Abstract
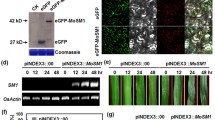
Elicitors are molecules that stimulate defense responses in plants. Previously, an elicitor-encoding gene, named pemG1, was isolated from Magnaporthe grisea. To assess the function of pemG1 in rice (Oryza sativa L. cv. Nipponbare), the gene was cloned under a constitutive maize ubiquitin promoter and introduced into Nipponbare cultivar. The resultant plants showed stable integration and constitutive expression of the pemG1 gene. The expression of defense-related gene for phenylalanine ammonia-lyase was triggered and proline content was also increased in pemG1-expressing plants. The pemG1-expressing plants showed enhanced resistance against rice blast after inoculation with M. grisea spores, suggesting that the pemG1 expression enhances disease resistance in transgenic rice.







Similar content being viewed by others
References
Amero SA, James TC, Elgin SCR (2002) Production of antibodies using proteins in gel bands. In: Walker JM (ed) The protein protocols handbook, 2nd edn, Part VII edn. Humana Press, Totowa, pp 941–944
Arlat M, Gijsegem FV, Huet JC, Pernollet JC, Doucher CA (1994) PopA1, a protein, which induces a hypersensitivelike response on specific petunia genotypes, is secreted via the Hrp pathway of Pseudomonas solanacearum. EMBO J 13:43–553
Baillieul F, Genetet I, Kopp MA, Saindrenan P, Fritig B, Kauffmann S (1995) A new elicitor of the hypersensitive response in tobacco: a fungal glycoprotein elicits cell death, expression of defense genes, production of salicylic acid, and induction of systemic acquired resistance. Plant J 8:551–560
Boissy G, Fprtelle DL, Kahn R (1996) Crystal structure of a fungal elicitor secreted by Phytophthora cryptogea, a member of a novel class of plant necrotic protein. Structure 4:1429–1432
Christensen AH, Quail PH (1996) Ubiquitin promoter-based vectors for high-level expression of selectable and/or screenable marker genes in monocotyledoneus plants. Transgenic Res 5:216–218
Coca M, Bortolotti C, Rufat M, Penas G, Eritja R, Tharreau D, Pozo AM, Messeguer J, Segundo BS (2004) Transgenic rice plants expressing the antifungal AFP protein from Aspergillus giganteus show enhanced resistance to the rice blast fungus Magnaporthe grisea. Plant Mol Biol 54:245–259
Coca M, Peñas G, Gómez J, Campo S, Bortolotti C, Messeguer J, Segundo BS (2006) Enhanced resistance to the rice blast fungus Magnaporthe grisea conferred by expression of a cecropin A gene in transgenic rice. Planta 223:392–406
Dixon RA, Paiva NL (1995) Stress-induced phenylpropanoid metabolism. Plant Cell 7:1085–1097
Fang LJ, Fu XQ, Ye QS, Wu T, Wang ZC, Chen X, Zhu ZK, Zhang XF, Fu CK (2007) Effects of p-glycoprotein inhibitor and elicitor on the salt tolerance of rice seedlings. J Integr Plant Biol 49:588–597
Gandikota M, Kochko A, Chen L, Ithal N, Fauquet C, Reddy AR (2001) Development of transgenic rice plants expressing maize anthocyanin genes and increased blast resistance. Mol Breed 7:73–83
Ger MJ, Chen CH, Wang SY, Huang HE, Podile AR, Dayakar BV, Feng TY (2002) Constitutive expression of hrap gene in transgenic tobacco plant enhances resistance against virulent bacterial pathogens by induction of a hypersensitive response. Mol Plant Microbe Interact 15:764–773
Guo B, Liang YC, Li ZJ, Guo W (2007) Role of salicylic acid in alleviating cadmium toxicity in rice roots. J Plant Nutr 30:427–439
He SY, Huang HC, Collmer A (1993) Pseudomonas syringae pv. syringae Harpinpss: a protein that is secreted via the Hrp pathway and elicits the hypersensitive response in plants. Cell 73:1255–1266
Keller H, Pamboukdjian N, Ponchet M, Poupet A, Delon R, Verrier JL, Roby D, Ricci P (1999) Pathogen-induced elicitin production in transgenic tobacco generates a hypersensitive response and nonspecific disease resistance. Plant Cell 11:223–235
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–683
Levine A, Tenhaken R, Dixon R, Lamb C (1994) H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance response. Cell 79:583–593
Maheshwari R, Dubey RS (2007) Nickel toxicity inhibits ribonuclease and protease activities in rice seedlings: protective effects of proline. Plant Growth Regul 51:231–243
Maruthasalam S, Kalpana K, Kumar KK, Loganathan M, Poovannan K, Raja JAJ, Kokiladevi E, Samiyappan R, Sudhakar D, Balasubramanian P (2007) Pyramiding transgenic resistance in elite indica rice cultivars against the sheath blight and bacterial blight. Plant Cell Rep 26:791–804
Moreno AB, Penas G, Rufat M, Bravo JM, Estopa M, Messeguer J, Segundo BS (2005) Pathogen-induced production of the antifungal AFP protein from Aspergillus giganteus confers resistance to the blast fungus Magnaporthe grisea in transgenic rice. Mol Plant-Microb Interact 18:960–972
Nespoulous C, Huet JC, Pernollet JC (1992) Strcture–function relationships of α and β elicitins, singal proteins involved in the plant–phytophthora interaction. Planta 186:551–557
Nishizawa Y, Nishio Z, Nakazono K, Soma M, Nakajima E, Ugaki M, Hibi T (1999) Enhanced resistance to blast (Magnaporthe grisea) in transgenic aponica rice by constitutive expression of rice chitinase. Theor Appl Genet 99:383–390
Nürnberger T, Nennstiel D, Jabs T, Sacks WR, Hahlbrock K, Scheel D (1994) High affinity binding of a fungal oligopeptide elicitor to parsley plasma membranes triggers multiple defense responses. Cell 78:449–460
Patkar RN, Chattoo BB (2006) Transgenic indica rice expressing ns-LTP-like protein shows enhanced resistance to both fungal and bacterial pathogens. Mol Breed 17:159–171
Peng JL, Bao ZL, Ren HY, Wang JS, Dong HS (2004) Expression of harpin(Xoo) in transgenic tobacco induces pathogen defense in the absence of hypersensitive cell death. Phytopathology 94:1048–1055
Pritilata N, Debabrata B, Sampa D, Asitava B, Dipankar G, Neeliyath A, Maloy G, Soumitra KS (1997) Transgenic elite indica rice plants expressing CryIAc ∂-endotoxin of Bacillus thuringiensis are resistant against yellow stem borer (Scirpophaga incertulas). PNAS 94:2111–2116
Ricci P (1997) Induction of the hypersensitive response and systemic acquired resistance by fungal proteins: the case of elicitins. In: Stacey G, Keen NT (eds) Plant–microbe interactions, vol 3. Chapman and Hall, New York
Ricci P, Bonnet P, Huet JC, Sallantin M, Beauvais CF, Bruneteau M, Billard V, Michel G, Pernollet JC (1989) Structure and activity of proteins from pathogenic fungi Phytophthora eliciting necrosis and acquired resistance in tobacco. Eur J Biochem 183:555–563
Saha P, Majumder P, Ray IDT, Roy SC, Das S (2006) Transgenic rice expressing Allium sativum leaf lectin with enhanced resistance against sap-sucking insect pests. Planta 223:1329–1343
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd ed edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, pp 931–944
Shah K, Dubey RS (1998) Effect of cadmium on proline accumulation and ribonuclease activity in rice seedlings: role of proline as a possible enzyme protectant. Biol Plant 40:121–130
Shao M, Wang J, Dean RA, Lin Y, Gao X, Hu S (2008) Expression of a harpin-encoding gene in rice confers durable nonspecific resistance to Magnaporthe grisea. Plant Biotechnol J 6(1):73–81
Shigeru T, Mitsuo O, Yusuke J, Hisakazu Y, Hanae K, Naoto S, Eiichi M (2006) Induction of resistance against rice blast fungus in rice plants treated with a potent elicitor, N-acetylchitooligosaccharide. Biosci Biotechnol Biochem 70:1599–1605
Sorho F, Pinel A, Traoré O, Bersoult A, Ghesquière A, Hébrard E, Konaté G, Séré Y, Fargette D (2005) Durability of natural and transgenic resistances in rice to rice yellow mottle virus. Eur J Plant Pathol 112:349–359
Southern EM (1975) Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol 98:503
Takakura Y, Che FS, Ishida Y, Tsutsumi F, Kurotani K, Usami S, Isogai A, Imaseki H (2008) Expression of a bacterial flagellin gene triggers plant immune responses and confers disease resistance in transgenic rice plants. Mol Plant Pathol 9(4):525–529
Tang KX, Hu QA, Sun XF, Wan BL, Qi HX, Lu XG (2001) Development of transgenic rice pure lines with enhanced resistance to rice brown planthopper. In Vitro Cell Dev Biol Plant 37:334–340
Toki S, Hara N, Ono K, Onodera H, Tagiri A, Oka S, Tanaka H (2006) Early infection of scutellum tissue with Agrobacterium allows high-speed transformation of rice. Plant J 47:969–976
Umemura K, Tanino S, Nagatsuka T, Koga J, Iwata M, Nagashima K, Amemiya Y (2004) Cerebroside elicitor confers resistance to Fusarium disease in various plant species. Phytopathology 94:813–818
Veit S, Wörle JM, Nürnberger T, Koch W, Seitz HU (2001) A novel protein elicitor (PaNie) from pythium aphanidermatum induces multiple defense responses in carrot, Arabidopsis, and tobacco. Plant Physil 127:832–841
Wei ZM, Laby RJ, Zumoff CH, Bauer DW, He SY, Collmer A, Beer SV (1992) Harpin, elicitor of the hypersensitive response produced by the plant pathogen, Erwinia amylovora. Science 257:85–88
Wei YP, Yao FY, Zhu CX, Jiang MS, Li GX, Song YZ, Wen FJ (2007) Breeding of transgenic rice restorer line for multiple resistance against bacterial blight, striped stem borer and herbicide. Biol plant 51:340–342
Xu F, Yang Y, Xie FJ, Liu Z, Qiu DW, Yang XF (2006) Effect of the activator protein from Magnaporthe grisea on plant growth and physiological activities. Acta Agriculturae Boreali-Sinica 21:1–5
Acknowledgments
We thank Zachary Huang of the Michigan State University for editing assistance and Zhu Haiying at the University of Washington for technical assistance. This work was supported by grants from the Ministry of Science and Technology of China, project numbers 973 (2003CB114204) and 863 (2006AA10A210), and the Beijing Grand Project (D706005040431) as well.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by A. Feher.
DQ and JM contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Qiu, D., Mao, J., Yang, X. et al. Expression of an elicitor-encoding gene from Magnaporthe grisea enhances resistance against blast disease in transgenic rice. Plant Cell Rep 28, 925–933 (2009). https://doi.org/10.1007/s00299-009-0698-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-009-0698-y