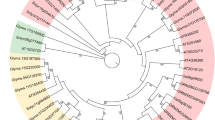
Short internodes-related sequence (SRS) family genes are important regulators in multiple growth and development programs. However, the SRS gene family in tomato (Solanum lycopersicum), an important model plant for investigating fleshy fruits, has not been whole-genome identified and analyzed, and our knowledge about their functions in abiotic stresses is still limited. Here, eight tomato SRS proteins were identified and assigned to three groups based on a phylogenetic tree. Gene structure and conserved protein motifs results exhibited that the same subfamily had similar introns/exon modes and motif compositions. There was a total of 33 cis-acting elements related to light response, plant hormone response, tissue-specific expression, and stress response found in the 2000 bp promoter regions of tomato SRS genes. Protein–protein interaction results showed that seven SlSRS proteins formed a complex protein interaction network. The results of expression patterns suggested that tomato SRS genes showed distinct differences in various tissues and organs. Furthermore, their expression patterns in response to diverse hormone and abiotic stress treatments were also studied by RT-qPCR technology. These results will provide meaningful information for further study of the biological functions of SRS family members and directing the tomato genetic breeding in the future.










Similar content being viewed by others
References
Bailey TL, Johnson J, Grant CE, Noble WS (2015) The MEME Suite. Nucl Acids Res 43(W1):W39–W49. https://doi.org/10.1093/nar/gkv416
Bari R, Jones JD (2009) Role of plant hormones in plant defence responses. Plant Mol Biol 69(4):473–488. https://doi.org/10.1007/s11103-008-9435-0
Baylis T, Cierlik I, Sundberg E, Mattsson J (2013) SHORT INTERNODES/STYLISH genes, regulators of auxin biosynthesis, are involved in leaf vein development in Arabidopsis thaliana. New Phytol 197(3):737–750. https://doi.org/10.1111/nph.12084
Büyük İ, Okay A, Aras S (2022) Identification and characterization of SRS genes in Phaseolus vulgaris genome and their responses under salt stress. Biochem Genet 60(2):482–503. https://doi.org/10.1007/s10528-021-10108-0
Chen C, Chen H, Zhang Y, Thomas HR, Frank MH, He Y, Xia R (2020) TBtools: an integrative toolkit developed for interactive analyses of big biological data. Mol Plant 13(8):1194–1202. https://doi.org/10.1016/j.molp.2020.06.009
Chou KC, Shen HB (2010) Plant-mPLoc: a top-down strategy to augment the power for predicting plant protein subcellular localization. PLoS ONE 5(6): e11335. https://doi.org/10.1371/journal.pone.0011335
Duan E, Wang Y, Li X, Lin Q, Zhang T, Wang Y, Zhou C, Zhang H, Jiang L, Wang J, Lei C, Zhang X, Guo X, Wang H, Wan J (2019) Osshi1 regulates plant architecture through modulating the transcriptional activity of ipa1 in rice. Plant Cell 31(5):1026–1042. https://doi.org/10.1105/tpc.19.00023
Expósito-Rodríguez M, Borges AA, Borges-Pérez A, Pérez JA (2008) Selection of internal control genes for quantitative real-time RT-PCR studies during tomato development process. BMC Plant Biol 8(1): 131. https://doi.org/10.1186/1471-2229-8-131
Finn RD, Bateman A, Clements J, Coggill P, Eberhardt RY, Eddy SR, Heger A, Hetherington K, Holm L, Mistry J, Sonnhammer ELL, Tate J, Punta M (2014) Pfam: the protein families database. Nucl Acids Res 42(D1):D222–D230. https://doi.org/10.1093/nar/gkt1223
Fridborg A, Kuusk S, Moritz T, Sundberg E (1999) The Arabidopsis dwarf mutant shi exhibits reduced gibberellin responses conferred by overexpression of a new putative zinc finger protein. Plant Cell 11(6):1019–1031. https://doi.org/10.1105/tpc.11.6.1019
Fridborg I, Kuusk S, Robertson M, Sundberg E (2001) The Arabidopsis protein SHI represses gibberellin responses in Arabidopsis and barley. Plant Physiol 127(3):937–948. https://doi.org/10.1104/pp.010388
Gomariz-Fernández A, Sánchez-Gerschon V, Fourquin C, Ferrándiz C (2017) The role of SHI/STY/SRS genes in organ growth and carpel development is conserved in the distant eudicot species Arabidopsis thaliana and Nicotiana benthamiana. Front Plant Sci 8. https://doi.org/10.3389/fpls.2017.00814
Greb T, Clarenz O, Schäfer E, Müller D, Herrero R, Schmitz G, Theres K (2003) Molecular analysis of the LATERAL SUPPRESSOR gene in Arabidopsis reveals a conserved control mechanism for axillary meristem formation. Gene Dev 17(9):1175–1187. https://doi.org/10.1101/gad.260703
Hajheidari M, Huang S-SC (2022) Elucidating the biology of transcription factor–DNA interaction for accurate identification of cis-regulatory elements. Curr Opin Plant Biol 68. https://doi.org/10.1016/j.pbi.2022.102232
He B, Shi P, Lv Y, Gao Z, Chen G (2020) Gene coexpression network analysis reveals the role of SRS genes in senescence leaf of maize (Zea mays L.). J Genet 99(1). https://doi.org/10.1007/s12041-019-1162-6
Hossain MS, Shrestha A, Zhong S, Miri M, Austin RS, Sato S, Ross L, Huebert T, Tromas A, Torres-Jerez I, Tang Y, Udvardi M, Murray JD, Szczyglowski K (2016) Lotus japonicus NF-YA1 plays an essential role during nodule differentiation and targets members of the SHI/STY gene family. Mol Plant Microbe Interact 29(12):950–964. https://doi.org/10.1094/MPMI-10-16-0206-R
Huang W, Xian Z, Kang X, Tang N, Li Z (2015) Genome-wide identification, phylogeny and expression analysis of GRAS gene family in tomato. BMC Plant Biol 15:209. https://doi.org/10.1186/s12870-015-0590-6
Jin J, Tian F, Yang DC, Meng YQ, Kong L, Luo J, Gao G (2017) PlantTFDB 4.0: toward a central hub for transcription factors and regulatory interactions in plants. Nucl Acids Res 45(D1): D1040–D1045. https://doi.org/10.1093/nar/gkw982
Kim SG, Lee S, Kim YS, Yun DJ, Woo JC, Park CM (2010) Activation tagging of an Arabidopsis SHI-RELATED SEQUENCE gene produces abnormal anther dehiscence and floral development. Plant Mol Biol 74(4):337–351. https://doi.org/10.1007/s11103-010-9677-5
Kuusk S, Sohlberg JJ, Long JA, Fridborg I, Sundberg E (2002) STY1 and STY2 promote the formation of apical tissues during Arabidopsis gynoecium development. Development 129(20):4707–4717. https://doi.org/10.1242/dev.129.20.4707
Kuusk S, Sohlberg JJ, Magnus Eklund D, Sundberg E (2006) Functionally redundant SHI family genes regulate Arabidopsis gynoecium development in a dose-dependent manner. Plant J 47(1):99–111. https://doi.org/10.1111/j.1365-313X.2006.02774.x
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23(21): 2947–2948. https://doi.org/10.1093/bioinformatics/btm404
Laxa M, Müller K, Lange N, Doering L, Pruscha JT, Peterhänsel C (2016) The 5ʹUTR intron of Arabidopsis GGT1 aminotransferase enhances promoter activity by recruiting RNA polymerase II. Plant Physiol 172(1):313–327. https://doi.org/10.1104/pp.16.00881
Lescot M, Déhais P, Thijs G, Marchal K, Moreau Y, Van De Peer Y, Rouzé P, Rombauts S (2002) PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucl Acids Res 30(1):325–327. https://doi.org/10.1093/nar/30.1.325
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Lu S, Wang J, Chitsaz F, Derbyshire MK, Geer RC, Gonzales NR, Gwadz M, Hurwitz DI, Marchler GH, Song JS, Thanki N, Yamashita RA, Yang M, Zhang D, Zheng C, Lanczycki CJ, Marchler-Bauer A (2020) CDD/SPARCLE: the conserved domain database in 2020. Nucleic Acids Res 48(D1):D265-d268. https://doi.org/10.1093/nar/gkz991
Magnus Eklund D, Ståldal V, Valsecchi I, Cierlik I, Eriksson C, Hiratsu K, Ohme-Takagi M, Sundström JF, Thelander M, Ezcurra I, Sundberg E (2010) The Arabidopsis thaliana STYLISH1 protein acts as a transcriptional activator regulating auxin biosynthesis. Plant Cell 22(2):349–363. https://doi.org/10.1105/tpc.108.064816
Marchler-Bauer A, Lu S, Anderson JB, Chitsaz F, Derbyshire MK, DeWeese-Scott C, Fong JH, Geer LY, Geer RC, Gonzales NR, Gwadz M, Hurwitz DI, Jackson JD, Ke Z, Lanczycki CJ, Lu F, Marchler GH, Mullokandov M, Omelchenko MV, Robertson CL, Song JS, Thanki N, Yamashita RA, Zhang D, Zhang N, Zheng C, Bryant SH (2011) CDD: a conserved domain database for the functional annotation of proteins. Nucl Acids Res 39(SUPPL 1):D225–D229. https://doi.org/10.1093/nar/gkq1189
Nicot N, Hausman J-F, Hoffmann L, Evers D (2005) Housekeeping gene selection for real-time RT-PCR normalization in potato during biotic and abiotic stress. J Exp Bot 56(421):2907–2914. https://doi.org/10.1093/jxb/eri285
Riechmann JL, Heard J, Martin G, Reuber L, Jiang CZ, Keddie J, Adam L, Pineda O, Ratcliffe OJ, Samaha RR, Creelman R, Pilgrim M, Broun P, Zhang JZ, Ghandehari D, Sherman BK, Yu GL (2000) Arabidopsis transcription factors: genome-wide comparative analysis among eukaryotes. Science 290(5499):2105–2110. https://doi.org/10.1126/science.290.5499.2105
Salinas M, Xing S, Höhmann S, Berndtgen R, Huijser P (2011) Genomic organization, phylogenetic comparison and differential expression of the SBP-box family of transcription factors in tomato. Planta 235(6):1171–1184. https://doi.org/10.1007/s00425-011-1565-y
Shaul O (2017) How introns enhance gene expression. Int J Biochem Cell Biol 91(Pt B):145–155. https://doi.org/10.1016/j.biocel.2017.06.016
Singh S, Yadav S, Singh A, Mahima M, Singh A, Gautam V, Sarkar AK (2020) Auxin signaling modulates LATERAL ROOT PRIMORDIUM1 (LRP1) expression during lateral root development in Arabidopsis. Plant J 101(1):87–100. https://doi.org/10.1111/tpj.14520
Smith DL, Fedoroff NV (1995) LRP1, a gene expressed in lateral and adventitious root primordia of Arabidopsis. Plant Cell 7(6):735–745
Sohlberg JJ, Myrenås M, Kuusk S, Lagercrantz U, Kowalczyk M, Sandberg G, Sundberg E (2006) STY1 regulates auxin homeostasis and affects apical-basal patterning of the Arabidopsis gynoecium. Plant J 47(1):112–123. https://doi.org/10.1111/j.1365-313X.2006.02775.x
Szklarczyk D, Franceschini A, Wyder S, Forslund K, Heller D, Huerta-Cepas J, Simonovic M, Roth A, Santos A, Tsafou KP, Kuhn M, Bork P, Jensen LJ, Von Mering C (2015) STRING v10: protein–protein interaction networks, integrated over the tree of life. Nucl Acids Res 43(D1):D447–D452. https://doi.org/10.1093/nar/gku1003
Tamura K, Stecher G, Kumar S (2021) MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 38(7):3022–3027. https://doi.org/10.1093/molbev/msab120
Waadt R, Seller CA, Hsu PK, Takahashi Y, Munemasa S, Schroeder JI (2022) Plant hormone regulation of abiotic stress responses. Nat Rev Mol Cell Biol 23(10):680–694. https://doi.org/10.1038/s41580-022-00479-6
Wallace EWJ, Beggs JD (2017) Extremely fast and incredibly close: cotranscriptional splicing in budding yeast. RNA 23(5):601–610. https://doi.org/10.1261/rna.060830.117
Wang Y, Zhang J, Hu Z, Guo X, Tian S, Chen G (2019) Genome-wide analysis of the MADS-box transcription factor family in Solanum lycopersicum. Int J Mol Sci 20(12): 2961. https://doi.org/10.3390/ijms20122961
Wilkins MR, Gasteiger E, Bairoch A, Sanchez JC, Williams KL, Appel RD, Hochstrasser DF (1999) Protein identification and analysis tools in the ExPASy server. Methods Mol Biol 112:531–552
Wu J, Wang F, Cheng L, Kong F, Peng Z, Liu S, Yu X, Lu G (2011) Identification, isolation and expression analysis of auxin response factor (ARF) genes in Solanum lycopersicum. Plant Cell Rep 30(11):2059–2073. https://doi.org/10.1007/s00299-011-1113-z
Yang J, Xu P, Yu D (2020) Genome-wide identification and characterization of the SHI-related sequence gene family in rice. Evol Bioinform 16: 1176934320941495. https://doi.org/10.1177/1176934320941495
Yang Y, Qi L, Nian L, Zhu X, Yi X, Jiyu Z, Qiu J (2021) Genome-wide identification and expression analysis of the SRS gene family in Medicago sativa. DNA Cell Biol 40(12):1539–1553. https://doi.org/10.1089/dna.2021.0462
Yuan TT, Xu HH, Li J, Lu YT (2020) Auxin abolishes SHI-RELATED SEQUENCE5-mediated inhibition of lateral root development in Arabidopsis. New Phytol 225(1):297–309. https://doi.org/10.1111/nph.16115
Yuan TT, Xu HH, Zhang Q, Zhang LY, Lu YT (2018) The cop1 target shi-related sequence5 directly activates photomorphogenesis-promoting genes. Plant Cell 30(10):2368–2382. https://doi.org/10.1105/tpc.18.00455
Zhang H, Gao S, Lercher MJ, Hu S, Chen WH (2012) EvolView, an online tool for visualizing, annotating and managing phylogenetic trees. Nucl Acids Res 40(W1):W569–W572. https://doi.org/10.1093/nar/gks576
Zhang Y, Behrens IV, Zimmermann R, Ludwig Y, Hey S, Hochholdinger F (2015) LATERAL ROOT PRIMORDIA 1 of maize acts as a transcriptional activator in auxin signalling downstream of the Aux/IAA gene rootless with undetectable meristem 1. J Exp Bot 66(13):3855–3863. https://doi.org/10.1093/jxb/erv187
Zhang Z, Chen X, Guan X, Liu Y, Chen H, Wang T, Mouekouba LD, Li J, Wang A (2014) A genome-wide survey of homeodomain-leucine zipper genes and analysis of cold-responsive HD-Zip I members’ expression in tomato. Biosci Biotechnol Biochem 78(8):1337–1349. https://doi.org/10.1080/09168451.2014.923292
Zhao SP, Song XY, Guo LL, Zhang XZ, Zheng WJ (2020) Genome-wide analysis of the SHI-related sequence family and functional identification of GmSRS18 involving in drought and salt stresses in soybean. Int J Mol Sci 21(5): 1810. https://doi.org/10.3390/ijms21051810
Zhao W, Liu YW, Zhou JM, Zhao SP, Zhang XH, Min DH (2016) Genome-wide analysis of the lectin receptor-like kinase family in foxtail millet (Setaria italica L.). Plant Cell Tiss Org 127(2): 335–346. https://doi.org/10.1007/s11240-016-1053-y
Zhu X, Wang B, Wang X, Wei X (2022) Genome-wide identification, structural analysis and expression profiles of short internodes related sequence gene family in quinoa. Front Genet 13. https://doi.org/10.3389/fgene.2022.961925
Acknowledgements
This research work was financially supported by the Natural Science Foundation of Jiangxi Province (Grant no. 20202BABL215009) and National Natural Science Foundation of China (No. 31960605).
Author information
Authors and Affiliations
Contributions
S.Z. designed the experiments and wrote the manuscript. F.L. performed the experiments. B.C. and Z.Z. revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Handling Editor: Ravinder Kumar.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, F., Cui, B., Zhu, Z. et al. Genome-Wide Identification, Structural Analysis, and Expression Patterns of Tomato SRS Gene Family. J Plant Growth Regul 43, 60–75 (2024). https://doi.org/10.1007/s00344-023-11054-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-023-11054-4