Abstract
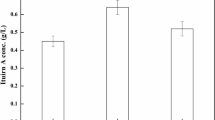
The industrially important organic compound 1,3-propanediol (1,3-PDO) is mainly used as a building block for the production of various polymers. In the present study, response surface methodology protocol was followed to determine and optimize fermentation conditions for the maximum production of 1,3-PDO using marine-derived Klebsiella pneumoniae HSL4. Four nutritional supplements together with three independent culture conditions were optimized as follows: 29.3 g/L glycerol, 8.0 g/L K2 HPO4, 7.6 g/L (NH4)2 SO4, 3.0 g/L KH2 PO4, pH 7.1, cultivation at 35°C for 12 h. Under the optimal conditions, a maximum 1,3-PDO concentration of 14.5 g/L, a productivity of 1.21 g/(L·h) and a conversion of glycerol of 0.49 g/g were obtained. In comparison with the control conditions, fermentation under the optimized conditions achieved an increase of 38.8% in 1,3-PDO concentration, 39.0% in productivity and 25.7% in glycerol conversion in flask. This enhancement trend was further confirmed when the fermentation was conducted in a 5-L fermentor. The optimized fermentation conditions could be an important basis for developing lowcost, large-scale methods for industrial production of 1,3-PDO in the future.
Similar content being viewed by others
References
Biebl H, Menzel K, Zeng A P, Deckwer W D. 1999. Microbial production of 1,3-propanediol. Appl. Microbiol. Biotechnol., 52: 289–297.
Daniel R, Stuertz K, Gottschalk G. 1995. Biochemical and molecular characterization of the oxidative branch of glycerol utilization by Citrobacter freundii. J. Bacteriol., 15: 4 392–4 401.
Gunawan E R, Basri M, Rahman M B D, Salleh A B, Rahman R N Z A. 2005. Study on response surface methodology (RSM) of lipase-catalyzed synthesis of palm-based wax ester. Enzyme Microb. Technol., 37: 739–744.
Hong E, Yoon S, Kim J, Kim E, Kim D, Rhie S, Ryu Y W. 2013. Isolation of microorganisms able to produce 1,3-propanediol and optimization of medium constituents for Klebsiella pneumoniae AJ4. Bioprocess Biosyst. Eng., 6: 835–843.
Manohar B, Divakar S. 2004. Applications of surface plots and statistical designs to select lipase catalysed esterification reactions. Process Biochem., 39: 847–853.
Montgomery D C. 1991. Design and Analysis of Experiments (3nd edn). John Wiley & Sons, New York.
Oh B R, Seo J W, Choi M H, Kim C H. 2008. Optimization of culture conditions for 1,3-Propanediol production from crude glycerol by Klebsiella pneumoniae using Response Surface Methodology. Biotechnol. Bioproc. E., 13: 666–670.
Oh B R, Seo J W, Heo S Y, Hong W K, Luo L H, Kim S H, Park D H, Kim C H. 2012. Optimization of culture conditions for 1,3-Propanediol production from glycerol using a mutant strain of Klebsiella pneumoniae. Appl. Biochem. Biotechnol., 166: 127–137.
Paul G C, Kent C A, Thomas C R. 1992. Quantitative characterization of vacuolization in Penicillium chyrsogenum using automatic image analysis. Trans. I Chem. E., 70: 13–20.
Raynaud C, Sarçabal P, Meynial-Salles I, Croux C, Soucaille P. 2003. Molecular characterization of the 1,3-propanediol (1,3-PDO) operon of Clostridium butyricum. PNAS, 9: 5 010–5 015.
Saxena R K, Anand P, Saran S, Jasmine I S A R. 2009. Microbial production of 1,3-propanediol: Recent developments and emerging opportunities. Biotechnol. Adv., 27: 895–913.
Sim J H, Kamaruddin A H, Long W S, Najafpour G. 2007. Clostridium aceticum-A potential organism in catalyzing carbon monoxide to acetic acid: application of response surface methodologyy. Enzyme Microb. Technol., 140: 1 234–1 243.
Yazdani S S, Gonzalez R. 2007. Anaerobic fermentation of glycerol: a path to economic viability for the biofuels industry. Curr. Opin. Biotechnol., 18: 213–219.
Zeng A P, Hanno B. 2002. Bulk chemicals from biotechnology: the case of 1,3-propanediol production and the new trends. Biochem. Eng. Biot., 74: 239–259.
Zeng A P, Wael S. 2011. Microbial production of diols as platform chemicals: recent progresses. Curr. Opin. Biotechnol., 22: 749–757.
Zhang Y P, Li Y, Du C Y, Liu M, Cao Z A. 2006. Inactivation of aldehyde dehydrogenase: a key factor for engineering 1,3-propanediol production by Klebsiella pneumoniae. Metab. Eng., 8: 578–586.
Zheng Y Y, Cao Y, Fang B S. 2004. Cloning and sequence analysis of the dha T gene of the 1,3-propanediol regulon from Klebsiella pneumoniae. Biotechnol. Lett., 3: 251–255.
Zheng Z M, Hu Q L, Hao J, Xu F, Guo N N, Sun Y, Liu D H. 2008. Statistical optimization of culture conditions for 1,3-propanediol by Klebsiella pneumoniae AC 15 via central composite design. Bioresource Technology, 99: 1 052–1 056.
Zheng Z M, Xu Y Z, Wang T P, Dong C Q, Yang Y P, Liu D H. 2010. Ammonium and phosphate limitation in 1,3-propanediol production by Klebsiella pneumoniae. Biotechnol. Lett., 32: 289–294.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the Scientific Research Project of Marine Public Welfare Industry of China (No. 201205020-4), the Knowledge Innovation Project of Chinese Academy of Sciences (No. KSCX2-EW-G-12B), and the Administration of Ocean and Fisheries of Guangdong Province (No. GD2012-D01-002)
Rights and permissions
About this article
Cite this article
Li, L., Zhou, S., Ji, H. et al. Optimization of fermentation conditions for 1,3-propanediol production by marine Klebsiella pneumonia HSL4 using response surface methodology. Chin. J. Ocean. Limnol. 32, 1036–1045 (2014). https://doi.org/10.1007/s00343-014-3267-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00343-014-3267-3