Abstract
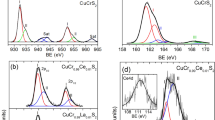
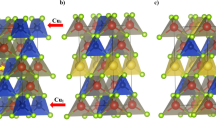
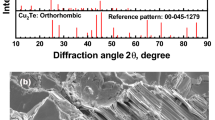
The study of the valence state, local environment structure and magnetic properties of novel type CuCr0.99Ln0.01S2 (Ln = La, Ce) solid solutions was carried out using X-ray absorption spectroscopy, finite difference method simulations and static magnetic susceptibility measurements. The good agreement between experimental and calculated data indicates that cationic substitution does not lead to significant changes in the copper, chromium and sulfur local environment character and electronic density distribution. The copper atoms were found to be in Cu+ oxidation state, the sulfur atoms—in S2− oxidation state and the chromium atoms—in Cr3+ state. The cationic substitution of chromium by lanthanum and cerium in CuCrS2 does not significantly affect the effective magnetic moment and exchange interactions character. The lanthanum-doped CuCr0.99Ln0.01S2 solid solution demonstrates the Seebeck coefficient value 4 times greater than for CuCrS2-matrix at 500 K.








Similar content being viewed by others
References
Y.-X. Chen, B.-P. Zhang, Z.-H. Ge, P.-P. Shang, Preparation and thermoelectric properties of ternary superionic conductor CuCrS2. J. Solid State Chem. 186, 109–115 (2012). https://doi.org/10.1016/j.jssc.2011.11.040
A. Kaltzoglou, P. Vaqueiro, T. Barbier, E. Guilmeau, A. Powell, Ordered-defect sulfides as thermoelectric materials. J. Electron. Mater. 43, 2029–2034 (2014). https://doi.org/10.1007/s11664-013-2941-0
E.V. Korotaev, M.M. Syrokvashin, IYu. Filatova, K.G. Pelmenev, V.V. Zvereva, N.N. Peregudova, J. Electron. Mater. 47, 3392–3397 (2018). https://doi.org/10.1007/s11664-018-6230-9
R.F. Al’mukhametov, R.A. Yakshibaev, E.V. Gabitov, Phys. Solid State 41, 1327–1328 (1999). https://doi.org/10.1134/1.1130992
R.F. Al’mukhametov, R.A. Yakshibaev, E.V. Gabitov, A.R. Abdullin, R.M. Kutusheva, Phys. Stat. Sol. B 236, 29–33 (2003). https://doi.org/10.1002/pssb.200301413
G.R. Akmanova, A.D. Davleshina, Lett. Mater. 3, 76–78 (2013)
A. Karmakar, K. Dey, S. Chatterjee, S. Majumdar, S. Giri, Appl. Phys. Lett. 104, 052906 (2014). https://doi.org/10.1063/1.4863937
F.M.R. Engelsman, G.A. Wiegers, F. Jellinek, B. van Laar, J. Solid State Chem. 6, 574–582 (1973). https://doi.org/10.1016/S0022-4596(73)80018-0
R. Miyawaki, F. Hatert, M. Pasero, S. Mills, Miner. Mag. 83, 887–893 (2019). https://doi.org/10.1180/mgm.2019.73
G.M. Abramova, G.A. Petrakovskii, Low Temp. Phys. 32, 725–734 (2006). https://doi.org/10.1063/1.2219495
G. Abramova, A. Pankrats, G. Petrakovskii, J.C.E. Rasch, M. Boehm, A. Vorotynov, V. Tugarinov, R. Szumszak, A. Bovina, V. Vasil’ev, J. Appl. Phys. 107, 931 (2010)
N. Tsujii, H. Kitazawa, G. Kido, Phys. Stat. Sol. (C) 3, 4417–4418 (2006). https://doi.org/10.1002/pssc.200669659
N. Le Nagard, G. Collin, O. Gorochov, Mater. Res. Bull. 14, 1411–1417 (1979). https://doi.org/10.1016/0025-5408(79)90083-7
I.G. Vasileva, V.V. Kriventsov, J. Synch. Investig. 4, 640–644 (2010). https://doi.org/10.1134/S1027451010040178
Inorganic crystal structure database. Version 2.1.0/FIZ Karlsruhe, Germany
Experimental station “EXAFS spectroscopy”. http://ssrc.inp.nsk.su/CKP/eng/stations/passport/8/. Accessed 18 June 2018
P.S. Zavertkin, D.V. Ivlyushkin, M.R. Mashkovtsev, A.D. Nikolenko, S.A. Sutormina, N.I. Chkhalo, Optoelectron. Instrum. Proc. 55, 107–114 (2019). https://doi.org/10.3103/s8756699019020018
S.A. Guda, A.A. Guda, M.A. Soldatov, K.A. Lomachenko, A.L. Bugaev, C. Lamberti, W. Gawelda, C. Bressler, G. Smolentsev, A.V. Soldatov, Y. Joly, J. Chem. Theory Comput. 11, 4512–4521 (2015). https://doi.org/10.1021/acs.jctc.5b00327
Y. Joly, Phys. Rev. B 63, 125120–125129 (2001). https://doi.org/10.1103/PhysRevB.63.125120
G.S. Henderson, F.M. de Groot, B.J. Moulton, Rev. Miner. Geochem. 78, 75–138 (2014). https://doi.org/10.2138/rmg.2014.78.3
J.W. Niemantsverdriet, Spectroscopy in Catalysis: An Introduction, 3rd, Completely Revised and Enlarged Edition (Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2007), p. 326
G.J. Schmitz, U. Prahl, Handbook of Software Solutions for ICME (Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2017), p. 595
N. Tsujii, H. Kitazawa, Substitution effect on the two-dimensional triangular-lattice system CuCrS2. J. Phys. Condens. Matter 19, 145245 (2007). https://doi.org/10.1088/0953-8984/19/14/145245
M.M. Syrokvashin, E.V. Korotaev, IYu. Filatova et al., Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 205, 593–596 (2018). https://doi.org/10.1016/j.saa.2018.07.053
O. Bunau, Y. Joly, J. Phys. Condens. Matter 21, 345501 (2009). https://doi.org/10.1088/0953-8984/21/34/345501
J.D. Bourke, C.T. Chantler, Phys. Rev. Lett. 104, 206601 (2010). https://doi.org/10.1103/PhysRevLett.104.206601
J.D. Bourke, C.T. Chantler, Y. Joly, J. Synchrotron Rad. 23, 551–559 (2016). https://doi.org/10.1107/S1600577516001193
S. Blundell, Magnetism in Condensed Matter (Oxford University Press, New York, 2001)
P. Selwood, Magnetochemistry, 2nd edn. (Interscience Publishers, New York, 1956)
Acknowledgements
The study was carried out with a funding from the Russian Science Foundation (Project No. 19-73-10073). The X-ray absorption spectra were measured using the shared research center SSTRC on the basis of the Novosibirsk VEPP-4 - VEPP-2000 complex at BINP SB RAS, using equipment supported by Project RFMEFI62119X0022.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Korotaev, E.V., Syrokvashin, M.M., Filatova, I.Y. et al. XANES investigation of novel lanthanide-doped CuCr0.99Ln0.01S2 (Ln = La, Ce) solid solutions. Appl. Phys. A 126, 537 (2020). https://doi.org/10.1007/s00339-020-03715-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-020-03715-y