Abstract
Key message
Two RNAi constructs were designed targeting chilli-infecting begomoviruses and associated betasatellites. Broad-spectrum resistance was achieved against multiple begomoviruses associated with leaf curl disease of chillies in India.
Abstract
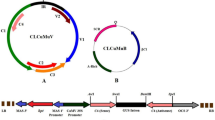
Chilli leaf curl disease (ChiLCD) caused by begomoviruses (family: Geminiviridae) has emerged as one of the most devastating viral diseases of chilli, especially in the Indian sub-continent. The severity of disease incidence is expanding at an alarming rate due to the emergence of new begomoviruses with greater ability to infect this crop in almost all the major chilli producing regions of India. In this study, we applied the RNA interference (RNAi) based strategies to control infection of chilli-infecting begomoviruses (CIBs). For this, we have generated transgenic Nicotiana benthamiana plants harboring two different intron hairpin RNAi constructs [designated as TR1 (AC1/AC2) and TR2 (AC1/AC2/βC1)] using conserved regions of viral genome and associated betasatellite. During our study, we observed that, two lines harboring TR1 construct (13-1 and 2-4) and one line harboring TR2 construct (5-1) have shown resistance to the most predominant Indian CIBs like Chilli leaf curl virus-Pakistan isolate Varanasi, Tomato leaf curl New Delhi virus-isolate chilli, and a newly identified begomovirus species, Chilli leaf curl Vellanad virus. Resistant lines accumulated transgene-specific siRNAs, confirming RNAi-mediated resistance against these viruses. Furthermore, these resistant lines also displayed delayed symptom appearance and milder symptoms, as compared to virus-inoculated non-transgenic plants. Average viral DNA accumulation in the resistant lines was reduced up to 90 % as compared to non-transgenic plants. Thus, our study demonstrated the application of RNAi-mediated approach in providing resistance against diverse monopartite and bipartite begomoviruses associated with ChiLCD.






Similar content being viewed by others
References
Akhter A, Qazi J, Saeed M, Mansoor S (2009) A severe leaf curl disease on chilies in Pakistan is associated with multiple begomovirus components. Plant Dis 93:962
Basu S, Sharma VK, Bhattacharyya D, Chakraborty S (2014) An overview of antiviral RNA silencing in plant: biogenesis, host–virus interaction and potential applications. In: Gaur RK, Sharma P (eds) Approaches to plant stress and their management. Springer, India, pp 317–337
Bonfim K, Faria JC, Nogueira EO, Mendes EA, Aragao FJ (2007) RNAi-mediated resistance to Bean golden mosaic virus in genetically engineered common bean (Phaseolus vulgaris). Mol Plant Microbe Interact 20:717–726
Chakraborty S, Vanitharani R, Chattopadhyay B, Fauquet CM (2008) Supervirulent pseudorecombination and asymmetric synergism between genomic components of two distinct species of begomovirus associated with severe tomato leaf curl disease in India. J Gen Virol 89:818–828
Chattopadhyay B, Singh AK, Yadav T, Fauquet CM, Sarin NB et al (2008) Infectivity of the cloned components of a begomovirus: DNA beta complex causing chilli leaf curl disease in India. Arch Virol 153:533–539
Chellappan P, Masona MV, Vanitharani R, Taylor NJ, Fauquet CM (2004) Broad spectrum resistance to ssDNA viruses associated with transgene-induced gene silencing in cassava. Plant Mol Biol 56:601–611
Cheng X, Wang X, Wu J, Briddon RW, Zhou X (2011) βC1 encoded by Tomato yellow leaf curl China betasatellite forms multimeric complexes in vitro and in vivo. Virology 409:156–162
Chyi Y-S, Jorgensen RA, Goldstein D, Tanksley SD, Loaiza-Figueroa F (1986) Locations and stability of Agrobacterium-mediated T-DNA insertions in the Lycopersicon genome. Mol Gen Genet 204:64–69
Cui X, Li G, Wang D, Hu D, Zhou X (2005) A begomovirus DNAβ-encoded protein binds DNA, functions as a suppressor of RNA silencing, and targets the cell nucleus. J Virol 79:10764–10775
Dellaporta SL, Wood J, Hicks JB (1983) A plant DNA minipreparation: Version II. Plant Mol Biol Rep 1:19–21
Dhanraj KS, Seth ML (1968) Enation in Capsicum annuum L. (chilli) caused by a new strain of leaf-curl virus. Indian J Hortic 25:70–71
Dong X, van Wezel R, Stanley J, Hong Y (2003) Functional characterization of the nuclear localization signal for a suppressor of posttranscriptional gene silencing. J Virol 77:7026–7033
Elmer JS, Brand L, Sunter G, Gardiner WE, Bisaro DM et al (1988) Genetic analysis of the tomato golden mosaic virus. II. The product of the AL1 coding sequence is required for replication. Nucleic Acids Res 16:7043–7060
Fedorov Y, Anderson EM, Birmingham A, Reynolds A, Karpilow J, Robinson K, Leake D, Marshall WS, Khvorova A (2006) Off-target effects by siRNA can induce toxic phenotype. RNA 12:1188–1196
Feinberg AP, Vogelstein B (1983) A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem 132:6–13
Guo W, Jiang T, Zhang X, Li G, Zhou X (2008) Molecular variation of satellite dna molecules associated with malvastrum yellow vein virus and their role in pathogenicity. Appl Environ Microbiol 74:1909–1913
Haley A, Zhan X, Richardson K, Head K, Morris B (1992) Regulation of the activities of African cassava mosaic virus promoters by the AC1, AC2, and AC3 gene products. Virology 188:905–909
Hamilton AJ, Baulcombe D (1999) A species of small antisense RNA in posttranscriptional gene silencing in plants. Science 286:950–952
Hofgen R, Willmitzer L (1988) Storage of competent cells for Agrobacterium transformation. Nucleic Acids Res 16:9877
Horsch RB, Fry JE, Hoffmann NL, Eichholtz D, Rogers SG, Tb Fraley R (1985) A simple and general method for transferring genes into plants. Science 227:1229–1231
Jackson AL, Bartz SR, Schelter J, Kobayashi SV, Burchard J, Mao M, Li B, Cavet G, Linsley PS (2003) Expression profiling reveals off-target gene regulation by RNAi. Nat Biotechnol 21:635–637
Jones L, Ratcliff F, Baulcombe DC (2011) RNA-directed transcriptional gene silencing in plants can be inheritated independently of the RNA trigger and requires Met1 for maintenance. Curr Biol 11(10):747–757
Khan MS, Raj SK, Singh R (2006) First report of tomato leaf curl New Delhi virus infecting chilli in india. Plant Pathol 55:289
Kumar Y, Hallan V, Zaidi AA (2011) Chilli leaf curl Palampur virus is a distinct begomovirus species associated with a betasatellite. Plant Pathol 60:1040–1047
Kumar RV, Sharma VK, Chattopadhyay B, Chakraborty S (2012) An improved plant regeneration and Agrobacterium-mediated transformation of red pepper (Capsicum annuum L.). Physiol Mol Biol Plants 18:357–364
Kumari P, Singh AK, Sharma VK, Chattopadhyay B, Chakraborty S (2011) A novel recombinant tomato-infecting begomovirus capable of transcomplementing heterologous DNA-B components. Arch Virol 156:769–783
Kushwaha N, Sahu PP, Prasad M, Chakraborty S (2015) Chilli leaf curl virus infection highlights the differential expression of genes involved in protein homeostasis and defense in resistant chilli plants. Appl Microbiol Biotech. doi:10.1007/s00253-015-6415-6
Li F, Huang C, Li Z, Zhou X (2014) Suppression of RNA silencing by a plant DNA virus satellite requires a host calmodulin-like protein to repress RDR6 expression. PLoS Pathog 10:e1003921
Liu W, Parrott WA, Hildebrand DF, Collins GB, Williams EG (1990) Agrobacterium induced gall formation in bell pepper (Capsicum annuum L.) and formation of shoot-like structures expressing introduced genes. Plant Cell Rep 9:360–364
MacNeish RS (1964) Ancient mesoamerican civilization. Science 143:531–537
Mette MF, Aufsatz W, van der Winden J, Matzke MA, Matzke AJM (2000) Transcriptional silencing and promoter methylation triggered by double-stranded RNA. EMBO J 19:5194–5201
Mishra MD, Raychaudhari SP, Jha A (1963) Virus causing leaf curl of chilli (Capsicum annuum L.). Indian J Microbiol 3:73–76
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Nahid N, Amin I, Briddon RW, Mansoor S (2011) RNA interference-based resistance against a legune mastrevirus. Virol J 8:499
Patil BL, Ogwok E, Wagaba H, Mohammed IU, Yadav JS et al (2011) RNAi-mediated resistance to diverse isolates belonging to two virus species involved in Cassava brown streak disease. Mol Plant Pathol 12:31–41
Powell PA, Sanders P, Tumer N, Fraley RT, Beachy RN (1990) Protection against tobacco mosaic virus infection in transgenic plants requires accumulation of coat protein rather than coat protein RNA sequences. Virology 175:124–130
Saeed M, Behjatnia SAA, Mansoor S, Zafar Y, Hasnain S et al (2005) A single complementary-sense transcript of a geminiviral DNA β satellite is determinant of pathogenicity. Mol Plant Microbe Interact 18:7–14
Sambrook J, Maccallum P, Russel D (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Springs Harbour Press, New York, p 133
Saunders K, Bedford ID, Briddon RW, Markham PG, Wong SM et al (2000) A unique virus complex causes Ageratum yellow vein disease. Proc Natl Acad Sci 97:6890–6895
Senanayake DMJB, Mandal B, Lodha S, Varma A (2007) First report of Chilli leaf curl virus affecting chilli in India. Plant Pathol 56:343
Sharma VK, Kushwaha N, Basu S, Singh AK, Chakraborty S (2015) Identification of siRNA generating hot spots in multiple viral suppressors to generate broad-spectrum antiviral resistance in plants. Physiol Mol Biol Plants 21(1):9–18
Shih SL, Tsai WS, Green SK, Singh D (2007) First report of Tomato leaf curl Joydebpur virus infecting chilli in India. Plant Pathol 56:341
Simonne AH, Simonne EH, Eitenmiller RR, Mills HA, Green NR (1997) Ascorbic acid and provitamin A contents in unusually colored bell peppers (Capsicum annuumL.). J Food Compos Anal 10:299–311
Smith NA, Singh SP, Wang MB, Stoutjesdijk PA, Green AG et al (2000) Total silencing by intron-spliced hairpin RNAs. Nature 407:319–320
Sunter G, Bisaro DM (1992) Transactivation of geminivirus AR1 and BR1 gene expression by the viral AL2 gene product occurs at the level of transcription. Plant Cell 4:1321–1331
Topfer R, Schell J, Steinbiss HH (1988) Versatile cloning vectors for transient gene expression and direct gene transfer in plant cells. Nucleic Acids Res 16:8725
Trinks D, Rajeswaran R, Shivaprasad PV, Akbergenov R, Oakeley EJ et al (2005) Suppression of RNA silencing by a geminivirus nuclear protein, AC2, correlates with transactivation of host genes. J Virol 79:2517–2527
van WR, Dong X, Liu H, Tien P, Stanley J et al (2002) Mutation of three cysteine residues in Tomato yellow leaf curl virus-China C2 protein causes dysfunction in pathogenesis and posttranscriptional gene-silencing suppression. Mol Plant Microbe Interact 15:203–208
Vanderschuren H, Akbergenov R, Pooggin MM, Hohn T, Gruissem W et al (2007) Transgenic cassava resistance to African cassava mosaic virus is enhanced by viral DNA-A bidirectional promoter-derived siRNAs. Plant Mol Biol 64:549–557
Vinoth Kumar R, Singh AK, Chakraborty S (2012) A new monopartite begomovirus species, Chilli leaf curl Vellanad virus, and associated betasatellites infecting chilli in the Vellanad region of Kerala India. New Dis Rep 25:20
Wallroth M, Gerats AGM, Rogers SG, Fraley RT, Horsch RB (1986) Chromosomal localization of foreign genes in Petunia hybrida. Mol Gen Genet 02:6–15
Waterhouse PM, Helliwell CA (2003) Exploring plant genomes by RNA-induced gene silencing. Nat Rev Genet 4:29–38
Waterhouse PM, Graham MW, Wang MB (1998) Virus resistance and gene silencing in plants can be induced by simultaneous expression of sense and antisense RNA. Proc Natl Acad Sci 95:13959–13964
Waterhouse PM, Wang M-B, Lough T (2001) Gene silencing as an adaptive defence against viruses. Nature 411:834–842
Xie ZX, Johansen LK, Gustafson AM, Kasschau KD, Lellis AD, Zilberman D, Jacobsen SE, Carrington JC (2004) Genetic and functional diversification of small RNA pathways in plants. PLoS Biol 2:642–652
Yang SJ, Carter SA, Cole AB, Cheng NH, Nelson RS (2004) A natural variant of a host RNA-dependent RNA polymerase is associated with increased susceptibility to viruses by Nicotiana benthamiana. Proc Natl Acad Sci 101(16):6297–6302
Yang JY, Iwasaki M, Machida C, Machida Y, Zhou X, Chua NH (2008) βC1, the pathogenicity factor of TYLCCNV, interacts with AS1 to alter leaf development and suppress selective jasmonic acid responses. Gene Dev 22:2564–2577
Zhang P, Vanderschuren H, Futterer J, Gruissem W (2005) Resistance to cassava mosaic disease in transgenic cassava expressing antisense RNAs targeting virus replication genes. Plant Biotechnol J 3:385–397
Zhang Z-Y, Fu F-L, Gou L, Wang H-G, Li W-C (2010) RNA interference-based transgenic maize resistant to Maize dwarf mosaic virus. J Plant Biol 53:297–305
Zhang Z-Y, Yang L, Zhou S-F, Wang H-G, Li W-C et al (2011) Improvement of resistance to Maize dwarf mosaic virus mediated by transgenic RNA interference. J Biotechnol 153:181–187
Zhang Z-Y, Wang Y-G, Shen X-J, Li L, Zhou S-F et al (2013) RNA interference-mediated resistance to Maize dwarf mosaic virus. Plant Cell Tiss Organ Cult 113:571–578
Zhou Y, Yuan Y, Yuan F, Wang M, Zhong H et al (2012) RNAi-directed down-regulation of RSV results in increased resistance in rice (Oryza sativa L.). Biotechnol Lett 34:965–972
Zilberman D, Cao XF, Johansen LK, Xie ZX, Carrington JC, Jacobsen SE (2004) Role of Arabidopsis ARGONAUTE4 in RNA-directed DNA methylation triggered by inverted repeats. Curr Biol 14:1214–1220
Zrachya A, Kumar PP, Ramakrishnan U, Levy Y, Loyter A et al (2007) Production of siRNA targeted against TYLCV coat protein transcripts leads to silencing of its expression and resistance to the virus. Transgenic Res 16:385–398
Acknowledgments
Veerendra Kumar Sharma thanks the Council of Scientific and Industrial Research, Govt of India, for providing Senior Research Fellowship. The authors gratefully acknowledge the financial support from the Department of Biotechnology, India (BT/PR13295/AGR/36/663/2009). They thank Pranav Pankaj Sahu and Devendran Ragunathan for their suggestions and critical comments on the manuscript.
Conflict of interest
Authors declare that there is no competing financial interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Prasad.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sharma, V.K., Basu, S. & Chakraborty, S. RNAi mediated broad-spectrum transgenic resistance in Nicotiana benthamiana to chilli-infecting begomoviruses. Plant Cell Rep 34, 1389–1399 (2015). https://doi.org/10.1007/s00299-015-1795-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-015-1795-8