Abstract
Borate glass samples containing chromium and erbium were prepared. According to the density and molar volume, the Cr2O3-free glass sample had an expanded glass structure. Cr has three distinct absorption bands, and according to the measured optical absorption characteristics, a band was observed at 688 nm due to the 4A2g(F) → 2Eg(G) transition, indicating the presence of Cr6+. Cr3+ was observed in the bands at 446 and 620 nm, which were attributed to 4A2g (F) → 4T2g (F) and 4A2g (F) → 4T1g (F). Using the absorption spectra of the glass samples, the Judd–Ofelt theory was used to calculate the three parameters for glass: Ω2, Ω4, and Ω6. The slow transformation of chromium ions in these glasses from Cr6+ to Cr3+ disturbs the local symmetry and adds coordinated bond defects, which affect the surroundings of Er3+ ions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The world is concentrating on finding ways to conserve energy while lowering pollution. Solid-state lighting is popular because of its eco-friendly attributes, compact size, high efficiency, low-temperature performance, extended lifespan, and low energy consumption. This could help reduce carbon emissions in places where it is utilized. A material with transparency, ease of preparation, and homogeneous structural characterization that emphasizes the amorphous aspect of glass preparation is desirable from the perspective of material manufacturing [1,2,3,4].
Owing to its superior mechanical qualities, low melting point, high thermal and chemical stability, and good solubility of rare earth elements, borate glass is often used to fabricate transparent glasses.
As the T.M. (Transition metal) has several valence states and the d-d transition, while the R.E. (rare earth) has greater emission efficiencies because of its 4f–4f and 4f–5d electronic transitions, both rare earth and transition metal-doped glass are employed to increase the optical and electrical properties [3, 5]. Er exhibits three luminescence transitions in the visible region: violet (2H9/2 → 4I15/2), green (4S3/2 → 4I15/2), and red (4F9/2 → 4I15/2). This makes it a suitable candidate for the rare-earth element category. The transition in near-IR 1.5 µm [6] can be applicable, especially for eye-safe lasers [1, 5], and in mid-IR emission at 2.7 µm (4I11/2 → 4I13/2) in the visible range [7].
Among other transition metals, Cr ions were chosen because of their multiple oxidation states and their ability to behave in small amounts as paramagnetic substances [8]. Cr ions can be found in glass in three different states: Cr3+ with CrO6 structural units functioning as modifiers, Cr5+ and Cr6+ with and structural units acting as glass formers [2, 9,10,11].
The Cr3+ luminescence is a superposition of a broadband and sharp emission created from the absorption transition 4A2 → 4T2 in the visible range [2, 12] when Cr3+ ions occupy low- and high-field sites in glasses [13].
In their investigation of the physical and optical characteristics of soda-lime silicate glass doped with low concentrations of Cr2O3, Meejitpaisan et al. [14] found that the Cr2O3 concentration primarily affected the density and molar volume, while the molar electron polarizability and molar refraction decreased as the refractive index increased. The presence of Cr in the hexavalent phase is shown by the peaks at 330, 350, and 370 nm, and by the minuscule peaks in trivalentat 641, 660, and 688 nm.
Pisarski et al. [15] studied composition dependent lead borate glasses doped with transition metal and rare earth ions using absorption and luminescence spectroscopy, and the presence of trivalent Cr3+, Eu3+, and Dy3+ ions as spectroscopic probe in glass samples. Spectral analysis indicated that Cr3+ ions occupy intermediate field sites; both sites coexist and are emitted from the 4T2 (low-field) and 2E (high-field) states.
Using Cr2O3 as the nucleating agent, Suresh et al. [16] investigated the effects of local structural disorders on the spectroscopic characteristics of multicomponent CaF2–Bi2O3–P2O5–B2O3 glass ceramics and the partial conversion of Cr3+ ions into Cr6+ ions with an increase in the concentration of the nucleating agent. Glass ceramics become more compact or polymerized as the Cr2O3 content increases. BO4 and PO43− structural components alternate in the presence of Cr6+ ions.
Vijayalakshmi et al. [17] investigated different Er3+ doping concentrations in lithium fluoro zinc borate glasses (0.3, 0.5, 1.0, and 1.5 mol%). The intensity parameters were examined using the Judd Ofelt hypothesis. It was noted that the glass produced would be a good option for white-light photonic applications, optical telecommunication windows, and amplifiers.
KNbO3 phosphors that were Er-doped and Er/Cr-co-doped were investigated by Kim et al. [18]. Emissions appeared in both systems at 555 nm and 405 nm excitation. Up-conversion emissions from the 4S3/2 and 4F9/2 states of Er3+ and the 4T2 state of Cr3+ ions were observed in the green, red, and infrared spectra of the co-doped phosphors.
I. Arul Rayappan and K. Marimuthu [19] examined the luminescence spectra and structure of Er3+ doped alkali borate and fluoroborate glasses and found that Er is an ionic species with a surrounding ligand, indicating a higher degree of asymmetry in the ion site with two parameter value.
Under 378 nm excitation, the title glasses showed strong green emission associated with the 4S3/2 → 4I15/2 transition. Sample glass, which has a composition of 49.5 Na2O, 49.5 B2O3, and 1 Er2O3, is suitable for use with green lasers.
The optical absorption, excitation, fluorescence properties, and emission lifetimes of chromium (III) in various glass formers were studied by Lachheb et al. [10]. The weak crystal field strengths were calculated for all glasses. The increased fluorescence intensity of Cr3+ is affected mainly by the low optical basicity, spectral position of the Cr3+ absorption, and emission. The optical absorption spectra showed the existence of Cr in various oxidation stages, depending on the former. The Cr3+ concentration affected the maximum fluorescence emission intensity.
Li2B4O7-doped glasses with Er3+ have also been studied [20]. Conventional glass synthesis was used to create high-quality glass samples from the matching polycrystalline compounds that contained 0.5 and 1.0 mol% Er2O3. The Er3+ luminescence center at the Li sites of the glass network had a local structure. The Judd–Ofelt theory was used to calculate the oscillator strength and experimental oscillator strength of Li2B4O7: Er glass.
The aim of this study was to understand the oxidation state of Cr when replacing boron and the effect of different oxidation states of Cr ions on the emissions from glass samples containing Cr and Er ions.
2 Experimental work
Glass samples with the formula (70–x) B2O3–30Li2O–xCr2O3 doped with 1Er2O3 mol% were made, with x = 0, 0.1, 0.5, and 1 mol%. The mixture of raw materials was taken into a platinum crucible. The samples were maintained at 400 °C for 30 min, followed by an hour at 1000 °C. At normal temperature, the molten substance was poured between the two copper plates and dispersed. The glassy samples were verified by XRD analysis carried out using a Philips Analytical X-ray diffractometer (PW3710). The generator current was 30 mA, the generator tension was 40 kV. 10° was the start angle (2θ) and 70° was the end angle.
To measure the optical transmission spectra, a computerized recording spectrophotometer was used to measure the wavelengths between 190 and 2500 nm (JASCO, V-570). According to the formula ρ (g/cm3) = [a/(a – b)] X (density of the toluene), the density is estimated using the Archimedes technique, where a is the weight of the glass sample in air and b is the weight in toluene.
The room temperature emission spectra recorded by a JASCO FP-8300, Japan spectrofluorometer were produced by a 150 W Xenon arc lamp.
3 Results and discussion
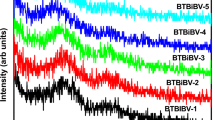
Figure 1 displays the X-ray diffraction patterns of samples examined to identify the type of samples material. In Fig. 1, the amorphous structure is indicated by the absence of sharp peaks.
Changes in geometry, hole focus, and number coordination can have a considerable impact on density, which is a crucial attribute for any alteration induced by the glass structure [8, 21, 22]. Figure 2A and B depict the relationship between the density, molar volume, and Cr2O3 concentration.
An increased density was observed in comparison to Cr2O3 because boron was swapped out for an element with a lower molecular weight [23, 24]. The glass becomes more compact when Cr2O3 is added because the molar volume changes from BO4 with a radius of 0.25oA to BO3 with a radius of 0.15oA [21]. Glass compactness can be assessed using OPD (oxygen packing density) and dB–B (distance separation between B–B) techniques.
According to the OPD values listed in Table 1, the value of [22] OPD changes slightly, even with an increase in oxygen in the samples under study. In addition, for dB–B, the substitution by the oxygen number did not change, resulting in values with a slight change.
An optical absorption analysis can be used to determine the oxidation status of TM [25] and any splitting in the d–d transition of TM and the F–F transition of RE. Figure 3A and B shows the optical absorption spectra of the glass samples containing both TM and RE. After the initial addition of Er, peaks corresponding to the transition from the ground state to the states 2G7/2, 2K15/2, 2G9/2, 4F3/2–4F5/2, 4F7/2, 2H11/2, 4S3/2, 4F9/2, 4I9/2, 4I11/2, and 4I13/2 were seen at 356, 378, 390, 448, 486, 518, 532, 684, 784, 960, and 1514 nm [26].
When examining the growth of Cr2O3 in Er-doped glass samples, the creation of peaks at 350 nm [27, 28], which represent the synthesis of Cr6+ acts as a former, and an increase in Cr2O3 concentration up to 0.5 mol percent were noted. A broad signal at 610 nm [29] indicates that Cr3+ acts as a modifier (4A2g(f)–4T2g (F)).
Additionally, it exhibits the peaks at 669 and 697 nm [16, 30], which are attributed to the transitions 4A2g → 2T1g (2G) and 4A2g → 2E (2G) [31], which are both spin-forbidden. The Cr6+/Cr3+ ratio in the sample was high, up to 5/0.7, and the higher the ratio, the more Cr was added up to 1 mol percent. Cr6+ sharp peaks convert predominantly to Cr3+ in an intermediate state of Cr5+ formation [29], as shown by the broad band formation, and the formed peak at 457 nm represents 4A2g → 4T1g(F) [16]. Peaks in the UV range and a small range in the visible range overcome the Er peak, as shown in the diagram.
The structural modifications that occur based on stoichiometry [9, 22] are attributed to the differences in the Eg values. In real-world applications, the optical bandgap (Eg) is calculated by measuring the optical absorption coefficient close to the absorption edge [32]. According to a previous report, powerful optical fields are favored by glasses with high refractive indices and minimal energy gaps.
One of the primary aspects that can be assessed by altering absorbance is optical band gap analysis, which may be useful in assessing the electronic band structure in the created material [22]. The optical bandgap was investigated using the absorption coefficient of the relationship.
Figure 4 shows the relationship between (αhv)2/3 and hv (eV). The optical bandgap values at (hv) = 0 were generated via extrapolation, as displayed in Table 1. The Eg values for glass containing Er were similar to those reported in other studies [22, 32,33,34]. Alternatively, it can be thought of as the insertion of a Cr sublevel above the Fermi level [24, 35], as Cr lowers the Eg values [9, 36]. At this level, the BOS increases, whereas the valance-conduction distances decrease [9, 37]. Cr2O3 enters the glass network in two valence states, Cr3+ and Cr6+,which significantly affect the insulating quality and optical transmission [20, 33, 36], with CrO6 and CrO4 structural units, respectively. In the octahedral symmetry, the optical absorption spectrum of Cr3+ displays two broad visible bands.
The transition 4A2g(F) → 4T1g (P) is also associated with two wide UV absorption bands, although it is less intense because of the double electron jump t32 g t12g e2g and one of the transitions 4A2g(F) → 4T2g(F) and 4A2g(F) → 4T1g (F).
The intensity of peaks increased as the increase of chromium oxide, and several bands in the spectrum are difficult to identify because they overlap with Er3+ and Cr3+ absorption bands.
A faint, weak band at 370 nm in the spectra of the glass sample with 0.1 mol% Cr2O3 shows that these glasses contain some chromium ions in the Cr6+ form [36]. Additionally, a 465 nm absorption band was predicted for Cr5+ ions [38]. The spectra of the glass sample showed a decrease in the strength of the narrow, weak band at 370 nm, which indicates a decrease in the concentration of Cr6+ ions at the expense of Cr3+ ions. These ions play a role in the glass network, causing an increase in the disorder of the glass network. The amount of non-bridging oxygen (NBO) in the glass matrix increases as the octahedral Cr3+ ion concentration increases.
Consequently, the degree of electron localization increases, which reduces the number of donor centers in the glass matrix. As demonstrated in Table 1 and Fig. 2, the increased donor centers increase the optical band tail and move the absorption edge towards lower wavelengths.
For Cr3+ doped lithium tetraborate glass, the crystal field Dq and the Racach parameters (B) were calculated from the relation using the measured energies of the absorption bands [39].
where
The Dq was calculated to be 1612.9 cm−1. B was estimated to have a value of 625 cm−1, which is much less than the value of the free ion Cr3+, where Bfree = 918 cm−1.
When examining the ionic/covalent characteristics of the bonding between the Cr–O ligands, the value of B, which measures the interelectronic repulsion in the d-shell, is helpful. The value of Dq/B is greater than 2.3 which represents the presence of Cr3+ in a high-field site [40]. As a result, Cr3+ Centers in the high-field sites dominate the luminescence spectra of the glasses. These centers have narrow emission bands because of the 2Eg → 4A2g or 2T2g → 4A2g spin-forbidden transitions, which produce conventional laser action.
The Judd–Ofelt theory is helpful in calculating the oscillator strength in the theoretical manner, and the root mean square deviation [41].
The spectral strength (fexp) of absorption bands of glass composition doped with rare earth calculated from the integrated area under the peaks according to the relation:
With the Judd–Ofelt theory, the spectral strength calculated theatrically according to the relation:
where Ω is the Judd–Ofelt intensity parameters and h is Plank constant, n is the refractive index, m is the mass of electron, c is the speed of light and v is frequency of transition. U are the reduced matrix elements due to the J–J transition of Er3+. The quality fit between the results obtained experimentally and theoretically obtained from the root mean square equation according to the relation:
where N is the number energy level.
The root mean square deviation can be used to determine the correctness of the experiment, which is determined by the area of the absorption peak and the calculated oscillator strength.
The data are summarized in Table 2. The transitions 4I15/2 → 2H11/2 and 4G11/2, which are classified as hypersensitive transitions, obey the selection |S|= 0 and |∆L|≤ 2 and |∆J|≤ 2 [33, 34], have high values. The RMS values were smaller, which contradicts the validity of applying the Judd–Ofelt theory.
The Judd–Ofelt parameters that were created using the least-squares method are also included in Table 2. These outcomes could be contrasted with those of earlier investigations [33, 34, 38].
These features can be used to describe the radiative properties of RE, as well as the structural changes, covalency, and rigidity of the glass samples. While Ω4 is utilized to assess the stiffness of a created glass sample and has an opposing view of covalency, Ω2 is crucial for describing the covalency and asymmetry of the RE with the surrounding ligand. The Ω 6 parameter is primarily responsible for controlling the electron density between the 5d and 4f levels and is utilized to calculate the stiffness and viscosity of a sample [42]. Because each glass exhibits solitary behavior, the obtained value demonstrates the high effect of adding Cr.
The behavior of is dictated by the covalency of the samples containing 0 or 0.5 mol% Cr2O3, which allows for luminescence. The stiffness of the sample containing 0.1Cr2O3 was very high. Based on the acquired spectral values and absorption curves, the values were directly related to the Cr3+/Cr6+ ratio. The glass sample has a higher Cr3+ content, which may result in maximum fluorescence emission intensity [10].
To explore the optical qualities, the excitation and emission characteristics of the manufactured glass samples can be used. The glass sample excitation spectra recorded at 550 nm emission wavelength are shown in Fig. 4.
In the two broad bands centered at 396 nm, bands (4I15/2 → 4G9/2(336) and 4G11/2 (378) overlap, whereas bands (4I15/2 → 4F3/2(443), 4F5/2(451), and 4F7/2 (488) overlap in the broad band centered at 430 nm [43]. The band at 378 nm was used as the pump wavelength to evaluate the visible-luminescence properties of the glass samples.
Figure 5 illustrates the development of bands at 468, 541, 570, and 667 nm, which reflect the emission from the excited states 4F5/2, 2H11/2 (green), 4S3/2 (green), and 4F9/2 (red) to the ground state 4I15/2 with various emission colors.
The addition of Cr3+ reduces the peak intensity in the broad version [18] because Cr3+ ions absorb some of the excitation energy and produce near-infrared light.
The band at 450 nm disappears from the sample containing the 0.5 mol% Cr2O3 emission curve, and peaks at 410 and 430 nm arise instead. This is explained by the appearance of the emission peak 2H9/2 → 4I15/2(violet) [19] for Er, or it could be attributed to the reduction of Cr6+ to Cr3+, which is consistent with the absorption curve.
The CIE1931 chromaticity diagram's monochromatic (x, y) color coordinates were established using the fluorescence emission spectra produced by excitation at 378 nm. The results shown in Fig. 6 and the calculations in Table 3 indicate that the glass emits light that is almost entirely white. It was compared with other data [1, 7, 17, 44,45,46] and other results. The related color temperature was calculated using the chromaticity coordinates (x, y) of samples tabulated in Table 3 and [5, 17].
where n = (x – 0.332)/(y – 0.186).
The Kelvin ranges indicate that the glass samples under investigation were used as follows:
The optimum sample for use in display areas and workplaces that require extremely bright illumination is that with 1 mol% Cr2O3 and 1 mol% Er2O3. This sample emitted a bright amount of blue-white light, similar to that of daylight.
A strong bluish color of light is produced by samples with 1 mol% Er2O3 and 0, 0.1, or 0.5 mol% Cr2O3, which is frequently seen in commercial settings and is ideal for bright work lighting [7, 44] (Fig. 7).
4 Conclusion
Lithium borate glass containing x Cr2O3—1 mol% Er2O3 (where x = 0, 0.1, 0.50, and 1 mol%) were prepared. The increase in density, decrease in molar volume, and values of the optical band gap with the addition of chromium oxide indicate the compaction of the glass samples under study. The Racach parameters and crystal field Dq were established. The number Dq/B represents Cr3+ in a high field. The Judd–Ofelt parameters (Ω2, Ω4, and Ω6) are strongly influenced by the symmetry of the Eu3+ site, the lithium borate composition containing Cr2O3, and the surrounding Eu3+ions (Cr3+, O–B groups). Except for samples containing 0.5 mol% Cr3+ doped glass matrix, the trend of the JO intensity parameters is Ω2 > Ω6 > Ω4.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
N. Deopa et al., Enhanced visible green and 1.5 μm radiative emission of Er3+ ions in Li2O-PbO-Al2O3-B2O3 glasses for photonic applications. J. Rare Earths 39(5), 520–525 (2021). https://doi.org/10.1016/j.jre.2020.05.002
C. Lin et al., Study on the structure, thermal and optical properties in Cr2O3-incorporated MgO-Al2O3-SiO2-B2O3 glass. J. Non. Cryst. Solids 500, 235–242 (2018). https://doi.org/10.1016/j.jnoncrysol.2018.08.004
P.P. Pawar, S.R. Munishwar, R.S. Gedam, Intense white light luminescent Dy3+ doped lithium borate glasses for W-LED: A correlation between physical, thermal, structural and optical properties. Solid State Sci. 64, 41–50 (2017). https://doi.org/10.1016/j.solidstatesciences.2016.12.009
A.M. Othman, Z.M.A. El-fattah, M. Farouk, A.M. Moneep, M.A. Hassan, Optical spectroscopy of chromium doped bismuth-lithium borate glasses. J. Non. Cryst. Solids 558, 120665 (2021). https://doi.org/10.1016/j.jnoncrysol.2021.120665
M. Mariyappan, S. Arunkumar, K. Marimuthu, Effect of Bi2O3 on JO parameters and spectroscopic properties of Er3+ incorporated sodiumfluoroborate glasses for amplifier applications. J. Non. Cryst. Solids (2019). https://doi.org/10.1016/j.jnoncrysol.2020.119891
B.C. Jamalaiah et al., Visible and near infrared luminescence properties of Er 3 + -doped LBTAF glasses for optical amplifiers. Opt. Mater. (Amst) 34(5), 861–867 (2012). https://doi.org/10.1016/j.optmat.2011.11.023
N.S. Prabhu et al., Spectroscopic study of Er3+ doped borate glass system for green emission device, NIR laser, and optical amplifier applications. J. Lumin. (2021). https://doi.org/10.1016/j.jlumin.2021.118216
V. Bhatia et al., Structural, optical and thermoluminescence properties of newly developed MnKB: Er3+ glass system. J. Non. Cryst. Solids 543, 120113 (2020). https://doi.org/10.1016/j.jnoncrysol.2020.120113
E. Ebrahimi, M. Rezvani, “Optical and structural investigation on sodium borosilicate glasses doped with Cr2O3. Spectrochim Acta Part A Mol. Biomol. Spectrosc. 190, 534–538 (2018). https://doi.org/10.1016/j.saa.2017.09.031
R. Lachheb, A. Herrmann, K. Damak, C. Rüssel, R. Maâlej, Optical absorption and photoluminescence properties of chromium in different host glasses. J. Lumin. 186, 152–157 (2017). https://doi.org/10.1016/j.jlumin.2017.02.030
S. Yusub, D. KrishnaRao, The role of chromium ions on dielectric and spectroscopic properties of Li2O-PbO-B2O3-P2O5 glasses. J. Non. Cryst. Solids 398–399, 1–9 (2014). https://doi.org/10.1016/j.jnoncrysol.2014.04.022
G. Venkateswara Rao, N. Veeraiah, Study on certain physical properties of R2O–CaF2–B2O3:Cr2O3 glasses. J. Alloys Compd. 339, 54–64 (2002)
X. He et al., Glass forming ability, structure and properties of Cr2O3–Fe2O3 co-doped MgO–Al2O3–SiO2–B2O3 glasses and glass-ceramics. J. Non. Cryst. Solids 529, 2–9 (2020). https://doi.org/10.1016/j.jnoncrysol.2019.119779
P. Meejitpaisan, J. Kaewkhao, P. Limsuwan, C. Kedkaew, Physical and optical properties of the SLS glass doped with low Cr2O3 concentrations. Procedia Eng. 32, 787–792 (2012). https://doi.org/10.1016/j.proeng.2012.02.013
W.A. Pisarski, J. Pisarska, G. Dominiak-Dzik, W. Ryba-Romanowski, Transition metal (Cr3+) and rare earth (Eu3+, Dy3+) ions used as a spectroscopic probe in compositional-dependent lead borate glasses. J. Alloys Compd. 484(1–2), 45–49 (2009)
S. Suresh et al., Influence of local structural disorders on spectroscopic properties of multi-component CaF2–Bi2O3–P2O5–B2O3 glass ceramics with Cr2O3 as nucleating agent. Spectrochim. Acta - Part A Mol. Biomol. Spectrosc. 153, 281–288 (2016). https://doi.org/10.1016/j.saa.2015.08.025
L. Vijayalakshmi, K. Naveen Kumar, K.S. Rao, P. Hwang, Bright up-conversion white light emission from Er3+ doped lithium fluoro zinc borate glasses for photonic applications. J. Mol. Struct. 1155, 394–402 (2018). https://doi.org/10.1016/j.molstruc.2017.11.025
T.H. Kim, D.J. Kim, W.Y. Jang, A. Moon, K.S. Lim, M. Lee, Upconversion emission in Er and Cr co-doped KNbO3 phosphors. Jpn. J. Appl. Phys. 50(6 PART 2), 1–5 (2011). https://doi.org/10.1143/JJAP.50.06GH11
I. Arul Rayappan, K. Marimuthu, Luminescence spectra and structure of Er3+ doped alkali borate and fluoroborate glasses. J. Phys. Chem. Solids 74(11), 1570–1577 (2013). https://doi.org/10.1016/j.jpcs.2013.05.025
B.V. Padlyak, R. Lisiecki, W. Ryba-Romanowski, Spectroscopy of the Er-doped lithium tetraborate glasses. Opt. Mater. (Amst) 54, 126–133 (2016). https://doi.org/10.1016/j.optmat.2016.02.025
A.R. Babu, S. Yusub, P.M.V. Teja, P.S. Rao, V. Aruna, D.K. Rao, Effect of Cr2O3 on the structural, optical and dielectric studies of LiF-SrO-B2O3 glasses. J. Non. Cryst. Solids 520, 119428 (2019). https://doi.org/10.1016/j.jnoncrysol.2019.05.004
G. Chandrashekaraiah, N. Sivasankara Reddy, B. Sujatha, R. Viswanatha, C. Narayana Reddy, Role of Er3+ and Bi3+ ions on thermal and optical properties of Li2B4O7 glasses: structural correlation. J. Non. Cryst. Solids 498, 252–261 (2018). https://doi.org/10.1016/j.jnoncrysol.2018.06.034
N.S. Prabhu, V. Hegde, M.I. Sayyed, O. Agar, S.D. Kamath, Investigations on structural and radiation shielding properties of Er 3+ doped zinc bismuth borate glasses. Mater. Chem. Phys. 230, 267–276 (2019). https://doi.org/10.1016/j.matchemphys.2019.03.074
M.S. Al-Buriahi, Y.S.M. Alajerami, A.S. Abouhaswa, A. Alalawi, T. Nutaro, B. Tonguc, Effect of chromium oxide on the physical, optical, and radiation shielding properties of lead sodium borate glasses. J. Non. Cryst. Solids 544, 120171 (2020). https://doi.org/10.1016/j.jnoncrysol.2020.120171
V. Felice, B. Dussardier, J.K. Jones, G. Monnom, D.B. Ostrowsky, Chromium-doped silica optical fibres: Influence of the core composition on the Cr oxidation states and crystal field. Opt. Mater. (Amst) 16(1–2), 269–277 (2001). https://doi.org/10.1016/S0925-3467(00)00087-2
I. Kashif, A. Ratep, Luminescence in Er3+ co-doped bismuth germinate glass–ceramics for blue and green emitting applications. J. Korean Ceram. Soc. (2023). https://doi.org/10.1007/s43207-022-00281-2
S. Kaczmarek et al., Controlling of the charge states in laser crystals. Biul. Wojsk. Akad. Tech. 48(2), 105–116 (1999)
P. Meejitpaisan, J. Kaewkhao, P. Limsuwan, C. Kedkaew, Physical and optical properties of the SLS glass doped with low Cr2O3 concentrations. Procedia Eng. 32, 787–792 (2012). https://doi.org/10.1016/j.proeng.2012.02.013
S.R. Ramanan, D. Ganguli, Spectroscopic studies of Cr-doped silica gels. J. Non. Cryst. Solids 212(2–3), 299–302 (1997). https://doi.org/10.1016/S0022-3093(97)00106-3
A.V. Lalitha Phani, B. Srinivas, A. Hameed, M. Narasimha Chary, J.L. Rao, M.D. Shareefuddin, Comparative studies on physical and spectroscopic properties of alumino bismuth borate glasses containing Pb, Zn & Cd ions. Chin J. Phys. 58, 303–319 (2019). https://doi.org/10.1016/j.cjph.2018.12.021
W. Kuznik et al., Absorption, fluorescence and second harmonic generation in Cr3+-doped BiB3O6 glasses. Spectrochim. Acta - Part A Mol. Biomol. Spectrosc. 145, 325–328 (2015). https://doi.org/10.1016/j.saa.2015.03.041
Z.A.S. Mahraz, E.S. Sazali, M.R. Sahar, Spectral and dielectric characteristics of Er3+-doped multicomponent tellurite glasses. Optik (Stuttg) 239, 166776 (2021). https://doi.org/10.1016/j.ijleo.2021.166776
K. Annapoorani, C. Basavapoornima, N. SuriyaMurthy, K. Marimuthu, Investigations on structural and luminescence behavior of Er3 + doped lithium zinc borate glasses for lasers and optical amplifier applications. J. Non. Cryst. Solids 447, 273–282 (2016). https://doi.org/10.1016/j.jnoncrysol.2016.06.021
J. Rajagukguk, Fitrilawati, B. Sinaga, J. Kaewkhao, Structural and spectroscopic properties of Er3+ doped sodium lithium borate glasses. Spectrochim. Acta - Part A Mol. Biomol. Spectrosc. 223, 117342 (2019). https://doi.org/10.1016/j.saa.2019.117342
T. Narendrudu, S. Suresh, G. Chinna Ram, N. Veeraiah, D. Krishna Rao, Spectroscopic and structural properties of Cr3+ ions in lead niobium germanosilicate glasses. J. Lumin. 183, 17–25 (2017). https://doi.org/10.1016/j.jlumin.2016.11.005
J. Santhan Kumar, J. Lakshmi Kumari, M. Subba Rao, S. Cole, EPR, optical and physical properties of chromium ions in CdO-SrO-B 2O3-SiO2 (CdSBSi) glasses. Opt. Mater. (Amst) 35(7), 1320–1326 (2013). https://doi.org/10.1016/j.optmat.2013.01.012
G. El-Damrawi, A.M. Abdelghany, A.H. Oraby, M.A. Madshal, Structural and optical absorption studies on Cr2O3 doped SrO-P2O5 glasses. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 228, 117840 (2020). https://doi.org/10.1016/j.saa.2019.117840
D. Rajesh, Y.C. Ratnakaram, A. Balakrishna, Er3+-doped strontium lithium bismuth borate glasses for broadband 1.5 μm emission-structural and optical properties. J. Alloys Compd. 563, 22–27 (2013). https://doi.org/10.1016/j.jallcom.2013.02.055
I. Kashif, A. Ratep, Blue, red, and green emission from chromium and copper metal doped lithium borate glass. IOP Conf. Ser.: Mater. Sci. Eng. 956, 012013 (2020). https://doi.org/10.1088/1757-899X/956/1/012013
B.V. Padlyak, W. Ryba-Romanowski, R. Lisiecki, V.T. Adamiv, Y.V. Burak, I.M. Teslyuk, Opt. Mater. (Amst). 34, 2112–2119 (2012)
I. Kashif, A. Ratep, Judd–Ofelt and luminescence study the characterization of dysprosium-doped lithium borosilicate glasses for lasers and w-LEDs. J. Span. Ceram. Glass Soc. 61, 622–633 (2022). https://doi.org/10.1016/j.bsecv.2021.06.001
A.L. Martins, C.A.C. Feitosa, W.Q. Santos, C. Jacinto, C.C. Santos, Influence of BaX2 (X = Cl, F) and Er2O3 concentration on the physical and optical properties of barium borate glasses. Phys. B Condens. Matter 558, 146–153 (2019). https://doi.org/10.1016/j.physb.2019.01.038
S. Kaur, O.P. Pandey, C.K. Jayasankar, N. Chopra, Effect of gamma irradiation on physical, optical, spectroscopic and structural properties of Er 3 + -doped vitreous zinc borotellurite. J. Lumin. 235, 118031 (2021). https://doi.org/10.1016/j.jlumin.2021.118031
K.A. Naseer, S. Arunkumar, K. Marimuthu, The impact of Er3+ ions on the spectroscopic scrutiny of bismuth bariumtelluroborate glasses for display devices and 1.53 μm amplification. J. Non. Cryst. Solids 520, 119463 (2019). https://doi.org/10.1016/j.jnoncrysol.2019.119463
N. Luewarasirikul, Y. Ruangthaweep, J. Kaewkhao, ScienceDirect preparation and spectroscopic studies of Cr 3 + -doped aluminium calcium sodium borate glasses. Mater. Today Proc. 17, 1831–1836 (2019). https://doi.org/10.1016/j.matpr.2019.06.220
M. Mariyappan, S. Arunkumar, K. Marimuthu, Effect of Bi2O3 on JO parameters and spectroscopic properties of Er3+ incorporated sodiumfluoroborate glasses for amplifier applications. J. Non. Cryst. Solids (2020). https://doi.org/10.1016/j.jnoncrysol.2020.119891
Acknowledgements
I. Kashif thanks Prof. Dr. A. M. Sanad for his support throughout my scientific career during his stay with us.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
AR: Investigation, Writing—original draft, Methodology, Formal analysis. IK: Writing—review & editing, administration, Formal analysis, Investigation. We, the authors of this paper, agree to send it to your journal and that IK is the corresponding author for submitting the research and addressing the journal.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
This paper meets the ethical standards of this journal.
Research involving human participants and/or animals
Not applicable.
Consent to participate
All authors agree with the review of this paper in this journal.
Consent for publication
This manuscript has not been published, was not, and is not being submitted to any other journal. All necessary permissions for publication were secured prior to submission of the manuscript. All authors listed have made a significant contribution to the research reported and have read and approved the submitted manuscript, and furthermore, all those who made substantive contributions to this work have been included in the author list. All authors have seen and approved the final version of the manuscript being submitted. The article is the original work, hasn't received prior publication and isn't under consideration for publication elsewhere.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kashif, I., Ratep, A. Optical properties of borate glasses containing chromium and erbium oxide. Appl. Phys. A 129, 489 (2023). https://doi.org/10.1007/s00339-023-06764-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-023-06764-1