Abstract
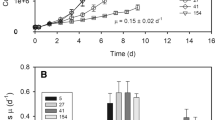
While global climate change in polar regions is expected to cause significant warming, the annual cycle of light and dark will remain unchanged. Cultures of three species of Antarctic sea ice diatoms, Fragilariopsis cylindrus (Grunow) Krieger, Thalassiosira antarctica Comber and Entomoneis kjellmanii (P.T. Cleve) Poulin and Cardinal, were incubated in the dark and exposed to differing temperatures. Maximum dark survival times varied between 30 and 60 days. Photosynthetic parameters, photosynthetic efficiency (α), maximum quantum yield (Fv/Fm), maximum relative electron transport rate (rETRmax) and non-photochemical quenching (NPQ), showed that dark exposure had a significant impact on photoacclimation. In contrast, elevated temperatures had a relatively minor impact on photosynthetic functioning during the dark exposure period but had a considerable impact on dark survival with minimal dark survival times reduced to only 7 days when exposed to 10°C. Recovery of maximum quantum yield of fluorescence (Fv/Fm) was not significantly impacted by temperature, species or dark exposure length. Recovery rates of Fv/Fm ranged from −5.06E−7 ± 2.71E−7 s−1 to 1.36E−5 ± 1.53E−5 s−1 for monthly experiments and from −9.63E−7 ± 7.71E−7 s−1 to 2.65E−5 ± 2.97E−5 s−1 for weekly experiments. NPQ recovery was greater and more consistent than Fv/Fm recovery, ranging between 5.74E−7 ± 8.11E−7 s−1 to 7.50E−3 ± 7.1E−4 s−1. The concentration of chl-a and monosaccharides remained relatively constant in both experiments. These results suggest that there will probably be little effect on Antarctic microalgae with increasing water temperatures during the Antarctic winter.





Similar content being viewed by others
References
Alderkamp AC, Buma AGJ, van Rijssel M (2007) The carbohydrates of Phaeocystis and their degradation in the microbial food web. Biogeochem 83:99–118
Andreoli C, Moro I, La Rocca N, Dalla Valle L, Masiero L, Rascio N, Dalla Vecchia F (2000) Ecological, physiological and biomolecular surveys on microalgae from Ross Sea (Antarctica). Ital J Zool 67:147–156
Antia NJ (1976) Effects of temperature on the darkness survival of marine microplanktonic algae. Microb Ecol 3:41–54
Antia NJ, Cheng JY (1970) The survival of axenic cultures of marine planktonic algae from prolonged exposure to darkness at 20°C. Phycol 9:179–183
Baldisserotto C, Ferroni L, Andreoli C, Fasulo MP, Bonora A, Pancaldi S (2005a) Dark-acclimation of the chloroplast in Koliella antarctica exposed to a simulated austral night condition. Arctic Antarctic Alpine Res 37:146–156
Baldisserotto C, Ferroni L, Moro I, Fasulo MP, Pancaldi S (2005b) Modulations of the thylakoid system in snow xanthophycean alga cultured in the dark for two months: comparison between microspectrofluorimetric responses and morphological aspects. Protoplasma 226:125–135
Bilger W, Björkman O (1990) Role of the xanthophyll cycle in photoprotection elucidated by measurements of light-induced absorbance changes, fluorescence and photosynthesis in leaves of Hedera canariensis. Photosynth Res 25:173–185
Botsford LW, Castilla JC, Peterson CH (1997) The management of fisheries and marine. Ecosys Sci 277:509–515
Bunt JS, Lee CC (1972) Data on the composition and dark survival of four sea-ice microalgae. Limnol Oceanog 17:458–461
Cullen JJ, Davis FF (2003) The blank can make a big difference in oceanographic measurements. Limnol Oceanograph Bull 12:29–35
Davison IR (1991) Environmental effects on algal photosynthesis: temperature. J Phycol 27:2–8
Demmig-Adams B, Adams WW (1992) Photoprotection and other responses of plants to high light stress. Ann Rev Plant Physiol Plant Mol Biol 43:599–626
Doucette GJ, Fryxell GA (1983) Thalassiosira antarctica: vegetative and resting stage chemical composition of an ice-related marine diatom. Marine Biol 78:1–6
Eicken H (1992) The role of sea ice in structuring antarctic ecosystems. Polar Biol 12:3–13
Falkowski PG, Raven JA (2007) Aquatic photosynthesis, 2nd edn. Blackwell Science, Oxford
Ferroni L, Baldisserotto C, Zennaro V, Soldani C, Fasulo M, Pancaldi S (2007) Acclimation to darkness in the marine chlorophyte Koliella antarctica cultured under low salinity: hypotheses on its origin in the polar environment. Eur J Phycol 42:91–104
Fiala M, Oriol L (1990) Light-temperature interactions on the growth of Antarctic diatoms. Polar Biol 10:629–636
Franklin DJ, Choi CJ, Hughes C, Malin G, Berges JA (2009) Effect of dead phytoplankton cells on the apparent efficiency of photosystem II. Mar Ecol Prog Ser 382:35–40
Granum E, Myklestad SM (1999) Effects of NH4 + assimilation on dark carbon fixation and β-1, 3-glucan metabolism in the marine diatom Skeletonema costatum (Bracillariophyceae). J Phycol 35:1191–1199
Griffith GP, Vennell R, Lamare MD (2009) Diadinoxanthin cycle of the bottom ice algal community in spring in McMurdo Sound, Antarctica. Polar Biol 32:623–636
Guillard RR (1975) Culture of phytoplankton for feeding marine invertebrates. In: Smith WL, Chanley MH (eds) Culture of marine invertebrate animals. Plenum Press, New York, pp 26–60
Gunnar Ö (1983) Effects of low temperature on photosynthesis. Plant Cell Environ 6:281–300
Holm-Hansen O, Riemann B (1978) Chlorophyll a determination: improvements in methodology. Oikos 30:438–447
Holm-Hansen O, Lorenzen CJ, Holmes RW, Strickland JDH (1965) Fluorometric determination of chlorophyll. ICES J Mar Sci 30:3
Hung CC, Santschi PH (2001) Spectrophotometric determination of total uronic acids in seawater using cation-exchange separation and pre-concentration lyophilization. Anal Chim Acta 427:111–117
Jasby AD, Platt T (1976) Mathematical formulation of the relationship between photosynthesis and light for phytoplankton. Limnol Oceanog 21:540–547
Knox GA (2006) Biology of the southern Ocean. CRC Press, Baton Roca
Kok B (1956) On the inhibition of photosynthesis by intense light. Biochim Biophys Acta 21:234
Kottmeier ST, Sullivan CW (1988) Sea ice microbial communities (SIMCO). IX. Effects of temperature and salinity on rates of metabolism and growth of autotrophs and heterotrophs. Polar Biol 5:293–304
Kudoh S, Imura S, Kashino Y (2003) Xanthophyll-cycle of ice algae on the sea ice bottom in Saroma Ko lagoon, Hokkaido, Japan. Polar Biosci 16:86–97
Lavaud J (2007) Fast regulation of photosynthesis in diatoms: mechanisms, evolution and ecophysiology. Funct Plant Sci Biotechnol 1:267–287
Lavaud J et al (2004) General features of photoprotection by energy dissipation in planktonic diatoms (Bacillariophyceae). J Phycol 40:130–137
Lizotte MP (2001) The contributions of sea ice algae to Antarctic marine primary production. Am Zool 41:57–73
Lizotte MP, Sullivan CW (1992) Biochemical-composition and photosynthate distribution in sea ice microalgae of McMurdo-Sound, Antarctica—evidence for nutrient stress during the spring bloom. Antarctic Sci 4:23–30
Lüder UH, Knoetzel J, Wiencke C (2001) Acclimation of photosynthesis and pigments to seasonally changing light conditions in the endemic Antarctic red macroalga Palmaria decipiens. Polar Biol 24:598–603
Lüder UH, Wiencke C, Knoetzel J (2002) Acclimation of photosynthesis and pigments during and after six months of darkness in Palmaria decipiens (Rhodophyta)-a study to simulate Antarctic winter sea ice cover. J Phycol 38:904–913
McConville MJ (1985) Chemical composition and biochemistry of sea ice microalgae. In: Horner RA (ed) Sea ice biota. CRC Press, Boca Raton, Florida, pp 105–129
McMinn A, Hattori H (2006) Effect of time of day on the recovery from light exposure in ice algae from Saroma Ko lagoon, Hokkaido. Polar Biosci 20:30–36
McMinn A, Hegseth EN (2004) Quantum yield and photosynthetic parameters of marine microalgae from the southern Arctic Ocean, Svalbard. J Mar Biol Assoc UK 84:865–871
McMinn A, Martin A, Ryan K (2010) Phytoplankton and sea ice biomass and physiology during the transition between winter and spring (McMurdo Sound, Antarctica). Polar Biol 33:1547–1556
Meiners K, Papadimitriou S, Thomas D, Norman L, Dieckmann G (2009) Biogeochemical conditions and ice algal photosynthetic parameters in Weddell Sea ice during early spring. Polar Biol 32:1055–1065
Mock T, Hoch N (2005) Long-term temperature acclimation of photosynthesis in steady-state cultures of the polar diatom Fragilariopsis cylindrus. Photosynth Res 85:307–317
Morgan-Kiss RM, Ivanov AG, Williams J, Khan M, Huner NPA (2002) Differential thermal effects on the energy distribution between photosystem II and photosystem I in thylakoid membranes of a psychrophilic and a mesophilic alga. BBA Biomembranes 1561:251–265
Morgan-Kiss RM, Priscu JC, Pocock T, Gudynaite-Savitch L, Huner NPA (2006) Adaptation and acclimation of photosynthetic microorganisms to permanently cold environments. Microbiol Mol Biol Rev 70:222–252
Myklestad SM, Skånøy E, Hestmann S (1997) A sensitive and rapid method for analysis of dissolved mon- and polysaccharides in seawater. Mar Chem 56:279–286
Oeltjen A, Krumbein WE, Rhiel E (2002) Investigations on transcript sizes, steady state mRNA concentrations and diurnal expression of genes encoding fucoxanthin chlorophyll a/c light harvesting polypeptides in the centric diatom Cyclotella cryptica. Plant Biol 4:250–257
Oliver RL, Whittington J, Lorenz Z, Webster IT (2003) The influence of vertical mixing on the photoinhibition of variable chlorophyll a fluorescence and its inclusion in a model of phytoplankton photosynthesis. J Plank Res 25:1107–1129
Oquist G, Huner NPA (2003) Photosynthesis of overwintering evergreen plants. Ann Rev Plant Biol 54:329–355
Palmisano AC, Sullivan CW (1982) Physiology of sea ice diatoms. 1. Response of 3 polar diatoms to a simulated summer-winter transition. J Phycol 18:489–498
Palmisano AC, Sullivan CW (1983) Sea ice microbial communities (SIMCO) I. Distribution, abundance and primary production of microalgae in McMurdo Sound, Antarctica. J Phycol 18:489–498
Palmisano AC, Soohoo JB, Sullivan CW (1985) Photosynthesis-irradiance relationships in sea ice microalgae from McMurdo Sound, Antarctica. J Phycol 21:341–346
Panagiotopoulos C, Sempéré R (2005) Analytical methods for the determination of sugars in marine samples: a historical perspective and future directions. Limnol Oceanog 3:419–454
Pankowski A, McMinn A (2009) Development of immunoassays for the iron- regulated proteins Ferredoxin and flavodoxin in polar microalgae. J Phycol 45:771–783
Peck LS (2005) Prospects for surviving climate change in Antarctic aquatic species. Frontiers Zool 2:2–9
Peters E (1996) Prolonged darkness and diatom mortality: II. Marine temperate species. J Experimen Mar Biol Ecol 207:43–58
Peters E, Thomas DN (1996) Prolonged darkness and diatom mortality I: Marine Antarctic species. J Experimen Mar Biol Ecol 207:25–41
Platt T, Gallegos CL, Harrison WG (1980) Photoinhibition of photosynthesis in natural assemblages of marine phytoplankton. J Mar Res 38:687–701
Ralph PJ, Gademann R (2005) Rapid light curves: a powerful tool to assess photosynthetic activity. Aquatic Bot 82:222–237
Ralph PJ, McMinn A, Ryan KG, Ashworth C (2005) Short-term effect of temperature on the photokinetics of microalgae from the surface layers of Antarctic pack ice. J Phycol 41:763–769
Schreiber U (2004) Pulse-amplitude-modulation (PAM) fluorometry and saturation pulse method: an overview. In: Papageorgiou GC (ed) Chlorophyll a fluorescence a signature of photosynthesis. Springer, Dordrecht, The Netherlands, pp 279–319
Thomas DN, Dieckmann GS (2002) Antarctic sea ice—a habitat for extremophiles. Science 295:641–644
Trenerry LJ, McMinn A, Ryan KG (2002) In situ oxygen microelectrode measurements of bottom-ice algal production in McMurdo Sound, Antarctica. Polar Biol 25:72–80
Turner J, Colwell SR, Marshall GJ, Lachlan-Cope TA, Carleton AM, Jones PD, Lagun V, Reid PA, Iagovkina S (2005) Antarctic climate change during the last 50 years’. Intern J Climatol 25:279–294
van Oijen T (2004) Iron and light limitation of carbohydrate production by phytoplankton in the Southern Ocean. Doctoral thesis. University of Groningen, The Netherlands
van Oijen T, van Leeuwe MA, Gieskes WWC (2003) Variation of particulate carbohydrate pools over time and depth in a diatom-dominated plankton community at the Antarctic Polar Front. Polar Biol 26:195–201
van Oijen T, Veldhuis MJW, Gorbunov MY, Nishioka J, van Leeuwe MA, de Baar HJW (2005) Enhanced carbohydrate production by Southern Ocean phytoplankton in response to in situ iron fertilization. Mar Chem 93:33–52
Wulff A, Roleda MY, Zacher K, Wiencke C (2008) Exposure to sudden light burst after prolonged darkness—a case study on benthic diatoms in Antarctica. Diatom Res 23:519–532
Acknowledgments
Funding for this project was from an Australian Research Council (ARC) Discovery Grant. We would also like to thank Helen Bond (IMAS) for assistance with culturing.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Reeves, S., McMinn, A. & Martin, A. The effect of prolonged darkness on the growth, recovery and survival of Antarctic sea ice diatoms. Polar Biol 34, 1019–1032 (2011). https://doi.org/10.1007/s00300-011-0961-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-011-0961-x