Abstract
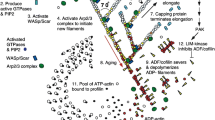
Filopodia are bundles of actin filaments that extend out ahead of the leading edge of a crawling cell to probe its upcoming environment. In vitro experiments (Vignjevic et al. in J Cell Biol 160:951–962, 2003) have determined the minimal ingredients required for the formation of filopodia from the dendritic-like morphology of the leading edge. We model these experiments using kinetic aggregation equations for the density of growing bundle tips. In mean field, we determine the bundle size distribution to be broad for bundle sizes smaller than a characteristic bundle size above which the distribution decays exponentially. Two-dimensional simulations incorporating both bundling and cross-linking measure a bundle size distribution that agrees qualitatively with mean field. The simulations also demonstrate a nonmonotonicity in the radial extent of the dendritic region as a function of capping protein concentration, as was observed in experiments, due to the interplay between percolation and the ratcheting of growing filaments off a spherical obstacle.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walter P (2007) Molecular biology of the cell. Taylor and Francis, New York
Amman KJ, Pollard TD (2001) Direct real-time observation of actin filament branching mediated by Arp2/3 complex using total internal relfection fluorescence microscopy. Proc Natl Acad Sci USA 98: 15009-15-13
Atiligan E, Wirtz D, Sun SX (2006) Mechanics and dynamics of actin-driven thin membrane protrusion. Biophys J 90: 65–76
Bicout DJ, Burkhardt TW (2000) Absorption of a randomly accelerated particle: gambler’s ruin in a different game. J Phys A Math Gen 33: 6835–6841
Borisy GG, Svitkina TM (2000) Actin machinery: pushing the envelope. Curr Opin Cell Biol 12: 104–112
Carlsson AE (2003) Growth velocities of branched actin networks. Biophys J 84: 2907–2918
Carlsson AE (2007) Disassembly of actin networks by filament severing. New J Phys 9: 1–15
Carlsson AE, Sept D (2008) Mathematical modeling of cell migration. In: Correira JJ, Detrich HW (eds) Biological tools for biologists, vol 1 in vitro techniques (Methods in cell biology, vol 84). Elsevier, New York, pp 911–937 2008
Carlsson AE, Wear MA, Cooper JA (2004) End versus side branching by Arp2/3 complex. Biophys J 86: 1074–1081
Chelakkot R, Lipowsky R, Gruhn T (2008) Self-assembling network and bundle structures in systems of rods and cross-linkers: a Monte-Carlo study. Soft Matter 5: 1504–1513
Connaughton C, Rajesh R, Zaboronski O (2006) Cluster-cluster aggregation as an analogue of a turbulent cascade: Kolmogorov phenomenology, scaling laws and the breakdown of self-similarity. Physica D 222: 97–115
Cueille S, Sire C (1997) Nontrivial polymerdisperity exponents in aggregation models. Phys Rev E 5: 5465–5478
Davies SC, King JR, Wattis JAD (1999) The Smoluchowski equation with continuous injection. J Phys A Math Gen 32: 7745–7763
Dawes AT, Ermentrout GB, Cytrynbaum EN, Edelstein-Keshet L (2006) Actin filament branching and protrusion velocity in a simple 1D model of a motile cell. J Theor Biol 242: 265–279
Edelstein-Keshet L, Ermentrout B (1989) Models for branching networks in two dimensions. SIAM J Appl Math 49: 1136–1157
Fournier N, Laurencot P (2005) Existence of Self-similar solutions to Smoluchowski’s coagulation equation. Commun Math Phys 256: 589–609
Fujiwara I, Suetsugu S, Uemura S, Takenawa T, Ishiwata S (2002) Visualization and force measurement of branching by Arp2/3 complex and N-WASP in actin filament. Biochem Biophys Res Commun 293: 1550–1555
Gopinathan A, Lee K-C, Schwarz JM, Liu AJ (2007) Branching, capping, and severing in dynamic actin structures. Phys Rev Lett 99:058103, pp 4
Grason GM, Bruinsma RF (2007) Chirality and equilibrium biopolymer bundles. Phys Rev Lett 99:098101, pp 4
Ha B-Y, Liu AJ (1999) Kinetics of bundle growth in DNA condensation. Europhys Lett 46: 624–630
Haviv L, Brill-Karniely Y, Mahaffy R, Backouche F, Ben-Shaul A, Pollard TD, Bernheim-Groswasser A (2006) Reconstitution of the transition from lamellipodium to filopodium in a membrane-free system. Proc Natl Acad Sci USA 103: 4906–4911
Haviv L, Gov N, Ideses Y, Bernheim-Groswasser A (2008) Thickness distribution of actin bundles in vitro. Eur Biophys J 37: 447–454
Henle ML, Pincus PA (2005) Equilibrium bundle size of rodlike polyelectrolytes with counterion-induced attractive interactions. Phys Rev E 71:060801, pp 4
Hoshen J, Kopelman R (1976) Percolation and cluster distribution: cluster multiple labeling technique and critical concentration algorithm. Phys Rev B 14: 3438–3445
Ichetovkin I, Grant W, Condeelis J (2002) Cofilin produces newly prolymerized actin filaments that are preferred for dendritic nucleation by the Arp2/3 complex. Curr Biol 12: 79–84
Ideses Y, Brill-Karniely Y, Haviv L, Ben-Shaul A, Bernheim-Groswasser A (2008) Arp2/3 branched actin network mediates filopodia-like bundle formation in vitro. PloS ONE 3(9):e3297. doi:10.1371/journal.pone.0003297
Kang K, Redner S, Meakin P, Leyvraz F (1986) Long-time crossover phenomena in coagulation kinetics. Phys Rev E 33: 1171–1182
Kierfeld J, Gutjahr P, Kuhne T, Kraikivski P, Lipowsky R (2006) Buckling, bundling, and pattern formation: from semi-flexible polymers to assemblies of interacting filaments. J Comput Theor Nanosci 3: 898–911
Korobova F, Svitkina T (2008) Arp2/3 complex is important for filopodia formation, growth cone motility, and neuritogenesis in neuronal cells. Mol Biol Cell 19: 1561–1574
Kraikivski P, Slepchenko BM, Novak IL (2008) Actin bundling: initiation mechanisms and kinetics. Phys Rev Lett 101:128102, pp 4
Krapivsky PL, Mendes JFF, Redner S (1999) Influence of island diffusion on submonolayer epitaxial growth. Phys Rev B 59: 15950–15958
Lan Y, Papoian GA (2008) The stochastic dynamics of filopodial growth. Biophys J 94: 3839–3852
Le Clainche C, Carlier MF (2008) Regulation of actin assembly associated with protrusion and adhesion in cell migration. Physiol Rev 88: 489–513
Leyvraz F (2003) Scaling theory and exactly solved models in the kinetics of irreversible aggregation. Phys Rep 383: 95–212
Lieleg O, Schmoller KM, Cyrn CJ, Luan Y, Wall WA, Bausch AR (2009) Structural polymorphism in heterogeneous cytoskeletal networks. Soft Matter 5: 1796–1803
Liu AP, Richmon DL, Maibaum L, Pronk S, Geissler PL, Fletcher DA (2008) Membrane-induced bundling of actin filaments. Nat Phys 4: 789–793
Mallavarapu A, Mitchinson T (1999) Regulated actin cytoskeleton assembly at filopodium tips controls their extension and retraction. J Cell Biol 146: 1097–1106
Mejillano MR, Kojima S, Applewhite DA, Gertler FB, Svitkina TM, Borisy GG (2004) Lamellipodia versus filopodial mode of the actin nanomachinery: Pivotal role of the filament barbed end. Cell 118: 363–373
Mogilner A (2009) Mathematics of cell motility: have we got it’s number?. J Math Biol 58: 105–134
Mogilner A, Edelstein-Keshet L (1999) Regulation of actin dynamics in rapidly moving cells: a quantitative analysis. Biophys J 83: 11324–11329
Mogilner A, Rubenstein B (2005) The physics of filopodial protrusion. Biophys J 89: 782–795
Mullins RD, Heuser JA, Pollard TD (1998) The interaction of Arp2/3 complex with actin: nucleation, high affinity pointed end capping and formation of branching networks of filaments. Proc Natl Acad Sci 95: 6181–6186
Pollard TD, Blanchoin L, Mullins RD (2000) Molecular mechanisms controlling actin filament dynamics in nonmuscle cells. Annu Rev Biophys Biomol Struct 29: 545–576
Saito N, Takahashi K, Yunoki Y (1967) The statistical mechanical theory of stiff chains. J Phys Soc Jpn 22: 219–226
Schaus TE, Taylor EW, Borisy GG (2007) Self-organization of actin filament orientation in the dendritic-nucleation/array-treadmilling model. Proc Natl Acad Sci USA 104: 7086–7091
Stewman SF, Dinner AR (2007) Lattice model for self-assembly with application to the formation of cytoskeletal-like structures. Phys Rev E 76:016103, pp 9
Svitkina TM, Borisy GG (1999) Arp2/3 complex and actin depolymerizing factor/cofiln in dendritic organization and treadmilling of actin filaments array in lamellipodia. J Cell Biol 145: 1009–1026
Svitkina TM, Bulanova EA, Chaga OY, Vignjevic DM, Kojima S, Vasiliev JM, Borisy GG (2003) Mechanism of filopodia initation by reorganization of a dendritic network. J Cell Biol 160: 409–421
Tang JX, Janmey PA (1996) The polyelectrolyte nature of F-actin and the mechanism of actin bundle formation. J Biol Chem 271: 8556–8563
Vavylonis D, Yang QB, O’Shaughnessy B (2005) Actin polymerization kinetics, cap structure, and fluctuations. Proc Natl Acad Sci USA 102: 8543–8548
Vignjevic D, Yarar E, Welch MD, Peloquin J, Svitkina T (2003) Formation of filopodia-like bundles in vitro from a dendritic network. J Cell Biol 160: 951–962
Volkmann N, Amann KJ, Stoilova-McPhie CE, Winter DC, Hazelwood L, Heuser JE, Li R, Pollard TD (2001) Structure of Arp2/3 complex in its activated state and in actin filament branch junctions. Science 293: 2456–2459
von Dongen PGJ, Ernst MH (1985) Dynamic scaling in the kinetics of clustering. Phys Rev Lett 54: 1396–1399
von Smoluchowski M (1916) Drei vortrage uber diffusion Brownsche molekular bewegung und koagulation von kolliodteichen. Z Phys 17: 557
Witten TA, Sander LM (1981) Diffusion-limited aggregation, a kinetic critical phenomenon. Phys Rev Lett 47: 1400–1403
Yang L, Sept D, Carlsson AE (2006) Energetics and dynamics of constrained actin filament bundling. Biophys J 90: 4295–4304
Yu XP, Carlsson AE (2004) Kinetics of filament bundling with attractive interactions. Biophys J 87: 3679–3689
Zhuravlev P, Papoian GA (2009) Molecular noise of capping protein induces macroscopic instability in filopodial dynamics. Proc Natl Acad Sci USA 106: 11570–11575
Acknowledgments
The authors would like to acknowledge helpful conversations with Andrea Liu, Ron Maimon, and Tatyana Svitkina during the early stages of this work. The authors gratefully acknowledge Louise Yang, an undergraduate summer intern who helped conduct some of the preliminary simulations in this work. The authors would also like to acknowledge the hospitality of the Aspen Center for Physics where some of this work was completed. Finally, AG acknowledges support from the James S. McDonnell Foundation and JMS acknowledges support from NSF-DMR-0645373.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Lee, KC., Gopinathan, A. & Schwarz, J.M. Modeling the formation of in vitro filopodia. J. Math. Biol. 63, 229–261 (2011). https://doi.org/10.1007/s00285-010-0371-7
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-010-0371-7