Abstract
In recent years, considerable progress has been made in increasing the knowledge of tumour biology and drug resistance mechanisms in urothelial cancer. Therapeutic strategies have significantly advanced with the introduction of novel approaches such as immune checkpoint inhibitors and Fibroblast Growth Factor Receptor inhibitors. However, despite these novel agents, advanced urothelial cancer is often still progressive in spite of treatment and correlates with a poor prognosis. The introduction of antibody–drug conjugates consisting of a target-specific monoclonal antibody covalently linked to a payload (cytotoxic agent) is a novel and promising therapeutic strategy. In December 2019, the US Food and Drug Administration (FDA) granted accelerated approval to the nectin-4-targeting antibody–drug conjugate, enfortumab vedotin, for the treatment of advanced or metastatic urothelial carcinomas that are refractory to both immune checkpoint inhibitors and platinum-based treatment. Heavily pre-treated urothelial cancer patients reported a significant, 40% response to enfortumab vedotin while other antibody–drug conjugates are currently still under investigation in several clinical trials. We have comprehensively reviewed the available treatment strategies for advanced urothelial carcinoma and outlined the mechanism of action of antibody–drug conjugate agents, their clinical applications, resistance mechanisms and future strategies for urothelial cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The treatment options for advanced urothelial cancer (UC) have been rapidly developing over the last few years. This development began with the approval of anti Fibroblast Growth Factor Receptor (FGFR) and various immune checkpoint inhibitors (ICIs), followed by the Food and Drug Administration (FDA) approving enfortumab vedotin (EV), an antibody–drug conjugate (ADC) for the treatment of advanced urothelial carcinoma in 2019 [1]. Whilst EV is the first ADC to gain FDA approval for the treatment of UC, their use is not novel and are commonly used for the treatment of breast cancer [2, 3] and hematologic malignancies [4, 5]. Given the positive results observed with EV in patients with advanced UC, several clinical trials are underway with the aim of demonstrating the improved efficacy of ADCs and with the ultimate goal of approving the ADC use in earlier lines of therapy. This review will provide an overview of current treatment options for advanced urothelial carcinoma and details of the clinical development of various ADCs being studied for use in this cancer type.
Urothelial carcinoma
Urothelial cancer is the fourth most common cancer among American males with over 80,000 cases diagnosed in 2020 [6]. UC normally occurs in older patients (7th decade and older). Risk factors for development include a genetic predisposition (such as Lynch syndrome), chemical and environmental exposure (cyclophosphamide, aromatic amines), cigarette smoking and male sex [7]. The vast majority of urothelial cancers arise within the bladder and are found to have not yet invaded the muscle at diagnosis [8]. First-line treatment for non-muscle invasive bladder cancer (NMIBC) (Ta/Tis/T1) is generally surgical via transurethral resection of bladder tumour (TURBT) with additional intravesical therapy using bacillus Calmette-Guerin (BCG) or mitomycin, in order to prevent either disease relapse or regression [9, 10].
Even though a large number of patients benefit from the aforementioned strategy, a fraction of these patients will progress to muscle-invasive bladder carcinoma (MIBC), a locally advanced disease stage that is associated with a high rate of lymph node spread and distant metastasis [11]. In these cases, the gold standard treatment involves neoadjuvant cisplatin-based chemotherapy (although still poorly adopted compared to adjuvant chemotherapy) followed by radical cystectomy, a surgical procedure associated with non-trivial mortality and a significant effect on quality of life [12]. As a result, it is clear that in certain cases there is a significant unmet clinical need and thus an opportunity for future drug development.
Treatment of advanced muscle-invasive urothelial carcinoma
Systemic treatment is required for patients with advanced or metastatic urothelial cancer (mUC). Until recently, cisplatin-based combination therapies were the only option available. Examples included: methotrexate, vinblastine, adriamycin and cisplatin (MVAC), or gemcitabine and cisplatin (GC). In a phase III trial, both regimens demonstrated a similar response rate of approximately 40–50% and a 5-year survival rate of 10–15%. However, GC regimens reported lower toxicity and are therefore currently the treatment of choice in ongoing trials [13]. Conversely, regimens containing carboplatin (usually gemcitabine plus carboplatin), are the preferred treatment for those deemed cisplatin-ineligible, with similar response rates to GC regimens, but with poorer survival outcomes [14, 15].
Further research in this setting has led to the development of immune checkpoint inhibitors, such as atezolizumab, avelumab, durvalumab, nivolumab and pembrolizumab; all of which are now can be used in patients with UC [16]. Phase III trials recently demonstrated that pembrolizumab treatment resulted in a 3 months benefit in terms of survival when compared to standard of care taxane or vinflunine in platinum-ineligible patients [17, 18]. Notably, in another phase III trial, atezolizumab showed no benefit on overall survival. Nonetheless, both atezolizumab and pembrolizumab were granted FDA approval for their use as first-line agents for platinum-ineligible patients with UC [19].
In addition to immunotherapy options, second-line regimens including antifolates and taxanes represent a valid option for patients who have had disease progression on platinum-based regimens, although these regimens do report poor response rates of approximately 15% [17]. Additionally, a vinflunine agent reported a debatable improvement in survival when compared to the standard of care in a phase II trial, achieving European Medicines Agency but not FDA approval [20].
Moreover, significant treatment advancements have been made for those metastatic UC patients (20%) harbouring mutations in the FGFR pathway. Erdafinitib, an oral FGFR-inhibitor, reported a response rate of 40% in patients with FGFR alterations in phase II single study, leading to accelerated FDA approval for use in patients who have progressed on platinum-based therapy [21, 22]. Despite these developments, a large number of patients still relapse following platinum-based regimens as well as immunotherapy. Consequently, this unmet clinical need led to the generation of antibody–drug conjugates, which have generated a robust interest within the scientific community.
Antibody–drug conjugates (ADCs)
ADCs are small molecule anticancer agents covalently linked to a monoclonal antibody (mAb). Specific antigens expressed on tumour surfaces are targeted by the mAb, resulting in selective delivery of the anticancer agent to tumour cells [23] (Table 1). Excessive toxicity or poor handling limits some chemotherapeutic drugs from being used as classical chemotherapeutic agents [24]. ADCs can overcome this limitation by selectively delivering cytotoxic agents to tumour targets, reducing toxicity, and increasing efficacy. The early development of ADCs involved the use of murine antibodies conjugated with chemotherapeutic agents such as methotrexate, doxorubicin, and vinblastine, even though these agents have limited selectivity and strong immunogenicity [25]. As the technology has evolved and humanised antibodies developed, these agents have become more effective and specific, leading to improved potency and reduced immunogenicity [26]. Three components make up an ADC; an antibody specific for the target antigen, a linker domain and then the cytotoxic agent.
Antibody identification
ADCs are not required to elicit an immune response after linking with the cytotoxic payload [27]. Presently, immunoglobulin G forms the integral structure and is composed of four subclasses (IgG1, IgG2, IgG3, and IgG4), each of which differ from each other in the structure of the constant domain and hinge regions [28, 29]. Most immunotherapies, including ADCs, utilise IgG1 as it can stimulate immune effector functions (receptor binding, endocytosis and downstream activation of immune pathways). IgG1 also has the advantage of high stability in serum, and low molecular weight and is well distributed in the intra- and extravascular space [26] (Fig. 1).
The mAb should be targeted against an antigen strongly expressed on malignant cells, and absent on non-malignant cells, this is crucial in reducing systemic toxicity and widening the therapeutic window. Examples of such antigens within UC cells include HER2, Nectin-4 and Trop-2. Furthermore, there should be limited antigen cross-reactivity, with strong binding affinity to the target to ensure effective internalisation and stability [30, 31].
Drug-mAb linker
The linker acts to join the cytotoxic agent to the antibody via the conjugation sites in the antibody heavy chains. Two crucial characteristics must be present for the Linker to be functional. Firstly, the linker must ensure the antibody and cytotoxic agent remain firmly bound, particularly in the plasma during circulation. An unstable linker may lead to premature delivery of the cytotoxic agent into the systemic circulation leading to unwanted toxicity and reduced therapeutic efficacy [32]. Secondly, the linker must be able to deliver the drug once at the tumour site [33].
There are two main sub-classes of linkers; cleavable and non-cleavable. Cleavable linkers rely on factors in the tumour microenvironment to stimulate the breakdown and release of the ADC cytotoxic agent [34]. Mechanisms of linker cleavage are diverse; one such mechanism is driven by glutathione (highly represented in the cytoplasm compared to the extracellular space), which leads to the release of the cytotoxic agent via breakage of disulphide bonds [35, 36]. A second type of mechanism involves linkers that cleave in environments with an acidic pH, such as hydrazone. This type of linker exploits the acidic pH found in endosomes and lysosomes. However, premature cleavage of these linkers into the circulation may lead to hepatotoxicity such as that described in gemtuzumab ozogamicin [32, 37]. A third type of linker is those that are protease-dependent and are degraded by lysosomal proteases after recognition of a specific peptide sequence. These linkers allow the ADC to be remarkably stable within the plasma and thus avoid a premature release of the cytotoxic agent. Examples of this type of linker include enfortumab vedotin, ADC directed against nectin-4, and sacituzumab govitecan (SG) directed against the human trophoblast cell surface antigen 2 (Trop-2) [38].
Cleavable linkers are less stable than non-cleavable linkers. Non-cleavable linkers rely on the degradation of the complete antibody-linker complex to release the cytotoxic agent. Examples of ADCs with non-cleavable linkers are belantamab mafodotin and trastuzumab emtansine (T-DM1) [39].
Payloads
The cytotoxic agents in ADCs are often referred to as the payload, and these drugs are usually heavily toxic molecules [40]. The antibody acts as the delivery mechanism of this payload to the tumour target. The early ADCs could deliver classical chemotherapeutic agents, such as methotrexate, vinca alkaloids and doxorubicin [41, 42]. However, ADCs delivering these agents did not demonstrate higher efficacy than when these agents were delivered as standard chemotherapeutic agents [43].
ADC payloads can be split into different macro-categories. The most important of which are the agents that destabilise microtubules, such as auristatins and maytansins, which are derived from natural bacteria. Monomethyl auristatin E and F (MMAE, MMAF) are examples of auristatins and are synthetic derivatives of the dolastatin 10 peptide which is isolated from Dolabella auricularia [44]. These drugs act by inhibiting the polymerisation of tubulin, resulting in cell cycle arrest and then apoptosis. Maytansins such as DM1 act in a similar fashion and target tubulin via the vinca alkaloid binding site, subsequently leading to a blockade of mitotic replication and then cell cycle arrest and apoptosis [45, 46].
Other types of payload include those that act directly on DNA damage, examples include cuocarmicins and pyrrolobenzodiazepines. These agents can generate DNA double helix damage and act as alkylating agents leading to disruption of transcription, causing DNA double helix breakage and apoptosis [47,48,49]. Further examples of this type of payload include the camptothecin analogues, such as SN-38, which can inhibit topoisomerase I resulting in DNA damage and breakage.
Additionally, ADC activity relies on a well-balanced drug-to-antibody ratio (DAR). A high DAR can negatively impact pharmacokinetics [50], whereas a low DAR may reduce ADC potency. ADCs with high DARs may show greater efficacy and internalisation, but this may also lead to increased clearance [51,52,53]. Importantly, ADCs with lower immunogenicity have a key advantage in that these are less likely to lead to the development of anti-drug antibodies, the presence of which can suppress drug efficacy [54].
Antitumour activity in urothelial carcinoma
Enfortumab vedotin
Enfortumab vedotin is a novel ADC composed of a fully human antibody, targeting Nectin-4 and the potent microtubule-disrupting agent monomethyl auristatin E [55]. Nectin-4 is a junction protein implicated in cell–cell adhesion [56]; it is involved in a variety of biological processes such as tumour-cell growth, proliferation, immune modulation and viral entry [57]. In a previous study, it has been found that Nectin-4 mRNA, a poliovirus receptor-related protein-4 (PVRL4), is highly expressed in cancer cells, especially in bladder cancer (BC) [58] and that such aberrant expression is associated with cancer progression and poor prognosis. Due to its central role in tumorigenesis and lymphangiogenesis, it has emerged as a potential biomarker and promising targeted therapy. In 2020, a phase III trial has investigated the efficacy of EV versus investigator-choice chemotherapy (docetaxel, paclitaxel, and vinflunine) in 608 patients progressing after platinum-containing chemotherapy and ICI [55]. At the prespecified interim analysis, the primary endpoint was met with longer overall survival (OS) in the enfortumab vedotin group than in the chemotherapy group [median OS, 12.8 vs. 8.9 months; hazard ratio (HR) for death, 0.70; 95% confidence interval (CI), 0.56–0.89; p = 0.001). After these results, enfortumab vedotin was granted EMA and FDA breakthrough therapy approval for the treatment of patients previously treated with platinum-containing chemotherapy and ICI [59].
Moreover, in the first-line setting, EV had a synergistic effect when combined with ICI, based on the results of the ongoing phase Ib/2 EV-103 trial, demonstrating an objective response rate (ORR) of 73%, with 15.6% of complete responses (CR) and median progression-free survival (PFS) of 12.3 months in cisplatin-unfit patients [60]. It is noteworthy to mention that EV is correlated with severe cutaneous adverse reactions, including fatal cases of Steven Johnson Syndrome or Toxic Epidermal Necrolysis, especially during the first cycle of treatment but may occur later, as well as hyperglycemia, pneumonitis, peripheral neuropathy, ocular disorders, infusion-site extravasation, and embryofetal toxicity [55, 61].
Sacituzumab govitecan
Sacituzumab govitecan is a humanized anti-Trop2 monoclonal IgG1k coupled to the cytotoxic payload, SN-38, the active metabolite of irinotecan and a topoisomerase I inhibitor [62] via a cleavable linker [63, 64]. Trop-2 is a 40-kDa transmembrane glycoprotein that was first discovered in human trophoblast and choriocarcinoma cell line [65]. Due to its short intracytoplasmic tail, it is correlated with several pathways regulating cellular functions such as cell–cell adhesion, cell proliferation and mobility [65, 66]. Moreover, a high Trop-2 expression has been found in different cancers including urothelial cancer where it is associated with aggressive progression and poor survival outcome [67]. The TROPHY-U-01 study is an open-label, single-arm phase II study designed to confirm the SG antitumor activity in patients with metastatic UC who progressed after prior platinum-based and checkpoint inhibitor-based therapies [68].
Among the 113 patients who received SG, central review evidenced an ORR of 27% with an mPFS and mOS of 5.4 months and 10.9 months, respectively; thus confirming the results from the prior phase I/II study showing that SG has significant anticancer activity in heavily pretreated patients [69]. Regarding the adverse events (AEs), it is worth mentioning that SG is generally well tolerated and the observed grade 3 or greater AEs were neutropenia (35%) followed by leukopenia (18%), anaemia (14%), diarrhoea (10%), and febrile neutropenia (10%). Based on this preliminary data, SG received accelerated approval in heavily pretreated patients with mUC who had progressed on platinum and ICIs.
Sirtratumab vedotin (ASG15-ME)
Sirtratumab vedotin is an ADC composed of a SLITRK6-specific human gamma 2 antibody (Igγ2) conjugated to a small molecule microtubule disrupting agent, monomethyl auristatin E (MMAE) via a protease-cleavable linker [70]. It enables the release of this MMAE to tumours expressing SLITRK6 [71]. This protein belongs to a neuronal transmembrane protein family regulating the growth and survival of neuronal cells in the inner ear that transmit auditory signals. Therefore, mutations in this gene lead to myopia and progressive auditory neuropathy in humans and mice [72, 73]. Several immunohistochemical studies have demonstrated that SLITRK6 is expressed in a variety of epithelial tumours, including lung cancer, glioblastoma and breast cancer, and that it is moderately negatively correlated with tumour malignancy [74].
The first study that reported data on SV anti-tumour activity was a phase I study that included 51 metastatic urothelial cancer patients. SLITRK6 expression was evaluated by immunohistochemistry and results demonstrated it to be positive in 93% of patients. Among the 42 patients treated with a therapeutic dose (> 0.5 mg per kg), 1 showed CR at 39 weeks and 13 had a partial response (PR), resulting in an ORR of 33%. The median duration of response (DOR) and mPFS were 15 and 16 weeks, respectively. SV was generally well tolerated; fatigue was the most common grade 3 or higher AEs, evaluated in 44% of patients [71]. Ten patients experienced reversible ocular toxicities with one grade 3 toxicity. Despite these results, no current ongoing trials are evaluating the SV efficacy in UC metastatic setting.
Human epidermal growth factor receptor 2 (HER2)—ADCs in bladder cancer
HER2 has a firmly established oncogenic potential in both preclinical and clinical settings, especially in breast cancer [75]. When overexpressed, it leads to the autophosphorylation of tyrosine residues within the cytoplasmic domain of the heterodimer and triggers a complex pathway, resulting in a strong pro-tumorigenic signalling cascade [76]. Recently, various ADCs targeting HER2-positive BC have been investigated, leading to a significant improvement in survival outcomes [77]. Beyond breast and gastric cancer, urothelial carcinoma is the third most prevalent cancer with HER2 overexpression, showing potential utility for HER2-targeting therapy in mUC. Notably, it has been shown that HER2 overexpression was observed in 9.2–12.4% of invasive bladder carcinoma, with 5.1% of those demonstrating a HER2 gene amplification [78]. In addition, Fleischmann et al. demonstrated that HER2 amplification was significantly more frequent in lymph node metastases (15.3%) than in matched primary bladder cancers as well as being more apparent in the luminal than in the basal subtypes [79]. Moreover, previous studies demonstrated that in bladder cancer, HER2 overexpression strongly correlated with tumour progression and poor prognosis and, unlike BC, HER2 genomic amplification is not a common mechanism [80, 81]. While in BC the role of HER2-targeting agents has been well defined in both metastatic and adjuvant settings, the efficacy of HER2-targeting agents in bladder carcinomas still remains a challenge.
Trastuzumab emtansine (TDM-1).T-DM1 is a HER2-targeted antibody–drug conjugate, combining a monoclonal antibody with an anticancer drug called emtansine, a microtubule inhibitor [82].
Although T-DM1 showed promising antitumor effects in preclinical models of HER2 overexpressing bladder cancers [83], the multi-histology phase II, basket trial of TDM-1 in patients with HER2 amplified cancers failed to demonstrate a significant activity of this drug in patients with mUC..
Trastuzumab deruxtecan (T-DXd). T-DXd (DS-8201) is an antibody–drug conjugate that is composed of a humanized monoclonal antibody specifically targeting HER2 linked to potent topoisomerase I inhibitor as the cytotoxic drug (payload) [3]. In the DESTINY-Breast01 trial, DS-8201 showed durable antitumor activity in a pretreated patient population with HER2-positive metastatic breast cancer [3]. Moreover, DS8201 have demonstrated a satisfactory efficacy in patients with metastatic BC HER2 low-expressing [3, 84].
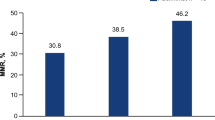
Disitamab vedotin.Disitamab vedotin, previously known as RC-48, is a novel ADC consisting of a humanized monoclonal antibody directed against HER-2 conjugated to MMAE via a cleavable linker [85]. In 2021 Shent et al., in a phase II study, evaluated the efficacy and safety of RC-48 in 43 patients with HER2 + (IHC 3 + and 2 +) locally advanced or metastatic UC refractory to standard therapies. They demonstrated a promising efficacy of RC-48 observing an ORR of 51%, an mPFS and mOS of 6.9 and 13.9 months, respectively, with a manageable safety profile [80]. This trial observed a higher ORR compared to historic response rates of currently available ICIs in the second-line setting. Indeed, another phase II trial, enrolling 100 patients, is underway to evaluate whether RC-48 works to treat HER2 expressing urothelial cancer (NCT04879329).
Mechanism of resistance
Little is currently known about potential resistance mechanisms against ADC treatment in UC. Further investigation is needed to shed light on drug-intrinsic mechanisms and streamline the identification of predictive biomarkers of drug efficacy. Preliminary results link ADC resistance to various biochemical mechanisms including alteration of the cell cycle, loss of payload efficacy, alteration of vesicle pathways and prevention of antibody attachment and loss of target antigen [16, 33].
Impairment of cell-cycle
It is well established that the cell cycle plays a pivotal role in generating novel resistance mechanisms and a recent study showed that the expression of cyclin B is significantly increased in TDM-1-resistant cells [86]. Furthermore, modifications of the apoptosis pathway might interfere with the efficacy of ADCs. There is evidence of overexpression and mutation of BCL-X and BCL-2, plus impairment of protein regulation of BAX and BAK pathways in patients treated with Gemtuzumab ozogamicin [87].
Inhibition of payload efficacy
A common mechanism of resistance has been shown to arise following mutations occurring in the molecular target of the payload. For example, the decreased success of SG treatment might be due to resistance mutations in topoisomerase-1 [88]. In addition, ATP-binding cassette (ABC) transporters are deemed to be a frequent mechanism of chemotherapy resistance, acting to increase drug discharge from the cell microenvironment [89]. Several ADC payloads are targeted against ABC efflux transporters, thus conferring resistance to ADC treatment [90, 91]. Myatansinoids and auristatin analogues have been previously reported to be substrates for ABC transporters including multidrug resistance-1 (MDR-1) in preclinical data. Exposing the cell to these agents can result in the overexpression of MDR-1 efflux transporters [92].
Impairment of vesicle pathways
There is preclinical evidence of decreased treatment sensitivity resulting from the internalization of TDM-1 into caveolin-1-coated vesicles [93]. Although the antibody internalisation into the cell (by endocytosis) is required to promote ADC efficacy, this process might curb payload efficacy. It has been shown that the internalization process might take place by means of clathrin-caveolin-independent, clathrin-mediated, and caveolin-mediated endocytosis mechanisms [94].
Loss of target antigen
Loss or reduction of target tumour antigen can occur due to a multitude of reasons. Examples include; gene mutation resulting in antigen concealment to the immune system or downregulation of target gene expression or clone selection of those tumour cells with lower target antigen expression. These mechanisms are a common hurdle to maximising ADC treatment efficacy [95] as the loss or reduction of cancer target antigen might result in the release of payload or loss of antibody binding. A study carried out on patients with metastatic triple-negative breast cancer (TNBC) showed that the loss of Trop-2 expression was associated with decreased response to SG treatment [88]. The ASCENT trial compounded that finding by demonstrating that metastatic TNBC patients with high Trop-2 expression, treated with SG, reported better outcomes when compared to those with low or absent Trop-2 expression [96]. Comparably, the EMILIA trial (results of which led to TDM-1 approval for HER-2-positive metastatic cancer patients) observed that patients with higher expression of HER-2 mRNA reported better outcomes when compared to those with lower HER-2 mRNA levels [97]. Nonetheless, preliminary research studies are underway to verify the efficacy of bispecific antibodies (those able to target multiple antigens) with the ultimate goal of overcoming this particular resistance mechanism.
The role of the tumour microenvironment (TME)
Recent evidence showed that the TME plays a pivotal role in regulating tumour progression, metastasis, immune escape, and it is involved in acquired resistance of tumours to various therapies, resulting in reduced treatment efficacy [98, 99]. Several mechanisms within the TME are deemed to lead to drug resistance. For instance, hypoxia and impaired blood supply, which results from the uncontrolled proliferation of tumour, are a cornerstone of TME in all solid tumours [100]. Hypoxia and impaired blood supply result in abnormal angiogenesis, inflammation and desmoplasia, all of which contribute to tumour progression and therapeutic resistance [101]. Additionally, hypoxia promotes decreased pH in the TME which supports multi-drug-resistances strategies including reduced apoptotic rate, increased activity of multidrug transported p-glycoprotein (P-gp/MDR1)(“drug efflux pump”), genetic alterations such as p53 mutations, and decreased concentration of the drug due to “ion trapping”—namely the inability for charged drugs to diffuse through cells [102].
Future options to overcome resistance and optimise ADC-based therapy
Although three new ADCs have been recently granted approval for the treatment of solid cancers, a major limit to ADC clinical success is resistance to these drugs. Nonetheless, ADC modular structure and the biochemical improvements will allow soon the development of new agents capable of overcoming resistance. Indeed, ADCs engineering has recently introduced new payloads, linkers and the development of a novel generation of ADCs with an increased drug-to-antibody ratio (DAR) and solid bystander effects [103]. The use of novel cleavable linkers in association with membrane-permeable payload can improve the efficacy of the bystander effect, enabling ADCs to be active against target-negative cells, namely expanding ADC efficacy to cancer with low target expression or on heterogeneous tumours [36, 104]. Another strategy that can be used is the increase of linker hydrophilicity, which can impair drug resistance as P-gp/MDR1 binds to hydrophobic compounds more efficiently than hydrophilic compounds [105]. Other studies have attempted to improve the stability of ADCs in plasma by altering the composition of linkers, focusing on replacing the most susceptible to degradation linker components with more stable substitutes [106].
Additional strategies have recently been investigated to expand the group of patients who might benefit from the newer generation of ADCs. Noticeably, new potential targets including proteins expressed by cancer stem cells (CSC), such as PTK7, ephrin-A4, 5T4 and in the TME, such as CD205, CD25, B7-H3, are under investigation with some of these that already reached clinical phases of drug development [107, 108]. Bispecific and biparatopic antibodies are also under investigation in preclinical studies. While bispecific antibodies can recognize two different antigens on the same antigen, biparatopic antibodies bind two non-overlapping epitopes of the same antigen. Additionally, a newer thread of research is focusing on smarter vehicles for payloads [109]. Probody drug conjugates are a novel group of ADC prodrugs that can be activated following proteolytic cleavage by TME proteases to minimise on-target/off-tumour toxicity [110].
More importantly, the most promising results are coming from several ongoing trials investigating the efficacy of novel ADCs in combination with several targeted agents such as immune checkpoint inhibitors (ICIs).The advent of ICIs and the more recent introduction of FGFR-targeted therapy have significantly altered the treatment paradigm of advanced urothelial cancer. Additionally, the even more recent introduction of ADCs (and the FDA approval of EV) are at the forefront of urothelial cancer treatment with encouraging preliminary and clinical data. ADCs have a multitude of benefits as treatment options. Firstly, response rates are promising (so far demonstrated to be between 30 and 60%) and comparable to current cisplatin-based first-line regimens. Secondly, response to ADCs has been shown to be durable with manageable toxicity. Finally given the universal expression of urothelial cancer targets, a large fraction of patients would most likely be considered eligible or appropriate for ADC treatment. With that in mind, what does the future hold for ADC treatment in urothelial cancer? It is highly likely that additional ADC agents will be granted approval for advanced urothelial cancer treatment, particularly as interest moves toward studying the outcome of ADC therapy in treatment-naïve patients.
Furthermore, the combination of ICIs and ADCs is a strategy that needs to be considered, as the biochemical effect is potentially synergistic with no overlapping toxicity. For example, ADCs can promote cell death, resulting in cancer antigen release, leading to immune system activation and an increase of antigen-presenting cells [111]. At the same time, ICIs can regulate immunosuppression within the tumour microenvironment by modulating cytokines, enzymes and T immune cells with immunomodulatory functions [112]. Therefore, ADC agents have potential synergistic activity in combination with ICIs. The ability of ADCs to modulate the immune system is under investigation in preclinical studies. Gardai et al. reported that ADCs with an MMAE payload can trigger anti-tumour immunity and stimulate immune cell death by induction of damage-associated molecular patterns (DAMPs) on the cell surface which, in turn, can trigger the immune system [113].
Further research in murine models has shown that tumour shrinkage was increased with a PD1 inhibitor and brentuximab combination treatment, corroborating the potential synergism of the two drug classes [114]. According to this preclinical data, it can be fairly confidently assumed that a combination of ADC and ICI might lead to a stronger anti-tumour response in vivo. Notably, combination treatment with ICIs and ADCs has already been granted regulatory approval for several cancers, and different combinations of these two agents are currently under clinical trial investigation. The EV-103 trial, a multi-arm phase Ib/II study investigating the efficacy of EV alone or in association with chemotherapy and/or pembrolizumab in locally advanced urothelial cancer, has reported a greater benefit in terms of ORR (73%) and mPFS (12.3 months) in the first-line setting when combinations are used [115].
In addition, the EV-304 trial, a randomized phase III open-label study in cisplatin-eligible patients is underway for the investigation of early-setting efficacy in patients treated with either EV plus pembrolizumab or neoadjuvant cisplatin in combination with gemcitabine [116]. Another phase III global study, VOLGA, is testing the efficacy and safety of neoadjuvant treatment with EV plus durvalumab and tremelimumab or EV plus durvalumab in cisplatin-ineligible MIBC [117]. As far as the development of novel ADCs is concerned, different agents including integrin β6, EGFR, B7-H1 and CD25 are being evaluated in early phase basket trials with the ultimate goal of better understanding and overcoming primary and acquired resistance mechanisms as well as limiting toxicity (Table 2).
Conclusion
Until recently, chemotherapy was the only treatment available for advanced or mUC. However, over recent years, UC treatment has benefited from multiple advances, and now more targeted therapy exists, in the form of immunotherapy. However, the outcome for these patients remains poor in the long term. ADCs are innovative drug agents, which allow conventional cytotoxic therapies to be transformed into highly targeted chemotherapeutics, potentially enabling better outcomes and reduced toxicity. ADCs offer particular promise in UC as we know that multiple tumour-specific antigens are highly expressed. As a result of this specificity and potential efficacy, ADCs offer a renewed hope for those malignancies with limited therapeutic strategies such as locally advanced or metastatic UC. The ongoing drug development within approved clinical trials will elucidate the optimal sequencing or combination of these drugs. The end goal is a personalized approach to the treatment of urothelial cancer, resulting in improved outcomes for patients.
References
Lattanzi M, Rosenberg JE (2020) The emerging role of antibody-drug conjugates in urothelial carcinoma. Expert Rev Anticancer Ther. https://doi.org/10.1080/14737140.2020.1782201
Verma S, Miles D, Gianni L et al (2012) Trastuzumab emtansine for HER2-positive advanced breast cancer. N Engl J Med 367:1783–1791. https://doi.org/10.1056/NEJMOA1209124
Modi S, Saura C, Yamashita T et al (2020) Trastuzumab deruxtecan in previously treated HER2-positive breast cancer. N Engl J Med 382:610–621. https://doi.org/10.1056/NEJMOA1914510/SUPPL_FILE/NEJMOA1914510_DATA-SHARING.PDF
Connors JM, Jurczak W, Straus DJ et al (2018) Brentuximab vedotin with chemotherapy for stage III or IV Hodgkin’s lymphoma. N Engl J Med 378:331–344. https://doi.org/10.1056/NEJMOA1708984/SUPPL_FILE/NEJMOA1708984_DISCLOSURES.PDF
Kantarjian HM, DeAngelo DJ, Stelljes M et al (2016) Inotuzumab ozogamicin versus standard therapy for acute lymphoblastic leukemia. N Engl J Med 375:740–753. https://doi.org/10.1056/NEJMOA1509277
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J Clin 71:209–249. https://doi.org/10.3322/CAAC.21660
Ravi P, McGregor BA (2021) Antibody-drug conjugates for the treatment of urothelial carcinoma. Expert Opin Biol Ther 21:1–8. https://doi.org/10.1080/14712598.2020.1789096
van Rhijn BWG, Burger M, Lotan Y et al (2009) Recurrence and progression of disease in non-muscle-invasive bladder cancer: from epidemiology to treatment strategy. Eur Urol 56:430–442. https://doi.org/10.1016/J.EURURO.2009.06.028
Babjuk M, Oosterlinck W, Sylvester R et al (2011) EAU guidelines on non-muscle-invasive urothelial carcinoma of the bladder, the 2011 update. Eur Urol 59:997–1008. https://doi.org/10.1016/J.EURURO.2011.03.017
Fuge O, Vasdev N, Allchorne P, Green JS (2015) Immunotherapy for bladder cancer. Res reports Urol 7:65–79. https://doi.org/10.2147/RRU.S63447
Sternberg CN, Skoneczna I, Kerst JM et al (2015) Immediate versus deferred chemotherapy after radical cystectomy in patients with pT3-pT4 or N+ M0 urothelial carcinoma of the bladder (EORTC 30994): an intergroup, open-label, randomised phase 3 trial. Lancet Oncol 16:76–86. https://doi.org/10.1016/S1470-2045(14)71160-X
Shabsigh A, Korets R, Vora KC et al (2009) Defining early morbidity of radical cystectomy for patients with bladder cancer using a standardized reporting methodology. Eur Urol 55:164–176. https://doi.org/10.1016/J.EURURO.2008.07.031
Von der Maase H, Hansen SW, Roberts JT et al (2000) Gemcitabine and cisplatin versus methotrexate, vinblastine, doxorubicin, and cisplatin in advanced or metastatic bladder cancer: results of a large, randomized, multinational, multicenter, phase III study. J Clin Oncol 18:3068–3077. https://doi.org/10.1200/JCO.2000.18.17.3068
Galsky MD, Hahn NM, Rosenberg J et al (2011) Treatment of patients with metastatic urothelial cancer “unfit” for Cisplatin-based chemotherapy. J Clin Oncol 29:2432–2438. https://doi.org/10.1200/JCO.2011.34.8433
De Santis M, Bellmunt J, Mead G et al (2012) Randomized phase II/III trial assessing gemcitabine/carboplatin and methotrexate/carboplatin/vinblastine in patients with advanced urothelial cancer who are unfit for cisplatin-based chemotherapy: EORTC study 30986. J Clin Oncol 30:191–199. https://doi.org/10.1200/JCO.2011.37.3571
Ungaro A, Tucci M, Audisio A et al (2022) Antibody-drug conjugates in urothelial carcinoma: a new therapeutic opportunity moves from bench to bedside. Cells 11:1–20. https://doi.org/10.3390/cells11050803
Bellmunt J, de Wit R, Vaughn DJ et al (2017) Pembrolizumab as second-line therapy for advanced urothelial carcinoma. N Engl J Med 376:1015–1026
Powles T, Durán I, van der Heijden MS et al (2018) Atezolizumab versus chemotherapy in patients with platinum-treated locally advanced or metastatic urothelial carcinoma (IMvigor211): a multicentre, open-label, phase 3 randomised controlled trial. Lancet 391:748–757. https://doi.org/10.1016/S0140-6736(17)33297-X
Suzman DL, Agrawal S, Ning Y et al (2019) FDA approval summary: atezolizumab or pembrolizumab for the treatment of patients with advanced urothelial carcinoma ineligible for cisplatin-containing chemotherapy. Oncologist 24:563–569. https://doi.org/10.1634/THEONCOLOGIST.2018-0084
Bellmunt J, Théodore C, Demkov T et al (2009) Phase III trial of vinflunine plus best supportive care compared with best supportive care alone after a platinum-containing regimen in patients with advanced transitional cell carcinoma of the urothelial tract. J Clin Oncol 27:4454–4461. https://doi.org/10.1200/JCO.2008.20.5534
Kim YS, Kim K, Kwon GY et al (2018) Fibroblast growth factor receptor 3 (FGFR3) aberrations in muscle-invasive urothelial carcinoma. BMC Urol 18:1–7. https://doi.org/10.1186/S12894-018-0380-1/FIGURES/1
Loriot Y, Necchi A, Park SH et al (2019) Erdafitinib in locally advanced or metastatic urothelial carcinoma. N Engl J Med 381:338–348. https://doi.org/10.1056/NEJMOA1817323
Sliwkowski MX, Mellman I (2013) Antibody therapeutics in cancer. Science. 341(6151):1192–8. https://doi.org/10.1126/science.1241145. PMID: 24031011
Sievers EL, Senter PD (2013) Antibody-drug conjugates in cancer therapy. Annu Rev Med 64:15–29. https://doi.org/10.1146/annurev-med-050311-201823
Thomas A, Teicher BA, Hassan R (2016) Antibody–drug conjugates for cancer therapy. Lancet Oncol 17:e254–e262. https://doi.org/10.1016/S1470-2045(16)30030-4
Hoffmann RM, Coumbe BGT, Josephs DH et al (2017) Antibody structure and engineering considerations for the design and function of antibody drug conjugates (ADCs). OncoImmunology. https://doi.org/10.1080/2162402X.2017.1395127
Saunders KO (2019) Conceptual approaches to modulating antibody effector functions and circulation half-life. Front Immunol 10:1296. https://doi.org/10.3389/FIMMU.2019.01296/BIBTEX
Vidarsson G, Dekkers G, Rispens T (2014) IgG subclasses and allotypes: From structure to effector functions. Front Immunol 5:520. https://doi.org/10.3389/FIMMU.2014.00520/BIBTEX
Tiller KE, Tessier PM (2015) Advances in antibody design. Annu Rev Biomed Eng 17:191–216. https://doi.org/10.1146/ANNUREV-BIOENG-071114-040733
Alley SC, Okeley NM, Senter PD (2010) Antibody–drug conjugates: targeted drug delivery for cancer. Curr Opin Chem Biol 14:529–537. https://doi.org/10.1016/J.CBPA.2010.06.170
Polson AG, Ho WY, Ramakrishnan V (2010) Investigational antibody-drug conjugates for hematological malignancies. Expert Opin Investig Drugs 20:75–85. https://doi.org/10.1517/13543784.2011.539557
Jain N, Smith SW, Ghone S, Tomczuk B (2015) Current ADC linker chemistry. Pharm Res 32:3526–3540. https://doi.org/10.1007/S11095-015-1657-7/FIGURES/16
Drake PM, Rabuka D (2015) An emerging playbook for antibody–drug conjugates: lessons from the laboratory and clinic suggest a strategy for improving efficacy and safety. Curr Opin Chem Biol 28:174–180. https://doi.org/10.1016/J.CBPA.2015.08.005
Ducry L, Stump B (2009) Antibody−drug conjugates: linking cytotoxic payloads to monoclonal antibodies. Bioconjug Chem 21:5–13. https://doi.org/10.1021/BC9002019
Drago JZ, Modi S, Chandarlapaty S (2021) Unlocking the potential of antibody–drug conjugates for cancer therapy. Nat Rev Clin Oncol 186(18):327–344. https://doi.org/10.1038/s41571-021-00470-8
Tsuchikama K, An Z (2018) Antibody-drug conjugates: recent advances in conjugation and linker chemistries. Protein Cell 9:33–46. https://doi.org/10.1007/S13238-016-0323-0/TABLES/1
Joubert N, Beck A, Dumontet C, Denevault-Sabourin C (2020) Antibody-drug conjugates: the last decade. Pharmaceuticals (Basel). https://doi.org/10.3390/PH13090245
Jabbour E, Paul S, Kantarjian H (2021) The clinical development of antibody–drug conjugates—lessons from leukaemia. Nat Rev Clin Oncol 187(18):418–433. https://doi.org/10.1038/s41571-021-00484-2
Lu J, Jiang F, Lu A, Zhang G (2016) Linkers having a crucial role in antibody-drug conjugates. Int J Mol Sci. https://doi.org/10.3390/IJMS17040561
Teicher BA, Chari RVJ (2011) Antibody conjugate therapeutics: challenges and potentialantibody conjugate therapeutics. Clin Cancer Res 17:6389–6397. https://doi.org/10.1158/1078-0432.CCR-11-1417
Saleh MN, Sugarman S, Murray J et al (2000) Phase I trial of the anti-Lewis Y drug immunoconjugate BR96-doxorubicin in patients with lewis Y-expressing epithelial tumors. J Clin Oncol 18:2282–2292. https://doi.org/10.1200/JCO.2000.18.11.2282
Kanellos J, Pietersz GA, McKenzie IFC (1985) Studies of methotrexate-monoclonal antibody conjugates for immunotherapy. JNCI J Natl Cancer Inst 75:319–332. https://doi.org/10.1093/JNCI/75.2.319
Trail PA, Willner D, Lasch SJ et al (1993) Cure of Xenografted Human Carcinomas by BR96-Doxorubicin Immunoconjugates. Science. https://doi.org/10.1126/SCIENCE.8327892
Waight AB, Bargsten K, Doronina S et al (2016) Structural basis of microtubule destabilization by potent Auristatin anti-mitotics. PLoS One 11:e0160890. https://doi.org/10.1371/JOURNAL.PONE.0160890
Barok M, Joensuu H, Isola J (2014) Trastuzumab emtansine: mechanisms of action and drug resistance. Breast Cancer Res 16:1–12. https://doi.org/10.1186/BCR3621/TABLES/2
Yardley DA, Krop IE, LoRusso PM et al (2015) Trastuzumab emtansine (T-DM1) in patients With HER2-positive metastatic breast cancer previously treated with chemotherapy and 2 or more HER2-targeted agents: results from the T-PAS expanded access study. Cancer J (United States) 21:357–364. https://doi.org/10.1097/PPO.0000000000000144
Zein N, Sinha AM, Mcgahren WJ, Ellestad GA (1988) Calicheamicin γ1I: an antitumor antibiotic that cleaves double-stranded DNA site specifically. Science. https://doi.org/10.1126/SCIENCE.3240341
Smellie M, Kelland LR, Thurston DE et al (1994) Cellular pharmacology of novel C8-linked anthramycin-based sequence-selective DNA minor groove cross-linking agents. Br J Cancer 701(70):48–53. https://doi.org/10.1038/bjc.1994.248
Jenkins TC, Hurley LH, Neidle S, Thurston DE (1994) Structure of a covalent DNA minor groove adduct with a pyrrolobenzodiazepine dimer: evidence for sequence-specific interstrand crosslinking. J Med Chem 37:4529–4537. https://doi.org/10.1021/JM00052A012/SUPPL_FILE/JM00052A012_SI_001.PDF
Hamblett KJ, Senter PD, Chace DF et al (2004) Effects of drug loading on the antitumor activity of a monoclonal antibody drug conjugate. Clin Cancer Res 10:7063–7070. https://doi.org/10.1158/1078-0432.CCR-04-0789
Lucas AT, Price LSL, Schorzman AN et al (2018) Factors affecting the pharmacology of antibody-drug conjugates. Antibodies. https://doi.org/10.3390/ANTIB7010010
Sun X, Ponte JF, Yoder NC et al (2017) Effects of drug-antibody ratio on pharmacokinetics, biodistribution, efficacy, and tolerability of antibody-maytansinoid conjugates. Bioconjug Chem 28:1371–1381. https://doi.org/10.1021/ACS.BIOCONJCHEM.7B00062/ASSET/IMAGES/ACS.BIOCONJCHEM.7B00062.SOCIAL.JPEG_V03
King HD, Dubowchik GM, Mastalerz H et al (2002) Monoclonal antibody conjugates of doxorubicin prepared with branched peptide linkers: inhibition of aggregation by methoxytriethyleneglycol chains. J Med Chem 45:4336–4343. https://doi.org/10.1021/JM020149G/ASSET/IMAGES/JM020149G.SOCIAL.JPEG_V03
Hwang WYK, Foote J (2005) Immunogenicity of engineered antibodies. Methods 36:3–10. https://doi.org/10.1016/J.YMETH.2005.01.001
Powles T, Rosenberg JE, Sonpavde GP et al (2021) Enfortumab vedotin in previously treated advanced urothelial carcinoma. N Engl J Med 384:1125–1135. https://doi.org/10.1056/NEJMOA2035807
Rikitake Y, Mandai K, Takai Y (2012) The role of nectins in different types of cell–cell adhesion. J Cell Sci 125:3713–3722. https://doi.org/10.1242/JCS.099572
Samanta D, Almo SC (2015) Nectin family of cell-adhesion molecules: structural and molecular aspects of function and specificity. Cell Mol Life Sci 72:645–658. https://doi.org/10.1007/S00018-014-1763-4/FIGURES/8
Challita-Eid PM, Satpayev D, Yang P et al (2016) Enfortumab vedotin antibody-drug conjugate targeting nectin-4 is a highly potent therapeutic agent in multiple preclinical cancer models. Cancer Res 76:3003–3013. https://doi.org/10.1158/0008-5472.CAN-15-1313
Kaplon H, Muralidharan M, Schneider Z, Reichert JM (2020) Antibodies to watch in 2020. MAbs. https://doi.org/10.1080/19420862.2019.1703531
Mar N, Friedlander TW, Hoimes CJ et al (2020) Study EV-103: new randomized cohort testing enfortumab vedotin as monotherapy or in combination with pembrolizumab in locally advanced or metastatic urothelial cancer. J Clin Oncol 38:TPS5092–TPS5092
Avellini C, Licini C, Lazzarini R, Gesuita R, Guerra E, Tossetta G, Castellucci C, Giannubilo SR, Procopio A, Alberti S et al. (2017) The trophoblast cell surface antigen 2 and miR-125b axis in urothelial bladder cancer. Oncotarget 8:58642–58653.
Smith NF, Figg WD, Sparreboom A (2006) Pharmacogenetics of irinotecan metabolism and transport: an update. Toxicol Vitr 20:163–175. https://doi.org/10.1016/j.tiv.2005.06.045
Moon SJ, Govindan SV, Cardillo TM et al (2008) Antibody conjugates of 7-ethyl-10-hydroxycamptothecin (SN-38) for targeted cancer chemotherapy. J Med Chem 51:6916–6926. https://doi.org/10.1021/JM800719T
Goldenberg DM, Cardillo TM, Govindan SV et al (2015) Trop-2 is a novel target for solid cancer therapy with sacituzumab govitecan (IMMU-132), an antibody-drug conjugate (ADC). Oncotarget 6:22496–22512. https://doi.org/10.18632/ONCOTARGET.4318
Guerra E, Trerotola M, Aloisi AL et al (2013) The Trop-2 signalling network in cancer growth. Oncogene 32:1594–1600. https://doi.org/10.1038/ONC.2012.151
Mcdougall ARA, Tolcos M, Hooper SB et al (2015) Trop2: from development to disease. Dev Dyn 244:99–109. https://doi.org/10.1002/DVDY.24242
Avellini C, Licini C, Lazzarini R et al (2017) The trophoblast cell surface antigen 2 and miR-125b axis in urothelial bladder cancer. Oncotarget 8:58642–58653. https://doi.org/10.18632/ONCOTARGET.17407
Tagawa ST, Balar AV, Petrylak DP et al (2021) TROPHY-U-01: a phase II open-label study of sacituzumab govitecan in patients with metastatic urothelial carcinoma progressing after platinum-based chemotherapy and checkpoint inhibitors. J Clin Oncol 39:2474–2485. https://doi.org/10.1200/JCO.20.03489
Bardia A, Messersmith WA, Kio EA et al (2021) Sacituzumab govitecan, a Trop-2-directed antibody-drug conjugate, for patients with epithelial cancer: final safety and efficacy results from the phase I/II IMMU-132-01 basket trial. Ann Oncol 32:746–756. https://doi.org/10.1016/j.annonc.2021.03.005
Morrison K, Challita-Eid PM, Raitano A et al (2016) Development of ASG-15ME, a novel antibody-drug conjugate targeting SLITRK6, a new urothelial cancer biomarker. Mol Cancer Ther 15:1301–1310. https://doi.org/10.1158/1535-7163.MCT-15-0570/87093/AM/DEVELOPMENT-OF-ASG-15ME-A-NOVEL-ANTIBODY-DRUG
Petrylak D, Heath E, Sonpavde G et al (2016) Interim analysis of a phase I dose escalation trial of the antibody drug conjugate (ADC) AGS15E (ASG-15ME) in patients (Pts) with metastatic urothelial cancer (mUC). Ann Oncol. https://doi.org/10.1093/ANNONC/MDW373.08
D-Y Bang, Y-J (2019) HER2-targeted therapies —A role beyond breast cancer. Nat. Rev. Clin. Oncol 17:33–48
Morlet T, Rabinowitz MR, Looney LR et al (2014) A homozygous SLITRK6 nonsense mutation is associated with progressive auditory neuropathy in humans. Laryngoscope 124:E95–E103. https://doi.org/10.1002/LARY.24361
Liu X, Liu Y, Liu Z et al (2021) Identification of SLITRK6 as a Novel Biomarker in hepatocellular carcinoma by comprehensive bioinformatic analysis. Biochem Biophys Rep 28:101157. https://doi.org/10.1016/J.BBREP.2021.101157
Holbro T, Beerli RR, Maurer F et al (2003) The ErbB2/ErbB3 heterodimer functions as an oncogenic unit: ErbB2 requires ErbB3 to drive breast tumor cell proliferation. Proc Natl Acad Sci USA 100:8933–8938. https://doi.org/10.1073/PNAS.1537685100/ASSET/ACDDD61C-7E8C-470C-8DF8-C7D321FC9821/ASSETS/GRAPHIC/PQ1537685005.JPEG
Yan M, Schwaederle M, Arguello D et al (2015) HER2 expression status in diverse cancers: review of results from 37,992 patients. Cancer Metastasis Rev. https://doi.org/10.1007/s10555-015-9552-6
Ferraro E, Drago JZ, Modi S (2021) Implementing antibody-drug conjugates (ADCs) in HER2-positive breast cancer: state of the art and future directions. Breast Cancer Res 23:1–11. https://doi.org/10.1186/S13058-021-01459-Y/TABLES/4
Chen D, Ye Y, Guo S, Yao K (2021) Progress in the research and targeted therapy of ErbB/HER receptors in urothelial bladder cancer. Front Mol Biosci 8:1272. https://doi.org/10.3389/FMOLB.2021.800945/BIBTEX
Fleischmann A, Rotzer D, Seiler R et al (2011) Her2 amplification is significantly more frequent in lymph node metastases from urothelial bladder cancer than in the primary tumours. Eur Urol 60:350–357. https://doi.org/10.1016/J.EURURO.2011.05.035
Sheng X, Yan X, Wang L et al (2021) Open-label, multicenter, phase II study of RC48-ADC, a HER2-targeting antibody-drug conjugate, in patients with locally advanced or metastatic urothelial carcinoma. Clin Cancer Res 27:43–51. https://doi.org/10.1158/1078-0432.CCR-20-2488/274631/AM/OPEN-LABEL-MULTICENTER-PHASE-2-STUDY-OF-RC48-ADC-A
Krüger S, Weitsch G, Büttner H et al (2002) Overexpression of c-erbB-2 oncoprotein in muscle-invasive bladder carcinoma: relationship with gene amplification, clinicopathological parameters and prognostic outcome. Int J Oncol 21:981–987. https://doi.org/10.3892/IJO.21.5.981/HTML
Battisti NMLL, Rogerson F, Lee K et al (2020) Safety and efficacy of T-DM1 in patients with advanced HER2-positive breast cancer The Royal Marsden experience. Cancer Treat Res Commun 24:100188. https://doi.org/10.1016/J.CTARC.2020.100188
Hayashi T, Seiler R, Oo HZ et al (2015) Targeting HER2 with T-DM1, an antibody cytotoxic drug conjugate, is effective in HER2 over expressing bladder cancer. J Urol 194:1120–1131. https://doi.org/10.1016/J.JURO.2015.05.087
Modi S, Park H, Murthy RK et al (2020) Antitumor activity and safety of trastuzumab deruxtecan in patients with HER2-low-expressing advanced breast cancer: results from a phase Ib study. J Clin Oncol 38:1887–1896. https://doi.org/10.1200/JCO.19.02318
Jiang J, Li S, Shan X et al (2020) Preclinical safety profile of disitamab vedotin: a novel anti-HER2 antibody conjugated with MMAE. Toxicol Lett 324:30–37. https://doi.org/10.1016/J.TOXLET.2019.12.027
Sabbaghi MA, Gil-Gomez G, Guardia C et al (2017) Defective cyclin B1 induction in trastuzumab-emtansine (T-DM1) acquired resistance in HER2-positive breast cancer. Clin Cancer Res 23:7006–7019. https://doi.org/10.1158/1078-0432.CCR-17-0696/14707/AM/DEFECTIVE-CYCLIN-B1-INDUCTION-IN-TRASTUZUMAB
Jedema I, Barge RMY, van der Velden VHJ et al (2004) (2003) Internalization and cell cycle-dependent killing of leukemic cells by Gemtuzumab Ozogamicin: rationale for efficacy in CD33-negative malignancies with endocytic capacity. Leuk 182(18):316–325. https://doi.org/10.1038/sj.leu.2403205
Coates JT, Sun S, Leshchiner I et al (2021) Parallel genomic alterations of antigen and payload targets mediate polyclonal acquired clinical resistance to sacituzumab govitecan in triple-negative breast cancer. Cancer Discov 11:2436–2445. https://doi.org/10.1158/2159-8290.CD-21-0702/673837/AM/PARALLEL-GENOMIC-ALTERATIONS-OF-ANTIGEN-AND
Yu M, Ocana A, Tannock IF (2012) Reversal of ATP-binding cassette drug transporter activity to modulate chemoresistance: why has it failed to provide clinical benefit? Cancer Metastasis Rev 321(32):211–227. https://doi.org/10.1007/S10555-012-9402-8
Kovtun YV, Audette CA, Mayo MF et al (2010) Antibody-maytansinoid conjugates designed to bypass multidrug resistance. Cancer Res 70:2528–2537. https://doi.org/10.1158/0008-5472.CAN-09-3546/655827/P/ANTIBODY-MAYTANSINOID-CONJUGATES-DESIGNED-TO
Cianfriglia M (2013) The biology of MDR1-P-glycoprotein (MDR1-Pgp) in designing functional antibody drug conjugates (ADCs): the experience of gemtuzumab ozogamicin. Ann Ist Super Sanita 49:150–168. https://doi.org/10.4415/ANN_13_02_07
Lambert JM, Chari RVJ (2014) Ado-trastuzumab emtansine (T-DM1): An antibody-drug conjugate (ADC) for HER2-positive breast cancer. J Med Chem 57:6949–6964. https://doi.org/10.1021/JM500766W/ASSET/IMAGES/JM500766W.SOCIAL.JPEG_V03
Sung M, Tan X, Lu B et al (2018) Caveolae-mediated endocytosis as a novel mechanism of resistance to trastuzumab emtansine (T-DM1). Mol Cancer Ther 17:243–253. https://doi.org/10.1158/1535-7163.MCT-17-0403/87273/AM/CAVEOLAE-MEDIATED-ENDOCYTOSIS-AS-A-NOVEL-MECHANISM
Kalim M, Chen J, Wang S et al (2017) Intracellular trafficking of new anticancer therapeutics: antibody–drug conjugates. Drug Des Devel Ther 11:2265–2276. https://doi.org/10.2147/DDDT.S135571
Robertson AG, Kim J, Al-Ahmadie H et al (2017) Comprehensive molecular characterization of muscle-invasive bladder cancer. Cell 171:540-556.e25
Madhumathi J, Devilakshmi S, Sridevi S, Verma RS (2016) Immunotoxin therapy for hematologic malignancies: where are we heading? Drug Discov Today 21:325–332. https://doi.org/10.1016/J.DRUDIS.2015.05.002
Baselga J, Phillips GDL, Verma S et al (2016) Relationship between tumor biomarkers and efficacy in EMILIA, a phase III study of trastuzumab emtansine in HER2-Positive metastatic breast cancer. Clin Cancer Res 22:3755–3763. https://doi.org/10.1158/1078-0432.CCR-15-2499/128479/AM/RELATIONSHIP-BETWEEN-TUMOR-BIOMARKERS-AND-EFFICACY
Jing X, Yang F, Shao C et al (2019) Role of hypoxia in cancer therapy by regulating the tumor microenvironment. Mol Cancer 18:1–15. https://doi.org/10.1186/s12943-019-1089-9
Whatcott CJ, Han H, Von Hoff DD (2015) Orchestrating the tumor microenvironment to improve survival for patients with pancreatic cancer: normalization, not destruction. Cancer J 21:299–306. https://doi.org/10.1097/PPO.0000000000000140
Shao C, Yang F, Miao S et al (2018) Role of hypoxia-induced exosomes in tumor biology. Mol Cancer 17:1–8. https://doi.org/10.1186/S12943-018-0869-Y/FIGURES/2
Wu T, Dai Y (2017) Tumor microenvironment and therapeutic response. Cancer Lett 387:61–68. https://doi.org/10.1016/J.CANLET.2016.01.043
Zub KA, De Sousa MML, Sarno A et al (2015) Modulation of cell metabolic pathways and oxidative stress signaling contribute to acquired melphalan resistance in multiple myeloma cells. PLoS One. https://doi.org/10.1371/JOURNAL.PONE.0119857
Criscitiello C, Morganti S, Curigliano G (2021) Antibody–drug conjugates in solid tumors: a look into novel targets. J Hematol Oncol 14:1–18. https://doi.org/10.1186/s13045-021-01035-z
Beck A, Goetsch L, Dumontet C, Corvaïa N (2017) Strategies and challenges for the next generation of antibody-drug conjugates. Nat Rev Drug Discov 16:315–337. https://doi.org/10.1038/NRD.2016.268
Bargh JD, Isidro-Llobet A, Parker JS, Spring DR (2019) Cleavable linkers in antibody-drug conjugates. Chem Soc Rev 48:4361–4374. https://doi.org/10.1039/C8CS00676H
Boni V, Sharma MR, Patnaik A (2020) The resurgence of antibody drug conjugates in cancer therapeutics: novel targets and payloads. Am Soc Clin Oncol Educ book Am Soc Clin Oncol Annu Meet 40:e58–e74. https://doi.org/10.1200/EDBK_281107
Chau CH, Steeg PS, Figg WD (2019) Antibody-drug conjugates for cancer. Lancet (London, England) 394:793–804. https://doi.org/10.1016/S0140-6736(19)31774-X
Abel M, Burkenroad A, Sun A et al (2021) The evolving landscape of antibody-drug conjugates for urothelial carcinoma. Clin Genitourin Cancer 19:183–193. https://doi.org/10.1016/j.clgc.2020.11.006
Maruani A (2018) Bispecifics and antibody–drug conjugates: a positive synergy. Drug Discov Today Technol 30:55–61. https://doi.org/10.1016/j.ddtec.2018.09.003
García-Alonso S, Ocaña A, Pandiella A (2018) Resistance to antibody–drug conjugates. Cancer Res 78:2159–2165. https://doi.org/10.1158/0008-5472.CAN-17-3671
Gerber HP, Sapra P, Loganzo F, May C (2016) Combining antibody–drug conjugates and immune-mediated cancer therapy: what to expect? Biochem Pharmacol 102:1–6. https://doi.org/10.1016/J.BCP.2015.12.008
Di YuW, Sun G, Li J et al (2019) Mechanisms and therapeutic potentials of cancer immunotherapy in combination with radiotherapy and/or chemotherapy. Cancer Lett 452:66–70. https://doi.org/10.1016/J.CANLET.2019.02.048
Gardai SJ, Epp A, Law C-L (2015) Abstract 2469: brentuximab vedotin-mediated immunogenic cell death. Cancer Res 75:2469–2469. https://doi.org/10.1158/1538-7445.AM2015-2469
Cao AT, Law C-L, Gardai SJ, Heiser RA (2017) Abstract 5588: Brentuximab vedotin-driven immunogenic cell death enhances antitumor immune responses, and is potentiated by PD1 inhibition in vivo. Cancer Res 77:5588–5588. https://doi.org/10.1158/1538-7445.AM2017-5588
Rosenberg JE, Flaig TW, Friedlander TW et al (2020) Study EV-103: Preliminary durability results of enfortumab vedotin plus pembrolizumab for locally advanced or metastatic urothelial carcinoma. J Clin Oncol 38:441–441
Hoimes CJ, Bedke J, Loriot Y et al (2021) KEYNOTE-B15/EV-304: Randomized phase 3 study of perioperative enfortumab vedotin plus pembrolizumab versus chemotherapy in cisplatin-eligible patients with muscle-invasive bladder cancer (MIBC). J Clin Oncol 39:4587–4587
A phase 3, randomized, open-label, multicenter, global study of the efficacy and safety of durvalumab (D) + tremelimumab (T) + enfortumab vedotin (EV) or D + EV for neoadjuvant treatment in cisplatin-ineligible muscle-invasive bladder cancer (MIBC) (VOLGA). https://ascopubs.org/doi/abs/10.1200/JCO.2022.40.6_suppl.TPS579
Rosenberg J, Sridhar SS, Zhang J, Smith D, Ruether D, Flaig TW, Baranda J, Lang J, Plimack ER, Sangha R, Heath EI, Merchan J, Quinn DI, Srinivas S, Milowsky M, Wu C, Gartner EM, Zuo P, Melhem-Bertrandt A, Petrylak DP. EV-101: A Phase I Study of Single-Agent Enfortumab Vedotin in Patients With Nectin-4-Positive Solid Tumors, Including Metastatic Urothelial Carcinoma. J Clin Oncol. 2020 Apr 1;38(10):1041–1049. https://doi.org/10.1200/JCO.19.02044 Epub 2020 Feb 7. Erratum in: J Clin Oncol. 2022 May 20;40(15):1711. PMID: 32031899; PMCID: PMC7106979.
Yu EY, Petrylak DP, O'Donnell PH, Lee JL, van der Heijden MS, Loriot Y, Stein MN, Necchi A, Kojima T, Harrison MR, Hoon Park S, Quinn DI, Heath EI, Rosenberg JE, Steinberg J, Liang SY, Trowbridge J, Campbell M, McGregor B, Balar AV. Enfortumab vedotin after PD-1 or PD-L1 inhibitors in cisplatin-ineligible patients with advanced urothelial carcinoma (EV‑201): a multicentre, single-arm, phase 2 trial. Lancet Oncol. 2021 Jun;22(6):872–882.https://doi.org/10.1016/S1470-2045(21)00094-2 Epub 2021 May 12. Erratum in: Lancet Oncol. 2021 Jun;22(6):e239. PMID: 33991512.
Funding
This paper is not funded.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We know of no conflicts of interest associated with this publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
D’Angelo, A., Chapman, R., Sirico, M. et al. An update on antibody–drug conjugates in urothelial carcinoma: state of the art strategies and what comes next. Cancer Chemother Pharmacol 90, 191–205 (2022). https://doi.org/10.1007/s00280-022-04459-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-022-04459-7