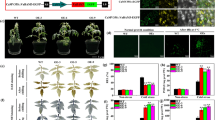
Abstract
Key message
Abscisic acid induced the expression of AsKIN during the recovery period of garlic cryopreservation. AsKIN was identified as a gene involved in cold and osmotic stress resistance.
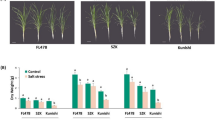
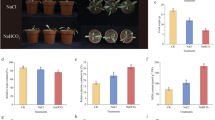
Cryopreservation has been proven to be effective in removing viruses from garlic. However, oxidative damage in cryopreservation has a significant impact on the survival after preservation. Abscisic acid (ABA) has been shown to reduce oxidative stress and promote the survival after cryopreservation. However, it is not clear which genes play important roles in this process. In this study, we added ABA to the dehydration step and analyzed the transcriptomic divergences between the ABA-treated group and the control group in three cryogenic steps (dehydration, unloading and recovery). By short time-series expression miner (STEM) analysis and weighted gene co-expression network analysis (WGCNA), the recovery step was identified as the period of significant changes in gene expression levels in cryopreservation. The addition of ABA promoted the upregulated expression of microtubule-related genes in the recovery step. We further identified AsKIN as a hub gene in the recovery step and verified its function. The results showed that overexpression of AsKIN enhanced the tolerance of Arabidopsis to oxidative stress in cryopreservation, influenced the expression of genes in response to cold and osmotic stress and promoted plant growth after stress. The AsKIN gene is likely to be involved in the plant response to cold stress and osmotic stress. These results reveal the molecular mechanisms of ABA in cryopreservation and elucidate the potential biological functions of the kinesin-14 subfamily.










Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the paper and within its supplementary materials published online.
References
Achard P, Gong F, Cheminant S, Alioua M, Hedden P, Genschik P (2008) The cold-inducible CBF1 factor-dependent signaling pathway modulates the accumulation of the growth-repressing DELLA proteins via its effect on gibberellin metabolism. Plant Cell 20:2117–2129
Ai TN, Naing AH, Yun BW, Lim SH, Kim CK (2018) Overexpression of RsMYB1 enhances anthocyanin accumulation and heavy metal stress tolerance in transgenic Petunia. Front Plant Sci 9:1388
Ashraf M (2010) Inducing drought tolerance in plants: recent advances. Biotechnol Adv 28:169–183
Baker SS, Wilhelm KS, Thomashow MF (1994) The 5′-region of Arabidopsis thaliana cor15a has cis-acting elements that confer cold-, drought-and ABA-regulated gene expression. Plant Mol Biol 24:701–713
Ballizany WL, Hofmann RW, Jahufer MZ, Barrett BA (2012) Multivariate associations of flavonoid and biomass accumulation in white clover (Trifolium repens) under drought. Funct Plant Biol 39:167–177
Banday MN, Lone FA, Rasool F, Rashid M, Shikari A (2017) Use of antioxidants reduce lipid peroxidation and improve quality of crossbred ram sperm during its cryopreservation. Cryobiology 74:25–30
Brunakova K, Zamecnik J, Urbanová M, Cellárová E (2011) Dehydration status of ABA-treated and cold-acclimated Hypericum perforatum L. shoot tips subjected to cryopreservation. Thermochim Acta 525:62–70
Burritt D (2008) Efficient cryopreservation of adventitious shoots of Begonia x erythrophylla using encapsulation-dehydration requires pretreatment with both ABA and proline. Plant Cell, Tissue Organ Cult 95:209–215
Chaves M, Flexas J, Pinheiro C (2008) Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell. Ann Bot 103:551–560
Chen C, Marcus A, Li W, Hu Y, Calzada JPV, Grossniklaus U, Cyr RJ, Ma H (2002) The Arabidopsis ATK1 gene is required for spindle morphogenesis in male meiosis. Development 129:2401–2409
Chen GQ, Ren L, Zhang D, Shen XH (2016) Glutathione improves survival of cryopreserved embryogenic calli of Agapanthus praecox subsp. orientalis. Acta Physiol Plant 38:1–12
Chinnusamy V, Ohta M, Kanrar S, Lee BH, Hong X, Agarwal M, Zhu JK (2003) ICE1: a regulator of cold-induced transcriptome and freezing tolerance in Arabidopsis. Genes Dev 17:1043–1054
Chinnusamy V, Zhu J, Zhu JK (2007) Cold stress regulation of gene expression in plants. Trends Plant Sci 12:444–451
Choi HI, Hong JH, Ha JO, Kang JJ, Kim SY (2000) ABFs, a family of ABA-responsive element binding factors. J Biol Chem 275:1723–1730
Cutler S, Rodriguez P, Finkelstein R, Abrams S (2010) Abscisic acid: emergence of a core signaling network. Annu Rev Plant Biol 61:651–679
Danova K (2010) Production of polyphenolic compounds in shoot cultures of Hypericum species characteristic for the Balkan flora. Bot Serb 34:29–36
Danova K, Denev B, Markovska Y (2012) Impact of pre-culture on short and long term in vitro recovery of the biosynthetic potential and enzymatic and non-enzymatic antioxidant defense of Hypericum rumeliacum Boiss after cryostorage. Plant Growth Regul 68:447–457
Gao C, Zhang K, Guiyan Y, Wang Y (2012) Expression analysis of four peroxiredoxin genes from Tamarix hispida in response to different abiotic stresses and exogenous abscisic Acid (ABA). Int J Mol Sci 13:3751–3764
Georgieva E, Petrova DH, Yordanova ZP, Kapchina-Toteva V, Cellárová E, Chaneva G (2014) Influence of cryopreservation on the antioxidative activity of in vitro cultivated Hypericum species. Biotechnol Biotechnol Equip 28:863–870
Gicking A, Swentowsky K, Dawe RK, Qiu W (2018) Functional diversification of the kinesin-14 family in land plants. FEBS Lett 592:1918–1928
Gilmour SJ, Artus NN, Thomashow MF (1992) cDNA sequence analysis and expression of two cold-regulated genes of Arabidopsis thaliana. Plant Mol Biol 18:13–21
Hazubska-Przybył T, Chmielarz P, Michalak M, Dering M, Bojarczuk K (2013) Survival and genetic stability of Picea abies embryogenic cultures after cryopreservation using a pregrowth-dehydration method. Plant Cell, Tissue Organ Cult 113:303–313
Heald R (2000) Motor function in the mitotic spindle minireview. Cell 102:399–402
Higgins DM, Nannas NJ, Dawe RK (2016) The maize divergent spindle-1 (dv1) gene encodes a kinesin-14A motor protein required for meiotic spindle pole organization. Front Plant Sci 7:1277
Hirokawa N, Noda Y, Tanaka Y, Niwa S (2009) Kinesin superfamily motor proteins and intracellular transport. Nat Rev Mol Cell Biol 10:682–696
Hu X, Jiang M, Zhang A, Lu J (2006) Abscisic acid-induced apoplastic H2O2 accumulation up-regulates the activities of chloroplastic and cytosolic antioxidant enzymes in maize leaves. Planta 223:57–68
Huang B, Zhang JM, Chen XL, Xin X, Yin GK, He JJ, Lu XX, Zhou YC (2018) Oxidative damage and antioxidative indicators in 48 h germinated rice embryos during the vitrification–cryopreservation procedure. Plant Cell Rep 37:1325–1342
Huang Y, Wang H, Huang X, Wang Q, Wang J, An D, Li J, Wang W, Wu Y (2019) Maize VKS1 regulates mitosis and cytokinesis during early endosperm development. Plant Cell 31:1238–1256
Hughes NM, Carpenter KL, Cannon JG (2013) Estimating contribution of anthocyanin pigments to osmotic adjustment during winter leaf reddening. J Plant Physiol 170:230–233
Hwarari D, Guan Y, Ahmad B, Movahedi A, Min T, Hao Z, Lu Y, Chen J, Yang L (2022) ICE-CBF-COR signaling cascade and its regulation in plants responding to cold stress. Int J Mol Sci 23:1549
Jiang M, Zhang J (2002) Role of abscisic acid in water stress-induced antioxidant defense in leaves of maize seedlings. Free Radic Res 36:1001–1015
John R, Anjum N, Sopory S, Akram N, Ashraf M (2016) Some key physiological and molecular processes of cold acclimation. Biol Plant 60:603–618
Kong L, von Aderkas P (2011) A novel method of cryopreservation without a cryoprotectant for immature somatic embryos of conifer. Plant Cell, Tissue Organ Cult 106:115–125
Kurkela S, Borg-Franck M (1992) Structure and expression of kin2, one of two cold- and ABA-induced genes of Arabidopsis thaliana. Plant Mol Biol 19:689–692
Landi M, Guidi L, Pardossi A, Tattini M, Gould KS (2014) Photoprotection by foliar anthocyanins mitigates effects of boron toxicity in sweet basil (Ocimum basilicum). Planta 240:941–953
Lawrence CJ, Dawe RK, Christie KR, Cleveland DW, Dawson SC, Endow SA, Goldstein LS, Goodson HV, Hirokawa N, Howard J (2004) A standardized kinesin nomenclature. J Cell Biol 167:19–22
Len JS, Koh WSD, Tan SX (2019) The roles of reactive oxygen species and antioxidants in cryopreservation. Biosci Rep 39:BSR20191601
Li M, Duan X, Gao G, Liu T, Qi H (2022) Running title: ABA pathway meets CBF pathway at CmADC. Hortic Res 9:uhac002
Liu X, Mou S, Cheng Z (2019) Effect of cryopreservation on plant growth, bulb characteristics, and virus reduction of garlic (Allium sativum L.). Cryo Lett 40:322–332
Lu ZW, Popova E, Wu CH, Lee EJ, Hahn E, Paek K (2009) Cryopreservation of Ginkgo biloba cell culture: effect of pretreatment with sucrose and ABA. Cryo Letters 30:232–243
Ma H, Liu M (2019) The microtubule cytoskeleton acts as a sensor for stress response signaling in plants. Mol Biol Rep 46:5603–5608
Marcus AI, Ambrose JC, Blickley L, Hancock WO, Cyr RJ (2002) Arabidopsis thaliana protein, ATK1, is a minus-end directed kinesin that exhibits non-processive movement. Cell Motil Cytoskelet 52:144–150
Mathew L, Burritt DJ, McLachlan A, Pathirana R (2019) Combined pre-treatments enhance antioxidant metabolism and improve survival of cryopreserved kiwifruit shoot tips. Plant Cell, Tissue Organ Cult 138:193–205
Naing AH, Kim CK (2021) Abiotic stress-induced anthocyanins in plants: their role in tolerance to abiotic stresses. Physiol Plant 172:1711–1723
Naing AH, Park KI, Ai TN, Chung MY, Han JS, Kang YW, Lim KB, Kim CK (2017) Overexpression of snapdragon Delila (Del) gene in tobacco enhances anthocyanin accumulation and abiotic stress tolerance. BMC Plant Biol 17:65
Naing AH, Ai T, Lee I, Lim KB, Kim C (2018) Overexpression of Rosea1 from snapdragon enhances anthocyanin accumulation and abiotic stress tolerance in transgenic tobacco. Front Plant Sci 9:1070
Nakabayashi R, Yonekura-Sakakibara K, Urano K, Suzuki M, Yamada Y, Nishizawa T, Matsuda F, Kojima M, Sakakibara H, Shinozaki K (2014) Enhancement of oxidative and drought tolerance in Arabidopsis by overaccumulation of antioxidant flavonoids. Plant J 77:367–379
Nakashima K, Yamaguchi-Shinozaki K, Shinozaki K (2014) The transcriptional regulatory network in the drought response and its crosstalk in abiotic stress responses including drought, cold, and heat. Front Plant Sci 5:170
Nayyar H, Bains T, Kumar S (2005) Low temperature induced floral abortion in chickpea: relationship to abscisic acid and cryoprotectants in reproductive organs. Environ Exp Bot 53:39–47
Parrano L, Afunian M, Pagliaccia D, Douhan G, Vidalakis G (2012) Characterization of viruses associated with garlic plants propagated from different reproductive tissues from Italy and other geographic regions. Phytopathol Mediterr 51:549–565
Petrussa E, Braidot E, Zancani M, Peresson C, Bertolini A, Patui S, Vianello A (2013) Plant flavonoids-biosynthesis, transport and involvement in stress responses. Int J Mol Sci 14:14950–14973
Pospíšilová J (2003) Participation of phytohormones in the stomatal regulation of gas exchange during water stress. Biol Plant 46:491–506
Raghavendra A, Gonugunta V, Christmann A, Grill E (2010) ABA perception and signaling. Trends Plant Sci 15:395–401
Ren L, Zhang D, Jiang XN, Gai Y, Wang WM, Reed BM, Shen XH (2013a) Peroxidation due to cryoprotectant treatment is a vital factor for cell survival in Arabidopsis cryopreservation. Plant Sci 212:37–47
Ren L, Zhang D, Chen GQ, Reed BM, Shen XH, Chen HY (2015) Transcriptomic profiling revealed the regulatory mechanism of Arabidopsis seedlings response to oxidative stress from cryopreservation. Plant Cell Rep 34:2161–2178
Ren L, Wang MR, Wang QC (2021) ROS-induced oxidative stress in plant cryopreservation: occurrence and alleviation. Planta 254:1–18
Ren L, Zhang D, Shen X, Reed B (2013b) Antioxidants and anti-stress compounds improve the survival of cryopreserved Arabidopsis seedlings. In: II International Symposium on Plant Cryopreservation 1039, pp 57-62
Richardson DN, Simmons MP, Reddy ASN (2006) Comprehensive comparative analysis of kinesins in photosynthetic eukaryotes. BMC Genom 7:18–18
Sablin EP, Jon Kull F, Cooke R, Vale RD, Fletterick RJ (1996) Crystal structure of the motor domain of the kinesin-related motor ncd. Nature 380:555–559
Sah SK, Reddy KR, Li J (2016) Abscisic acid and abiotic stress tolerance in crop plants. Front Plant Sci 7:571–571
Seung D, Webster MW, Wang R, Andreeva Z, Marc J (2013) Dissecting the mechanism of abscisic acid-induced dynamic microtubule reorientation using live cell imaging. Funct Plant Biol 40:224–236
Shao H, Chu LY, Lu ZH, Kang CM (2008) Primary antioxidant free radical scavenging and redox signaling pathways in higher plant cells. Int J Biol Sci 4:8–14
She ZY, Yang WX (2017) Molecular mechanisms of kinesin-14 motors in spindle assembly and chromosome segregation. J Cell Sci 130:2097–2110
Shu Y, Li W, Zhao J, Zhang S, Xu H, Liu Y, Guo C (2017) Transcriptome sequencing analysis of alfalfa reveals CBF genes potentially playing important roles in response to freezing stress. Genet Mol Biol 40:824–833
Stockinger EJ, Gilmour SJ, Thomashow MF (1997) Arabidopsis thaliana CBF1 encodes an AP2 domain-containing transcriptional activator that binds to the C-repeat/DRE, a cis-acting DNA regulatory element that stimulates transcription in response to low temperature and water deficit. Proc Natl Acad Sci U S A 94:1035–1040
Takatani S, Hirayama T, Hashimoto T, Takahashi T, Motose H (2015) Abscisic acid induces ectopic outgrowth in epidermal cells through cortical microtubule reorganization in Arabidopsis thaliana. Sci Rep 5:11364–11364
Tan ST, Dai C, Liu HT, Xue HW (2013) Arabidopsis casein kinase1 proteins CK1.3 and CK1.4 phosphorylate cryptochrome2 to regulate blue light signaling. Plant Cell 25:2618–2632
Taylor L, Grotewold E (2005) Flavonoids as developmental regulators. Curr Opin Plant Biol 8:317–323
Tian J, Han L, Feng Z, Wang G, Liu W, Ma Y, Yu Y, Kong Z (2015) Orchestration of microtubules and the actin cytoskeleton in trichome cell shape determination by a plant-unique kinesin. Elife 4:e09351
Toldi O, Tuba Z, Scott P (2009) Vegetative desiccation tolerance: Is it a goldmine for bioengineering crops? Plant Sci 176:187–199
Tseng KF, Wang P, Lee YR, Bowen J, Gicking A, Guo L, Liu B, Qiu W (2018) The preprophase band-associated kinesin-14 OsKCH2 is a processive minus-end-directed microtubule motor. Nat Commun 9:1067
Vale RD, Reese TS, Sheetz MP (1985) Identification of a novel force-generating protein, kinesin, involved in microtubule-based motility. Cell 42:39–50
Vieira RL, da Silva AL, Zaffari GR, Steinmacher DA, de Freitas Fraga HP, Guerra MP (2014) Efficient elimination of virus complex from garlic (Allium sativum L.) by cryotherapy of shoot tips. Acta Physiol. Plant 37:1733
Wang H, Georges F, Pelcher LE, Saleem M, Cutler AJ (1994) A 5.3-kilobase genomic fragment from Arabidopsis thaliana containing kin1 and cor6.6. Plant Physiol 104:291–292
Wang F, Kong W, Wong GKP, Fu L, Peng RH, Li ZJ, Yao Q (2016) AtMYB12 regulates flavonoids accumulation and abiotic stress tolerance in transgenic Arabidopsis thaliana. Mol Genet Genom 291:1545–1559
Wang F, Ren G, Li F, Qi S, Xu Y, Wang B, Yang Y, Ye Y, Zhou Q, Chen X (2018a) A chalcone synthase gene AeCHS from Abelmoschus esculentus regulates flavonoid accumulation and abiotic stress tolerance in transgenic Arabidopsis. Acta Physiol Plant 40:1–13
Wang MR, Chen L, Zhang Z, Blystad DR, Wang QC (2018b) Cryotherapy: a novel method for virus eradication in economically important plant species. Plant cell culture protocols. Springer, pp 257–268
Wang GL, Ren XQ, Liu JX, Yang F, Wang YP, Xiong AS (2019a) Transcript profiling reveals an important role of cell wall remodeling and hormone signaling under salt stress in garlic. Plant Physiol Biochem 135:87–98
Wang L, Sadeghnezhad E, Riemann M, Nick P (2019b) Microtubule dynamics modulate sensing during cold acclimation in grapevine suspension cells. Plant Sci 280:18–30
Wang X, Song Q, Liu Y, Brestic M, Yang X (2022) The network centered on ICEs play roles in plant cold tolerance, growth and development. Planta 255:81
Winkel B (2001) Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol 126:485–493
Winkel-Shirley B (2002) Biosynthesis of flavonoids and effects of stress. Curr Opin Plant Biol 5:218–223
Wisniewski M, Norelli J, Artlip T (2015) Overexpression of a peach CBF gene in apple: a model for understanding the integration of growth, dormancy, and cold hardiness in woody plants. Front Plant Sci 6:85
Xing W, Rajashekar C (2001) Glycine betaine involvement in freezing tolerance and water stress in Arabidopsis thaliana. Environ Exp Bot 46:21–28
Xiong L, Zhu JK (2003) Regulation of abscisic acid biosynthesis. Plant Physiol 133:29–36
Xu Z, Mahmood K, Rothstein S (2017) ROS Induces anthocyanin production via late biosynthetic genes and anthocyanin deficiency confers the hypersensitivity to ROS-generating stresses in Arabidopsis. Plant Cell Physiol 58:1364–1377
Yoshida K, Igarashi E, Mukai M, Hirata K, Miyamoto K (2003) Induction of tolerance to oxidative stress in the green alga, Chlamydomonas reinhardtii, by abscisic acid. Plant Cell Environ 26:451–457
Zandkarimi H, Ebadi A, Salami SA, Alizade H, Baisakh N (2015) Analyzing the expression profile of AREB/ABF and DREB/CBF genes under drought and salinity stresses in grape (Vitis vinifera L.). PloS one 10:e0134288
Zhang X, Henriques R, Lin SS, Niu QW, Chua NH (2006) Agrobacterium-mediated transformation of Arabidopsis thaliana using the floral dip method. Nat Protoc 1:641–646
Zhang D, Ren L, Chen GQ, Zhang J, Reed BM, Shen XH (2015) ROS-induced oxidative stress and apoptosis-like event directly affect the cell viability of cryopreserved embryogenic callus in Agapanthus praecox. Plant Cell Rep 34:1499–1513
Zhang Q, Zhai J, Shao L, Lin W, Peng C (2019) Accumulation of anthocyanins: an adaptation strategy of Mikania micrantha to low temperature in winter. Front Plant Sci 10:1049
Zhang D, Yang T, Ren L (2021) Y2SK2-and SK3-type dehydrins from Agapanthus praecox act as protectants to improve plant cell viability during cryopreservation. Plant Cell, Tissue Organ Cult 144:271–279
Zhou S, Chen Q, Li X, Li Y (2017) MAP65-1 is required for the depolymerization and reorganization of cortical microtubules in the response to salt stress in Arabidopsis. Plant Sci 264:112–121
Zhu H, Zhang TJ, Zheng J, Huang XD, Yu ZC, Cl P, Chow WS (2017) Anthocyanins function as a light attenuator to compensate for insufficient photoprotection mediated by nonphotochemical quenching in young leaves of Acmena acuminatissima in winter. Photosynthetica 56:445–454
Acknowledgements
The research was supported by National Natural Science Foundation of China (31372056; 31872125), Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), Fundamental Research Funds for the Central Universities (KJFP201702).
Funding
This study was funded by National Natural Science Foundation of China (31372056; 31872125), Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD) and Fundamental Research Funds for the Central Universities (KJFP201702).
Author information
Authors and Affiliations
Contributions
ZW and XDX initiated and designed the research; XDX, YHB, HYW, DZ and JJW performed the experiments; XDX analyzed the data; ML contributed reagents/materials/analysis tools; XDX wrote the paper; ZW, FLJ and RZ revised the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by Zheng-Yi Xu.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xing, X., Liu, M., Jiang, F. et al. Abscisic acid induces the expression of AsKIN during the recovery period of garlic cryopreservation. Plant Cell Rep 41, 1955–1973 (2022). https://doi.org/10.1007/s00299-022-02894-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-022-02894-7