Abstract
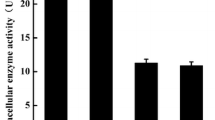
Leucine aminopeptidase (LAP), an enzyme used in the food industry, is an exopeptidase that removes an amino acid residue, primarily leucine (Leu), from the N-terminus of peptides and protein substrates. In this study, we focused on the leucine aminopeptidase A (lapA) gene from Aspergillus oryzae RIB40. To purify and characterize the LapA, lapA was overexpressed in A. oryzae RIB40 using the amyB promoter. LAP activity in the culture supernatant of one transformant harboring the lapA expression plasmid was 33 times that of the host strain. LapA was purified from the culture supernatant of this lapA-overexpressing strain by column chromatography. The purified recombinant LapA had a molecular mass of 33 kDa, and its N-terminal amino acid was the tyrosine at position 80 of the deduced amino acid sequence. Optimal enzyme activity was observed at 60°C and pH 8.5, and the enzyme was stable at temperatures up to 60°C and in the pH range 7.5–11. In transcriptional analysis, lapA was induced under alkaline conditions and expressed at a relatively low level under normal conditions. LapA showed maximum hydrolyzing activity for the substrate leucine para-nitroanilide (Leu-pNA), followed by substrates Phe-pNA (39% activity compared with Leu-pNA), Met-pNA, Lys-pNA, and Arg-pNA. In addition, LapA preferentially hydrolyzed peptides longer than tripeptides.


Similar content being viewed by others
References
Akao T, Sano M, Yamada O et al (2007) Analysis of expressed sequence tags from the fungus Aspergillus oryzae cultured under different conditions. DNA Res 14:47–57
Bradford MM (1976) A rapid and sensitive method for detecting microgram amounts of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chien HC, Lin LL, Chao SH et al (2002) Purification, characterization, and genetic analysis of a leucine aminopeptidase from Aspergillus sojae. Biochim Biophys Acta 1576:119–126
Clemente A (2000) Enzymatic protein hydrolysates in human nutrition. Trends Food Sci Technol 11:254–262
Denison SH (2000) pH regulation of gene expression in fungi. Fungal Genet Biol 29:61–71
Dohmoto M, Inoue Y, Kobayashi A et al (2010) Characterization of the palH gene from Aspergillus oryzae. Biosci Biotechnol Biochem 74:188–190
Exterkate FA (1984) Location of peptidases outside and inside the membrane of Streptococcus cremoris. Appl Environ Microbiol 47:177–183
Gomi K, Iimura Y, Hara S (1987) Integrative transformation of Aspergillus oryzae with a plasmid containing the Aspergillus nidulans argB gene. Agric Biol Chem 51:2549–2555
Gonzales T, Robert-Baudouy J (1996) Bacterial aminopeptidases: properties and functions. FEMS Microbiol Rev 18:319–344
Gotou T, Shinoda T, Mizuno S et al (2009) Purification and identification of proteolytic enzymes from Aspergillus oryzae capable of producing the antihypertensive peptide Ile-Pro-Pro. J Biosci Bioeng 107:615–619
Hanahan D (1983) Studies on the transformation of Escherichia coli with plasmids. J Mol Biol 166:557–580
Hubbard SC, Ivatt RJ (1981) Synthesis and processing of asparagine-linked oligosaccharides. Ann Rev Biochem 50:555–583
Izawa N, Tokuyasu K, Hayashi K (1997) Debittering of protein hydrolysates using Aeromonas caviae aminopeptidase. J Agric Food Chem 45:543–545
Kusumoto KI, Matsushita-Morita M, Furukawa I et al (2008) Efficient production and partial characterization of aspartyl aminopeptidase from Aspergillus oryzae. J Appl Microbiol 105:1711–1719
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:3397–3707
Machida M, Asai K, Sano M et al (2005) Genome sequencing and analysis of Aspergillus oryzae. Nature 438:1157–1161
Matsushita-Morita M, Furukawa I, Suzuki S et al (2010) Characterization of recombinant prolyl aminopeptidase from Aspergillus oryzae. J Appl Microbiol 109:156–165
Monod M, Léchenne B, Jousson O et al (2005) Aminopeptidases and dipeptidyl-peptidases secreted by the dermatophyte Trichophyton rubrum. Microbiology 151:145–155
Morita H, Okamoto A, Yamagata Y et al (2009) Heterologous expression and characterization of CpI, OcpA, and novel serine-type carboxypeptidase OcpB from Aspergillus oryzae. Appl Microbiol Biotechnol 85:335–346
Nakadai T, Nasuno S, Iguchi N (1972) The action of peptidase from Aspergillus oryzae on soybean proteins. Agric Biol Chem 36:261–268
Nakadai T, Nasuno S, Iguchi N (1973) Purification and properties of leucine aminopeptidase I from Aspergillus oryzae. Agric Biol Chem 37:757–765
Nakadai T, Nasuno S, Iguchi N (1973) Purification and properties of leucine aminopeptidase II from Aspergillus oryzae. Agric Biol Chem 37:767–774
Nakadai T, Nasuno S, Iguchi N (1973) Purification and properties of leucine aminopeptidase III from Aspergillus oryzae. Agric Biol Chem 37:775–782
Nakadai T, Nasuno S (1977) Purification and properties of leucine aminopeptidase IV from Aspergillus oryzae. Agric Biol Chem 41:1657–1666
Nakamura Y, Yamamoto N, Sakai K et al (1995) Antihypertensive effects of sour milk and peptides isolated from it that are inhibitors to angiotensin I-converting enzyme. J Dairy Sci 78:1253–1257
Sano J, Ohki K, Higuchi T et al (2005) Effect of casein hydrolysate, prepared with protease derived from Aspergillus oryzae, on subjects with high-normal blood pressure or mild hypertension. J Med Food 8:423–430
Strater N, Lipscomb WN (2004) Leucyl aminopeptidase (animal). In: Barrett AJ, Rawlings ND, Woesner JF (eds) Handbook of proteolytic enzymes, 2nd edn. Elsevier/Academic Press, San Diego, pp 896–899
Tatsumi H, Ogawa Y, Murakami S et al (1989) A full length cDNA clone for the alkaline protease from Aspergillus oryzae: structural analysis and expression in Saccharomyces cerevisiae. Mol Gen Genet 219:33–38
Tilburn J, Sarkar S, Widdick DA et al (1995) The Aspergillus PacC zinc finger transcription factor mediates regulation of both acid- and alkaline-expressed genes by ambient pH. EMBO J 14:779–790
Toldrá F, Aristoy AC, Flores M (2000) Contribution of muscle aminopeptidases to flavor development in dry-cured ham. Food Res Int 33:181–185
Acknowledgment
This study was supported by the Program for Promotion of Basic Research Activities for Innovative Biosciences (PROBRAIN)
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Matsushita-Morita, M., Tada, S., Suzuki, S. et al. Overexpression and Characterization of an Extracellular Leucine Aminopeptidase from Aspergillus oryzae . Curr Microbiol 62, 557–564 (2011). https://doi.org/10.1007/s00284-010-9744-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-010-9744-9