Abstract
Long-term movements of adult marine animals predicate spatial management measures. Sea cucumbers are intensely exploited worldwide, yet few studies have assessed their long-term movement potential. Elephant trunkfish, Holothuria (Microthele) fuscopunctata, is a large coral reef holothuroid. At four sites at Lizard Island, northern Great Barrier Reef, we photographically marked and georeferenced 36 H. fuscopunctata, ranging 1.4–5.1 kg in weight. In broader searches one year later, we recaptured 92% of the animals. Their displacements were comparable among sites and averaged 8.7 m yr−1. None of the movements could be characterised as nomadic; the maximum displacement was 21 m. Contrary to expectations, smaller animals tended to roam further than larger animals (p = 0.025). Our study shows that certain large-bodied sea cucumbers might not displace far over annual timescales. This empirical evidence of home ranging reshapes our understanding of echinoderm mobility and reveals a behaviour that would exacerbate Allee effects at low population densities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The movement of marine animals—i.e. their spatial displacements over time—allows them to undertake processes that shape their life-history (Pittman and McAlpine 2003). Finding mates for reproduction is one central purpose of mobility in marine animals (Allen et al. 2018), as broadcast spawners that are too far apart fail to reproduce successfully (Bell et al. 2008). Mobile animals are also able to access optimal areas for foraging. Since coral reef invertebrates often have affinities to soft-bottom or hard reef habitats (Tanita et al. 2022), their displacement for both reproduction and foraging might be restricted by the patchy seascapes within individual reefs.
A stunning range of movement patterns exists among marine fish and invertebrates (Allen et al. 2018). Nomadism involves movements over large areas that tend to be random or irregular but can include small-scale displacements during foraging (Grüss et al. 2011; Teitelbaum and Mueller 2019). In contrast, home ranging involves relatively short movements within a defined area for an animal’s routine activities (Moffitt et al. 2009). Both nomadic and home ranging movement behaviours can be observed among individuals of mobile marine species (Grüss et al. 2011; Purcell et al. 2016). These movement patterns influence resource management and conservation measures, which can be area-based and rely on limited animal mobility (Moffitt et al. 2009; Purcell 2010; Grüss et al. 2011). Knowledge about the extent of home ranging and nomadism is especially critical for exploited coral reef species because the marine reserves can often be rather small (Kramer and Chapman 1999).
Matching the spatial scale of movement with appropriate management and conservation measures requires knowledge of an animal’s displacement over time (Palumbi 2004; Grüss et al. 2011). Mark-recapture studies are used extensively to infer movement patterns of marine animals. ‘Marking’ may involve inserting a tag internally or externally, genetic fingerprinting, body etching or photographic marking. The literature abounds with mark-recapture studies on marine vertebrates, including sea turtles, sharks, and fishes, yet holds remarkably few on marine invertebrates (Palumbi 2004; Allen et al. 2018). On coral reefs, notable exceptions include spiny lobsters (Sharp et al. 1997; Frisch 2007; O’Malley 2009), predatory gastropods (Schlaff et al. 2020), herbivorous gastropods (Purcell et al. 2004), nautilus (Dunstan et al. 2011), and a range of reef echinoderms (echinoids, asteroids and holothuroids).
Echinoderms have been broadly characterised as relatively sedentary (Allen et al. 2018). However, on coral reefs, crown-of-thorns starfish and certain sea cucumbers can be highly mobile in the short-term, displacing 9–10 m per day at some sites (Keesing and Lucas 1992; Purcell et al. 2016). Nonetheless, such movements do not necessarily translate to long displacements over yearly timeframes (Purcell et al. 2016). Sea urchins can move metres per day and some are known to display seasonal migrations of much longer distances, but on tropical reefs their movement tends to be restricted (Lawrence 2020).
Sea cucumbers are extensively harvested worldwide for Asian seafood markets (Toral-Granda et al. 2008; Purcell et al. 2023). On coral reefs, many of the species are large-bodied and, where unfished, are among the most ubiquitous of benthic megafauna (Pierrat et al. 2022). The foraging on sediments by holothuroids contributes to the maintenance of sediment health (Purcell et al. 2016; Pierrat et al. 2022), and their daily foraging movements could contribute to ‘reef halos’ (Gray et al. 2022). Empirical evidence for their long-term movement is critically limited to inform spatial management measures and understand Allee effects at low population densities.
The movement ecology of sea cucumbers is known mostly from observations over hours or days (e.g. Shiell and Knott 2008; Navarro et al. 2013, 2014; Rodríguez-Barreras et al. 2014; Siegenthaler et al. 2015; Wolfe and Byrne 2017; Hammond et al. 2020). Conventional tagging methods have proved problematic due to high rates of tag loss and-or ill effects on the animals (Conand 1991; Cieciel et al. 2009; Purcell et al. 2006; Rodríguez-Barreras et al. 2014). Photographs of distinctive colour patterns and body features have proved effective for long-term mark-recapture studies on sea cucumbers, revealing home ranging and nomadic behaviours (e.g. Purcell et al. 2016; Hammond and Purcell 2023). This technique can be used for a small proportion of species with individual colour patterns or papillae, and a majority of species are unsuitable.
All of the sea cucumbers on tropical reefs of a large body size and thick body wall are heavily exploited throughout the Indo-Pacific (Purcell et al. 2023). Genetic fingerprinting indicated a low frequency of nomadic movements in the large-bodied Pacific black teatfish, Holothuria (Microthele) whitmaei (Uthicke et al. 2004). The elephant trunkfish H. (Microthele) fuscopunctata Jaeger, 1833, is equally large-bodied and can be found commonly on unexploited coral reefs from east Africa to the central south Pacific Ocean. Although attracting a low value in Asian dried seafood markets, this species is exploited in at least 20 countries and commercial harvesting is a threat to its populations (Conand et al. 2013). On the Great Barrier Reef, Australia, fishing for H. fuscopunctata is regulated by a rotational harvest strategy and a catch quota (Wolfe and Byrne 2022). Studies have documented that H. fuscopunctata occurs mostly on reef flats and outer reef slopes on soft sediments (Conand 1989; Eriksson et al. 2012; Bellchambers et al. 2011) and reproduces sexually in the austral summer (Conand 1993; Uthicke 1994a, b; Tessier and Letouze 2014). Little else is known of the ecology of this species, and we know nothing about its mobility in coral reef habitats.
This study aimed to use photographic mark-recapture to determine the year-long movement (i.e. displacement distances) of H. fuscopunctata across several coral reef sites. We were primarily interested to know if individuals would show long displacements (that could be a concern for spatial management measures), or home ranging, or a combination of the two. Measurements of water depth and animal body weight enable us to explore relationships with spatial displacements. These variables were examined because, for certain holothuroid species, larger individuals are known to displace further than smaller ones (Hammond and Purcell 2023) and movement rates appear to decline with water depth (Cieciel et al. 2009). The results of this study inform future fishery management planning such as the sizing of marine reserves and rotational harvest strategies (Pittman and McAlpine 2003; Grüss et al. 2011). Further, the findings enrich our modest understanding of macroinvertebrate mobility on coral reefs.
Methods
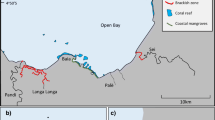
The fieldwork took place in February 2020 and February 2021 at Lizard Island on the northern Great Barrier Reef (GBR) in Queensland, Australia (14° 40′ S, 145° 27′ E). This location is within a no-take Scientific Research reserve in which fishing for sea cucumbers has not occurred for many decades. The 8-year long-term averages of sea temperatures (at 0.6 m depth) at this locality range from 23.6 °C in the austral winter to 29.4 °C in the austral summer (AIMS 2009). During 2020, the sea temperature range was slightly above this average. Following previous searches for Holothuria fuscopunctata around Lizard Island, four sites were selected in which the species was known to occur (Fig. 2).
Two snorkelers searched each site, at depths of 2–9 m, to locate animals irrespective of their body sizes and depth of occurrence. Thirty-six animals were located and measured at several sites in 2020 with body lengths ranging 31–47 cm and body weights ranging 1.44–5.11 kg. We then returned and searched broader areas, beyond the initial locations, in 2021, locating 84 animals (Fig. 2) (Table 1). Overall, 33 (92%) of the animals from 2020 were relocated and confidently matched by visual comparison of photographs in 2021 (Table 1).
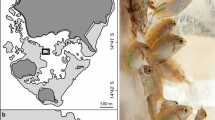
A handheld GPS towed on a dive float, to which dive watches were synchronised, recorded search tracks with a 30-s frequency that were later used to determine animal locations and record track lengths. Once located, the float and GPS were positioned directly over the animal at the water surface for one minute and the time was recorded. A labelled tent peg was placed next to the posterior end of each animal, which was photographed overhead. Sediment on the dorsal surface of the animals was gently wafted away before taking photographs. For the first few animals, we photographed the anterior end, but then photographed the whole animals thereafter to aid photographic identification. We weighed each animal to the nearest 10 g using a hanging balance in a boat after a 5 min draining period (see Skewes et al. 2004) and then returned each one to their original (marked) position on the reef.
Photographs of the animals were visually analysed and the deep dorsal-surface wrinkles, characteristic of this species (Purcell et al. 2023), used to match 2020 animals to 2021 recaptures (Fig. 1). We matched ten consecutive wrinkles between photos to confirm a successful match, yielding a false-positive rate of 0.098%, assuming approximately 50% chance of each wrinkle occurring in the same position on another animal by chance. The GPS locations of initial capture and recaptures were plotted in Google Earth Pro (2020), and the ruler tool was used to measure the one-year displacement to the nearest cm. Applying this procedure to 12 fixed positions on land (for methodological validation) revealed an average error of 1.28 m for the displacement distances. Very short animal displacements of a few metres therefore have a proportionally large measurement error and were interpreted cautiously.
Length-frequency and weight-frequency histograms were constructed from the data of the 84 animals located in 2021. This avoided any multiple measures for animals first recorded in 2020.
Depths were corrected to zero tidal data. One-year displacements were compared among sites using a one-way analysis of covariance (ANCOVA), with water depth and animal body weight as covariates. Inspection of box-and-whisker plots indicated that the data were normally distributed within sites. Levene’s test indicated moderate heteroscedasticity (p = 0.03), but ANOVA is robust to this departure given the large sample size. Relationships between the displacements and the water depth and body weight were examined by nonlinear regressions using DataFit™ (v9) software, choosing the best fitting two-parameter model according to r2 values to avoid overfitting.
Results and discussion
Long-term movement
To our knowledge, the recapture rate of 92% for Holothuria fuscopunctata is the highest documented of any mobile coral reef macroinvertebrate over long timeframes. The rate largely surpasses the 11% recaptures for Panulirus marginatus spiny lobsters and 12% for Scyllarides squammosus slipper lobsters up to five years in Hawai’i (O’Malley and Walsh 2013), 23% for Panulirus guttatus spiny lobsters in the Florida Keys over 2.1 years (Sharp et al. 1997), 57% for Panulirus versicolor painted crayfish on the Great Barrier Reef over 18 months (Frisch and Hobbs 2006), 10% for Nautilus pompilius nautilus in the Coral Sea from 1 to 10 years (Dunstan et al. 2011), and 36–47% for Trochus niloticus gastropods after 6 months in Vanuatu (Purcell et al. 2004). Other long-term studies on sea cucumber had recapture rates of 67% for Bohadschia argus and 53% for Thelenota ananas over 2 years on the Great Barrier Reef (Purcell et al. 2016), and 67–72% for Pearsonothuria graeffei over 1–2 years at the same locality (Hammond and Purcell 2023). The high recapture rate in our study can be attributed to the non-cryptic nature of the animals, the facility of matching individuals of this species and their site-attached behaviour.
Using a rough estimate of 8 m for the width of our 2021 searches over the tracks at each site, the track areas represent 0.88 to 1.30 ha per site. The densities of H. fuscopunctata within these track areas ranged from 6 ind ha−1 (at Palfrey Lagoon) to 50 ind ha−1 (Lagoon Channel). Even at this longstanding marine reserve at Lizard Island where the animals are unfished, the site with lowest densities of the sea cucumbers were lower than the proposed threshold ‘regional reference density’ of 10 ind ha−1 for ‘healthy’ populations for this species (Pakoa et al. 2014). At the other study sites, densities of H. fuscopunctata were above this reference density, although we note that our searches were within a specific habitat and not representative of all lagoon or reef flat areas.
Over half of the animals were found in a large ‘pickle’ (collective noun; Hammond and Purcell 2023) at the Lagoon Channel site, while other individuals were found alone, or in pairs or trios (Fig. 2). Although the recapture surveys extended up to a couple of hundred metres beyond the initial capture areas, within the depths in which this species was observed to occur at these sites, none of the recaptured animals had moved further than 21 m (Fig. 2). The average yearly displacement of H. fuscopunctata was 8.7 m (± 5.4 SD) (Table 1). The measurement error of the GPS represents 15% of this average distance. The displacements of three individuals were measured as less than 2 m, close to the error limits of the methodology but, in any case, show that the animals moved little from the site of initial capture. Summer sea temperatures during the study were warmer than the 8-year long-term average. In view that certain tropical holothuroids display elevated mobility at high seawater temperature (Chen et al. 2022), it is possible that H. fuscopunctata in our study were somewhat more mobile during the year of study compared to what they might be other years. We noticed some H. fuscopunctata close to the reef edge in the morning and foraging on sand further from the reef later in the day. Such behaviours have been recently reported for other large coral reef holothuroids (Gray et al. 2022) and might indicate that certain refuges are used and reinforce home ranging behaviours.
Aerial photographs (Google Earth Pro 2020) showing the location of the survey sites at Lizard Island. The Holothuria fuscopunctata initially photographed in 2020 (yellow) and recaptured in 2021 (red) are shown and their one-year displacement indicated by the yellow lines. Unmatched animals from 2020 (three animals) and 2021 (47 animals) are shown as grey dots. Extensive searches for recaptures at each site in 2021 were tracked by a GPS and illustrated as grey dashed lines
These findings provide empirical evidence of confined home ranging in a widely distributed and exploited sea cucumber. The remarkably short, one-year displacements indicate that Holothuria fuscopunctata is highly site attached and not nomadic. In reality, the movement distances could have been slightly shorter due to the 1.28 m measurement error, but this is not at a scale to reshape our findings. One implication from this finding is that small, strategically placed marine reserves may be adequate for their protection (Purcell 2010).
The average annual movement distance is the same as that of a smaller-bodied sea cucumber, Pearsonothuria graeffei (9 m y−1), which lives on hard-reef outcrops (Hammond and Purcell 2023). In contrast, invasively marked Holothuria arguinensis, a moderate-sized holothuroid, travel on average 8 m per day (Navarro et al. 2014). A short-term study in Western Australia showed that the large-bodied black teatfish (H. whitmaei) move a couple of metres per day (Shiell and Knott 2010), and a genetic fingerprinting study at Lizard Island in eastern Australia found that some of them could move 90 m in one year (Uthicke et al. 2004). Two other large-bodied sea cucumbers, Bohadschia argus and Thelenota ananas, exhibit both long-term home-ranging and nomadism, with displacements averaging tens of metres per year (Purcell et al. 2016). The present study reveals that large sea cucumber species do not necessarily move far.
Uthicke (1994a, b) found H. fuscopunctata spawning in December at our study locality. The small and large pickles (i.e. groups/aggregations) that we found could be reflective of reproductive behaviour. However, the apparent lack of mixing of animals between groups within the site (Fig. 2) suggests that it is unlikely that the animals moved far and all returned so close to their original locations and respective groups for spawning. Nonetheless, future studies involving seasonal monitoring (c.f. Wolfe and Byrne 2017) of holothuroid movements would be valuable.
The tight home ranging found in our study at least demonstrates that H. fuscopunctata might struggle to locate mates at low population density. The findings underscore the importance of management strategies that maintain reasonable densities of these species in order to avoid Allee effects (Bell et al. 2008). Our findings imply that the movement ecology of certain sea cucumbers might be a significant factor in depensatory effects that provoke local extinctions.
Displacement rates were statistically comparable among three of the sites where we had sufficient replication (ANCOVA, F(2,27) = 0.47, p = 0.63). The covariates of body weight and water depth were (linearly) non-significant in the ANCOVA test (p-values: 0.07 and 0.13, respectively) although we explored these further using nonlinear regression (below).
Relationships of long-term movement
The H. fuscopunctata measured in this study in 2021 were all large, with only one being less than 30 cm in length (Fig. 3a). The modal length in this study of 36–45 cm is smaller than the mode of 46–48 cm reported for the same species in New Caledonia in the 1980s by Conand (1989). Body weights of the animals in our study were mostly between 2.5 and 4 kg, and the maximum body weight (in 2021) was 5.67 kg (Fig. 3b). Compared with a weight at first sexual maturity reported for H. fuscopunctata from New Caledonia of 1.22 kg by Conand (1993), it would seem that all of the animals we studied had attained maturity.
Sites were pooled for regression analyses based on non-significant differences shown in the ANCOVA test. The nonlinear regression showed that body weight influenced the yearly movement (r2 = 0.152, p = 0.025), with larger animals tending to move less, commonly displacing < 10 m yr−1 (Fig. 4a). This result is contrary to a more common pattern of larger animals moving greater distances, found in sea urchins (Dumont et al. 2004) and in a smaller-bodied sea cucumber (Hammond and Purcell 2023). Body weights of H. fuscopunctata explained only 15% of the variation in annual displacements, so movement could be rather stochastic and likely influenced by other factors.
Relationships of body weight and water depth with the 1-year displacements of recaptured Holothuria fuscopunctata at Lizard Island (n = 33). a scatterplot of displacement and initial body weight with best-fitting two-parameter model (y = a + b*exp(−x)) (black trendline) and 95% confidence intervals (blue dashed lines); b scatterplot of displacement and initial water depth (adjusted to zero tidal datum)
Water depths (corrected to zero tide data) varied amongst sites, ranging 2–10 m (Table 1) with all recaptures in depths < 7 m. The yearly displacements were consistently short for animals in deeper waters and variable for those in shallower waters (Fig. 4b), but the effect of water depth was not statistically significant (p = 0.09). Certain sea cucumbers at higher latitudes might display twofold greater mobility in shallower waters, possibly because of differing conditions such as water temperature (Cieciel et al. 2009). Future studies on mobility of tropical species should best examine a wide range of depths and measure potential causal variables such as temperature, and the granulometry and organic content of sediments.
References
Allen RM, Metaxas A, Snelgrove PV (2018) Applying movement ecology to marine animals with complex life cycles. Annu Rev Mar Sci 10:19–42. https://doi.org/10.1146/annurev-marine-121916-063134
Australian Institute of Marine Science (AIMS) (2009). Northern Australia Automated Marine Weather and Oceanographic Stations. https://doi.org/10.25845/5c09bf93f315d
Bell JD, Purcell SW, Nash WJ (2008) Restoring small-scale fisheries for tropical sea cucumbers. Ocean Coast Manage 51:589–593. https://doi.org/10.1016/j.ocecoaman.2008.06.011
Bellchambers LM, Meeuwig JJ, Evans SN, Legendre P (2011) Modelling habitat associations of 14 species of holothurians from an unfished coral atoll: implications for fisheries management. Aquat Biol 14:57–66
Chen M, Sun S, Xu Q, Gao F, Wang H, Wang A (2022) Influence of water temperature and flow velocity on locomotion behavior in tropical commercially important sea cucumber Stichopus monotuberculatus. Front Mar Sci 9:931430
Cieciel K, Pyper BJ, Eckert GL (2009) Tag retention and effects of tagging on movement of the giant red sea cucumber Parastichopus californicus. N Am J Fish Manage 29:288–294
Conand C (1993) Reproductive biology of the holothurians from the major communities of the New Caledonian Lagoon. Mar Biol 116:439–450. https://doi.org/10.1007/BF00350061
Conand C, Purcell S, Gamboa R (2013) Holothuria fuscogilva. The IUCN red list of threatened species 2013: e.T200715A2681354. https://doi.org/10.2305/IUCN.UK.2013-1.RLTS.T200715A2681354.en
Conand C (1989) Les Holothuries Aspidochirotes du lagon de Nouvelle-Calédonie: biologie, e´cologie et exploitation. Etudes et The`ses. O.R.S.T.O.M., Paris, p 393
Conand C (1991) Long-term movements and mortality of some tropical sea-cucumbers monitored by tagging and recapture. In: Yanagisawa T, Yasumasu I, Oguro C, Suzuki N, Motokawa T (eds) Biology of Echinodermata. Proceedings of the 7th international echinoderm conference Blakema, Rotterdam, pp 169−175
Dumont C, Himmelman JH, Russell MP (2004) Size-specific movement of green sea urchins Strongylocentrotus droebachiensis on urchin barrens in eastern Canada. Mar Ecol Prog Ser 276:93–101
Dunstan AJ, Ward PD, Marshall NJ (2011) Nautilus pompilius life history and demographics at the Osprey Reef Seamount, Coral Sea. Australia Plos One 6:e16312
Eriksson H, Byrne M, de la Torre-Castro M (2012) Sea cucumber (Aspidochirotida) community, distribution and habitat utilization on the reefs of Mayotte, Western Indian Ocean. Mar Ecol Prog Ser 452:159–170
Frisch AJ (2007) Short-and long-term movements of painted lobster (Panulirus versicolor) on a coral reef at Northwest Island, Australia. Coral Reefs 26:311–317
Frisch AJ, Hobbs JP (2006) Long-term retention of internal elastomer tags in a wild population of painted crayfish (Panulirus versicolor [Latreille]) on the Great Barrier Reef. J Exp Mar Biol Ecol 339:104–110
Gray BC, Calvert LA, Purcell SW (2022) Short-term movement dynamics of the world’s largest sea cucumbers (genus Thelenota). Mar Ecol 43:e12705
Grüss A, Kaplan DM, Guénette S, Roberts CM, Botsford LW (2011) Consequences of adult and juvenile movement for marine protected areas. Biol Conserv 144:692–702. https://doi.org/10.1016/j.biocon.2010.12.015
Hammond AR, Purcell SW (2023) Limited long-term movement and slow growth of the sea cucumber Pearsonothuria graeffei. Mar Ecol Prog Ser 704:1–14. https://doi.org/10.3354/meps14240
Hammond AR, Meyers L, Purcell SW (2020) Not so sluggish: movement and sediment turnover of the world’s heaviest holothuroid. Thelenota Anax Mar Biol 167:60. https://doi.org/10.1007/s00227-020-3671-5
Keesing JK, Lucas JS (1992) Field measurement of feeding and movement rates of the crown-of-thorns starfish Acanthaster planci (L.). J Exp Mar Biol Ecol 156:89–91. https://doi.org/10.1016/0022-0981(92)90018-6
Kramer DL, Chapman MR (1999) Implications of fish home range size and relocation for marine reserve function. Environ Biol Fish 55:65–79
Lawrence JM (ed) (2020) Sea urchins: biology and ecology, 4th edn. Academic Press, London
Moffitt EA, Botsford LW, Kaplan DM, O’Farrell MR (2009) Marine reserve networks for species that move within a home range. Ecol Appl 19:1835–1847
Navarro PG, García-Sanz S, Barrio JM, Tuya F (2013) Feeding and movement patterns of the sea cucumber Holothuria sanctori. Mar Biol 160:2957–2966. https://doi.org/10.1007/s00227-013-2286-5
Navarro PG, García-Sanz S, Tuya F (2014) Contrasting displacement of the sea cucumber Holothuria arguinensis between adjacent nearshore habitats. J Exp Mar Biol Ecol 453:123–130. https://doi.org/10.1016/j.jembe.2014.01.008
O’Malley JM (2009) Spatial and temporal variability in growth of Hawaiian spiny lobsters in the Northwestern Hawaiian Islands. Mar Coast Fish Dyn Manag Ecosyst Sci 1:325–342
O’Malley JM, Walsh WA (2013) Annual and long-term movement patterns of spiny lobster, Panulirus marginatus, and slipper lobster, Scyllarides squammosus, in the Northwestern Hawaiian islands. Bull Mar Sci 89:529–549
Pakoa K, Friedman K, Moore B, Tardy E, Bertram I (2014) Assessing tropical marine invertebrates: A manual for Pacific Island resource managers. Secretariat of the Pacific Community, Noumea, New Caledonia
Palumbi SR (2004) Marine reserves and ocean neighborhoods: the spatial scale of marine populations and their management. Annu Rev Environ Resour 29:31–68. https://doi.org/10.1146/annurev.energy.29.062403.102254
Pierrat J, Bédier A, Eeckhaut I, Magalon H, Frouin P (2022) Sophistication in a seemingly simple creature: a review of wild holothurian nutrition in marine ecosystems. Biol Rev 97:273–298. https://doi.org/10.1111/brv.12799
Pittman S, McAlpine C (2003) Movements of marine fish and decapod crustaceans: process, theory and application. Adv Mar Biol 44:205–294
Purcell SW, Amos MJ, Pakoa K (2004) Releases of cultured sub-adult Trochus niloticus generate broodstock for fishery replenishment in Vanuatu. Fish Res 67:329–333
Purcell SW, Blockmans BF, Nash WJ (2006) Efficacy of chemical markers and physical tags for large-scale release of an exploited holothurian. J Exp Mar Biol Ecol 334:283–293. https://doi.org/10.1016/j.jembe.2006.02.007
Purcell SW, Piddocke TP, Dalton SJ, Wang Y-G (2016) Movement and growth of the coral reef holothuroids Bohadschia argus and Thelenota ananas. Mar Ecol Prog Ser 551:201–214. https://doi.org/10.3354/meps11720
Purcell SW, Lovatelli A, González-Wangüemert M, Solís-Marín FA, Samyn Y, Conand C (2023) Commercially important sea cucumbers of the world, 2nd edn. FAO Species Catalogue for Fishery Purposes No. 6, Rev. 1. Rome, FAO. p. 245 + viii
Purcell SW (2010) Managing sea cucumber fisheries with an ecosystem approach. FAO Fish Aquacult Tech Pap No. 520. FAO, Rome
Rodríguez-Barreras R, Serrano-Torres S, Macías-Reyes D (2014) A study of two tagging methods in the Caribbean sea cucumber Holothuria mexicana. Mar Biodivers Rec 7:118. https://doi.org/10.1017/S1755267214001171
Schlaff A, Menéndez P, Hall M, Heupel M, Armstrong T, Motti C (2020) Acoustic tracking of a large predatory marine gastropod, Charonia tritonis, on the Great Barrier Reef. Mar Ecol Prog Ser 642:147–161
Sharp WC, Hunt JH, Lyons WG (1997) Life history of the spotted spiny lobster, Panulirus guttatus, an obligate reef-dweller. Mar Freshwater Res 48:687–698
Shiell GR, Knott B (2008) Diurnal observations of sheltering behaviour in the coral reef sea cucumber Holothuria whitmaei. Fish Res 91:112–117. https://doi.org/10.1016/j.fishres.2007.12.010
Shiell GR, Knott B (2010) Aggregations and temporal changes in the activity and bioturbation contribution of the sea cucumber Holothuria whitmaei (Echinodermata: Holothuroidea). Mar Ecol Prog Ser 415:127–139. https://doi.org/10.3354/meps08685
Siegenthaler A, Cánovas A, González-Wangüemert M (2015) Spatial distribution patterns and movements of Holothuria arguinensis in the Ria Formosa (Portugal). J Sea Res 102:33–40. https://doi.org/10.1016/j.seares.2015.04.003
Skewes T, Smith L, Dennis D, Rawlinson N, Donovan A, Ellis N (2004) Conversion ratios for commercial beche-de-mer species in Torres Strait. AFMA Proj No. R02/1195 Final Rep, Torres Strait Research Program. Australian Fisheries Management Authority, Canberra
Tanita I, Hirohavi EB, Diau BA, Masaki K, Komatsu T, Ramofafia C (2022) Macro-and microhabitat use patterns of holothurians in Buena Vista, Nggela, Solomon Islands: rock climber and bottom crawler species. Mar Ecol Prog Ser 687:79–93
Teitelbaum CS, Mueller T (2019) Beyond migration: causes and consequences of nomadic animal movements. Trends Ecol Evol 34:569–581
Tessier E, Letouze P (2014) Mass spawning of Holothuria fuscopunctata in New Caledonia. SPC Beche-De-Mer Info Bull 34:54
Toral-Granda V, Lovatelli A, Vasconcellos M (2008) Sea cucumbers: a global review of fisheries and trade. FAO Fisheries Technical Paper. No. 516. Rome, FAO
Uthicke S (1994a) Spawning observations from the Lizard Island area. SPC Bêche-De-Mer Info Bull 6:12–14
Uthicke S (1994b) Spawning observations from Lizard Island area. SPC Beche-De-Mer Inf Bull 6:12–14
Uthicke S, Welch D, Benzie J (2004) Slow growth and lack of recovery in overfished holothurians on the Great Barrier Reef: evidence from DNA fingerprints and repeated large-scale surveys. Conserv Biol 18:1395–1404. https://doi.org/10.1111/j.1523-1739.2004.00309.x
Wolfe K, Byrne M (2017) Biology and ecology of the vulnerable holothuroid, Stichopus herrmanni, on a high-latitude coral reef on the Great Barrier Reef. Coral Reefs 36:1143–1156
Wolfe K, Byrne M (2022) Overview of the Great Barrier Reef sea cucumber fishery with focus on vulnerable and endangered species. Biol Conserv 266:109451
Acknowledgements
We thank the staff of the Lizard Island Research Station for facilitating the fieldwork. We also thank two anonymous reviewers for their constructive suggestions on the manuscript, which helped to improve the clarity of this article. This research was partially supported by the Marine Ecology Research Centre, Southern Cross University and conducted under Lizard Island Research Station collection permit GBRMPA G14/36625.1 and SAP permit GBRMPA G19/39553.1.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
SLR, ARH and SWP all declare that they have no conflicts of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. Within Australia, animal ethics permits are not required for research on echinoderms.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Purcell, S.W., Rallings, S.L. & Hammond, A.R. Long-term home ranging in the large sea cucumber, Holothuria fuscopunctata. Coral Reefs 42, 1059–1066 (2023). https://doi.org/10.1007/s00338-023-02413-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-023-02413-4