Abstract
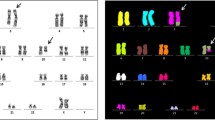
Deletion of the long arm of chromosome 20 is a recurrent abnormality observed in myelodysplastic syndromes (MDS) and in Philadelphia-chromosome-negative myeloproliferative disorders (MPD). Our objective was to characterize the deletion size among 38 MDS and MPD patients using fluorescence in situ hybridization (FISH) with bacterial artificial chromosome (BAC) probes and to define commonly deleted and retained regions on chromosome 20. Patients were distributed in three groups according to the World Health Organization classification: MDS (22 patients), MPD (12 patients) and myelodysplastic/myeloproliferative diseases (four patients). FISH with centromeric, subtelomeric, and unique sequence probes was performed to characterize the deletion whereas its size was delineated using BAC clones. All 38 deletions were found to be interstitial. A commonly deleted region was identified for each of the three groups; it varied from 6.62 to 10.4 Mb and showed considerable overlapping. Two commonly retained regions (CRR), also showing overlapping, were identified in all three groups, one in the centromeric region, the other in the telomeric region. The deletion size is highly variable, with no apparent recurrent breakpoint. The deletion may result in the loss of one or several tumor suppressor genes but the target genes remain unknown. Loss of genes plays an important part in the myeloid leukemic process associated with del(20q). However, genes located in the retained chromosomal regions may also play a role in the oncogenetic mechanisms.


Similar content being viewed by others
References
Knapp RH, Dewald GW, Pierre RV (1985) Cytogenetic studies in 174 consecutive patients with preleukemic or myelodysplastic syndromes. Mayo Clin Proc 60:507–516
Rege-Cambrin G, Mecucci C, Tricot G, Michaux JL, Louwagie A, Van Hove W, Francart H, Van den Berghe H (1987) A chromosomal profile of polycythemia vera. Cancer Genet Cytogenet 25:233–245
Bench AJ, Nacheva EP, Champion KM, Green AR (1998) Molecular genetics and cytogenetics of myeloproliferative disorders. Baillieres Clin Haematol 11:819–848
Testa JR, Kanofsky JR, Rowley JD, Baron JM, Vardiman JW (1981) Karyotypic patterns and their clinical significance in polycythemia vera. Am J Hematol 11:29–45
Demory JL, Dupriez B, Fenaux P, Lai JL, Beuscart R, Jouet JP, Deminatti M, Bauters F (1988) Cytogenetic studies and their prognostic significance in agnogenic myeloid metaplasia: a report on 47 cases. Blood 72:855–859
Djordjevic V, Dencic-Fekete M, Jovanovic J, Bizic S, Jankovic G, Bogdanovic A, Cemerikic-Martinovic V, Gotic M (2007) Cytogenetics of agnogenic myeloid metaplasia: a study of 61 patients. Cancer Genet Cytogenet 173:57–62
Brigaudeau C, Liozon E, Bernard P, Trimoreau F, Bordessoule D, Praloran V (1996) Deletion of chromosome 20q associated with hypereosinophilic syndrome. Cancer Genet Cytogenet 87:82–84
Vardiman JW, Harris NL, Brunning RD (2002) The World Health Organization (WHO) classification of the myeloid neoplasms. Blood 100:2292–2302
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR, Sultan C (1982) Proposals for the classification of the myelodysplastic syndromes. Br J Haematol 51:189–199
Murphy S (1999) Diagnostic criteria and prognosis in polycythemia vera and essential thrombocythemia. Semin Hematol 36:9–13
Herry A, Douet-Guilbert N, Morel F, Le Bris MJ, De Braekeleer M (2007) Redefining monosomy 5 by molecular cytogenetics in 23 patients with MDS/AML. Eur J Haematol 78:457–467
ISCN (2005) An international system for human cytogenetic nomenclature. Karger, Basel
Pinson L, Perrin A, Plouzennec C, Parent P, Metz C, Collet M, Le Bris MJ, Douet-Guilbert N, Morel F, De Braekeleer M (2005) Detection of an unexpected subtelomeric 15q26.2→qter deletion in a litte girl: clinical and cytogenetic studies. Am J Med Genet 138:160–165
Davis MP, Dewald GW, Pierre RV, Hoagland HC (1984) Hematologic manifestations associated with deletions of the long arm of chromosome 20. Cancer Genet Cytogenet 12:63–71
Le Beau MM, Westbrook CA, Diaz MO, Rowley JD (1985) c-src is consistently conserved in the chromosomal deletion (20q) observed in myeloid disorders. Proc Natl Acad Sci USA 82:6692–6696
Roulston D, Espinosa RI, Stoffel M, Bell GI, Le Beau MM (1993) Molecular genetics of myeloid leukemia: identification of the commonly deleted segment of chromosome 20. Blood 82:3424–3429
Asimakopoulos FA, White NJ, Nacheva E, Green AR (1994) Molecular analysis of chromosome 20q deletions associated with myeloproliferative disorders and myelodysplastic syndromes. Blood 84:3086–3094
Bench AJ, Aldred MA, Humphray SJ, Champion KM, Gilbert JGR, Asimakopoulos FA, Deloukas P, Gwilliam R, Bentley DR, Green AR (1998) A detailed physical and transcriptional map of the region of chromosome 20 that is deleted in myeloproliferative disorders and refinement of the commonly deleted segment. Genomics 49:351–362
Bench AJ, Nacheva EP, Hood TL, Holden JL, French L, Swanton S, Champion KM, Li J, Whittaker P, Stavrides G, Hunt AR, Huntly BJP, Campbell LJ, Bentley DR, Deloukas P, Green AR (2000) Chromosome 20 deletions in myeloid malignancies: reduction of the common deleted region, generation of a PAC/BAC contig and identification of candidate genes. UK Cancer Cytogenetics Group (UKCCG) Oncogene 19:3902–3913
Wang PW, Eisenbart JD, Espinosa RI, Davis EM, Larson RA, Le Beau MM (2000) Refinement of the smallest commonly deleted segment of chromosome 20 in malignant myeloid diseases and development of a PAC-based physical and transcription map. Genomics 68:28–32
Wang PW, Iannantuoni K, Davis EM, Espinosa R III, Stoffel M, Le Beau MM (1998) Refinement of the commonly deleted segment in myeloid leukemias with a del(20q). Genes, Chromosomes & Cancer 21:75–81
MacGrogan D, Alvarez S, DeBlasio T, Jhanwar S, Nimer SD (2001) Identification of candidate genes on chromosome band 20q12 by physical mapping of translocation breakpoints found in myeloid leukemia cell lines. Oncogene 20:4150–4160
Koga H, Matsui S, Hirota T, Takebayashi S, Okumura K, Saya H (1999) A human homolog of Drosophila lethal(3)malignant brain tumor (l(3)mbt) protein associates with condensed mitotic chromosomes. Oncogene 18:3799–3809
Li J, Bench AJ, Vassiliou GS, Fourouclas N, Ferguson-Smith AC, Green AR (2004) Imprinting of the human L3MBTL gene, a polycomb family mamver located in region of chromosome 20 deleted in human myeloid malignancies. Proc Natl Acad Sci USA 101:7341–7346
MacGrogan D, Kalakonda N, Alvarez S, Scandura JM, Boccuni P, Johansson B, Nimer SD (2004) Structural integrity and expression of the L3MBTL gene in normal and malignant hematopoietic cells. Genes, Chromosomes & Cancer 4:203–213
Ma Y, Lin D, Sun W, Xiao T, Yuan J, Han N, Guo S, Feng X, Su K, Mao Y, Cheng S, Gao Y (2006) Expression of targeting protein for Xklp2 associated with both malignant transformation of respiratory epithelium and progression of squamous cell lung cancer. Clin Cancer Res 12:1121–1127
Mackinnon RN, Patsouris C, Chudoba I, Campbell LJ (2007) A FISH comparison of variant derivatives of the recurrent dic(17;20) of myelodysplastic syndromes and acute myeloid leukemia: obligatory retention of genes on 17p and 20q may explain the formation of dicentric chromosomes. Genes, Chromosomes & Cancer 46:27–36
Pitti RM, Marsters SA, Lawrence DA, Roy M, Kischkel FC, Dowd P, Huang A, Donahue CJ, Sherwood SW, Baldwin DT, Godowski PJ, Wood WI, Gurney AL, Hillan KJ, Cohen RL, Goddard AD, Botstein D, Ashkenazi A (1998) Genomic amplification of a decoy receptor for Fas ligand in lung and colon cancer. Nature 396:699–703
Li H, Zhang L, Lou H, Ding I, Kim S, Wang L, Huang J, Di Sant'Agnese PA, Lei JY (2005) Overexpression of decoy receptor 3 in precancerous lesions and adenocarcinoma of the esophagus. Am J Clin Pathol 124:282–287
Garcia-Domingo D, Ramirez D, Gonzalez DB, Martinez A (2003) Death inducer-obliterator I triggers apoptosis after nuclear translocation and caspase upregulation. Mol Cell Biol 23:3216–3225
Futterer A, Campanero MR, Leonardo E, Criado LM, Flores JM, Hernandez JM, San Miguel JF, Martinez AC (2005) Dido gene expression alterations are implicated in the induction of hematological myeloid neoplasms. J Clin Invest 115:2351–2362
Rodier F, Kim SH, Nijjar T, Yaswen P, Campisi J (2005) Cancer and aging: the importance of telomeres in genome maintenance. Int J Biochem Cell Biol 37:977–990
Gilley D, Tanaka H, Herbert BS (2005) Telomere dysfunction in aging and cancer. Int J Biochem Cell Biol 37:1000–1013
De Lange T (1995) Telomere dynamics and genome instability in human cancer. In: Blackburn EH, Greider CW (eds) Telomeres. Cold Spring Harbor, Plainview, NY, pp 265–293
Murnane JP, Sabatier L (2004) Chromosome rearrangements resulting from telomere dysfunction and their role in cancer. BioEssays 26:1164–1174
Sabatier L, Ricoul M, Pottier G, Murnane JP (2005) The loss of a single telomere can result in instability of multiple chromosomes in a human tumor cell line. Mol Cancer Res 3:139–150
Fenaux P, Morel P, Lai JL (1996) Cytogenetics of myelodysplastic syndromes. Semin Hematol 33:127–138
Vallespi T, Imbert M, Mecucci C, Preudhomme C, Fenaux P (1998) Diagnosis, classification, and cytogenetics of myelodysplastic syndromes. Haematologica 83:258–275
Liu YC, Ito Y, Hsiao HH, Sashida G, Kodama A, Ohyashiki JH, Ohyashiki K (2006) Risk factor analysis in myelodysplastic syndrome patients with del(20q): prognosis revisited. Cancer Genet Cytogenet 171:9–16
Greenberg P, Cox C, LeBeau MM, Fenaux P, Morel P, Sanz G, Sanz M, Vallespi T, Hamblin T, Oscier D, Ohyashiki K, Toyama K, Aul C, Mufti G, Bennett J (1997) International socring system for evaluating prognosis in myelodysplastic syndromes. Blood 89:2079–2088
Brezinova J, Zemanova Z, Ransdorfova S, Sindelarova L, Siskova M, Neuwirtiva R, Cermak J, Michalova K (2005) Prognostic significance of del(20q) in patients with hematological malignancies. Cancer Genet Cytogenet 160:188–192
Ohyashiki K, Murakami T, Ohyashiki JH, Kodama A, Sakai N, Ito H, Toyama K (1992) Double 20q−Anomaly in myelodysplastic syndrome. Cancer Genet Cytogenet 58:174–176
Kubonishi I, Muneishi H, Hashimoto E, Hatakeyama N, Kuzume T, Miyoshi I (1996) Duplication of del(20)(q11) in a case of MDS. Cancer Genet Cytogenet 86:185–186
Kurtin PJ, Dewald GW, Shields DJ, Hanson CA (1996) Hematologic disorders associated with deletions of chromosome 20q: a clinicopathologic study of 107 patients. Am J Clin Pathol 106:680–688
Parlier V, Van Melle G, Beris P, Schmidt PM, Tobler A, Haller E, Bellomo HJ (1994) Hematologic, clinical, and cytogenetic analysis in 109 patients with primary myelodysplastic syndrome. Prognostic significance of morphology and chromosome findings. Cancer Genet Cytogenet 78:219–231
Campbell LJ, Garson OM (1994) The prognostic significance of deletion of the long arm of chromosome 20 in myeloid disorders. Leukemia 8:67–71
Falzetti D, Vermeesch JR, Hood TL, Nacheva EP, Matteucci C, Martelli MF, Van den Berghe H, Marynen P, Mecucci C (1999) Identification of multiple copies of a 20q−chromosome in a case of myelodysplastic syndrome: a FISH study. Leukemia Research 23:407–413
Li T, Xue Y, Wu Y, Pan J (2004) Clinical and molecular cytogenetic studies in seven patients with myeloid diseases characterized by i(20q−). Br J Haematol 125:337–342
Author information
Authors and Affiliations
Corresponding author
Additional information
Nathalie Douet-Guilbert and Audrey Basinko contributed equally to the study.
Rights and permissions
About this article
Cite this article
Douet-Guilbert, N., Basinko, A., Morel, F. et al. Chromosome 20 deletions in myelodysplastic syndromes and Philadelphia-chromosome-negative myeloproliferative disorders: characterization by molecular cytogenetics of commonly deleted and retained regions. Ann Hematol 87, 537–544 (2008). https://doi.org/10.1007/s00277-008-0462-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-008-0462-3